Abstract
Coarctation of aorta (CoA) is an important, treatable cause of heart failure in neonates and of secondary hypertension in adolescents and adults. Surgery has remained as the gold standard in neonates but catheter intervention has usurped it in children, adolescents, and adults. Introduction of covered stents has made the intervention safer. Since it usually appears to be a discrete lesion, at first glance, it attracts interventional cardiologists. However, CoA intervention is associated with significant immediate and long-term complications. Thus, it is imperative for the interventionist to understand the minutiae of the intervention, how to perform it correctly, and how to minimize complications. This review aims to provide complete description of intervention in neonates, adolescents, and adults in one place and also to guide interventional cardiologists perform safer intervention especially in subatretic and atretic CoA.
Coarctation of aorta (CoA) has been reported in 8% to 10% of congenital heart defects. 1 It may occur as an isolated lesion, but is commonly associated with other cardiac lesions—simple or complex. Bicuspid aortic valve is the most common association found in >50% CoA cases. When isolated, diagnosis is often missed in pediatric age group. Systemic hypertension is important reason for CoA intervention as well as being a surrogate marker of clinical outcome.
In 1945, Crafoord and Nyhlin 2 described successful surgical correction of CoA. Lock 3 documented the first balloon angioplasty in 1983. First stent was used by O’Laughlin et al 4 in 1993 and first covered stent in 1999 by Gunn et al. 5 Covered cheatham-platinum (CP) stent got the CE mark in 2003 and FDA approval in 2016.
CoA Anatomy for Interventionists
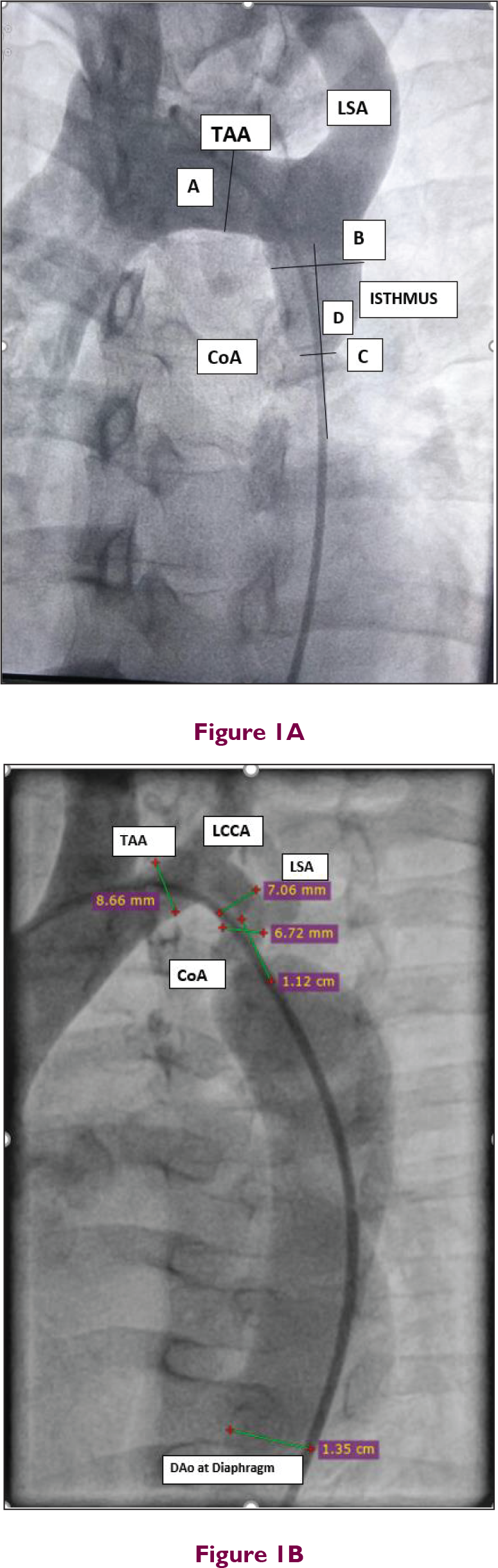
Most often, CoA segment is like a diaphragmatic shelf with indentation in the outer wall of aorta, opposite to insertion of ligamentum arteriosum. It may be very discrete (<5 mm long), or involve a longer tubular segment right from arch or left subclavian artery (LSA) causing tubular hypoplasia. Transverse aortic arch (TAA) is segment of arch between innominate artery and LSA. Isthmus is a portion of aorta distal to LSA. If TAA diameter is <60% of ascending aortic diameter, it is called hypoplastic. Isthmus is hypoplastic if its diameter is <40% of ascending aortic diameter. 6 Normally, aortic isthmus is 80% to 90% of TAA diameter and almost equal to that of aorta at diaphragm. Hence, this diameter is used for sizing stents.
