Abstract
Extracorporeal membrane oxygenation (ECMO) is a form of circulatory and respiratory support used for patients who have an almost complete loss of function of native lungs or heart. ECMO helps supplement organ function while treatment for the primary pathology is initiated. Initiation and maintenance of ECMO requires specialized skill, and knowledge of potential complications is necessary to ensure rapid and good return of organ function while minimizing patient morbidity and mortality. Weaning from ECMO requires complex studies to assess return of native cardiac function. Major complications include bleeding thrombosis and ischemia.
Introduction
Venoarterial extracorporeal membrane oxygenation (VA-ECMO) is a form of temporary mechanical circulatory support and simultaneous extracorporeal gas exchange. The initiation of VA-ECMO has emerged as a salvage intervention in patients with cardiogenic shock, even cardiac arrest refractory to standard therapies. Analogous to veno-venous ECMO for acute respiratory failure, VA-ECMO provides circulatory support and allows time for other treatments to promote recovery or may be a bridge to a more durable mechanical solution in the setting of acute or acute on chronic cardiopulmonary failure. In this review, we provide a brief overview of VA-ECMO, the attendant physiological considerations of peripheral VA-ECMO, and its complications.
Mechanics of ECMO
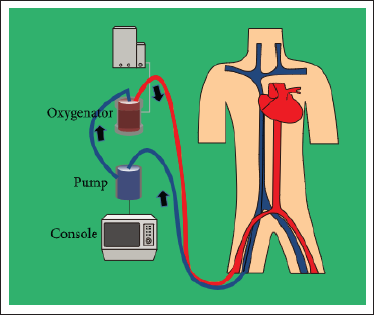
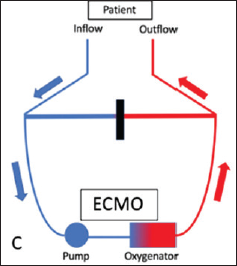
The ECMO machine is connected to a patient through wide-bore cannula. The cannulae are placed in large veins and arteries in the legs, neck, or chest. The ECMO machine drains blood from the patient’s body to an oxygenator that oxygenates and decarbonizes blood. This blood is then sent back to the patient via a centrifugal pump which may be used for circulatory support (Figure 1). The ECMO machine is controlled by a person called a perfusionist, or a nurse with advanced training called an ECMO nurse.
There are 2 major types of ECMO. The VA-ECMO is connected to both a vein and an artery and is used in cardiac dysfunction. The VV-ECMO is connected to one or more veins, usually near the heart, and is used for pulmonary support. There are also hybrid modes such as veno-arterio-venous (VAV) where support of both lungs and heart is required. SAVE score can help in the decision to initiate VA-ECMO and prognosticate the patient already on ECMO.
Indications for VA-ECMO
Young age, higher creatinine, long history of coronary artery disease with previous myocardial infarction, repeat operation, recent previous operation, unstable clinical status, and an emergency operation with incomplete revascularization without left mammary artery are potential predictors of the need for temporary cardiocirculatory support at the end of or after cardiac surgery procedures. 7
The most common post cardiotomy scenario is represented by intraoperative failure of:
Weaning from cardiopulmonary bypass (CPB) due to left or biventricular dysfunction, or refractory hypoxemia with or without associated global cardiac hypocontractility. Graft failure or persistent pulmonary hypertension is also potential contributor to the need for ECMO support after heart transplant.
The timing and indication of ECMO implant influence the type of approach, with central cannulation more frequently applied intraoperatively taking advantage of the cannula used for CPB. In contrast, the peripheral route, either percutaneous or with an open access, is more commonly used in case of delayed ECMO implant. Several groups have also adopted intraoperative peripheral cannulation to allow sternal closure and reduce the risk of bleeding.
The etiology is usually viral or other infective causes.
The efficacy of VA-ECMO in these cases has been consistently proven.
A published case series by Arlt and colleagues has reported use of ECMO in 10 patients during interventional procedures, all patients had heart activity restores and 2 TAVI patients were converted to surgical replacement on ECMO and all PCI procedures went on to completion on ECMO with an overall survival of 50% to hospital discharge. 10
It has also been used in pre op stabilization of patients with post infarct complications such as ventricular septal defect, papillary muscle defect, or free wall rupture.
