Abstract
Objective:
To evaluate the incidence rate of definite stent thrombosis and major adverse cardiovascular events (MACE) with Ticagrelor versus Clopidogrel in ST segment elevation myocardial infarction (STEMI) patients candidate for primary percutaneous coronary intervention (PCI).
Methods:
STEMI participants naïve to antiplatelets, with no history of coronary artery disease (CAD) and candidate for primary PCI were included, while participants with history of CAD were excluded. Two hundred consecutive participants were selected, divided into 2 groups of 100 participants each, received either Ticagrelor or Clopidogrel, and followed up at 3 and 6 months.
Results:
The percent of patients in the Ticagrelor group who developed definite stent thrombosis (in-hospital and total) was 0%, while the percent of patients in the Clopidogrel group who developed definite stent thrombosis (in-hospital and total) was 8% and 9%, respectively. There were statistically significant weak associations between the class of P2Y12 platelet inhibitors and definite stent thrombosis (in-hospital and total) (X2 = 8.33, P = .004, V = 0.204 and Χ2 = 9.424, P = .002, V = 0.217, respectively). The percent of patients in the Ticagrelor and Clopidogrel groups who developed in-hospital MACE was 1% and 9%, respectively. There was a statistically weak significant association between the class of P2Y12 platelet inhibitors and in-hospital MACE (X2 = 6.74, P = .009, V = 0.184).
Conclusion:
Ticagrelor is more efficacious than Clopidogrel in preventing definite stent thrombosis (in-hospital and total) and in-hospital MACE in STEMI patients.
Introduction
Acute coronary syndrome (ACS) is a major cause of death and disability-adjusted life years lost worldwide.1, 2 Clinically, ACS presents with acute persistent chest pain or other symptoms suggestive of cardiac ischemia and either ST segment elevation in at least 2 adjacent leads with abnormal cardiac biomarkers (ST elevation myocardial infarction or STEMI), ST segment depression, and/or T wave inversion with abnormal cardiac biomarkers (non-ST elevation myocardial infarction or NSTEMI), or ECG changes indicative of ischemia without abnormal cardiac biomarkers (unstable angina).3-5 As per the current American College of Cardiology/American Heart Association guidelines, STEMI and NSTEMI patients are recommended to receive dual antiplatelet therapy (aspirin and P2Y12 platelet inhibitor). 6 Ticagrelor and Clopidogrel are two P2Y12 platelet inhibitors commonly used in clinical practice. Ticagrelor is a reversible P2Y12 platelet inhibitor, while Clopidogrel is an irreversible one.7, 8 .The landmark trial which compared Ticagrelor to Clopidogrel was the PLATelet inhibition and patient Outcomes (PLATO) trial. Results of the PLATO trial showed that in both STEMI and NSTEMI patients, major adverse cardiovascular events (MACE; death from cardiovascular causes, myocardial infarction, or stroke) at 12 months were significantly reduced with Ticagrelor compared with Clopidogrel (9.8% versus 11.7%; hazards ratio [HR] 0.84 (95% confidence interval [CI] 0.77-0.92), P = <.001)), and among the subgroup of participants who underwent stent deployment, the rate of definite stent thrombosis at 12 months was significantly reduced with Ticagrelor compared with Clopidogrel (1.3% versus 1.9%; HR 0.67 [95% CI 0.50-0.91], P = .009]. 9 PLATO trial subgroup analysis study showed that in STEMI patients, MACE at 12 months were slightly nonsignificantly reduced with Ticagrelor compared with Clopidogrel (9.4% versus 10.8%; HR 0.87 [95% CI 0.75-1.01], P = .07), and the rate of definite stent thrombosis at 12 months was significantly reduced with Ticagrelor compared with Clopidogrel (1.6% versus 2.4%; HR 0.66 [95% CI 0.45-0.95], P = .03]. 10 Ticagrelor versus Clopidogrel in real-world patients with STEMI is another study which compared Ticagrelor and Clopidogrel. Results showed that in STEMI patients, in-hospital MACE were nonsignificantly reduced with Ticagrelor compared with Clopidogrel (4.9% versus 6.9%; odds ratio [OR] 0.69 [95% CI 0.28-1.7], P = .52), and there was no significant difference in the in-hospital stent thrombosis between Ticagrelor and Clopidogrel groups (1.4% versus 0.8%, P = .62). 11 We wanted to evaluate the efficacy outcomes of Ticagrelor versus Clopidogrel in STEMI patients naïve to antiplatelets, with no history of coronary artery disease (CAD) and candidate for revascularization with primary percutaneous coronary intervention (PCI).
