Abstract
Percutaneous coronary intervention for chronic total occlusion is a complex and high-risk intervention (CHIP). Even though persistent ischemia-induced mitral regurgitation is rare, ischemic mitral regurgitation is a frequent complication of an acute coronary crisis. Transcatheter mitral repair has the potential to supplant surgical repair or replacement as the gold-standard treatment for persistent mitral regurgitation. Currently, these interventions are only performed on high-risk surgical candidates, but the indications may eventually be expanded to include low-to-intermediate risk patients as well, in a manner like transcatheter aortic valve replacement.
In patients with ischemic cardiomyopathy who still had considerable viable myocardium, combining current guideline-directed pharmacological treatment with interventional complete revascularization decreased hospitalizations for heart failure. We performed a CHIP intervention on the left circumflex artery, which was chronically totally occluded, to address the severe ischemic mitral regurgitation that had been present for a long time.
Introduction
The treatment of chronic ischemic mitral regurgitation (IMR) is one of the most difficult and contentious issues in cardiomyopathy. Chronic IMR is not only common but also has a significant influence on the prognosis of the patient.1 The most common surgical therapy for chronic IMR is restrictive annuloplasty in combination with coronary artery bypass grafting (CABG). However, in other circumstances, the mitral valve mechanism does not exhibit any structural abnormalities. “Mitral regurgitation induced by a prior myocardial infarction (MI) and accompanied by normal mitral valve leaflets and chordae” is the definition of chronic IMR.2 Due to a lack of consideration for IMR’s pathophysiology, this definition (based only on MI history) is inadequate.
Case Reports
A 65-year-old female, hypertensive and diabetic presented to emergency room with sudden onset of breathlessness for 1 day. She has never experienced any medical problems in the past. She was tachypnoeic and diaphoretic on examination, with vital data showing a pulse rate of 122 beats per minute, blood pressure of 108/68 mmHg, and a respiratory rate of 33 per minute. There were bilateral rales in more than two-thirds of the lung fields and a pansystolic murmur grade III in the parasternal area with radiation to the back on auscultation. A clinical diagnosis of acute decompensated heart failure (ADHF) secondary to severe mitral regurgitation was made. Intravenous diuretics, vasodilators, and noninvasive breathing were employed to achieve medical stabilization.
An electrocardiogram revealed normal sinus rhythm with pathological Q-waves in the inferior and lateral leads, suggestive of a silent postero-lateral MI. Transthoracic echocardiography demonstrated regional wall motion abnormality in the form of hypokinesia of the lateral and posterior wall (preserved thickness) with mild left ventricular (LV) dysfunction (ejection fraction (EF) by modified Simpson’s equation—45%). The left atrial size was within normal limits (3.6 cm). The color flow mapping of the mitral valve revealed severe eccentric, posteriorly directed mitral regurgitation (MR). The mitral valve system seemed to be intact, however there was tethering of the anterolateral papillary muscle on close examination in multiple views.
Her laboratory parameters were normal, including high-sensitivity troponin I except the N-terminal prohormone B-type natriuretic peptide (2,334 pg/ml). The angiotensin receptor blocker neprilysin inhibitor (ARNI), empagliflozin [sodium glucose co-transporter-2 inhibitor (SGLT2i)], carvedilol, torsemide, eplerenone [mineralocorticoid receptor antagonists (MRA)], aspirin, ticagrelor, and atorvastatin were administered as guideline-guided medical treatment (GDMT). She was subjected to coronary angiography after medical stabilization demonstrated chronic total occlusion (CTO) of the proximal segment of the left circumflex artery, followed by the obtuse marginal artery at the bifurcation of the large second obtuse marginal (OM) branch and distal left circumflex (LCx) arteries (J-CTO score 2). The left anterior descending and right coronary arteries showed non-obstructive lesions.
