Abstract
Background–Aim:
Uric acid is considered a marker of high cardiovascular risk. We investigated the association between serum uric acid and coronary arterial disease (CAD) in pre-dialysis chronic kidney disease (CKD) patients.
Methods:
We enrolled 110 subjects on mean age 69.02 ± 14.3 years old. The participants were categorized for both estimated glomerular filtration rate (eGFR) and albuminuria according to criteria 2012 of the Kidney Disease Improving Global Outcomes. Estimated pulse wave velocity (ePWV) was calculated using an equation including the age and mean blood pressure. The CAD prevalence rate was recorded.
Results:
The patients were divided in two groups according to uric acid cutoff point value related to high ePWV. The patients with higher uric acid were older and they had significantly higher systolic blood pressure, pulse pressure, and parathyroid hormone, but significantly lower eGFR and 1,25(OH)2 Vit D3 levels. The association between high uric acid and both high ePWV and CAD occurrence was found to be significant (x2 = 6.7, P = .008 and x2 = 4.1, P = .03, respectively), although the relationship with albuminuria was found to be nonsignificant. In a built multifactorial model, the low serum uric acid rather than the high was found to be an independent predictor for CAD demonstration entering traditional and specific confounders.
Conclusion:
The low serum uric acid levels were proved to be a significant predictor for CAD accounting potential covariates, even though the high uric acid per se was found to be connected with cardiovascular disease characteristics including arterial stiffness in predialysis CKD patients.
Highlights
In this study, we noted a significant association between a high serum uric acid and both vascular stiffness defined by a high estimated pulse wave velocity and coronary arterial disease occurrence.
In multifactorial association, the low rather than the high serum uric acid levels were proved to be an important predictor of coronary disease in combination to old age and diabetes mellitus in presence of traditional and specific confounders.
Uric acid levels were not significantly correlated with albuminuria, even though high serum uric acid was combined with a low rate of renal function. It seems that a second “hit,” such as diabetes mellitus or something other factor, needs for this relationship.
Uric acid levels were significantly associated with low levels of vitamin D active form in our participants.
Introduction
Serum uric acid is considered to be a marker of high cardiovascular risk because elevated serum acid levels were significantly associated with known risk factors of cardiovascular disease (CVD) such as hypertension, chronic kidney disease (CKD), and metabolic syndrome.1-3 Epidemiological studies have revealed that serum uric acid is associated with the development of hypertension, dyslipidemia, diabetes mellitus, renal disease, and cardiovascular events including atrial fibrillation.4-6 However, the evidence for a causal link between hyperuricemia and these disorders is controversial and it has not been well elucidated. 7
Vascular stiffness may be one of the mechanisms by which hyperuricemia increases the CVD risk through specific pathophysiological routes including changes in elastic properties of vascular wall, endothelial dysfunction, hypertension, and organ injury. 8 Previously, it has been already reported a link between uric acid and arterial stiffness in apparently healthy women. 9 On the other hand, because of vascular stiffness is highly prevalent in CKD and elevated serum uric acid are a usual finding in CKD, a relationship between uric acid and vascular disease could partly explain the commonly observed vascular stiffening in CKD. Arterial stiffness was found to be associated with decline in glomerular filtration rate (GFR) and albuminuria. 10 It has been also reported that in type 2 diabetes mellitus patients the levels of albuminuria, but not the reduced estimated GFR (eGFR), were associated with high arterial stiffening and atherosclerosis. 11 The presence of albuminuria is an early indicator of the underlying kidney disease. 12
Furthermore, it has been already established that the leading cause of death is mainly cardiovascular disease in patients with end-stage renal disease. 13 Recently, it has been supported that cardiovascular risk is also increased in patients with early stage of CKD and that early features of myocardial and coronary arterial disease (CAD) are common in predialysis CKD patients.14, 15
In this study, we aimed to investigate the association between serum uric acid and CAD in predialysis CKD patients after clustering according to the rate of renal clearance and to urinary albumin excretion. We also considered metabolic biomarkers which could influence this relationship.
