Abstract
With a rise in the complexity and widening of its applicability in diagnostic procedures, a parallel rise has been witnessed in the use of contrast agents and information about contrast-induced acute kidney injury. Contrast media play a critical role in medical imaging procedures today, and physicians and diagnostic radiologists are responsible for assessing the potential risks of contrast media with diagnostic benefits. Physicians are currently focusing on preventive therapies and identifying risk factors because there is no particular treatment for CI-AKI. Although CI-AKI is uncommon in general, the risk of CI-AKI can increase up to 20% to 30% in individuals with risk factors. Numerous models have been used in clinical settings to evaluate CI-AKI risk factors, identify high-risk populations, and enhance the value of care provided to these patients. Over the last several years, effective preventive strategies have helped reduce the CI-AKI risk. However, several unanswered questions still remain due to the evolving diagnostic and therapeutic interventions paradigm. This review summarizes CI-AKI epidemiology, pathogenesis, risk factors, and prevention strategies to manage this complication in Indian populations effectively.
Introduction
Contrast-induced acute kidney injury (CI-AKI) remains a worrisome problem among patients given intravascular contrast agents. According to the European Society of Urogenital Radiology (ESUR) CI-AKI or “post-contrast acute kidney injury (PC-AKI) is defined as an increase in serum creatinine >0.3 mg/dL (or >26.5 µmol/L), or >1.5 times baseline, within 48 to 72 h of intravascular administration of a contrast agent.” 1 According to the most recent guidelines by the Kidney Disease Improving Global Outcomes working group, CI-AKI is defined as kidney injury following contrast injection if seen within 48−72 h after contrast media administration: (a) a rise in serum creatinine of ≥26.4 µmol/L (0.3 mg/dL); (b) rise in serum creatinine ≥1.5−1.9 times from baseline; (c) reduction in the urine output <0.5 mL/kg/h. 2 Physiological changes caused by the nephrotoxic outcome of contrast agents on tubular epithelial cells and the production of vasoactive molecules have been linked to the development of an oxidative stress state and resultant ischemic renal cell injury. 3 Patients with already present chronic kidney disease (CKD), cardiovascular disease (CVD), and type 2 diabetes mellitus (T2DM) have fewer functional nephrons and a diminished capacity to restore tubular epithelial cells. When exposed to contrast material (CM), these patients present a unique challenge to the vulnerable kidney. The prevalence of these diseases increases with age. By 2025, India is projected to have 157 million elderly populations. By 2050, India is projected to have 297 million senior citizens, representing 18% of the country’s population. Elderly patients also pose multiple challenges in the form of advanced complex coronary diseases and multiple comorbidities. 4
However, there are no effective treatments for contrast-induced nephropathy once it has developed; prevention is the cornerstone of management. 1 In recent years, intravenous hydration, pharmacological medicines, and renal replacement therapy have contributed to a decline in CI-AKI incidence. Iodinated contrast media are frequently administered intravenously during computerized tomography investigations and intra-arterially during angiographic operations. The high osmolarity of the contrast agents leads to the absorption of water from the extravascular space and dilutes the CM, reducing the overall vascular attenuation. Iso-osmolar contrast media (IOCM) could prevent fluid entry from the extravascular space and provide stable CM levels. 5 This article aims to offer physicians and radiologists a comprehensive understanding of the safe usage of contrast media in the Indian population by summarizing the epidemiology, pathogenesis, and protective approaches of CI-AKI.
