Abstract
Acute type A aortic dissection is a catastrophic disease that develops from a tear within the intima of the aortic wall, thereby creating a false lumen in the ascending aorta. Early suspicion, diagnosis, and prompt surgery play a key role in the survival of patients. It is a surgical emergency and requires replacement of the ascending aorta/aortic root with or without aortic arch replacement. Over the past decade the surgical outcomes have improved in specialized tertiary centers.
Introduction
Acute type A aortic dissection is a lethal disease with a reported incidence of 2.5 to 6 per 100 000 patient years. 1 The first reported aortic dissection and the concept of true and false lumen is attributed to Shekelton in the early 1800. If left untreated, a patient with acute type A aortic dissection has a 50% to 70% risk of dying within 48 h of the event. 2 For this reason, it is essential to operate immediately. Untreated patients usually die of rupture of false lumen, acute cardiac tamponade, acute high-grade aortic valve insufficiency, and malperfusion syndromes. Despite surgery, the overall 30-day mortality remains around 20% to 25% as reported by international registry of aortic dissection. 3 The international registry of acute aortic dissection (IRAD) is a multinational registry that initiated enrolling of patients in 1996. This project has provided contemporary insight into the short-term and long-term outcomes of acute aortic dissection and proposed therapeutic options.
The ideal surgery for acute type A aortic dissection should replace the diseased aortic segment as long as possible and obliterate the false lumen in the remaining aorta with minimal operative risks and reduction in the need for reintervention. Over the last decade, improved understanding of the dynamics of aortic dissection has led to lower mortality and morbidity in selected centers.
Pathogenesis and Classification
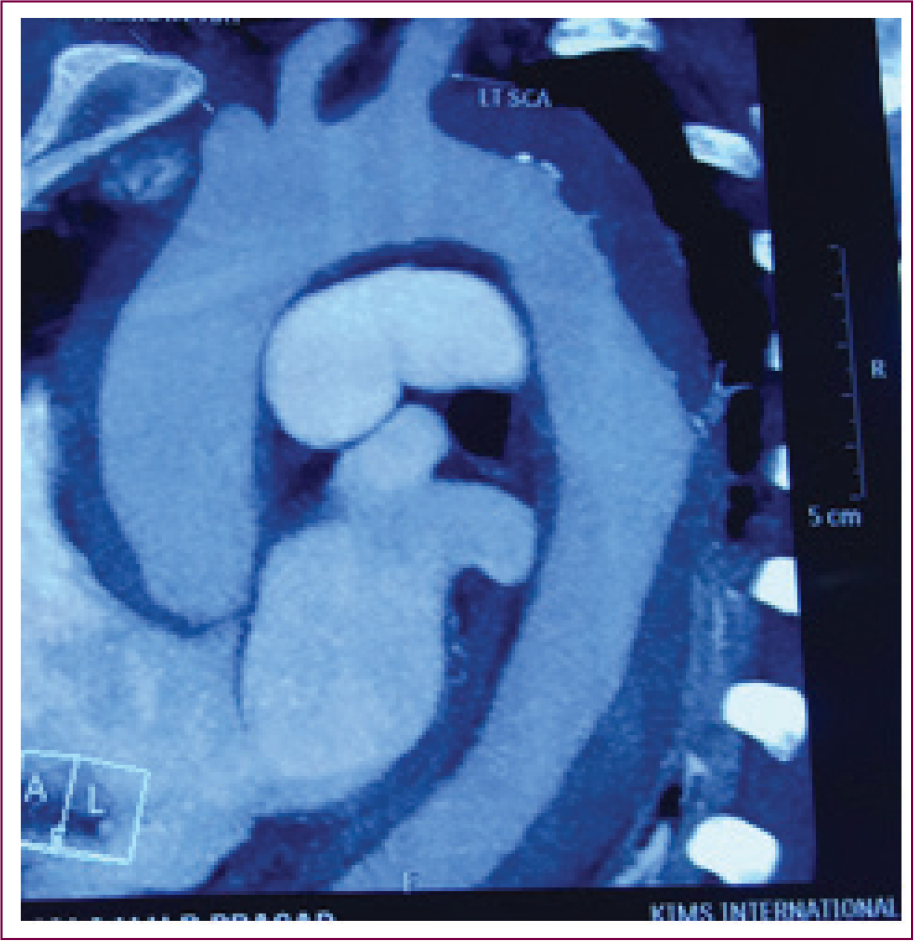
Acute type A aortic dissection develops from a tear within the intima of the aortic wall. Blood flows across the entry point into the weakened media, splitting the medial layer along the direction of blood flow, creating a new false lumen within the aortic media. Subsequently the false lumen can extend in both directions and could affect most of the branches of aorta including coronary, carotid, mesenteric, and limb arteries. Acute type A aortic dissection can also happen secondary to trauma, most often iatrogenic—during percutaneous interventions, aortic cannulation for cardiopulmonary bypass (CPB), cross-clamping of aorta in open heart surgery, endovascular interventions like transcatheter aortic valve implantation (TAVI), endovascular aneurysm repair (EVAR), or insertion of intra-aortic balloon pump (IABP). Intramural hematoma involving ascending aorta also falls into the category of type A aortic dissection.