For CoA intervention, diameter at the level of isthmus, diameter of TAA, narrowest segment diameter, length of CoA segment, relation to LSA, diameter at diaphragm, tortuosity of CoA segment, and nature of ascending aorta and aortic valve (bicuspid/tricuspid) are important to plan procedure (Figure 1).
Hardware: Balloon and Stents
Most of the CoA are dealt with peripheral balloons. These balloons are of lower profile. Care should be taken to avoid balloons more than 3 to 4 cm length. Of late, “Balloon-in-Balloon” (BIB; NuMED, NY, USA) balloon catheters are being used. It has an inner and an outer balloon. Outer balloon is 1-cm longer and twice the diameter of inner balloon. The BIB balloon is available in diameter of 12 to 30 mm and length of 2.5 to 5.5 cm and compatible with 8 to 11F introducer sheaths.
The CP Balloon Expandable Stent
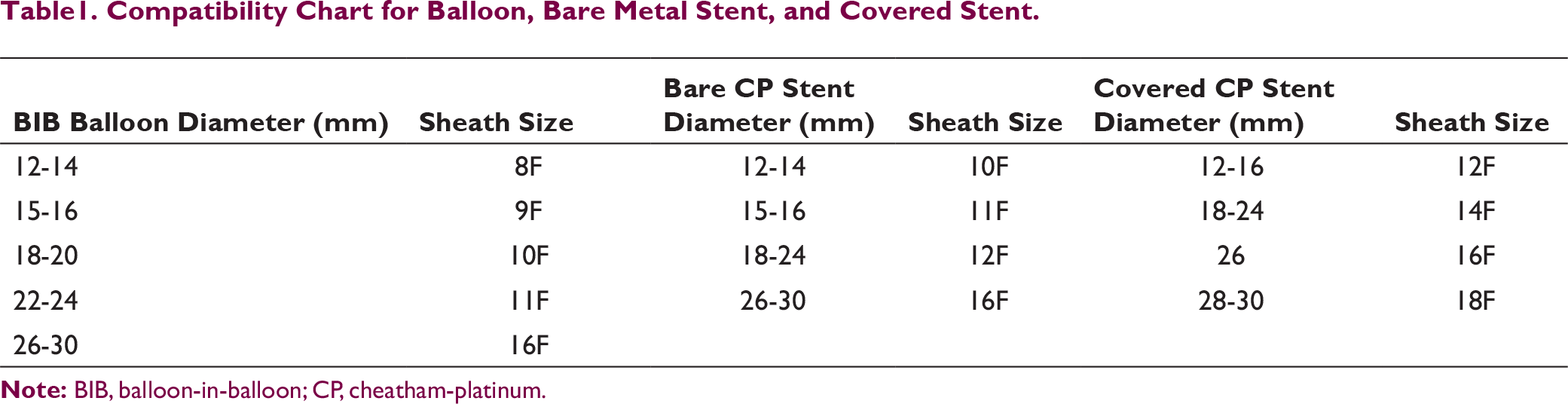
The CP stent is available as bare stent, covered stent, premounted CP stent over BIB balloon, and also as preloaded stent delivery system (NuDEL CP stent Delivery system; NuMED). CP stent is made of platinum-iridium wire arranged in 8 or 10 zig patterns. The covered CP stent is covered with ePTFE sleeve. Stents are available in 1.6 to 6.0 cm length and 12 to 30 mm diameter (Table 1). These are magnetic resonance imaging (MRI) compatible stents with high radial force and lesser foreshortening.