Risk Factors
A range of contributory factors predisposing CI-AKI reflects the mechanisms of contrast media-induced injury. The contrast medium is primarily excreted by glomerular filtration. The reduction in blood supply to the medullary region of the kidney exacerbates the contrast-induced toxicity to renal cells. Comorbidities worsen the fundamental etiology of the damage, which is contrast-induced vasoconstriction, resulting in decreased blood supply to the renal medulla. These comorbidities include T2DM, congestive heart failure, abrupt hypotension (requiring vasoconstrictors or intra-aortic balloon pump), myocardial infarction, and volume diminution. 4 Patients with chronic renal disease are susceptible to CI-AKI because compensatory processes to uphold filtration performance are decreased, and fewer nephrons are available to eliminate the contrast load. Additional risk factors include procedural considerations such as the measure and class of contrast used. The grading of risk factors (including both pre-existing comorbidities and procedural factors) has been used to guide the occurrence of CI-AKI, the need for renal replacement therapy, and long-term mortality. Patients with cancer have an increased chance of developing AKI (12%−17%) than those without cancer (5%−8%). 6 Cancer subtype, presence of other diseases, and use of several medicines contribute to increased risk (e.g., furosemide, antibiotics, chemotherapy, and iodinated CM administration). Patients treated with pemetrexed, ifosfamide, cisplatin, bevacizumab, and cetuximab, among other routinely used anticancer drugs, are highly susceptible to developing AKI. A high risk of AKI is seen when there is less time between chemotherapy and CM administration. 6
Recent Developments in Risk Assessment
Percutaneous cardiac intervention patients should undergo risk stratification efforts. Independent predictors of CI-AKI in patients following cardiac interventions include estimated glomerular filtration rate (eGFR), diabetes mellitus, and CM dosage. Renal impairment at the start is probably the most significant risk factor. The danger is amplified if the person is dehydrated because of insufficient fluid intake or the use of diuretic drugs. Adequate volume enhancement before and after the contrast administration may improve renal blood flow and avoid CI-AKI. Therefore, getting plenty of fluids, including near-isotonic and bicarb-based solutions, and stopping the use of nephrotoxic drugs before the treatment is recommended. There is a positive synergy between diabetes mellitus and low baseline GFR. Proteinuria is a strong predictor of CI-AKI because it indicates the presence of kidney injury that has already occurred. If a known risk factor (renal issues, T2DM, age >75, nephrotoxic drug use, gout, high blood pressure) is present or if risk factors are unknown, a quantitative estimate of GFR should be made using one of the validated formulae (such as the Cockcroft-Gault and Modification of Diet in Renal Disease [MDRD] equations, Chronic Kidney Disease Epidemiology Collaboration [CKD-EPI] creatinine equation) before angiography. AKI is more likely in patients with a GFR <60 mL/min/1.73 m2.6–12
Customized hydration approaches have an increased chance of dispersion, depicting the broad scope of volume infusion. All tailored strategies have been shown to have higher benefits compared to fixed hydration. A large reduction with reference to CI-AKI was reported for urine flow rate-directed and central venous pressure-directed hydration strategies. 13
A pooled analysis has shown that anemia increases the susceptibility to CI-AKI in patients experiencing coronary angiography. Besides, anemia raises oxygen-free radical injury and imbalances of vasoactive substances, which can encourage apoptosis and injury to immune cells in the kidney. 14
In addition, the individuals with aberrant BMI values have also shown an increased AKI risk for BMI <18.5 group and BMI >28.0. A rise in BMI beyond 24.0 significantly raises the chance of developing CS-AKI. 15
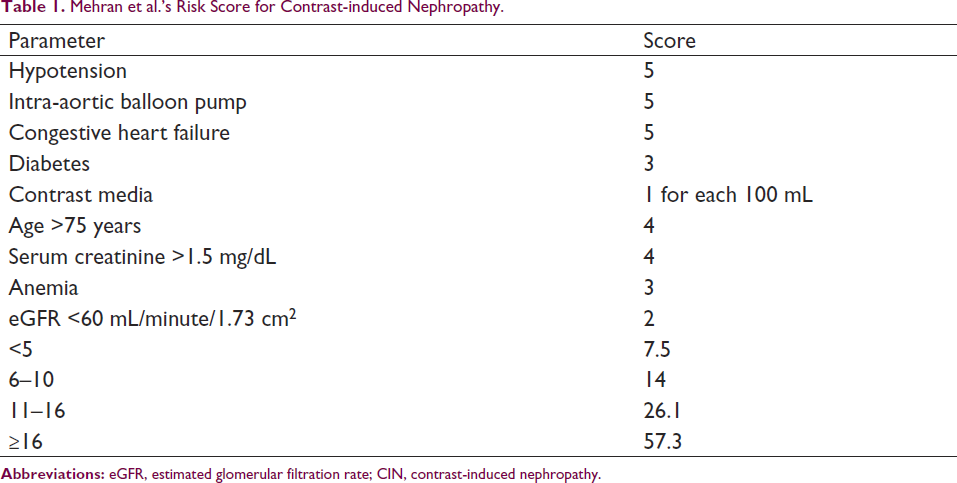
Acute coronary syndromes and myocardial infarction provide the greatest difficulties, especially when combined with hypotension or cardiogenic shock. Angiography and therapy must often be administered urgently. Patients with acute myocardial infarction have a high risk of CI-AKI. In these situations, physicians/radiologists may be compelled to utilize high CM doses without having enough time to prepare patients adequately. There are a variety of CI-AKI risk scores available, but the one produced by Mehran et al. is the most widely used (Table 1).8–12, 16, 17
Mehran et al.’s Risk Score for Contrast-induced Nephropathy.