Materials and Methods
Subjects
This is a single-center cross-sectional study in which we collected participants from the Department of Nephrology outpatient clinic of our Hospital.
A total of 110 subjects were enrolled, 67 males and 43 females on mean age 69.02 ± 14.3 years old.
We included patients in predialysis CKD according to criteria of the Kidney Disease Improving Global Outcomes (KDIGO) 2012. 16 The chronic renal disease had a time duration more than 3 months. We used the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation for the calculation of estimated glomerular filtration rate (eGFR). The enrolled subjects were also categorized according to albuminuria, which was defined as urinary albumin-to-creatinine ratio (UACR) ≥30 mg/gr based on KDIGO 2012 criteria using a spot urine sample. 16 This is an acceptable method for the calculation of albuminuria because UACR is correlated well with 24-h urinary albumin excretion. 17 The levels of albuminuria were stable for the past 6 months.
The underlying renal disease in our data included hypertensive nephrosclerosis in a ratio equal to 43.6% with duration of hypertension for more than 10 years and diabetic nephropathy in a ratio equal to 26.4%. Patients with an acute glomerulopathy proven by biopsy were excluded. Other exclusion criteria included the unstable angina, acute myocardial infarction, heart failure, or stroke within the past year.
Demographic data of the participants including age, gender, body mass index (BMI), peripheral blood pressure measurements, waist circumference measurements, and lifestyle characteristics regarding active smoking, alcohol drinking, and physical activity were recorded. We considered nondrinkers the subjects who declared no alcohol consumption during the past month and everyone who declared drinking was only a social drinker. We measured the physical activity/inactivity according to the World Health Organization (WHO) recommendations for healthy adults. 18 Waist circumference measurements made by our trained staff and the visceral obesity were determined using the International Diabetes Federation criteria for metabolic syndrome diagnosis. 19
Detailed individual medical histories were obtained from the participants. The occurrence of CAD (n = 51, 46.4%) was documented by clinical signs of angina pectoris, arrhythmia, a pre-existed old history of myocardial infarction, and coronary artery angioplasty or bypass surgery. Ischemic findings were also found out by electro- and echocardiographical examination and/or coronary computed angiography. In the suspicious subclinical cases of CAD, provocative tests were performed for consideration of classical clinical manifestations of vasospastic angina, documentation of myocardial ischemia during spontaneous episodes, and demonstration of coronary artery spasm using invasive coronary angiography to directly image coronary spasm. The existence of systolic and diastolic dysfunction during exposure period of the study was also considered. The patients with left ventricular hypertrophy (LVH) were excluded because they may have similar chest pain with CAD. The first and the current episodes of angina during the study were recorded as 1 event.
The current pharmacological therapy included similar hypoglycemic medications in all of diabetics. All participants were taking statin as hypolipidemic treatment, despite that being randomized. Everybody of the hypertensive was receiving the same category of antihypertensive medications including diuretics, calcium channel blockers, beta-blockers, and inhibitors of angiotensin II AT1 receptors. Forty of the subjects (n = 40, a ratio equal to 36.4%) were taking a complement of vitamin D. Nobody of our participants was taking phosphate binders. More than half of our participants (n = 64, a ratio of 58.2%) were taking allopurinol 100 mg or 150 mg per day for the regulation of serum uric acid. It should be ideal to be included participants without any therapy for the regulation of serum uric acid, but it was almost impossible, because our patients mostly had a renal dysfunction, which is commonly combined by serum uric acid disorders. However, the allopurinol treatment was stopped in total of our participants for 1 month, as study exposure period, before the statistical analysis after approval by ethical committee.