Recently, NuMED has introduced D’Vill 12 to 14F sheath and introducer. The entire covered CP stent with sheath is also available as NuDEL CP stent delivery system which eliminates need of separate sheath. It is available in 12 to 14F size preloaded with 14 to 24 mm diameter stents and usable length is 100 cm.
While using Palmaz stent, one has to be extremely careful when introducing this hand-crimped stent into the sheath as it has high chance of displacement. To avoid displacement, it is advisable to slightly inflate the balloon at <0.5 atmosphere so that the balloon beyond stent on both sides is a bit inflated and slightly larger than crimped stent diameter. While introducing in long sheath for delivery, it should be carefully monitored under fluoroscopy right from groin to detect dislodgement. Palmaz stent is not MRI compatible. In certain patients, self-expanding nitinol stents can be used.
Indications for Intervention for CoA in Adults and Adolescents
Significant native CoA or recoarctation is defined as mean systolic gradient ≥20 mmHg on Doppler, or mean systolic gradient ≥10 mmHg on Doppler Plus evidence of reduced left ventricular (LV) systolic function or aortic regurgitation (AR) or collaterals; or upper extremity and lower extremity systolic pressure gradient ≥10 mmHg on plethysmography Plus evidence of reduced LV systolic function or AR or collaterals.
Compatibility Chart for Balloon, Bare Metal Stent, and Covered Stent.
Class I
In a hypertensive patient with peak-to-peak gradient is ≥20 mmHg. Stenting is preferred over surgery when technically feasible.
Class IIa
In a hypertensive patient with peak-to-peak gradient is <20 mmHg, but aortic narrowing ≥50% relative to diameter of aorta at diaphragm, catheter treatment (stenting) should be considered when technically feasible. In a normotensive patient with peak-to-peak gradient ≥20 mmHg, catheter treatment (stenting) should be considered when technically feasible.
Class IIb
In a normotensive patient with peak-to-peak gradient <20 mmHg, but aortic narrowing ≥50% compared to diameter of aorta at diaphragm, catheter treatment (stenting) may be considered when technically feasible.
Procedure
Aortogram is performed in left anterior oblique 15-degree caudal 10-degree and left lateral angulations. An injector volume 35 ml with flow rate of 35 ml/sec is sufficient in most cases. Intravenous unfractionated heparin 100 U/Kg and antibiotics for endocarditis prophylaxis are given.
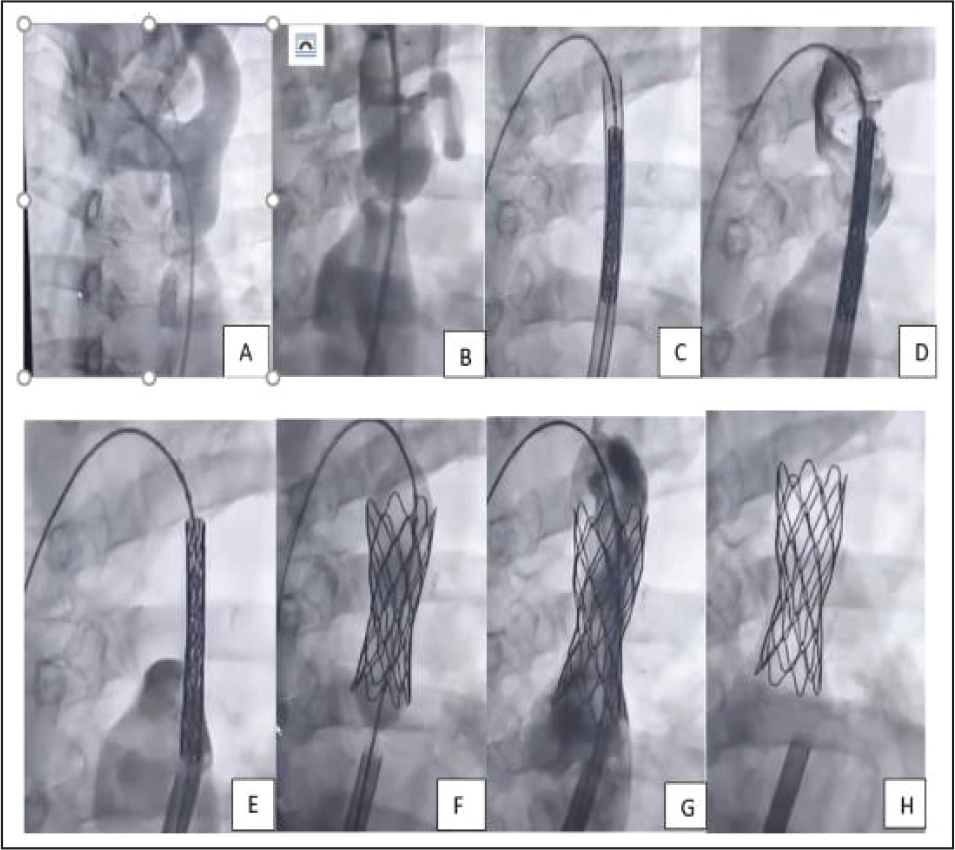
The standard technique involves crossing the lesion with 0.035-inch 260 cm long stiff Amplatz wire (AGA Medical Corp., MN, USA), placing compatible sheath across CoA segment, loading the stent, retracting the sheath, and deploying the stent at CoA site (Figure 2). Same sheath is used for injection during stent deployment. For CP stent, inner balloon is inflated first, and stent position is rechecked. If needed, stent can be repositioned and then it is followed by outer balloon inflation. It is recommended to reinflate outer balloon for optimal stent apposition. Further dilatation is usually not necessary, and it may be preferable to leave some waist if gradient disappeared. Distal stent will be hanging in dilated segment and it shouldn’t be post dilated (post dilatation on either side of waist is not recommended in order to avoid aortic wall injury). To remove balloon, gently advance sheath over the balloon and then withdraw the sheath. Excessive balloon, wire, or catheter manipulation should be avoided after stent deployment as such maneuvers can dislodge stent. After final aortogram, pigtail catheter should also be gently removed over 0.035-inch PTFE wire. In subatretic or atretic aorta, second arterial puncture can be obtained from radial artery. Rapid right ventricular pacing may be done at 180 to 220 beats per minute to improve precision of stent placement.