Risk Factors
Hypertension is present in nearly 75% of individuals with aortic dissection. Other risk factors include pre-existing ascending aortic aneurysms, 4 smoking, Marfan’s syndrome, hereditary TAA/D, Ehlers-Danlos syndrome, Turner’s syndrome, penetrating atherosclerotic ulcer, giant cell arteritis, Takayasu arteritis, Bechet’s disease, aortitis, cocaine use, and pregnancy. Genetically triggered disorders are important and often unrecognized causes of aortic dissection.
The first classification scheme of aortic dissection was designed by DeBakey, who separated dissections into the following types:
Type I: The dissection involves the ascending aorta, aortic arch, descending aorta, and often abdominal aorta.
Type II: The dissection involves ascending aorta but stops at the level of aortic arch and does not involve the aorta beyond the left subclavian artery take off.
Type III: The dissection starts distal to the left subclavian artery and extends to the entire descending thoracic aorta (IIIa) or the descending thoracic and abdominal aorta (IIIb).
The following Stanford classification has become popular, primarily because of its therapeutic implications.
Type A aortic dissection: Dissection involves ascending aorta and will most often extend into the arch, descending thoracic as well as abdominal aorta.
Pictorial Depiction of Aortic Dissection.
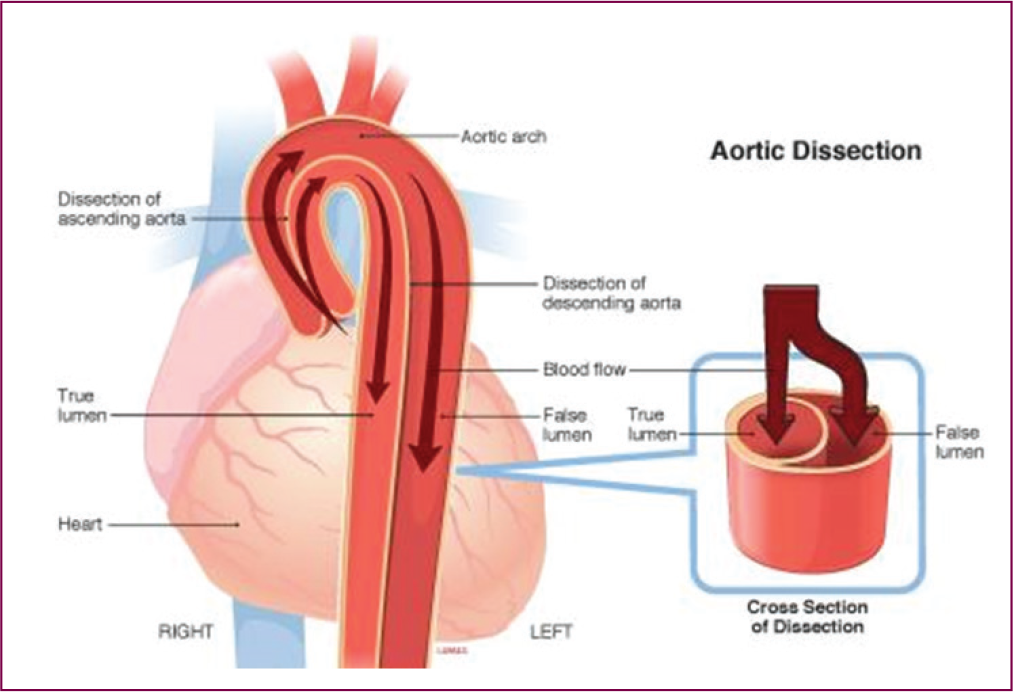
Classification of Aortic Dissection.