Procedure
Being placed on ECMO requires a surgical or percutaneous procedure. Percutaneous procedure is usually done bedside. The patient is sedated and given analgesia and anticoagulant, usually heparin 5000U, after guidewires are inserted to minimise blood clotting. Activated clotting time done at that time should be targeted around 300 to 400 s. A critical care physician/surgeon, assisted by a team, inserts the ECMO cannulae into an artery and veins. Fluoroscope/Ultrasound is then used to ensure the cannulae are in the correct position. Patients being initiated on ECMO are usually sedated and ventilated but there is a trend toward early initiation while the patient is awake. It is possible for patients to be completely dependent on ECMO and not be on ventilator.11-13
Femoral access is preferred for VA-ECMO because of ease of insertion. The main drawback of femoral access is ischemia of the ipsilateral lower extremity. The likelihood of this complication can be decreased by inserting an additional arterial cannula (minimum of 7 Fr) distal to the femoral artery cannula and redirecting a portion of the infused blood to the additional cannula for reperfusion of the extremity. Alternatively, a cannula can be inserted into the posterior tibial artery for retrograde flow to the extremity.
Occasionally, the femoral vessels are unsuitable for cannulation for VA-ECMO (eg, patients with severe occlusive peripheral artery disease or prior femoral arterial reconstruction). In such circumstances, the right common carotid artery or subclavian artery can be used. In our experience, there is a 5% to 10% risk of a large watershed cerebral infarction when the right common carotid artery is used.
For postcardiotomy ECMO, the cannula employed for CPB can be transferred from the heart-lung machine to the ECMO circuit, with blood drained from the right atrium and reinfused into the ascending aorta.
Monitoring on VA-ECMO
Patient
Monitoring patients on ECMO requires special training of staff and well-equipped intensive care unit (ICU) for early identification and management of complications
SpO2 >98%
Hb >10 g/dL
Platelets >50,000/cu mm (daily monitoring early identification of heparin-induced thrombocytopenia [HIT])
Fibrinogen
Plasma-free Hb%
Lactates <1 mmol/L
Bleeding from cannula sites
Position of cannula
2d Echo
TDss >6 cm
EF: daily changes
Aortic valve VTI >10
LA pressure <10 mm of Hg
New onset of shunts
New onset of regurgitations especially on aortic valve, left ventricular (LV) distension (daily monitoring of LV diameter)
Evidence of ischemia in the distal limb
NIRS on both lower limbs to identify compartment syndrome
NIRS for neuromonitoring can be used (Figure 2)
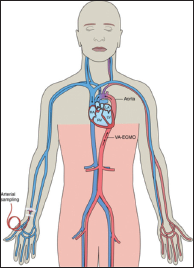
Right radial artery pulsatility and saturation
Neuromonitoring: regular examination of pupils and decision for imaging given high risk of bleeding and thrombosis
Urine output and color, to identify rhabdomyolysis
Anticoagulation Monitoring
All patients requiring VA-ECMO need therapeutic levels of anticoagulation. The most commonly used anticoagulant is unfractionated heparin because of its long usage experience and short half-life. Anticoagulation needs to be monitored frequently with either activated partial thromboplastin time (>1.5 times normal) or activated coagulation time (160-180) being normally used. Newer evidence suggests that using anti-Xa may actually lead to decrease in doses of heparin and also decreasing incidence of bleeding. The targets for anti-Xa are 0.6 to 1.0 IU/mL.
Presumptive diagnosis of HIT requires workup with 4T score and if found to have high likelihood, then changing anticoagulant to either bivalirudin or argatroban.
Machine (ECMO/Ventilator/HCU)
ECMO
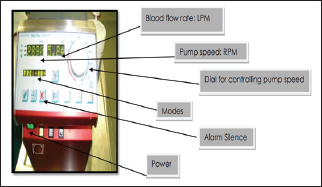
Prepump pressure p1 (Figure 3)
Premembrane pressure p2
Postmembrane pressure p3
Transmembrane pressure (p2-p3)
A rise in transmembrane pressure along with fall in post membrane arterial blood gas (ABG) indicates circuit failure/oxygenator failure due to mechanical causes.
If membrane is unable to oxygenate at least 4 times premembrane value, it is an indication of oxygenator change.
Flow and corresponding RPM, separate flow sensor in return cannula and in reperfusion cannula Fast flush test of oxygenator to check membrane efficiency Flash light test to look for clots and fibrin deposits (Figure 4) Battery indicator/po Power supply
Ventilator
FiO2
Peak pressure
Tv (tidal volume )
Compliance
Ventilation targets:
Lung protective ventilation in the form of 6 to 8 mL/kg ideal body weight Plateau pressures <30 cm water pH >7.3 SpO2 >88%
HCU
Temperature difference in heater-cooler unit (HCU) (early indicator of sepsis/fever) (Figure 5).