Materials and Methods
Study Design
Our study was a single tertiary care cardiac center prospective, open-labeled, nonrandomized, controlled cohort study. Investigators weren’t blinded to the study groups. The study design was approved by the hospital ethics committee and all participants signed written informed consents. The total duration of the study was 2 years from November 2015 to November 2017.
Study Participants
Study participants were patients referred to the Sabah Al-Ahmad Cardiac Center at Al-Amiri Hospital in Kuwait City, Kuwait. They were subjected to history taking and data collection for age, gender, nature and course of ischemic symptoms, and risk factors especially hypertension, diabetes mellitus, smoking and dyslipidemia, comprehensive clinical examination, 12 leads electrocardiogram (ECG), and laboratory investigations including complete blood picture, coagulation profile, kidney function tests, and cardiac biomarkers (including high sensitivity cardiac troponin T). Screened participants were enrolled if they were naïve to antiplatelets (aspirin and adenosine diphosphate receptor antagonist), had STEMI, candidate for primary PCI, and with no history of CAD. Screened participants with history of CAD or candidates for rescue PCI were excluded from the study.
Study Treatment
Two hundred eligible participants were nonrandomly consecutively assigned with 1:1 allocation ratio to receive either Ticagrelor or Clopidogrel, administered in an open-labeled, unblinded fashion. The estimated sample size, after consulting the literature, is based on a minimum anticipated observed treatment effect (difference in the rate of in-hospital and total definite stent thrombosis with Ticagrelor versus Clopidogrel) equivalent to 0.02 (2.0%), standard deviation of rate of in-hospital, and total definite stent thrombosis with Ticagrelor or Clopidogrel equivalent to 0.05, type I error = 0.05, type II error = 0.20, and power of 0.8 (80%). The calculated sample size is 200. The enrolled study participants were divided into 2 groups of 100 participants each, received a loading dose of 300 mg acetyl salicylic acid chewed in the emergency room (ER) followed by a maintenance dose of 81 mg per oral (PO) once daily (OD), Ticagrelor (loading dose 180 mg in the ER prior to the primary PCI followed by maintenance dose 90 mg PO twice daily), or Clopidogrel (loading dose 600 mg in the ER prior to the primary PCI followed by maintenance dose 75 mg PO OD), heparin intravenously in the catheterization laboratory according to the activated clotting time, and anti-ischemic treatment prescribed according to the participant’s conditions and comorbidities including beta blockers or calcium channel blockers, statins and nitrates, angiotensin converting enzyme inhibitors or angiotensin II receptor blockers. All study participants underwent primary PCI. Data documented with the primary PCI and during hospital stay included door to balloon time, culprit vessel, angiographic thrombus burden classification, culprit lesion length, reference diameter of the culprit vessel, presence or absence of calcification in culprit vessel, number of nonculprit diseased vessels, type, diameter, length, and number of the deployed stents in culprit and nonculprit vessels, angiographic procedural success, in-hospital complications (eg, definite stent thrombosis, bleeding) and MACE, and predischarge echocardiography. 12 Outpatient visits were scheduled at 3 and 6 months for follow-up. Data documented with the outpatient visits included ischemic symptoms as chest pain with details of its character, duration, frequency, and requirements of nitrates per day and per week, dyspnea, syncope, effort intolerance, and other symptoms that may be of cardiac origin, bleeding requiring blood transfusion, MACE, clinical examination, and 12 leads ECG. 13
End Points
The study assessed the difference in the rate of in-hospital and total definite stent thrombosis with Ticagrelor versus Clopidogrel in STEMI patients postprimary PCI and examined the association between the class of P2Y12 platelet inhibitors and total definite stent thrombosis. Definite stent thrombosis is acute thrombosis with angiographic or pathological confirmation of thrombus in the immediate vicinity of the deployed stent. 14 In-hospital definite stent thrombosis includes acute definite stent thrombosis occurring within 24 h of primary PCI and subacute definite stent thrombosis occurring beyond the first 24 h until the first 30 days after primary PCI. 15 Total definite stent thrombosis includes in-hospital definite stent thrombosis and late definite stent thrombosis which occurs between 30 days and 1 year after primary PCI. 16 Secondary objectives included assessment of the difference in the rate of in-hospital, 3 months, and 6 months MACE with Ticagrelor versus Clopidogrel in STEMI patients postprimary PCI.