Blood Pressure Measurements
The blood pressure was measured at home double per day as systolic blood pressure (SBP) and diastolic blood pressure (DBP) using an automatic sphygmomanometer (OMRON M4-I Co Ltd, Kyoto, Japan). Peripheral mean blood pressure was calculated as: pMBP = DBP + 0.4 (SBP − DBP). We calculated the pulse pressure (PP) as the difference between SBP and DBP (PP = SBP – DBP). The definition of hypertension became according to recent American guidelines. 20
Calculation of Estimated Pulse Wave Velocity
We calculated estimated pulse wave velocity (ePWV) using the mathematic equation described in the study by Greve et al, 21 which was derived by the Reference Values for Arterial Stiffness’ Collaboration. 22 The age and mean blood pressure (MBP) were entered in the equation as: ePWV = 9.587 − 0.402 × age + 4.560 × 10−3 × age2 − 2.621 × 10−5 × age2 × MBP + 3.176 × 10−3 × age × MBP − 1.832 × 10−2 × MBP.
Electro- and Echocardiographical Assessment
A 12-lead electrocardiographical (ECG) examination was used to estimate the ischemic findings. Echocardiographical assessment was conducted by a Hewlett Packard SONOS 2500 device using a 2.25-MHz transducer. Two cardiologists examined our patients with the method of conventional M-mode and two-dimensional echocardiography for LVH estimation, assessment of systolic cardiac function, and ischemic findings consideration according to the recommendations of the American Society of Echocardiography. 23 LVH existence was defined by a thickness of interventricular septum >11.5 mm. The systolic function of the left ventricle (LV) was assessed by the measurement of ejection fraction (EF) and the systolic dysfunction was defined as EF <50%. We used tissue Doppler imaging and pulsed wave Doppler techniques for the evaluation of LV diastolic function. 24 Transmitral early and late diastolic (E and A waves) velocities were measured and the ratio of E/A was calculated. We also measured the peak early (e′) diastolic mitral annular velocity and E/e′ ratio was calculated. E/e′ ratio ≥15 and a ratio of E/A ≤1 were used to determine the existence of diastolic dysfunction, even if the E/A ratio is greatly influenced by hemodynamic status.
Biochemical Measurements
Overnight fasting plasma creatinine, glucose, calcium (Ca), phosphate (P), triglycerides, low-density lipoprotein-cholesterol (LDL-C), and high-density lipoprotein-cholesterol (HDL-C) were recorded using the results from a blood test in the end of study exposure period. These biochemical markers were measured using spectrophotometric technique by Chemistry Analyzer (MINDRAY BS-200; Diamond Diagnostics, Holliston, MA, USA) and were represented as mg/dL. The Ca × P products were calculated.
Because serum uric acid is fluctuated throughout the time, 25 we considered 4 values of serum uric acid sequentially in fasting state, 1 measurement per week, during the study exposure period equal to 1 month and the average of them was used in statistical analysis.
The concentration of intact-parathyroid hormone (i-PTH) and insulin levels were measured by radioimmunoassays (CIS Bio International/France and BioSource Europe SA, Belgium, respectively). 25(OH)VitD3 and 1,25(OH)2VitD3 were assessed using high-performance liquid chromatography 26 and chemiluminescent immunoassay, respectively.
Insulin resistance was calculated using the homeostasis model assessment of insulin resistance as: HOMA-IR = insulin (µU/mL) × glucose (mmol/dL)/22.8. 27
Spot urine samples from the first morning void were used for the measurement of albumin and creatinine concentrations by the Chemistry Analyzer.
Data Analysis
Data were analyzed using SPSS 25.0 statistical package for Windows (SPSS Inc, Chicago, IL, USA) and expressed as mean ± standard deviation or as median value (interquartile range) for data that showed skewed distribution. First, we determined the cutoff point ePWV value related to the presence of albuminuria and we changed the numerical ePWV to a categorical variable. Then, we separated our participants in 2 groups according to ROC curve uric acid cutoff point value related to categorical ePWV equal to 6.62 mg/dL (greater, n = 49 or lower, n = 61 than 6.62 mg/dL). Differences between mean values were assessed by using unpaired t-test for 2 groups and data that showed skewed distributions were compared with Mann-Whitney U test.