Complications
Complications can include the following.
Problems with coagulation:
Bleeding (intracranial/retroperitoneal) are life-threatening complication with an incidence of 1% to 2%. Bleeding from cannula sites and tracheostomy sites can usually be controlled with pressure Clots in oxygenator Deep vein thrombosis Embolic stroke
Limb gangrene (need to have distal perfusion cannula with a flow min of 200 mL/min)
Infections (blood stream is most common)
Circuit failure (identified by pre- and postmembrane PaO2 levels)
Pulmonary edema/LV distension (needs LV venting strategies which includes:
Inotropes Decreasing flow in ECMO Intra-aortic balloon pump Atrial septostomy Drainage cannula in LV Surgical/Percutaneous LVAD Converting to central VA ECMO
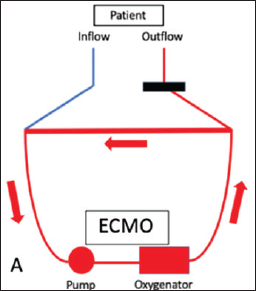
As seen in Figure 6, it’s mainly because of a recovering heart pumping deoxygenated/mixed blood from native lung. Identified by early pulsatile waveform in right radial artery, drop occurs in right hand saturation and PaO2 levels in ABG. It is most commonly seen due to impending respiratory failure on a recovering heart.
It can be managed by increasing FiO2 on ventilator, or conversion to VAV-ECMO in refractory cases.
Arterial pseudo aneurysms, dissections at insertion sites


Weaning
Invasive hemodynamic monitoring must be used whenever possible to guide therapy in VA-ECMO. A pulmonary artery catheter provides valuable information about LV loading conditions. A right upper extremity arterial line should be maintained in all patients with femoral cannulation to facilitate monitoring of pulsatility and oxygenated blood flow to the arch of the aorta.
Targets to consider before attempting weaning:
Cardiac index >2.2 L/min/m2 Mean arterial pressure (MAP) 65 to 80 mmHg Central venous pressure 8 to 12 mmHg, and pulmonary capillary wedge pressure <18 mmHg ABG, lactate, hepatic, and renal function are followed in serial laboratory measurements to assess adequacy of end-organ perfusion.
In the event of concomitant use of a LV venting device, IABP, or percutaneous left ventricular assist device (pLVAD), weaning and removal of VA-ECMO must be prioritized, given its propensity to increase afterload on a failing myocardium and relatively high complication rate. One exception is when complications arise directly related to the ancillary device necessitating its removal. When used as a LV venting device, IABP must be maintained at 1:1 for unloading and ensuring adequate washing of the aortic root. Concomitant pLVADs are generally maintained at relatively low levels of flow (1.5-2 L per min [LPM]) as their primary use in this setting is LV decompression rather than circulatory support.
Prior to weaning VA-ECMO flow, weaning of vasoactive medications is advantageous.
The following criteria should be met prior to weaning VA-ECMO:
The patient phenotype is compatible with recovery End-organ function is recovering PaO2/FiO2 >100 Vasopressors and inotropes are at reasonably low levels (for instance norepinephrine ≤4 µg/min or dobutamine <5 mcg/kg/min) Pulse pressure should be >20 mm Hg
When these criteria are met, a 3-part approach to weaning should be initiated. This includes the following:
Daily weaning study Bedside assessment for decannulation Final assessment
Daily Weaning Study
Daily transient reductions in ECMO flow rate in all patients is done to assess suitability for weaning. Flows are reduced at increments of 0.5 LPM to a minimum of 2 LPM, adequate time is given to assess the effect on MAP and intracardiac pressures. If the MAP falls more than 10 to 15 mmHg or below 65 mmHg, the patient is not yet ready to wean to that level.14-17
Significant increases in right-sided filling pressures during weaning may also constitute failure especially in the setting of predominant RV failure. At the end of the study, the blood flow rate is set at the lowest level achieved in the weaning study required to maintain stable MAP and intracardiac pressures. ABG, lactate, and a full set of invasive hemodynamics are obtained at this new flow rate to detect any impact on hemodynamics, tissue perfusion, or respiratory status. Because a subset of patients who tolerate reduced support transiently are unable to tolerate the decreased support over a prolonged duration, lower flow rates are maintained for a minimum of 8 h prior to further weaning attempts.