Routine predilataion is not recommended. However, it is required in atretic or subtretic CoA. This predilataion should be done with smaller balloons (up to 6-8 mm diameter) just to make room for smooth passage of long sheath. The prospective US protocol for CoA stenting (COAST) trial 8 included predilataion to assess compliance of CoA segment. It used a compliant balloon 2 mm smaller than intended stent diameter and inflated at low pressures (<4 atm). It was used as diagnostic procedure rather than angioplasty procedure. If the waist disappeared, it was stented. If waist remained, a lower size balloon was used and complete expansion of stent was delayed up to 6 months. Most of native CoA lesions are compliant, and wherever room is adequate for safe passage of delivery sheath, predilatation is not necessary.
Post procedure, it is important to monitor blood pressure and electrocardiogram of the patient. Aspirin 2 to 5 mg/kg/day is given for 6 months. Blood pressure medications are adjusted time to time. Some centers repeat CT angiogram after 24 hours to look for any dissection, aneurysm, or pseudoaneurysm formation. Routine CT angiogram is recommended 6 to 12 months after uncomplicated procedure and then after 3 years.
Native Coarctation in Neonates and Infants
Indications of CoA intervention in neonates and infants include hypertension, heart failure, and LV dysfunction secondary to CoA. Though surgical intervention is mainly preferred in this age group, balloon treatment is used in sick babies. Femoral arterial approach, femoral venous approach (if VSD is present or transposed great vessels) or umbilical approach may be considered based on expertise. In most of small babies, a transfemoral approach with 3 to 4F sheath and percutaneous transluminal coronary angioplasty (PTCA) hardware is sufficient. Lesion can easily be crossed with 0.014-inch PTCA wire and dilated sequentially with 3 to 6 mm noncompliant mono rail PTCA balloon. Balloon diameter should not be more than 1 to 2 mm of diameter at isthmus, or 50% of aortic diameter at the level of diaphragm, or 2 to 3 times the narrowed segment diameter and length should be less than 3 cm.9, 10 Balloon is gently inflated at 3 to 4 atmosphere or until waist disappears for 5 seconds, 2 to 3 times 5 minutes apart. If balloon waist doesn’t disappear at 1 to 2 atmosphere, don’t inflate it further, as it may lead to catastrophe. Instead, downsize the balloon and try. Result can be checked with pressure gradients. In case of need of higher balloons, 0.014-inch wire can be exchanged with 0.021- or 0.025-inch wire. Such exchanges should be done first by passing a 4F multipurpose catheter over 0.014-inch wire across CoA segment, remove 0.014-inch wire, and replace it with 0.021 or another desired wire. Temptation to cross CoA segment directly with another wire should be avoided. Excessive use of contrast and radiation should be avoided. Currently, stent use is not recommended in this age group.