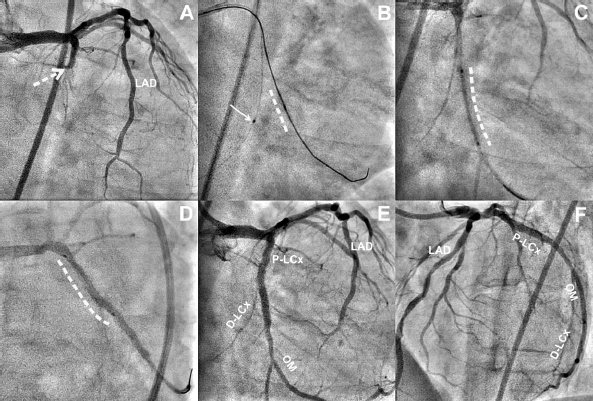
Despite the presence of severe IMR, the heart team decided to perform percutaneous coronary intervention (PCI) in the LCx territory due to the presence of heart failure and involvement of single vessel disease. PCI was performed through a transfemoral approach with a 7fr. EBU guiding catheter. Fielder XT guide wire (Terumo Corporation, Japan) could cross both the chronic total occlusions (CTOs), but 1.25 × 8 mm balloon was unable to cross at the distal segment of the CTO. The proximal segment of the CTO was predilated with a 1.5 × 10 mm balloon. BMW guide wire (Abbott Vascular, USA) was placed into LCX and was anchored using another 1.5 × 8 mm balloon. Subsequently, a 1.25 × 8 mm balloon could cross over Fielder XT into OM2. After sequential predilatation with 2 × 12 mm, 2.5 × 12 mm balloons, the two overlapping sirolimus-eluting stents—proximal (2.75 × 20 mm) and distal (2.5 × 20 mm) segments—were implanted. The final angiography revealed TIMI-3 flow into the OM2 and LCX with no residual stenosis (Figure 1).
Coronary angiography revealed chronic total occlusion of the proximal segment of the left circumflex (P-LCx) branch, which extended to the proximal segment of the major second obtuse marginal (OM) branch (dashed arrow, Panel A). Nonobstructive plaques were seen in the left anterior descending artery (LAD). The CTO segment was crossed with the use of a microcatheter and a Gaia guide wire assembly. The proximal segment was predilated, allowing access to the distal LCX (D-LCx); however, the balloon could not track into the distal segment. As a consequence, anchoring balloon technique using 1.5 mm balloon (thin solid arrow) into D-LCX could result in crossing of the 1.25 mm balloon (dashed line) which was successively predilated (Panel B). The proximal segment of the OM2 deployed a sirolimus-eluting stent (dashed line, Panel C), which overlapped with other SES (dashed line, Panel D). The final angiography revealed thrombolysis in myocardial infarction 3 flow with residual stenosis in the right anterior oblique caudal (Panel E) and left anterior cranial (Panel F) views.
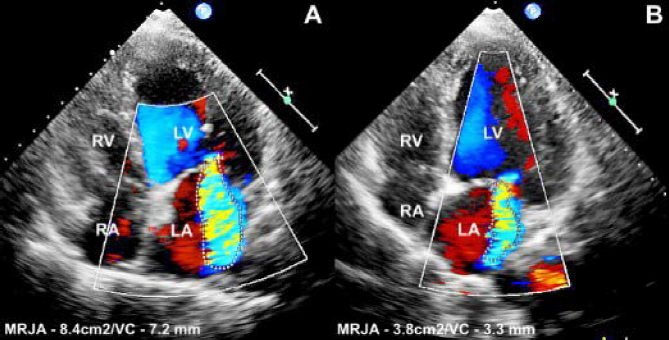
The pharmacoinvasive strategy resulted in reverse remodeling of LV, reducing her symptoms to NYHA class I. At 6-month follow-up, there was a >50% decrease in the severity of the MR (Figure 2) and a 50% improvement in LV function. At 1-year follow-up, the patient was asymptomatic on regular GDMT.
Before intervention, transthoracic echocardiography with color flow mapping revealed severe mitral regurgitation [mitral jet area (MRJA): 8.4 cm 2 and vena contract (VC): 7.2 mm; Panel A]. In addition, the left ventricle and left atrium were dilated whereas the right ventricle and atrium were normal in size and function. Mitral regurgitation was significantly reduced after intervention (MRJA: 3.8 cm 2 and vena contract: 3.3 mm; Panel B).
Discussion
IMR may occur after an acute or prior MI. IMR is a prevalent consequence of coronary artery disease and contributes to a dismal outlook for patients with this condition. The development of significant IMR following acute or subacute MI leads to ADHF and cardiogenic shock.3 Papillary muscle dysfunction or rupture is a possible cause of acute mitral regurgitation after a MI.