Statistical Analysis
The efficacy outcomes were coded, and the data was analyzed with the Statistical Package for the Social Sciences software (SPSS) version 25. Quantitative data was expressed as means and standard deviations, while qualitative data was expressed as medians and ranges. Comparisons between parametrically distributed quantitative variables were done with Independent t-test, between nonparametrically distributed quantitative variables with Mann-Whitney test, and between qualitative variables with Chi-square test, respectively.17, 18 The CI was set to 95% and the margin of error accepted was set to 5%. Any comparison considered statistically significant was at P < .05 or less and highly significant at P < .01. Final data analysis was as per the intention to treat analysis.
Results
Study Participants, Medications, and Procedures
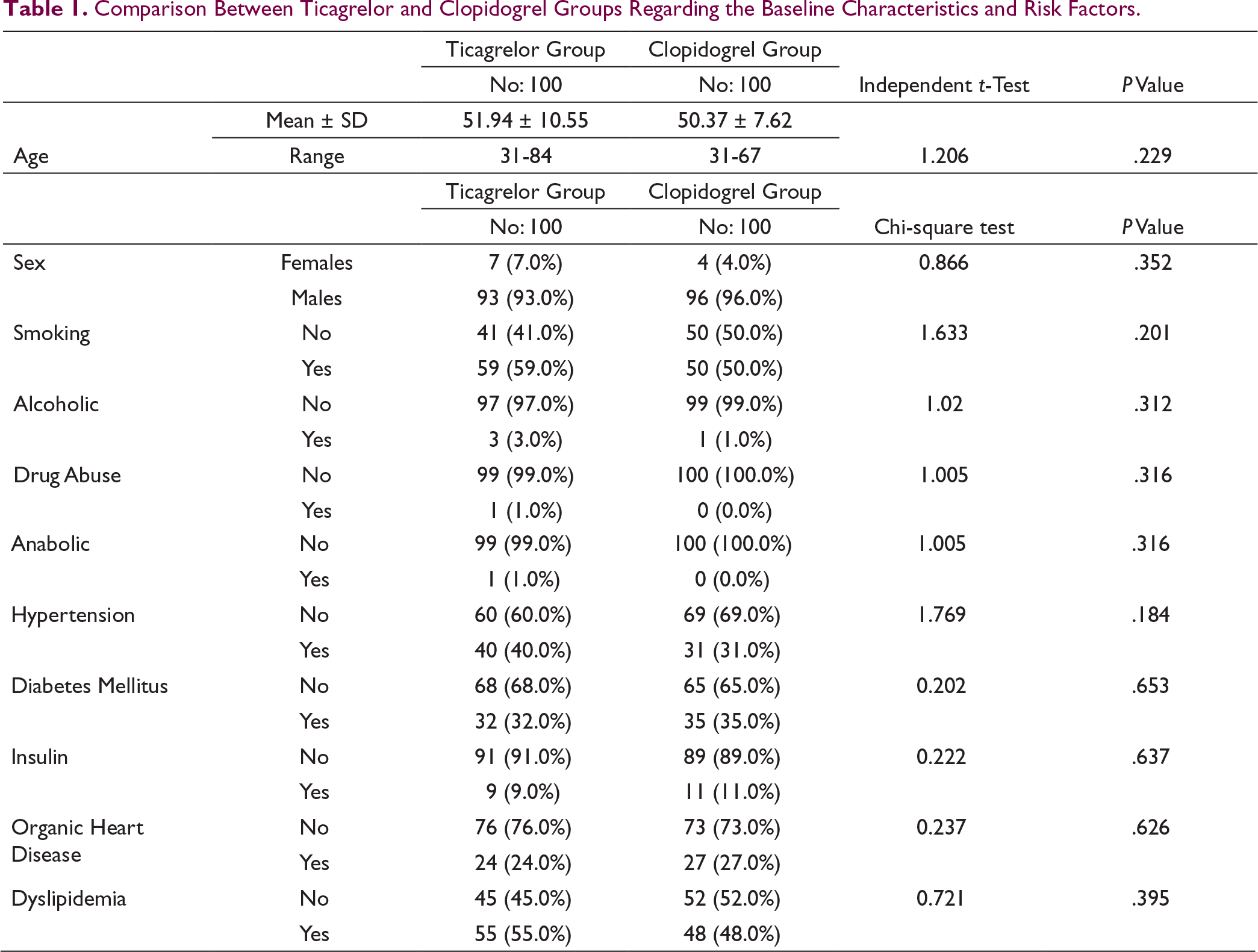
Comparison Between Ticagrelor and Clopidogrel Groups Regarding the Baseline Characteristics and Risk Factors.