Native Coarctation in Adolescents
After crossing the CoA segment, 260 cm Amplatz 0.035 in stiff wire is placed in right subclavian artery or LSA according to anatomical convenience. CoA segment is dilated sequentially with peripheral balloons, starting with lower profile. If initial CoA segment diameter is less than 3 mm, it is better not to aim for optimal result in first sitting. In such cases, it is better to restrict to maximum diameter of balloon to approximately 6 to 8 mm, that is, 50% to 60% of aortic diameter at diaphragm. Redilatation can be done after 3 months with higher balloon. It’s better to avoid elective stenting in this age group. In case of bailout, stents can be used which can be redilated when patient has grown up.
Covered stents are indicated in (provided the weight of patient is more than 20 kg) subatretic or totally atretic CoA, CoA in association of PDA, Turner Syndrome, as a “bail-out” procedure during plain balloon angioplasty (rupture, failed procedure), or recurrent CoA in adolescents.
Native Coarctation in Adults
Procedure has been described in section 4.0. Any bare metal stent, covered stent, or self-expanding stent may be sufficient in most of the cases based on operator experience and comfort. Aggressive pre or post dilation should be avoided.11, 12 Multiple injector injections can increase aortic trauma. If needed, stent can be redilated after 3 months. At every step, gradient across CoA segment should be documented carefully. A successful procedure is defined as peak-to-peak systolic gradient less than 10 mmHg.
In subatretic CoA (narrowest point diameter 1-3 mm), it’s better to span the entire procedure in 2 to 3 settings over a gap of 6 months. Start with 4 to 6 mm mono rail balloon first to get adequate room for stent placement. A covered stent is preferred in this group. The diameter of stent is approximately equal to aortic diameter at isthmus or diaphragm. In case of bare metal stents in such situation (not a desired strategy, but financial compulsion), it’s better to dilate it to lower diameter in first sitting (Figure 3).