Correlations between variables were defined by Spearman coefficient and the relationships between categorical variables were defined by chi-square tests. P values less than .05 were considered significant.
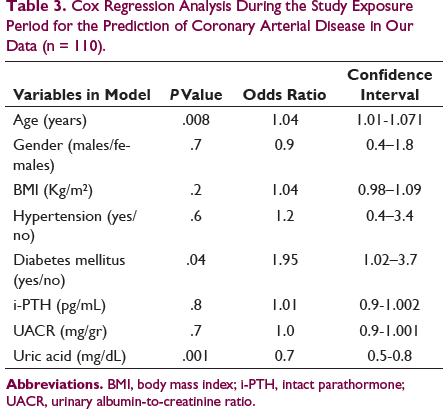
We built a model using Cox-regression analysis during the study exposure period equal to 30 days, in order to investigate the important predictors for the manifestation of coronary artery disease entering traditional and specific covariates in our data including the age, gender, hypertension, BMI, diabetes mellitus, i-PTH, and albuminuria.
Results
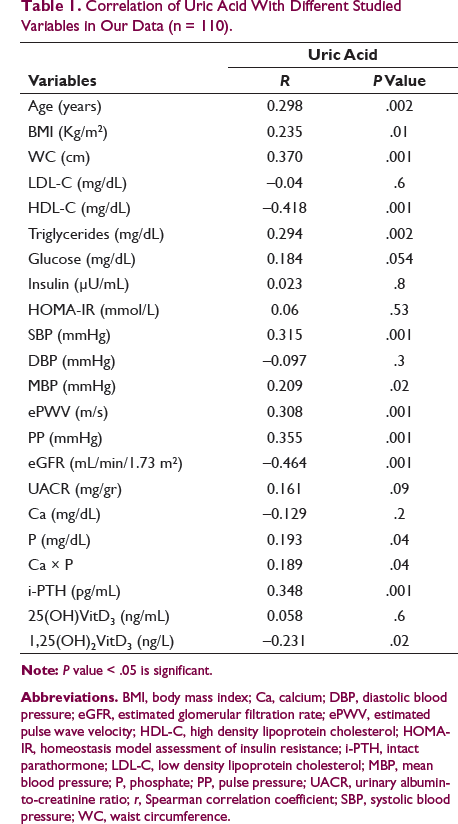
Correlation of Uric Acid With Different Studied Variables in Our Data (n = 110).
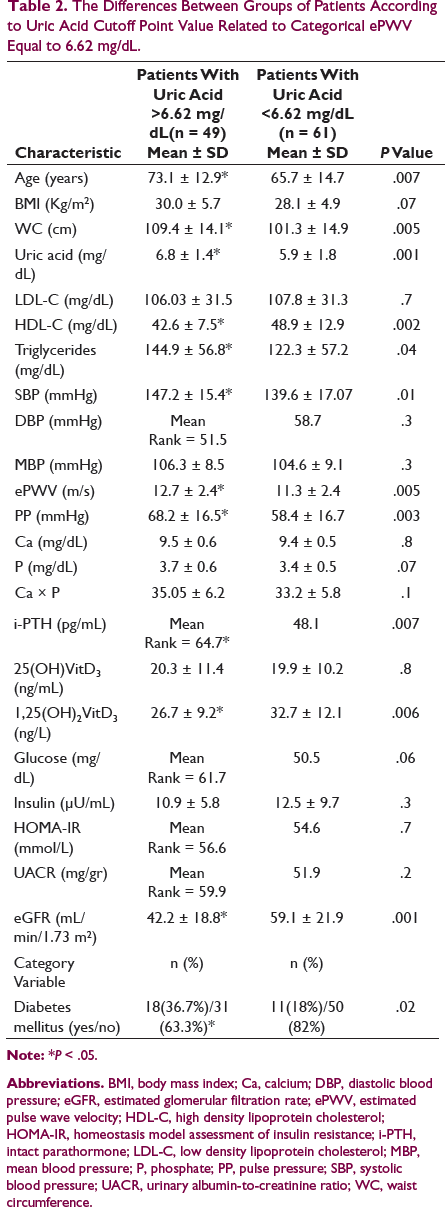
The Differences Between Groups of Patients According to Uric Acid Cutoff Point Value Related to Categorical ePWV Equal to 6.62 mg/dL.