Prevalence of CI-AKI in India
The burden of CI-AKI, its risk factors, and the development of preventative measures can all be improved with a more profound knowledge of the disease’s origins. However, incidence rates of AKI in different regions of India are poorly understood. There are no national statistics on the prevalence of AKI in Indian Intensive Care Units (ICUs). The data are derived from numerous single-center investigations and are highly fragmented. According to prior studies, the incidence of AKI in the Indian ICU seems equally high compared to the global data. Hospitalized patients with AKI have significantly higher rates of healthcare resource utilization. The prevalence of CI-AKI in India has been summarized in Table 2 for the convenience of the readers.18–31
Epidemiology of CI-AKI in India.
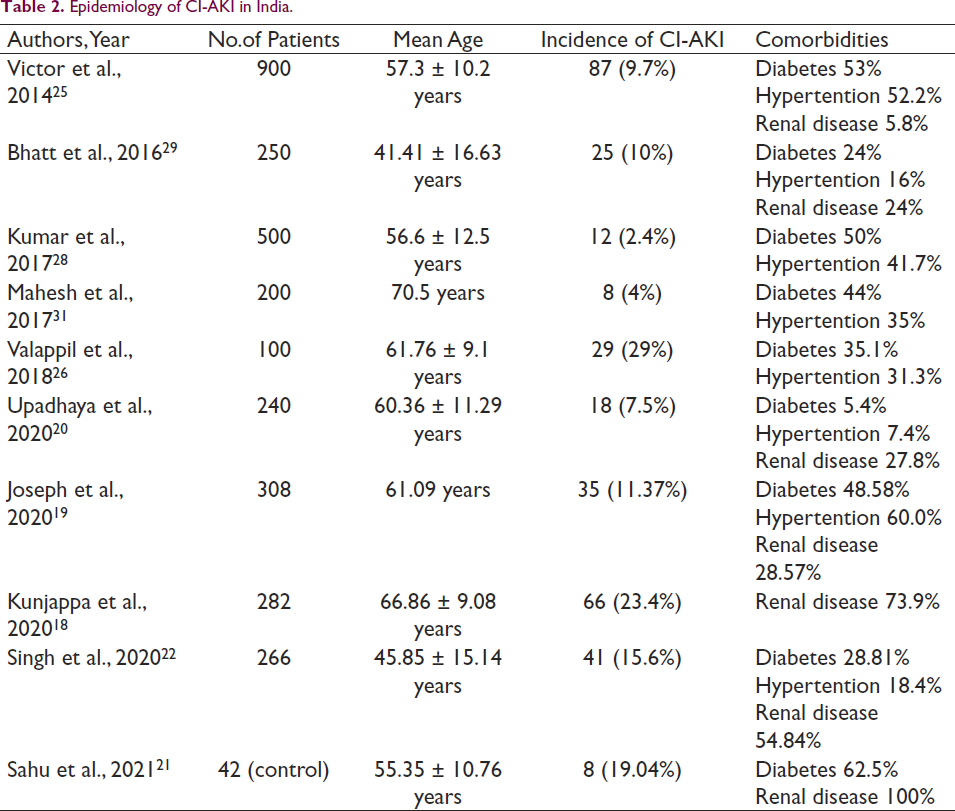
Indians have the highest rates of coronary artery disease (CAD) incidence, 23 and percutaneous coronary intervention (PCI) is thus a lifesaving option for many CAD patients in India and shows a crucial role in interventional cardiology. 24 According to the National Interventional Council of the Cardiological Society of India, approximately 4,38,351 PCIs are performed each year in India, and this number is on the rise. 24 As the frequency of coronary procedures increases, so do the consequences. Hence, the identification of high-risk patients through risk stratification is crucial. 23 In Indian patients undergoing PCI, diabetes combined with microvascular complications such as retinopathy or neuropathy are major risk factors of CI-AKI, and the overall contrast-induced nephropathy incidence in Indian patients varies between 2.4% and 29% (Table 2). Other proven risk factors include renal impairment, peripheral vascular disease, and a high amount of contrast medium. Comorbidity is highly prevalent among patients undergoing PCI.18–31
Preventive Strategies
Grouping of CI-AKI patients based on risk factors is of great importance. However, it is always better to give enhanced attention to the clinical management of patients with the clinical characteristics associated with an increased risk of CI-AKI before and after the procedure, such as pre-existing kidney disease(defined as eGFR ≤60 mL/min/1.73 m2), diabetes, advanced age (≥75 years), heart failure, reduced ejection fraction, and hemodynamic instability.4, 8, 9 It has been suggested that zero-contrast PCI also reduces CI-AKI, thereby improving the outcome of CKD patients undergoing PCI.