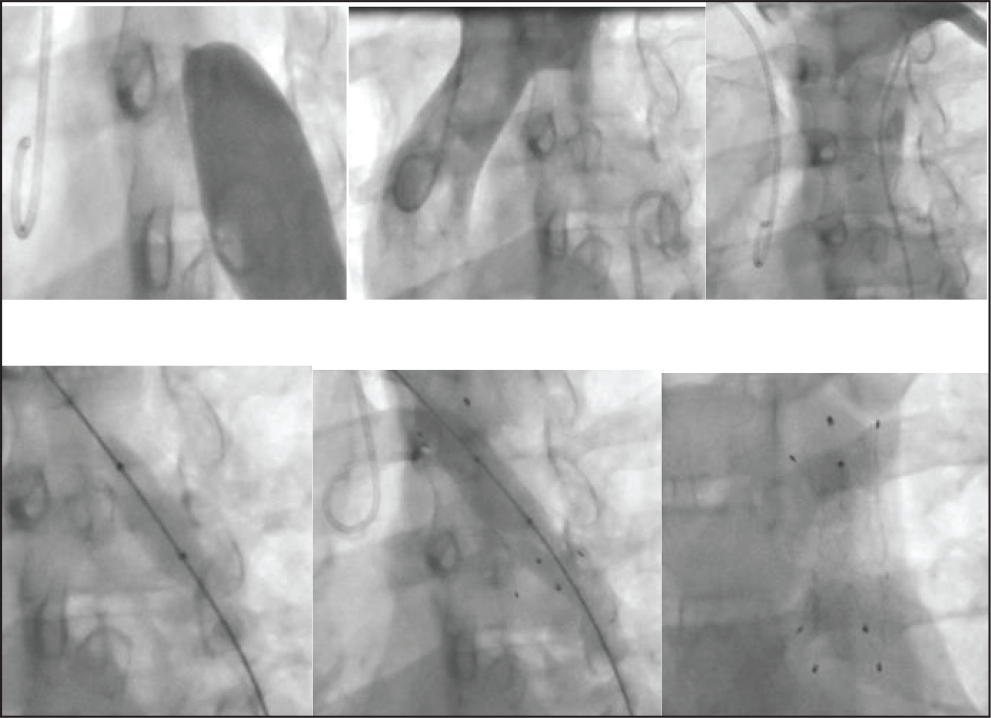
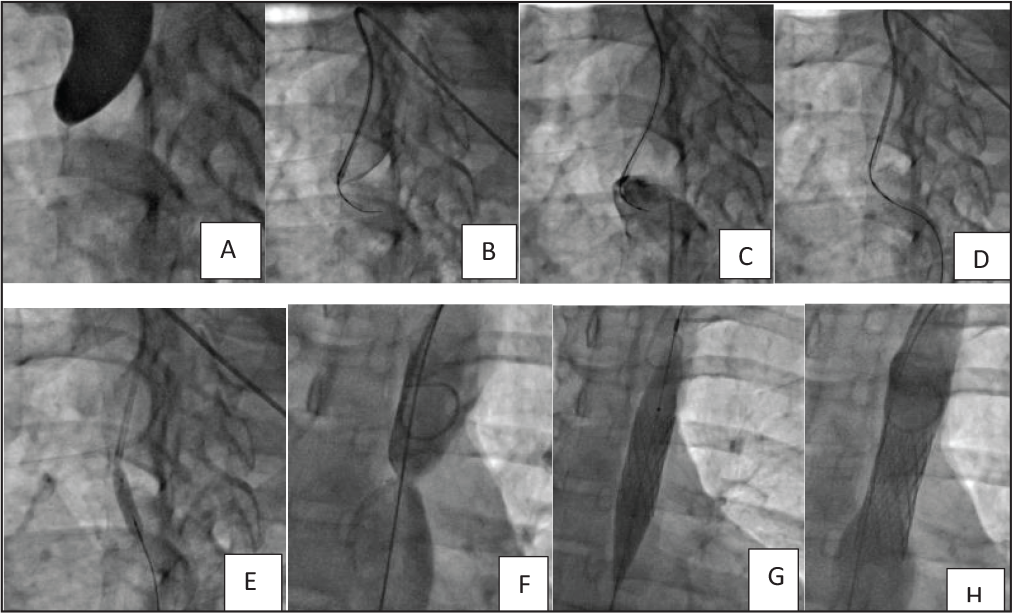
In case of totally occluded CoA (atretic CoA), access left radial artery and right femoral artery. To puncture femoral artery if arterial pulse is not well palpable, wire femoral vein first. Then femoral artery can be easily punctured lateral to it. A 6F Mullin’s dilator with straight tip Terumo wire can be used to cross the occluded segment. Some operators have successfully used hard end of PTCA wire to cross such occlusions. If this approach fails, antegrade approach with 6F right coronary Judkins guide near the blind pouch can be tried (Figure 4). Once crossed, wire is snared through femoral sheath and ballooning done from femoral side using 2, 4, and 6 mm mono rail balloons sequentially and then exchange 0.014 wire with 0.035-inch 260 cm Amplatz super stiff wire using a catheter. Then use over the wire 8 mm peripheral balloon for further dilatation. After checking the result, long sheath is advanced and, in case of bare metal stent, stent is implanted over a 12-mm balloon and deployed. Further dilatation is done after 3 months. In case of self-expanding stents, dilate up to 8 to 12 mm followed by stenting and redilate after 3 months. Covered stents are ideal choice for atretic CoA. Covered stents can be dilated to higher diameter in first sitting itself or can be redilated later. In case LSA is jailed by a covered stent, it can be perforated and dilated to make adequate room. Whenever this complication is anticipated, one should do vertebral artery angiogram on both sides and make sure that right vertebral artery is not hypoplastic.