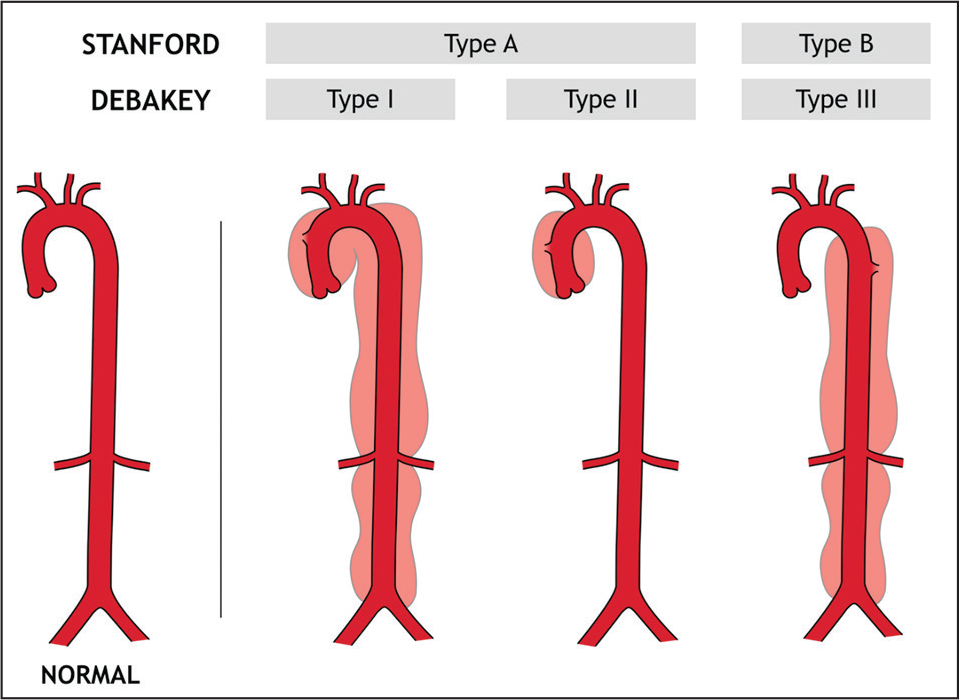
Type B aortic dissection: Dissection starts beyond aortic arch usually at the subclavian artery, progresses distally thus not involving ascending aorta or the arch.
Dissection based on timing of presentation 5 :
Acute aortic dissection: Patient presenting within 2 weeks from the onset of pain.
Subacute aortic dissection: Patient presenting between 2 and 6 weeks of onset of pain.
Chronic aortic dissection: Patient presenting after 6 weeks from the onset of pain.
Epidemiology
Acute type A aortic dissection constitute about 60% of all aortic dissections. About 49% of these patients die before they reach the hospital. The incidence of acute type A aortic dissection has been reported to be 2.5 to 6 cases per 100 000 patient years. The increasing life expectancy of population is probably contributing to the higher incidence. Men are affected about 4 times more commonly than women.
Clinical Presentation
The aortic media has high density of nerve endings, 6 so when medial disruption takes place the acute onset of sharp pain is a uniform presenting symptom. The pain is classically described as acute ripping, tearing, or knife-like chest pain, which then migrates to the upper back and often progresses down to the lower back depending on the dynamics of growth of false lumen. Once the false lumen is fully established and has stabilized, the acute pain may change into a more persistent and dull pain, which is often not so easy to localize and may involve chest and back in variable patterns with additional elements of nausea, abdominal pain, diaphoresis, and shortness of breath.