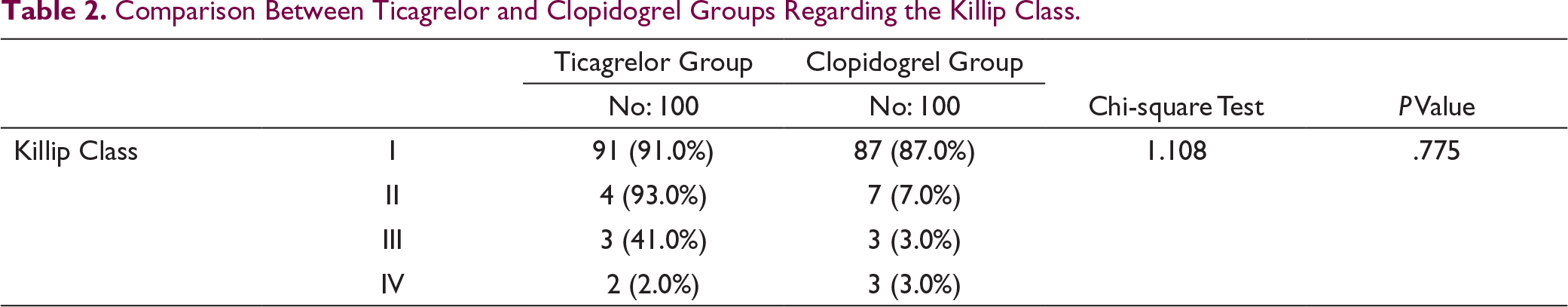
Comparison Between Ticagrelor and Clopidogrel Groups Regarding the Killip Class.
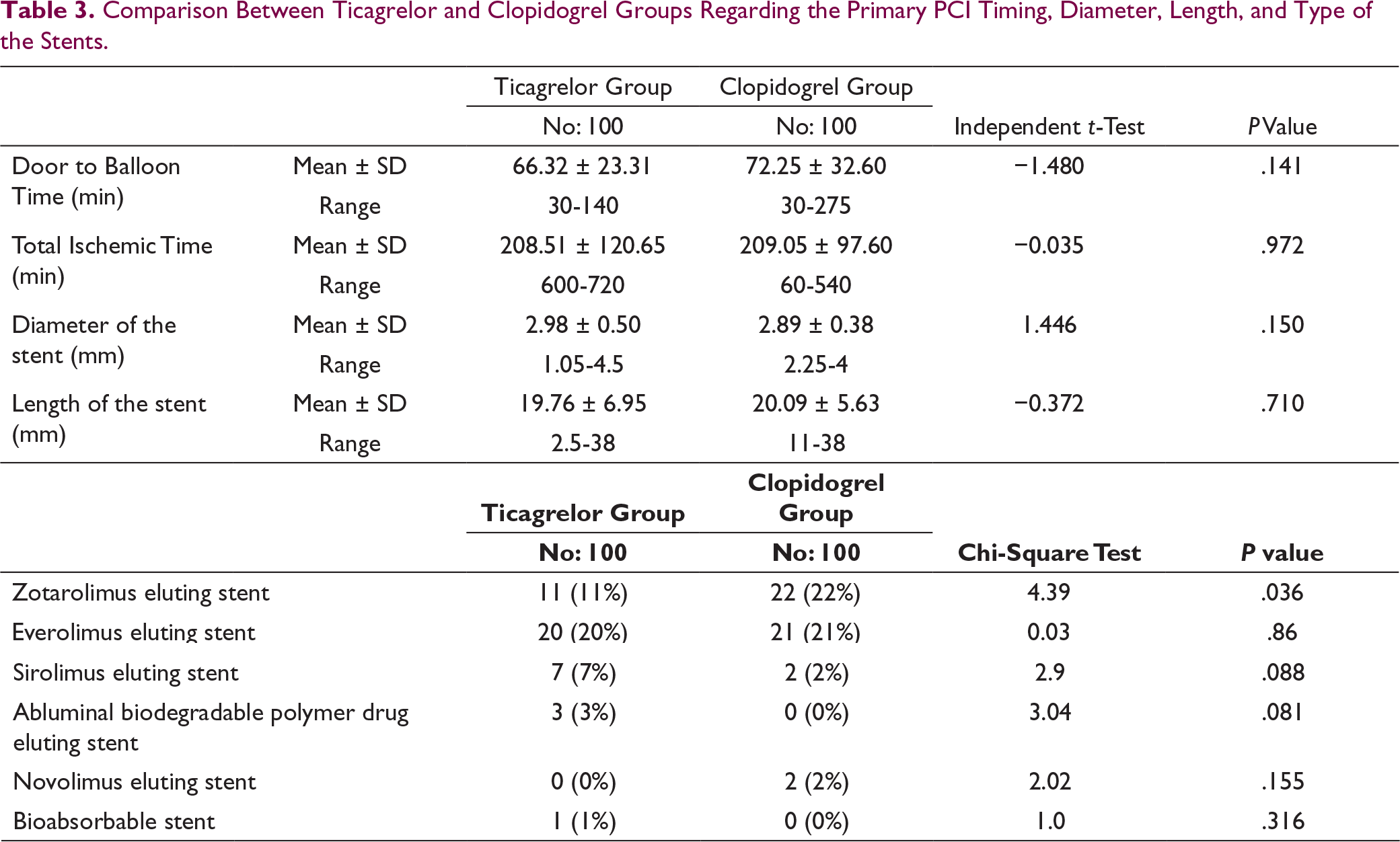
Comparison Between Ticagrelor and Clopidogrel Groups Regarding the Primary PCI Timing, Diameter, Length, and Type of the Stents.
Efficacy
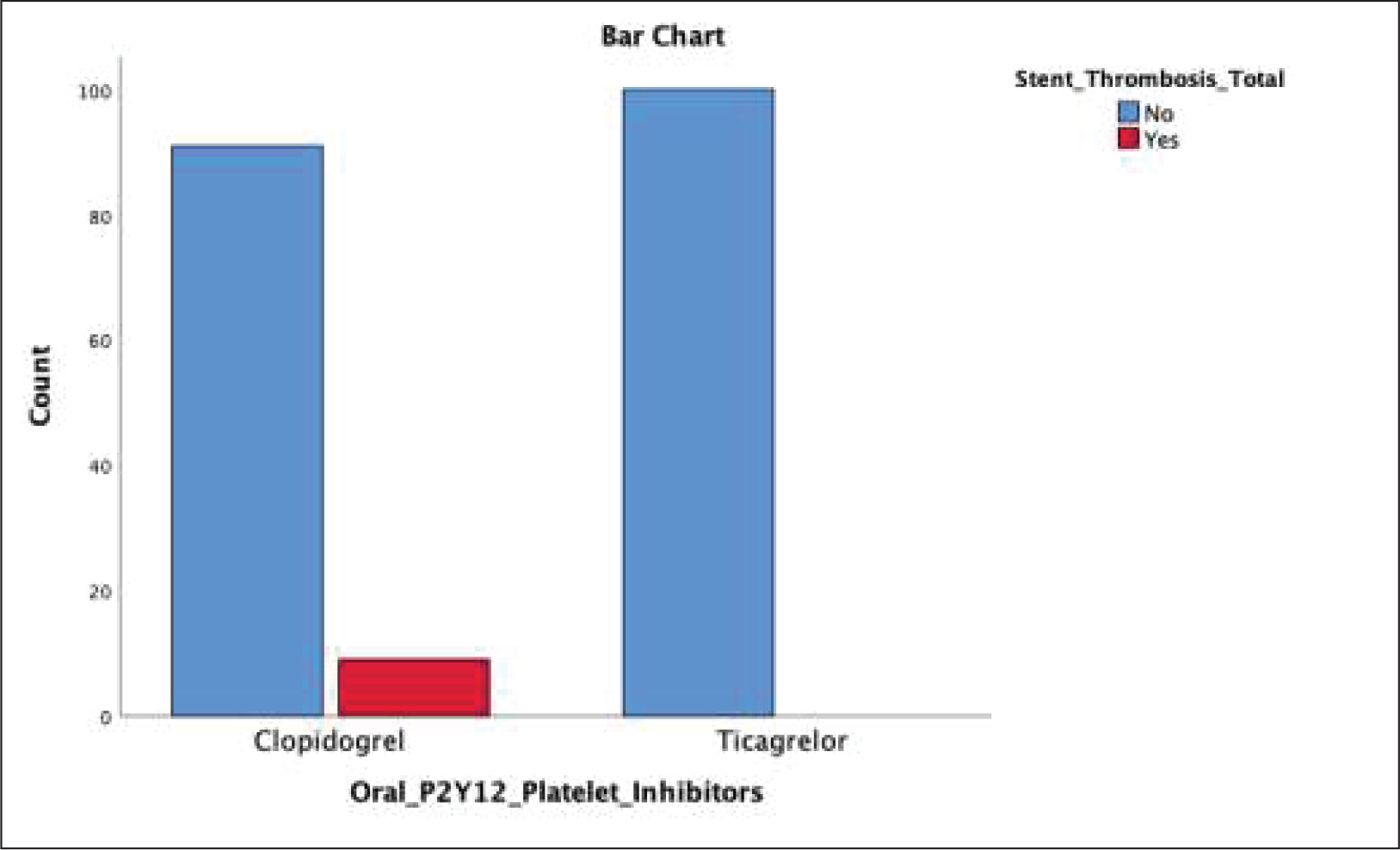
The percent of patients in the Ticagrelor group who developed definite stent thrombosis (in-hospital and total) was 0%, while the percent of patients in the Clopidogrel group who developed definite stent thrombosis (in-hospital and total) was 8% and 9%, respectively (Figure 1). There was a statistically significant difference in the rate of definite stent thrombosis (in-hospital and total) between Ticagrelor and Clopidogrel groups (P = .004 and P = .002, respectively), with an 8% absolute risk reduction and 100% relative risk reduction of in-hospital definite stent thrombosis in the Ticagrelor group compared to the Clopidogrel group, a 9% absolute risk reduction and 100% relative risk reduction of total definite stent thrombosis in the Ticagrelor group compared to the Clopidogrel group, and a statistically significant weak association between the class of P2Y12 platelet inhibitors and total definite stent thrombosis (in-hospital and total) (Χ2 = 8.33, P = .004, V = 0.204 and Χ2 = 9.424, P =.002, V = 0.217, respectively).