Pharmacological
Angiotensin-converting enzyme inhibitors, nonsteroidal anti-inflammatory drugs, cyclooxygenase-2 inhibitors, metformin, and antivirals like acyclovir and foscarnet, increase the chance of developing AKI, when administered more than once within 72 h in unexposed patients.9, 10, 12, 18, 19 AKI can occur with higher doses. Ischemia from intermittent upper arm cuff inflation (remote ischemic preconditioning) can help high-risk CI-AKI patients. Few studies suggest that transient tissue or organ ischemia may benefit neighboring locations. 10
CI-AKI risk can be significantly reduced with a physiological volume expansion caused by normal saline. Saline increases urine flow, diluting contrast media and diluting it and reducing osmolality. Back pressure and intraluminal obstruction are reduced. Renal vasoconstriction is prevented by volume expansion, which suppresses the renin-angiotensin system. 32
Preventive intravenous saline volume is unclear and may vary per patient. Despite this uncertainty, the American College of Radiology advises 100 mL/hour of saline 6 to 12 h pre- and 4 to 12 h post-angiography. Volume expansion should be done judiciously in patients with poor left ventricular function and long-term renal illness.11, 12, 16
Alkalinizing urine with sodium bicarbonate (NaHCO3) reduces free radicals and neutralizes contrast-induced active oxygen species in renal tubules. 23 Saline and bicarbonate performed similarly in a large multicenter randomized controlled experiment. Thus, bicarbonate infusion alone cannot prevent CI-AKI. Antioxidants like N-acetylcysteine decrease tubule-produced free radicals, making them effective. The clinical practice uses N-acetylcysteine to minimize CI-AKI risk. Numerous studies assessing the protective effect of N-acetyl cysteine on CA-AKI have shown inconsistent results among patients; however, there is a lack of larger clinical trials or meta-analyses. 33 Statins (3-hydroxy-3-methylglutaryl–coenzyme A reductase inhibitors) are antioxidant and anti-inflammatory agents. Statins have been shown to be beneficial for CI-AKI prevention in some studies. Due to inconsistent study methodologies and outcomes, these are not extensively used despite their benefits. Calcium channel blockers, allopurinol, fenoldopam, dopamine, and prostaglandin E1 have been explored to prevent CI-AKI, although insufficient evidence exists to use them clinically.12, 32, 34
The amount, dosage, and type of contrast agent used significantly impact the chance of contrast-induced AKI. Usually, the recommended contrast agent dose is no more than 100 mL in patients with an eGFR <60 mL/min/1.73 m2. Smaller quantities of contrast agents (approximately 30 mL) may lead to AKI in highly susceptible patients with risk factors. In patients with CKD who have serum creatinine concentration >1.8 mg/dL; a volume restriction of 5 mL/kg body weight standardized to the concentration of serum creatinine as a threshold for CI AKI. 35 A correction of anemia in high-risk patients is important to prevent CI AKI, considering the significant part of anemia in the inception and advance of CI AKI. 14 It is also suggested that injection-related features, including CM volume, CM concentration, injection rate, and patient-associated characteristics such as body weight, cardiac output, and fluid balance, determine the grade of arterial advancement in CTA. The study has reported that in overweight patients (BMI > 25), the bodyweight-customized range of 1 mL/kg and 1.5 mL/kg causes considerably higher improvement than the regular dose. In normal-weight patients (BMI ≤ 25), the patient-specific prescribed amount of 1.0 mL/kg and 1.5 mL/kg led to comparable enhancements as the customary dose. 36
Interventional
The mainstay of interventional approaches is underlined by specific strategies or focused devices. The strategies include invasive imaging like intravascular ultrasound or non-contrast-dependent optical coherence tomography. Other ancillary measures decreasing the risk of CI-AKI are radial access, remote ischemic conditioning, and hemodynamic support in highly susceptible patients. 37
Nonpharmacological
As of yet, CI-AKI has no cure. The condition is treated symptomatically like other forms of AKI. To this end, various measures have been proposed and implemented to avert the onset of CI-AKI. Identifying high-risk individuals and performing a risk-benefit analysis before providing contrast can help reduce the likelihood of adverse outcomes in those at risk for CI-AKI. Considering other imaging modalities to reduce contrast exposure is crucial if the dangers outweigh the benefits.9, 10
The following are effective preventative measures:9, 10
Minimizing contrast volume. Preventing recurrent exposure to contrast agent within 72 h. Recognizing and treating volume diminution to prevent the stimulation of the renal angiotensin pathway, which causes vasoconstriction. Hence, adequate hydration remains the cornerstone.