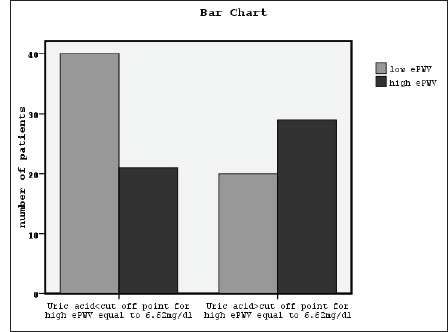
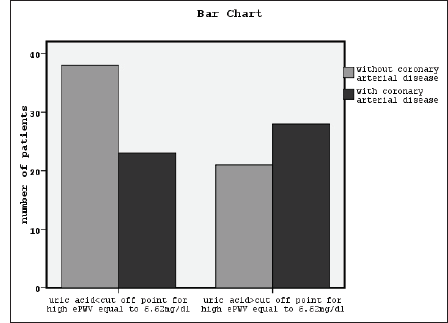
X2 tests showed significant association between a high serum uric acid and a high ePWV, classified eGFR, diabetes mellitus, and occurrence of CAD (x2 = 6.7, P = .008, Figure 1, x2 = 14.2, P = .003, x2 = 4.8, P = .02, and x2 = 4.1, P = .03, Figure 2, respectively). The association between serum uric acid and the presence of albuminuria was found to be nonsignificant. In the meantime, the relationship between coronary artery disease and ePWV, classified eGFR and classified albuminuria was found to be significant (x2 = 24.2, P = .001, x2 = 14.6, P = .002, and x2 = 9.7, P =.007, respectively). The association between ePWV and both classified eGFR and classified albuminuria was also found to be significant (x2 = 33.9, P = .001 and x2 = 12.7, P = .002, respectively).
The Association Between High Serum Uric Acid and High Estimated Pulse Wave Velocity Using Chi-Square, Phi, and Cramer’s V Correlation Test (x2 = 6.7, P = .008).
The Association Between High Serum Uric Acid and Occurrence of Coronary Arterial Disease Using Chi-Square, Phi, and Cramer’s V Correlation Test (x2 = 4.1, P = .03).
Cox Regression Analysis During the Study Exposure Period for the Prediction of Coronary Arterial Disease in Our Data (n = 110).
Discussion
Our principal findings included a significantly positive association between high serum uric acid and vascular stiffness, hypertension, CAD occurrence, metabolic syndrome components, bone disease characteristics, and a low rate of renal clearance. We did not find a significant relationship between high serum uric acid and albuminuria. We also observed that the low serum uric acid to be a significant risk factor for the prediction of CAD.
Uric acid is the final enzymatic product of purine metabolism in the human body regulated by the action of xanthine oxidase, and increased levels are mainly caused by the high intake of purine-rich foods or reduced urinary excretion. 28 The uric acid kinetic through the renal clearance explains our finding regarding the significant association of high uric acid with a low rate of renal function. However, serum uric acid levels are fluctuated throughout the time resulting in manifestation even of transient than permanent hyperuricemia and/or hyperuricosuria during the day and/or the time depended on different variables including blood pH, temperature, serum sodium concentrations, and anion overload. 25 In this study, we used the average of 4 consecutive measurements of serum uric acid during the study exposure period for our statistical analysis.
