Abstract
With growing expertise and evolving hardware chronic total occlusion (CTO) percutaneous coronary intervention (PCI) success and safety has been improved remarkably. The nuances of CTO wiring help operators to find novel solutions to the complications created during percutaneous coronary interventions. Accidental coronary perforations caused during CTO interventions can be sealed by wiring from other side and shifting interposing plaque or flap onto perforation. We report a case of saccular aneurysm around the proximal cap formed by antegrade wire escalation in earlier failed percutaneous coronary interventions, which is excluded by “retrograde plaque lift and shift.”
Introduction
Chronic total occlusion (CTO) intervention has been evolving over last 2 decades achieving best success rates (>90%) in various registries.1, 2, 3 CTO percutaneous coronary interventions (PCI) is also associated with increased incidence of complications like coronary perforations, dissections, and intramural hematoma (IMH). However, the accumulating experience of operators and evolving hardware brought novel solutions to tackle the complications created during CTO PCI. Here, we are reporting a case of antegrade wire escalation induced saccular aneurysm at proximal cap of right coronary artery (RCA) CTO excluded by retrograde wire escalation with controlled antegrade and retrograde tracking technique (CART) and stenting. To our knowledge this is the first such case reported in the literature.
Case Report
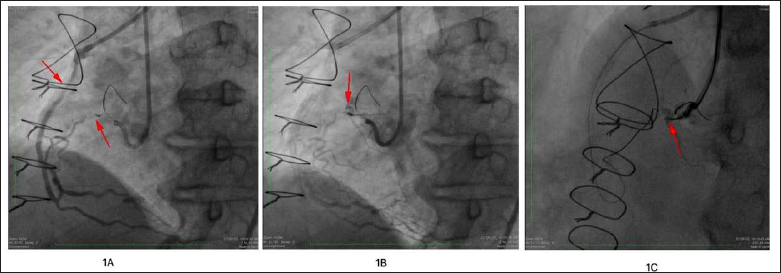
64 year male, hypertensive, status post coronary artery bypass grafting (CABG) with left internal mammary artery (LIMA) to left anterior descending artery (LAD), saphenous vein graft (SVG) to obtuse marginal (OM) and SVG to RCA, had SVG to RCA PCI done twice for exertional angina limiting his quality of life. During the second intervention operator tried to open the CTO of native RCA but could not cross proximal cap despite wire escalation; therefore, PCI to in-stent stenosis of SVG to RCA was done with placement of another stent (Figure 1A). Within 6 months of the last procedure, he came with exertional angina to our center; upon evaluation echo showed good left ventricular (LV) function and stress nuclear scan revealed inducible ischemia in RCA territory. Coronary angiography (CAG) done revealed CTO of proximal RCA with saccular aneurysm at the proximal cap which was not seen in the earlier angiograms (Figures 1B and 1C).
A: Dual angiography showing significant in-stent stenosis in SVG to and short proximal RCA CTO. B: Sinusoidal loop in Mid RCA distal to CTO. C: Saccular Aneurysm at proximal cap.