Cardiac symptoms are usually secondary to acute aortic regurgitation, myocardial ischemia, or cardiac tamponade. About 50% to 80% of patients with acute type A aortic dissection develop moderate to severe aortic regurgitation, due to prolapse of the intimal layer at the level of one or more commissures. In about 10% to 15% of the patients the false lumen can compromise coronary ostium (usually RCA), causing myocardial ischemia or infarct. These patients can present with heart failure or cardiogenic shock.
Nearly 10% to 40% of the patients with acute type A aortic dissection can have neurological symptoms, due to malperfusion of arch vessels/thromboembolism or hypotension. Neurological symptoms can be transient in about 50% of these patients.
Mesenteric ischemia secondary to gut vessel malperfusion can occur in about 5% of the patients and the reported mortality in these patients is as high as 70% to 80%. Flank pain can also be present, especially in patients who are suffering from an acute malperfusion of one of the kidneys. Additional presenting symptom can be sudden onset of a cold and pulseless extremity.
Medical Management
During the initial assessment, it is imperative to control pain and blood pressure in these patients to prevent aortic rupture. To achieve this, morphine and intravenous B-blockers (Labetalol, Esmolol) are administered. The target systolic blood pressure should be less than 120 mmHg and heart rate less than 60 beats/min. Intravenous B-blockers are favorable to reduce the force of left ventricular ejection (dP/dt), which will otherwise continue to weaken the aortic wall. Combined use of intravenous B-blockers and sodium nitroprusside might be required in some patients with severe hypertension. Patient with significant hemodynamic instability might need intubation and ventilation.
Diagnosis
A high degree of clinical suspicion leads to timely diagnosis of acute type A aortic dissection, which in turn leads to successful treatment. Screening test should include ECG and chest X-ray. ECG is often nondiagnostic and occasionally may show inferior wall changes suggestive of right coronary artery compromise by dissection. Chest X-ray may show a dilated mediastinum or a large cardiac silhouette possibly due to acute pericardial effusion. The next screening test should be a transthoracic echocardiogram. It might show a variable degree of aortic valve insufficiency with a dilated root or presence of intimal flap along with pericardial effusion if any.
Computed tomography (CT) aortogram, transesophageal echocardiogram (TEE), and magnetic resonance imaging (MRI) are highly accurate in the diagnosis of aortic dissection. The selection of specific imaging modality is influenced by individual patient characteristics, variabilities, institutional capabilities, and expertise.
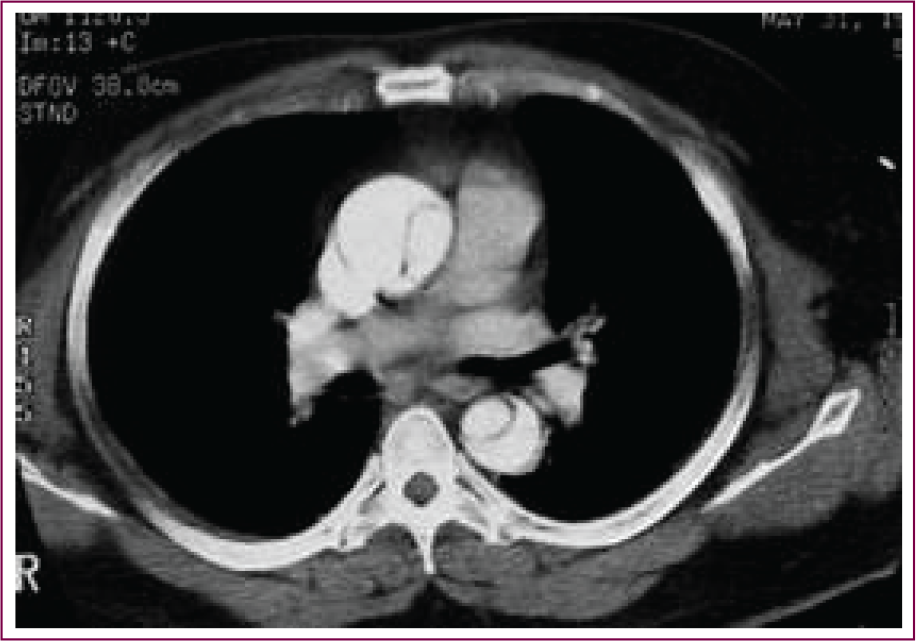
The best diagnostic modality for confirmation of an aortic dissection is CT aortogram. It has high sensitivity and specificity. 7 It will reveal the extent of dissection, true and false lumen, and the involvement of branch or arch vessels. Usually the false lumen is greater than the true lumen. CT aortogram also aids in decision making regarding the arterial cannulation site and surgical planning.