Previously, epidemiological observational studies showed a positive association between uric acid and vascular stiffness in apparently healthy women,9, 29-31 However some of the studies did not find any such association in subjects without co-morbidities, such as CKD, CVD, or diabetes mellitus. In this study, we observed a positive relationship between high serum uric acid and vascular stiffness. This may be due to renal failure and diabetes mellitus in enrolled subjects, as primary disease. In discrepancy, recent study did not find a significant correlation of uric acid with ba-PWV in advanced CKD patients. 32 Previous interventional studies also suggested that the treatment with allopurinol, an inhibitor of xanthine oxidase, reduced the levels of serum uric acid and it had a significant regression of left ventricular mass without a significant change in PWV in CKD or patients with heart disease. 33 In addition, it has been reported that the therapy with allopurinol improved endothelial function resulting in improved arterial compliance without a significant effect on blood pressure.33, 34 In this study, we applied stopping of allopurinol therapy in our participants for 1 month before the statistical analysis.
The underlying mechanisms for the linking between uric acid and vascular stiffness include the production of free radicals, oxidative stress, endothelial dysfunction, and inflammation.35-37 The inflammatory pathways, which are associated with uric acid, may play a role in the process of arterial stiffness even in healthy subjects. The hyperuricemia-induced microvascular damage in afferent arterioles of kidneys may lead to the manifestation of sodium sensitive hypertension, which in turn aggravates the vascular stiffening. 38 Indeed, in this study we noted that the group of patients with a high uric acid had higher blood pressure, and the bivariate correlation between uric acid and SBP, MBP, and PP was found to be significantly positive.
Recent studies have shown that hyperuricemia could predict metabolic syndrome in adolescents, elderly, and even children, and hyperuricemia may be a component of metabolic syndrome in accordance to our findings.3, 39-41
Furthermore, in this study we noted an important association between uric acid and bone disease characteristics including serum phosphate, Ca × P products, i-PTH, and active form of vitamin D (1,25(OH)2VitD3). Previously, 1 study reported a positive relationship between PTH and uric acid levels in men suggesting that PTH is able to affect the transport of uric acid in the proximal renal tubules playing a role in secretive and absorptive transporters at the cellular membranes.41, 42 Recent study also reported that uric acid was an important predictor of low serum 25(OH)VitD3 form of vitamin D in predialysis CKD patients. 43 None of the study reported a correlation between serum uric acid and 1,25(OH)2VitD3 to our knowledge until now. 1,25(OH)2VitD3 or calcitriol is the physiologically active form of vitD, but 25(OH)VitD3 is the main form in the blood stream and the best indicator of vitD status because 1,25(OH)2VitD3 levels are influenced by PTH abnormalities. 44 However, in renal disease both forms of vitD is required to measure. Low levels of vitD have been reported to be associated not only with metabolic disorders but also with endothelial dysfunction and inflammation causing cardiovascular morbidity. 45
Bone metabolism abnormalities, inflammation, oxidative stress, hypertension, and a disordered renin-angiotensin-aldosterone system are involved in the pathophysiology of early CKD-associated cardiomyopathy, stiffness of LV, and coronary artery disease even nonflow limiting. 46 Recently, it has been reported that serum uric acid levels may serve as a predictor of the severity of coronary artery stenosis in nondialysis CKD patients with coronary disease. 47 In agreement, in our data we observed a significant unadjusted relationship between high serum uric acid and CAD occurrence. Furthermore, in this study coronary disease was significantly associated with vascular stiffness, low rate of renal function, and albuminuria. Previously, it has been suggested that chronic renal dysfunction and albuminuria may be important factors determining the severity and occurrence of coronary disease in accordance to our findings.48-50
Nevertheless, even though we noted a significant association between high serum uric acid and classified eGFR, we did not reveal such a significant association between high serum uric acid and classified albuminuria. In discordance, previously, it has been reported that uric acid was independently associated with albuminuria in type 2 and type 1 diabetes mellitus.51, 52 This discrepancy may be attributed to our participants, who included a relatively low ratio of diabetics. It may be required the combination of a second “hit,” such as diabetes mellitus, thus uric acid to be associated with urinary albumin excretion.