Primary IMR occurs because of ischemic heart disease-related structural and functional abnormalities in the mitral valve apparatus of the LV.1,2 Secondary MR, which is not a valve disease in and of itself but rather a sign of LV dysfunction. IMR is a predictable consequence of LV pathological remodeling due to chronic coronary artery disease or dilated cardiomyopathy.1, 2 If papillary muscle rupture is not present, then severe MR may result from leaflet tethering due to left ventricular failure. 3 In the context of cardiomyopathy or coronary artery disease, secondary MR is characterized as functional MR resulting from LV remodeling. Secondary functional MR is known as IMR in this context.1
Valuckiene et al 4 discovered that MR was considerably greater in women, had a lower EF, and was related with multi-vessel coronary artery disease (CAD) and well-developed collateral supply to the infarct site, independent of the duration of the ischemia, in a study of 173 ST-segment elevation myocardial infarction (STEMI) patients.4 IMR is classified by Carpentier based on the leaflet margin’s movement in relation to the annular plane. To explain IMR, it is preferable to use this classification. In the majority of cases of IMR, Type IIIb manifests as restricted motion of the leaflet margins during systole. Type I dysfunction occurred in our instance, where there was normal leaflet motility with isolated annular dilatation, which is less typical in the context of isolated basilar MI.5 This type of IMR may be alleviated by revascularization of the culprit vessels with PCI or CABG, according to current studies. After 3 months, PCI may be a suitable time span in which to evaluate the likelihood of IMR recovery.2 Previous studies have demonstrated that the MitraClip® (Abbott Vascular) is a promising alternative for treating IMR after MI, but it is not cost-effective and faces several difficulties before being widely used.3
The success of isolated revascularization, on the contrary, is contingent on the amount of viable myocardium. Revascularization may cause the left ventricle to reverse its remodeling, resulting in less regurgitation.2,6 CTO-PCI has grown and evolved tremendously as new procedures and equipment have been developed, and trained professionals have been trained to use them. It is because of these considerations that the success rates of various treatments have risen dramatically.7 The collateral circulation, which may be created by angiogenesis or by the activity of endothelial progenitor cells, maintains the survival of the myocardium irrigated by the blocked coronary artery. Although coronary perfusion may be assessed via angiography, it is not the most reliable way for predicting collateral vessel function, which is responsible for the development of IMR.8 An examination of fractional flow reserve (FFR) data challenges the conventional wisdom that CTO ischemia may be prevented by an occluded vessel’s “enough and sufficient collaterals.”
Staggering anterograde wires is the most popular method of recanalization in CTO intervention. To cross the CTO, a microcatheter is advanced to the proximal cap, and appropriate guidewires are used according to the cap’s anatomy. As a rule, the procedure begins with the insertion of a polymer-coated, 1.0 g soft guidewire. Knowing the course of the guidewire is essential for both success and avoiding difficulties. Using two orthogonal projections, the microcatheter is pushed through into the occluded area and the guidewire is replaced with a conventional one, which is followed by balloon angioplasty and stent insertion if the guidewire accesses the true distal lumen.7
The goal of medical therapy for IMR patients is to avoid, delay, or reverse LV remodeling, heart failure, and myocardial ischemia. Aside from the standard pharmacological treatment for heart failure, which includes ARNI, beta-blockers (BB), spironolactone, SGLT2i, nitrates, and diuretics, there are no additional recommendations for IMR patients.9,10 In those with secondary functional MR, sacubitril/valsartan reduced MR more than valsartan. According to our findings, patients with heart failure and functional MR may benefit from an angiotensin receptor-neprilysin inhibitor. As with the paradigm effects of BB, ARNI, and MRA, SGLT2i causes significant improvements in symptoms, morbidity, mortality, and LV reverse remodeling.10 In those with chronic heart failure, GDMT reverses LV remodeling and lowers MR severity. The thickness of the posterolateral walls of the LV was shown to be maintained by echocardiogram, even though we were unable to execute a stress test to confirm this. Although mitral regurgitation and symptoms may have improved with medical treatment alone, we believe that CTO-PCI improved LV function, which helped reverse LV remodeling. Yousufzai et al 11 showed that in one third of their patients, PCI alone led to a 75% sustainable improvement in MR, which was related to reverse LV remodeling, and left atrial size is the most significant factor of the improvement in MR after PCI.11
To our knowledge, this is the first time a CTO intervention in combination with a newer GDMT for heart failure has been used to treat persistent severe mitral regurgitation. Our goal was to demonstrate clinical efficacy rather than to demonstrate favorable LV characteristics, and the intervention was done at a tertiary care center.
Conclusion
Patients with chronic ischemic mitral regurgitation who have an intact mitral valve apparatus and considerable viable and ischemic myocardium may benefit from CTO intervention. There is a substantial mortality and morbidity rate linked to long-term chronic mitral regurgitation. Studies of these novel percutaneous approaches for secondary MR, although promising, still lack large patient populations and long-term follow-up at this time.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