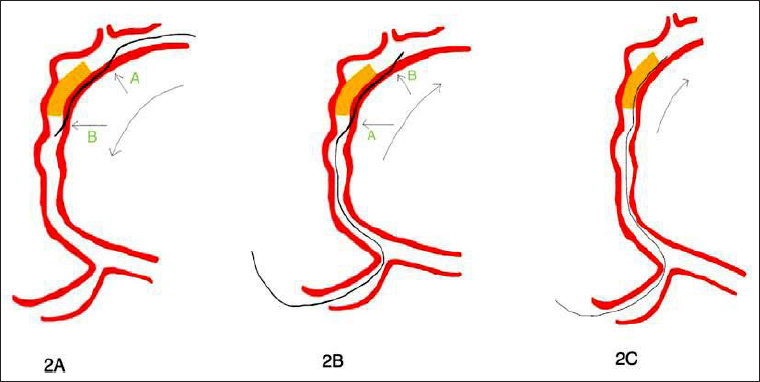
SVG to RCA showed significant in-stent stenosis and other grafts were normal. Recurrent in-stent stenosis in the 10 years old venous graft favored native RCA CTO PCI as the better therapeutic option in the heart team. Though the CTO segment was not long and the JCTO score is 1, the aneurysm at the proximal cap and the tortuosity of vessel (Figure 1A) distal to CTO were considered technically challenging. The aneurysm exclusion was considered while planning the wiring strategy which limited our options to those showed in Figures 2A–2C. SVG cannulated with 7FR MPA 1 from femoral access and RCA with 7FR AL 0.75 from radial access. In the wiring options described above, Option 1 tried first but the antegrade subintimal entry was not successful due to inadequate guide support despite using anchor balloon and too proximal CTO without much room for wire maneuvering. Hence, option 3 was attempted after caravel (Asahi Intecc, Japan) being placed at the distal cap (Figures 3A and 3B) retrograde wire escalated to GAIA 3 (Asahi Intecc, Japan) after failed progression with GAIA 2 (Asahi Intecc, Japan) and it is navigated along the wall away from aneurysm which was marked by antegrade wire loop (Figure 3B).
(A) Option 1—Antegrade dissection reentry with subintimal entry proximal to and opposite side of aneurysm at “A”. Reentry at (or) after point “B.” (B) Option 2: RDR—Retrograde dissection and reentry with subintimal entry at or distal to “A” and reentry proximal and opposite to Aneurysm at “B.” (C) Option 3: RWE—Navigating wire in the CTO segment away from Aneurysm and extended reverse CART to push the plaque on to Aneurysm.
A,B: Retrograde GAIA 3 navigated beneath the floor of the aneurysm (arrow 1) marked by antegrade wire loop (arrow 2). C: IVUS from SA nodal branch showing aneurysm (arrow 1) and retrograde wire (arrow 2). D: Retrograde wire (arrow 1) away from aneurysm (arrow 2) and in subintimal plane. E: Retrograde GAIA 3 in true lumen proximal to aneurysm
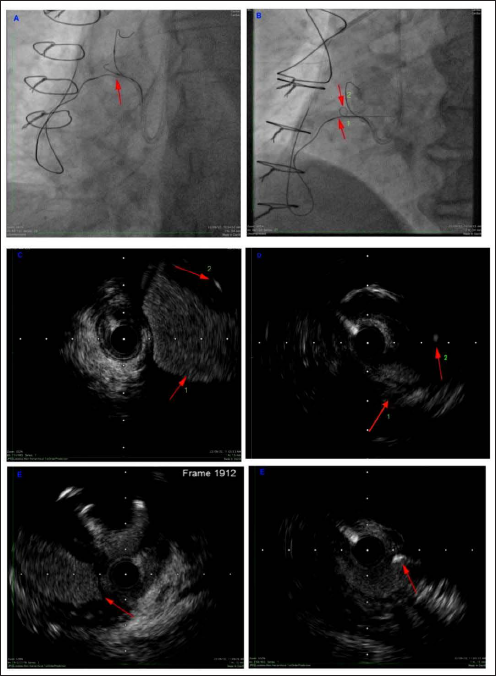
After retrograde wire advanced beyond proximal cap extended reverse CART performed and wire further navigated into aorta as shown in Figures 3A and 3B. Intravascular ultrasound (IVUS) performed over antegrade wire in Sino atrial (SA) nodal branch as shown in Figures 3C-3E to confirm retrograde wire course and its relation to aneurysm.