Immediate and Long-Term Results of Transcatheter Repair
Neonates and Infants
In a large center experience of 181 neonates and children who underwent surgical repair between 1986 and 2002, Wood et al 13 reported 30-day mortality of 0.5% and recoarctation rates of 2.2%. Of 181 patients, 135 were neonates and 107 had hypoplastic arch.
Percutaneous angioplasty was done by P S Rao et al10, 14 to control heart failure or hypertension in 51 infants and neonates less than 3 months of age. Additional surgery was required to achieve optimal gradient in 7.8% neonates. Angioplasty was successful in 92.2%. Recoarctation was noted in approximately 50%.
Children and Adolescents
In an observational study by Congenital Cardiovascular Interventional Study Consortium (CCISC) 2011, Forbes et al 15 reported good immediate and intermediate follow-up results in 61 children weighing ≥10 kg with mean age 9.0 ± 8.0 year from 36 institutions. Procedural complications occurred in 9.8%. During short-term follow-up, recoarctation was noted in 32.1%. In the subgroup analysis of children between 6 and 12 years of age who underwent surgery, Balloon angioplasty or stents, stent strategy was superior to surgery or balloon strategy. Immediate complication rates were 13.0% in surgical arm, 13.0% in balloon arm, and 1.8% in stent arm. However, 16.1% in stent group required reintervention, mainly redilatation.
Adults
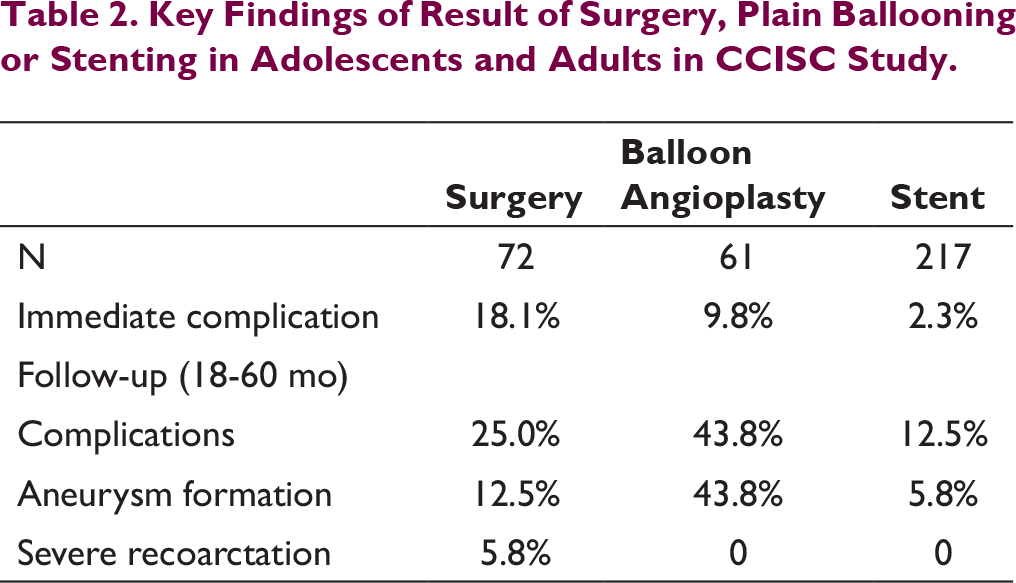
Table 2. Key Findings of Result of Surgery, Plain Ballooning or Stenting in Adolescents and Adults in CCISC Study.
In the series reported by Forbes et al,16, 17 out of 565 patients, initial stent was a bare metal stent in 93.3% cases and covered stent in 6.7% cases. Prestent ballooning was done in 17.6% cases, with median balloon to CoA segment diameter ratio of 2.0. Acute procedural success was 97.3%. Acute complication noted in 14.3%. Death was reported in 0.5% and aortic wall complication 4% patients. Technical complication and risk of aortic dissection was higher in age above 40 years. Stent migration noted in 5% patients. Most of stent migrations occurred when delivery balloon diameter was ≥15 mm. Thirteen of 28 stents were repositioned successfully, another 8 in suboptimal location requiring another stent in CoA segment.