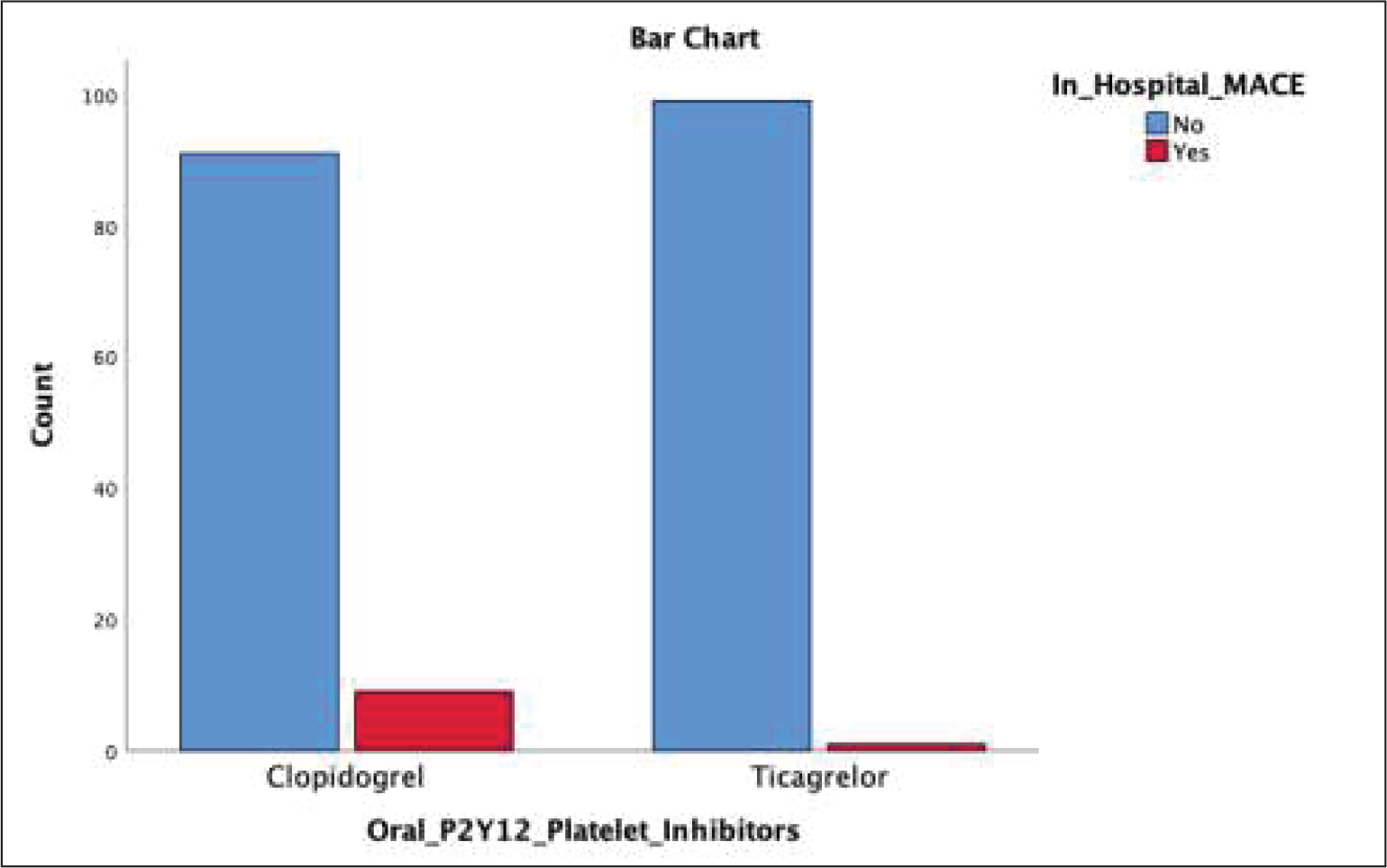
The percent of patients in the Ticagrelor group who developed MACE (in-hospital, 3 months, and 6 months) was 1%, 1%, and 0%, respectively, while the percent of patients in the Clopidogrel group who developed MACE (in-hospital, 3 months, and 6 months) was 9%, 2%, and 2%, respectively (Figure 2). There was a statistically significant difference in the rate of in-hospital MACE between Ticagrelor and Clopidogrel groups (P = .009), with an 8% absolute risk reduction and 89% relative risk reduction of in-hospital MACE in the Ticagrelor group compared to the Clopidogrel group, a nonsignificant difference in the rate of 3 months and 6 months MACE between Ticagrelor and Clopidogrel groups (P = .561 and P =.153, respectively), and a statistically significant weak association between the class of P2Y12 platelet inhibitors and in-hospital MACE (Χ2 = 6.74, P = .009, V = 0.184).
Comparison Between Ticagrelor and Clopidogrel Groups Regarding the Rate of Total Definite Stent Thrombosis.
Comparison Between Clopidogrel and Ticagrelor Regarding the Rate of In-Hospital MACE.
Safety
The percent of patients in the Ticagrelor group who developed in-hospital gastrointestinal bleeding and hematuria was 1% and 0%, respectively, while the percent of patients in the Clopidogrel group who developed in-hospital gastrointestinal bleeding and hematuria was 1% and 1%, respectively. There was a nonsignificant difference in the rate of in-hospital gastrointestinal bleeding and hematuria (P = 1.0 and P = .316, respectively) between Ticagrelor and Clopidogrel groups.