CT Aortogram Showing Type A Aortic Dissection.
CT Aortogram Showing Intramural Hematoma.
TEE has an advantage of being portable and can be performed at the bedside in an unstable patient. TEE is helpful in evaluation of aortic valve leaflets, pericardial effusion, and detection of intimal flap in the aortic root. TEE is usually performed as an intraoperative test after intubation for obtaining accurate and detailed information.
Because MRI imaging takes longer for image acquisition and leaves the patient relatively unmonitored, it is usually not the procedure of choice.
A question usually arises over the value of coronary artery evaluation after making the diagnosis of type A aortic dissection. Retrospective reviews suggest that the incidence of coronary artery disease in this population is sufficiently low. More patients are placed at risk by the delay and technical risks of cardiac catheterization. CT coronary angiogram or intraoperative coronary angiogram is an option in these patients.
It is important to understand malperfusion, before we proceed into further discussion about aortic dissection.
Malperfusion
Malperfusion means compromised blood flow to the organs resulting in organ ischemia. It is secondary to increased pressure in the false lumen which compresses and jeopardizes flow in the true lumen and its branch vessels. Branch vessel compromise can often be seen on CT aortogram, but it is the presence or absence of end organ ischemia that decides the prognosis.
Malperfusion occurs in the following scenarios:
Presurgery malperfusion: Malperfusion phenomenon might already be existing prior to surgery in the form of pulse deficit (carotid or peripheral) or organ ischemia.
Malperfusion on institution of cardiopulmonary bypass: Peripheral arterial cannulation at any site like common femoral artery or axillary artery can precipitate or worsen malperfusion on initiation of CPB. The retrograde arterial flow towards the heart, even when cannula lies within the true lumen, may lead to differential false lumen dominance via primary or secondary tear and cause cerebral, cardiac or other organ malperfusion.
Malperfusion during aortic cross-clamping: Malperfusion can also occur during cross-clamping of aorta. The existing communication between true and false lumen can be obstructed by the cross-clamp, resulting in increased pressure in the false lumen and can cause cerebral malperfusion or rupture of aorta.
Surgical Technique
There are 5 primary causes of death in acute type A aortic dissection. They are (1) aortic rupture, (2) congestive heart failure due to acute aortic valve insufficiency, (3) acute myocardial infarction secondary to malperfusion of coronary arteries, (4) stroke resulting from malperfusion of aortic arch vessels, and (5) mesenteric or other organ ischemia.
A successful operation for acute type A aortic dissection will leave the patient with a reconstructed or replaced aortic root, a well-functioning aortic valve, a completely replaced ascending aorta, a partially or completely replaced aortic arch with true lumen patency in the arch vessels and a distal type B aortic dissection in the residual aorta.
Once the patient has confirmed diagnosis of acute type A aortic dissection, an expeditious surgical repair may be done after arranging for blood and blood products. Patient is anesthetized under endotracheal general anesthesia. Blood pressure monitoring consists of bilateral radial artery catheterization or right radial artery and a femoral artery catheterization. The most common vessel to suffer malperfusion is the innominate artery. Therefore, the right radial artery would provide early warning of the most likely malperfusion syndrome affecting the brain. Left radial artery or femoral artery catheter monitoring will give further information about the presence of downstream and less common arch malperfusions. A wide bore peripheral cannula and a central line are inserted. Some centers would prefer to insert PA catheter as a routine in these patients.
Patient’s chest, abdomen, and both lower extremities are prepped and draped. It is advisable to perform peripheral arterial cannulation for CPB before performing the sternotomy.