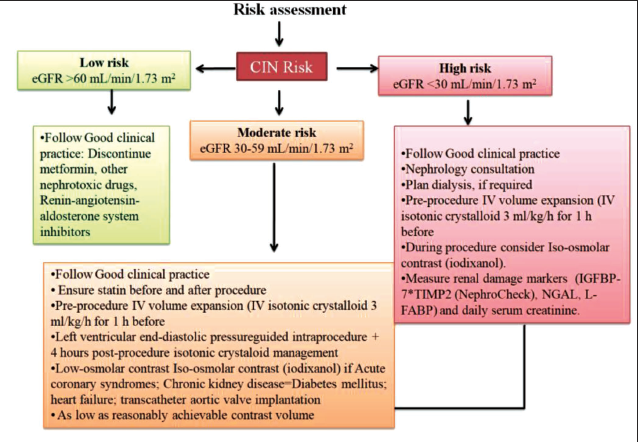
Hence, pre- and post-procedural clinical management are of paramount importance as a prevention strategy. The likelihood of resulting CI-AKI can be reduced with the prudent use of contrast media following a thorough risk evaluation and the implementation of relevant preventative measures (Figure 1). 38
Contrast Media Osmolality
Intra-arterially injected iodinated contrast medium (ICM) can efficiently opacify the arteries under examination and are well-tolerated by the patient. ICM, used in angiography, is a water-soluble compound produced from triiodobenzoic acid. It is categorized according to physicochemical properties (molecular structure, ionicity, and osmolarity) as monomer/dimer, ionic/nonionic, and with high-low-/iso-osmolarity. High-osmolarity ionic compounds were the first ICMs employed (high-osmolar contrast media, HOCM). These are commonly associated with unpleasant effects and are poorly accepted by patients. 39 Today’s intra-arterial contrast media are predominately nonionic chemicals with lower osmolarity than those employed historically (low-osmolar contrast media, LOCM). Iodixanol is the only available contrast media with osmolarity similar to blood (280–295 mOsm/kg H2O). Comparatively, the osmolarity of LOCM is twice or thrice folds compared to blood (521–915 mOsm/kg H2O). 39 Viscosity is an additional physicochemical property that differs between molecules. This is unique to each molecule and is determined by the size of the molecule and the iodine content. Viscosity also depends on temperature; warming the ICM to get its temperature close to that of the body significantly decreases viscosity.40, 41
Iodixanol has been shown to have a low risk for CI-AKI in patients with CKD and diabetes mellitus. 42 In a grouped analysis of 16 randomized trials (2,727 patients) of ICM, the occurrence of CI-AKI was considerably lesser with iodixanol than with LOCM. 43
Recently, Iacovelli et al. reported a prospective multicentric observational study. Six hundred ninety-seven patients who were not undergoing hemodialysis underwent transcatheter aortic valve implantation were enrolled. The prevalence of AKI was considerably lesser with IOCM (9.73%) than with LOCM (15.90%; P = .02), and this considerable change (P < .001) in a post-procedural difference of renal function factors lasted at discharge too. The occurrence of AKI was also considerably reduced with IOCM in younger patients. The authors concluded that IOCM has a positive effect on renal function regarding LOCM.44
Hiremath et al. showed that using iodixanol, rather than low-osmolar contrast media, for cardiac catheterization leads to a definite advantage in clinical results and savings in directly incurred health costs.45
Conclusion
Intra-arterially injected ICM can efficiently opacify the arteries under examination and be well-tolerated by the patient. Although much remains to be understood about CI-AKI, it is an established issue for patients undergoing contrast-enhanced investigations and procedures. Available evidence suggests that LOCM and IOCM should be preferred. However, IOCM may be preferred in high-risk patients undertaking interventional techniques.
Footnotes
Acknowledgements
The authors acknowledge and appreciate Dr. Debalina Das and Ms. Pooja Banerjee from IJCP Group for the medical writing support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not applicable, as this is a review article, and no patients were involved in the study.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Patient Consent
Not applicable, as this is a review article, and no patients were involved in the study.