Discussion
Despite the recommended usage of the newer reversible P2Y12 platelet inhibitor drugs in STEMI, there is still uncertainty about their efficacy compared with the more widely adopted irreversible P2Y12 platelet inhibitor drugs, especially in the STEMI patient population. 21 The STEMI patient population has reduced gastrointestinal absorption, increased vomiting, and frequent morphine administration, causing unpredictable pharmacokinetic profile and affecting drug bioavailability. 22 Results of the PLATO trial showed that in both STEMI and NSTEMI patients, MACE and definite stent thrombosis were significantly reduced with Ticagrelor compared with Clopidogrel without causing a significant concomitant increase in major bleeding. Our study’s results were consistent with the overall PLATO trial and the PLATO trial subgroup analysis results. As noted above, the reduction in morbidity and mortality rates with Ticagrelor compared to Clopidogrel in STEMI patient population (8% absolute risk reduction and 100% relative risk reduction of in-hospital definite stent thrombosis, 9% absolute risk reduction and 100% relative risk reduction of total definite stent thrombosis, and 8% absolute risk reduction and 89% relative risk reduction of in-hospital MACE) added health and survival benefits to Ticagrelor without increasing the risk of major bleeding. Given the PLATO trial subgroup analysis definite stent thrombosis event rate of 2.4% in the Clopidogrel group at 12 months, our study’s total definite stent thrombosis event rate of 9% in the Clopidogrel group at 12 months indicates possible Clopidogrel resistance in our study group and justifies consideration of Clopidogrel pharmacogenetic testing in future retrospective and prospective cohort studies. On the other hand, Ticagrelor versus Clopidogrel in real-world patients with STEMI study results showed that in STEMI patients, there was a nonsignificant difference in the in-hospital definite stent thrombosis and the in-hospital MACE between Ticagrelor and Clopidogrel groups. Enrolled participants of this study had history of CAD and weren’t naïve to antiplatelets. Our study’s results were inconsistent with the results of this study possibly because our study’s participants were naïve to antiplatelets and had no history of CAD.
Strengths and Limitations
Our study was conducted on a well-balanced cohort with regard to baseline characteristics, risk factors, and Killip class, didn’t have missing data allowing robust intention to treat analysis of the efficacy outcomes, and to the best of our knowledge, assessment of the difference in the rate of in-hospital and total definite stent thrombosis with Ticagrelor versus Clopidogrel in antiplatelet-naïve STEMI patient population postprimary PCI is a novel endpoint that wasn’t researched before. Despite its’ strengths, the study limitations require consideration. Lack of blinding of the investigator and absence of allocation concealment of both treatments and hence, the possibility of biased observation of the outcomes among participants who received Ticagrelor and Clopidogrel. To minimize observer bias, the outcome assessors who analyzed and reported the results were blind to the identity of the study participants and unaware of the treatment received by them. 23 In addition, the study was a single center study with a small sample size and small number of events in both treatment groups.
Conclusion
Ticagrelor is more efficacious than Clopidogrel in preventing in-hospital and total definite stent thrombosis and in-hospital MACE in STEMI patients. Reversible P2Y12 platelet inhibitor drugs, as Ticagrelor, are more potent alternatives to irreversible P2Y12 platelet inhibitor drugs, as Clopidogrel, in antiplatelet-naïve STEMI patients resulting in significant morbidity and mortality reduction without increasing the risk of major bleeding.
What This Study Adds (What’s New)
More in-hospital and total definite stent thrombosis in the Clopidogrel group than the Ticagrelor group (8% and 9% versus 0%).
Statistically significant weak associations between the class of P2Y12 platelet inhibitors and in-hospital definite stent thrombosis (Χ2 = 8.33, P = .004, V = 0.204), total definite stent thrombosis (Χ2 = 9.424, P = .002, V = 0.217), and in-hospital MACEs (Χ2 = 6.74, P = .009, V = 0.184), respectively.
Nonsignificant difference in the rate of in-hospital gastrointestinal bleeding and hematuria (P = 1.0 and P = .316, respectively) between Ticagrelor and Clopidogrel groups.