Theoretically, self-expanding stents offer gentle radial force, less trauma, less bulky, and conform more to aortic wall as compared to bare metal stents. 12 These can be used in hypoplastic arch across LSA without distorting aortic arch. Tyagi et al 18 compared balloon expandible stent (N = 5) with self-expanding stent (N = 16) implantation in 21 adults ranging 18 to 61 years of age. One stent embolization occurred in balloon-mounted stent group and none in self-expanding nitinol stent group. Over 12 to 71 months follow-up, there was no stent fracture, migration, or aneurysm formation in self-expanding stent group. Ali-Mohammad Haji-Zeinali et al 19 shared experience of self-expanding stents in 21 patients. Cephalad stent displacement occurred in 3/12 first generation stents but none in second or third generation stents with antijump markers. None had aortic wall-related complications.
Covered stents have taken over bare metal stents now. COAST 8 evaluated covered CP stent in children and adults with native CoA or recoarctation. Covered CP stent was implanted in 105 patients. Acute aortic wall injury occurred in 1 patient and 4 had evidence of aortic aneurysm after 1 year follow-up. These patients underwent another CP stent implantation. Stent radiation was done in 13% patients. At the time of implantation, there were no stent fracture, but at 1 year 2% patients and at 2 year 12.2% patients had stent fracture to some extent. However, none of these required further interventions. COAST II trial 20 further evaluated covered CP stent in 158 patients with aortic wall injury due to previous procedure, nearly atretic CoA, genetic syndromes like Marfan syndrome, Turner syndrome, bicuspid aortic valve or ascending aortic aneurysms, or CoA in patients >60 years of age. Complete coverage of aortic wall injury occurred in 92% patients. Seven patients had minor endoleaks which didn’t require further intervention.
Sohrabi et al 21 randomized 120 patients age group 12 years to 58 years with short segment post ductal severe native CoA treated with covered CP stent (N = 60) with bare CP stent (N = 60). Success rate was 100% in both groups. Recoarctation was noted in 6.7% in bare CP stent group but none in covered CP stent group. Pseudoaneurysm was noted in 3.3% of covered CP stent group but none in bare CP stent group. Blood pressure normalized in 73.3% of bare CP stent and 78.1% of covered CP stent group.
Features associated with suboptimal result includes hypoplastic TAA or isthmus, tortuous CoA segment, tubular hypoplasia, and post procedural residual gradient >10 mmHg. Rupture or aneurysm formation is mainly related to use of oversized balloons and aggressive ballooning. Stent migration is related to balloon diameter >15 mm, post dilation with oversized balloons. Stent fracture is mainly associated with balloon-mounted stents.
Long-Term Results
There are limited data on long-term follow-up of stent procedures. Butera et al 22 reported 15-year single center experience with bare metal and covered stents. Over a median follow-up of 81.7 months, 23.9% patients in bare metal stent group and 4.5% patients in covered stent group required reintervention. Reintervention in bare metal stent group was mainly for restenosis whereas covered stent group had reintervention due to late aneurysm formation. Predictors for reintervention were small initial stent balloon <14 mm, residual gradient >10 mmHg, and complex lesions. Stent fracture was more common in bare metal stent group (5%) and was important cause for reintervention. Somatic growth with age less than 15 years was associated with reintervention. Patients should be followed closely for blood pressure, stent fracture, and aneurysm formation on long term. It’s advisable to repeat CT angiogram at 6 month, 1 year, and then after 3 years post intervention.
Newer Devices
Biodegradable scaffolds, growth stents, and dilatable stents are future concepts for treatment of neonatal CoA. There are case reports of use of biodegradable scaffolds in treatment of neonatal CoA, but long-term follow-up is not available.
Conclusion
Surgery remains preferred modality in neonates, balloon with bailout stenting in children, and primary stenting remains the preferred modality of CoA treatment in adults. Percutaneous intervention for CoA needs patience and experience in order to avoid catastrophic complications. Covered CP stent remains first preference in almost all CoA in adolescents and adults. The correct picture of long-term outcome is missing; hence, it’s better to follow the intervention patient meticulously with echocardiography, fluoroscopy, and CT angiogram whenever required.
Footnotes
Declaration of Conflicting Interests
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