Weaning trials are performed at least every 24 h. Bedside echocardiography is used to provide additional information about native cardiac function as blood flow rates are reduced, particularly if the patient has failed prior weaning attempts. Prior literature has suggested the following parameters to be associated with successful weaning: aortic VTI ≥10 cm, LVEF >20% to 25%, and lateral mitral annulus peak systolic velocity >6 cm/s (Figure 7).
Bedside Assessment for Decannulation
After a patient has tolerated a trial of 2 LPM for a minimum of 8 h with stable end-organ function, a bedside assessment for decannulation is performed. Flow is gradually decreased to 1 LPM over the course of approximately 1 min to detect hemodynamic instability with minimal ECMO support. If tolerated, blood flow is then returned to 2 LPM, and plans are made for decannulation if the underlying cause of initial decompensation has been sufficiently addressed such that liberation from VA-ECMO is possible. If the patient fails the bedside assessment, then flow is returned to 2 LPM with plans to reassess every 24 h.
Final Assessment
When a patient has tolerated a trial of 1 to 1.5 LPM (20 mL/kg/min of blood flow for a minimum of 8 h with stable hemodynamics and end-organ function) and tolerates transient flow reduction to 1 LPM at the bedside, the patient is typically taken to the operating room for ECMO decannulation. The final assessment is performed at this time, wherein blood flows are gradually decreased and the cannulae are clamped; hemodynamic and ABG parameters are checked. Focused echocardiography is often undertaken to assess the impact of flow reduction on biventricular function. If acceptable, decannulation is performed.
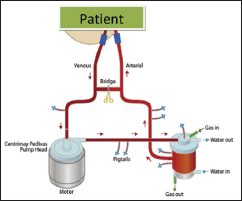
Another way to discontinuation of VA-ECMO is tubing an external bridge between the arterial and venous lines, which allows blood to circulate continually when clamping the arterial and venous cannulae (Figures 8 and 9).
Decannulation
Decannulation when the patient is ready must be done in theatre by a vascular surgeon as the arterial puncture must be closed under vision after ensuring patency of vessel. Post removal, it is advised to put a drain at the site to prevent compression of the artery by oozing blood. The venous cannula can be removed and percutaneous suture placed.
Post removal monitoring should include regular check of distal pulses, drain monitoring, and anticoagulation. Restart deep vein thrombosis prophylaxis as routine. Close monitoring of pulses along with frequent ultrasound of the groin will help us to identify early pseudoaneurysm.
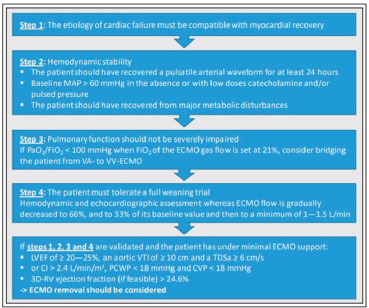
The following algorithm is followed in terms of weaning in cardiac failure in our ICU.
Precautions to be taken while incorporating bridge in ECMO circuit (Figures 8, 9 and 10):
Make sure bridge is always clamped. Unclamping of bridge to be done at regular intervals keeping a watch for clots in the circuit especially near the corners. Some centers incorporate a 3-way stop cock in bridge for regular flushing. Clamping bridge will ensure flow happens in circuit as shown in Figure 9.
Once all criteria for weaning are met, we can try clamping and unclamping technique before decannulation.
We need to clamp the cannulae (outflow and inflow) and unclamp the bridge so that blood flow is only in the circuit. If patient tolerates this, we can plan for decannulation. Flush the cannulae and bridge to prevent clot formation.
Another method known as pump controlled retrograde trial off can be used as an alternative to bridge method.
Future of ECMO Practice
The future of ECMO in critical care is vastly expanding with ever increasing indications such as severe pancreatitis/severe SIRS/poisoning (especially cardiac poisons)/SLE flare with MODS/autonomic dysfunction (any cause, like GBS)/E-CPR.
Intensivists and physicians due to increasing experience with ECMO are likely to initiate early ECMO before the onset of VILI/VOLU Trauma.
Conclusion
ECMO is a potential life-saving treatment which has not been explored enough due to its perceived complexity and cost implications, but a protocolized management of patients by staff with experience would lead to better outcomes. We hope that with time this ground-breaking therapy is used more commonly.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