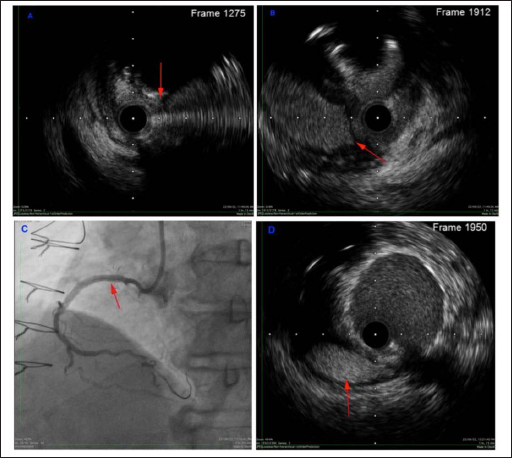
As the retrograde wire couldn’t be advanced into antegrade guide catheter, retrograde caravel pushed across the CTO by balloon trapping of retrograde GAIA 3 in ostial RCA, followed by wire de-escalation to work horse wire (Abbott) with longer curve. This wire could be navigated into the antegrade guide and externalization done with RG wire (Asahi Intecc, Japan). Predilation was done with 2 mm and 3 mm balloons followed by antegrade wiring into posterior left ventricular branches (PLVB) through double lumen catheter over RG wire. IVUS done over antegrade wire revealed extra aneurysmal course of wire and aneurysm exclusion with plaque and flap Figures 4A and 4B. Stenting completed over the antegrade wire with 2 stents 3/38 mm and 3.5/28 mm with overlap, after retrograde wire with microcatheter had withdrawn into venous graft to avoid jailing. Final IVUS and angiogram revealed complete exclusion of aneurysm as shown in Figures 4C and 4D, acceptable lumen areas, and good flow into distal vessel.
A, B: Arrow indicates flap separating aneurysm from true lumen. C: Final angiogram showing aneurysm exclusion. D: Final IVUS showing aneurysm (arrow indicates aneurysm) exclusion.
Discussion
CTO wires, having high tip load than the workhorse wires, can be more traumatizing to vessel wall. In CTO’s with tough and impenetrable proximal cap and poor guide support, antegrade escalated wire can slip into the vessel wall adjacent to the proximal cap. Injudicious repeated attempts to penetrate proximal cap might injure the adjacent vessel wall leading to perforation or aneurysm formation. The aneurysm formation and further expansion is facilitated by the direct exposure of subintimal space to aortic pressure, which commonly happens when the space created is around proximal cap than mid or distal CTO. In the above case antegrade wire escalation (AWE) with prolonged wire manipulation around the proximal cap must have created subintimal space and its exposure to the aorta pressure probably resulted in aneurysm formation and further expansion. Aneurysms or perforations can be excluded from lumen by using subintimal or intimal (away from the location of aneurysm or perforation) wire tracking and stenting. 4 For saccular aneurysm exclusion the wire should course along the intimal plaque or subintima away from the location of aneurysm (Figure 3C and 3D). If dissection/reentry is done, entry and exit of subintimal space must be located beyond the boundaries of aneurysm and wire is navigated along the opposite wall of aneurysm. IVUS from immediate proximal branch might help in understanding the relative position of wire with aneurysm and probability of its exclusion.
Conclusion
“Plaque and/or flap lift and shift” to seal saccular aneurysm or perforation can be used by experienced operators in complicated CTO PCI. However, executing this from retrograde or antegrade is decided by anatomical considerations for better sealing. Generally, if the complication is created by the antegrade it can be sealed by retrograde plaque or flap shift and vice versa.
Footnotes
Abbreviations
CABG: coronary artery bypass grafting
CAG: coronary angiography
CART: Controlled antegrade and retrograde tracking technique
CTO: chronic total occlusion
PCI: percutaneous coronary interventions
IMH: intramural hematoma
IVUS: intravascular ultrasound
LAD: left anterior descending artery
LIMA: left internal mammary artery
OM: obtuse marginal
PLVB: posterior left ventricular branches
SA: sino atrial
SVG: saphenous vein graft
Acknowledgements
I would like to acknowledge the Dr Chandana Poosala from the clinical research department for drafting the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The patient consent form is not applicable for this case report because the patient images, names, and initials are not taken for the publication.
This is a rare case which is describing a diagnostic and therapeutic challenge and providing a learning points for the reader.