In the meantime, we found a significant correlation between vascular stiffness and both classified eGFR and classified albuminuria beyond of our finding for the significant relationship between vascular stiffness and high serum uric acid. The mechanisms linking arterial stiffness, uric acid and renal decline may be realted to endothelial dysfunction. Endothelial dysfunction modifies the glomerular permeability leading to increased albumin excretion. 53 However, the role of uric acid per se on the pathogenesis of endothelial dysfunction is difficult to be determined because uric acid presents a strong association with other risk factors such as hypertension, metabolic syndrome, CKD, alcohol intake, and diuretics, which may also influence endothelial dysfunction. 54 On the other hand, it has been suggested by experimental findings a role of uric acid as antioxidant in plasma with an antiatherosclerotic effect through neutralizing radical species, maintaining superoxide dismutase activity, and formatting complexes with metals, such as Fe3+. 55 Recently, it has also been reported that both low and high serum uric acid levels were associated with increased all-cause mortality supporting a U-shaped association between uric acid and mortality. 56 In agreement, in this study in the built multifactorial model accounting the observed time (equal to one month), the low serum uric acid rather than the high serum uric acid in combination to old age and diabetes mellitus was found to be an important independent predictor for CAD in presence of traditional and specific confounders, such as gender, BMI, hypertension, i-PTH, and albuminuria.
This study showed that the low uric acid rather than the high could predict CAD in combination with known predictors including the old age and diabetes mellitus. However, the high serum uric acid per se was found to be significantly associated with characteristics of cardiovascular disease including vascular stiffness, which may be the connecting ring between uric acid, CAD, and renal disease. Based on these findings, we should not support the hypothesis that the strong reduction of uric acid levels using pharmaceutical therapy could protect the cardiac function decreasing the cardiovascular events in predialysis chronic renal disease patients. Furthermore, the continuous fluctuation of serum uric acid requires the average usage of more consecutive measurements rather than the use of 1 single value. Bigger studies need to verify the above findings and to clarify the association between serum uric acid and albuminuria in this population of patients independently on the presence of diabetes mellitus.
Limitations
The main limitation of this study is the fact that this is a cross-sectional study by one nephrological center, which does not allow the assessment of cause-effect relationships. Additionally, the inclusion of patients without any therapy for the regulation of serum uric acid in our data was unavailable.
Conclusion
The low serum uric acid levels were proved to be a significant risk factor for CAD prediction accounting potential covariates, even though the high serum uric acid per se was found to be connected with characteristics of cardiovascular disease including arterial stiffness in predialysis CKD patients. Metabolic disorders, bone disease abnormalities, and the low rate of renal clearance may influence this relationship.
Practical Application
Considering not only one single measurement, but the average of more consecutive values of serum uric acid, due to its fluctuation throughout the time, the modest reduction of uric acid levels using pharmacological therapy could improve the prevalence rate of CAD in predialysis chronic renal disease patients.
Footnotes
Authors’ Contributions
Vaia D Raikou: Research plan, data collection, biostatistic analyses, and manuscript writing. Giannis Vlaseros: Electro- and Echocardiographical assessments. Despina Kyriaki: biochemical measurements. Sotiris Gavriil: research plan, data collection, and manuscript writing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted and it was approved by the Hospital Institutional Review Board with an Institutional Review Board (IRB)/Ethics Committee approval number (A.P:7216). All procedures were performed in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed oral consent was obtained from all individual participants (or legal parent) included in the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