Because of the size and ease of access, common femoral artery is often used for arterial cannulation to establish cardiopulmonary bypass. But, retrograde perfusion through femoral artery can cause pressurization of false lumen and can compromise the true lumen flow resulting in malperfusion. Over the last decade, right axillary artery is being used more often. It is an excellent alternative site for peripheral arterial cannulation. 8 Usually an 8 mm Dacron graft is sown to the right axillary artery, which in turn is connected to the arterial circuit of CPB. It facilitates anterograde flow and has a decreased probability of creating cerebral malperfusion. Other alternative arterial cannulation sites include carotid artery, innominate artery, direct aortic cannulation (with the help of epiaortic scanning and Seldinger technique) and trans LV apical-aortic cannulation. However, cannulation at any of the aforementioned sites can still create malperfusion on establishment of CPB. Thus, it is very important to be extra vigilant during the institution of CPB. Detection of malperfusion at this time should prompt the surgeon to change the arterial cannulation site immediately to ensure true luminal flow.
Intraoperatively blood gas analysis, lactate measurement, and mixed venous saturation monitoring from CPB circuit will give an indication of a global organ perfusion. Evidence of ongoing profound lactic acidosis will alert the possibility of malperfusion phenomenon either related to limb or visceral ischemia.
Once the sternotomy is performed, the pericardium is opened. It is common to find a variable amount of blood tinged pericardial fluid or even moderate amount of blood under tension. Often the ascending aorta will be moderately dilated because aneurysm is a major risk factor leading to ascending aortic dissection. 4 We find the most common area of intimal tear is at mid ascending aortic level. Most often the blood enters the loose areolar tissue between the main pulmonary artery and the ascending aorta, sometimes dissecting down onto right ventricular outflow tract (as shown in the picture).
Type A Aortic Dissection—Appearance of Ascending Aorta.
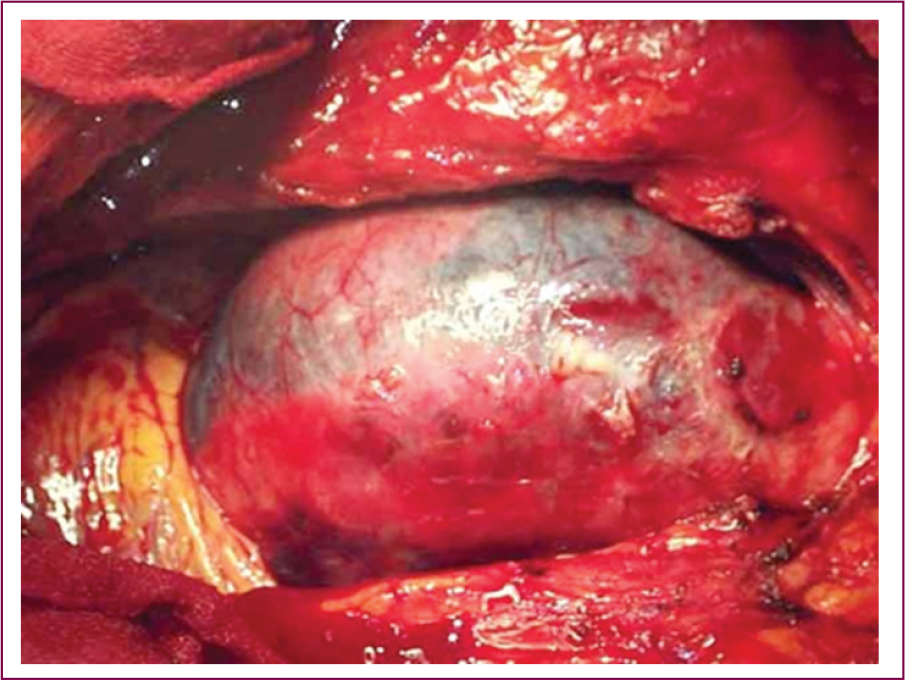
Type A Aortic Dissection Showing Clot Around RVOT.
It is important early in the process not to disturb this dissected tissue because it is easy to convert dissection process into free rupture. With arterial access obtained peripherally, right atrium is cannulated in routine manner. Patient is placed on full CPB. During institution of bypass, very close monitoring of arterial wave form, from both radial arteries, is used to detect any malperfusion syndrome. In several patients, significant aortic valve insufficiency will be present hence the left atrium and left ventricle are vented through right superior pulmonary vein. Patient is subjected for cooling with a goal of achieving nasopharyngeal temperature of 16°C.
Temperature monitoring: Organ protection constitutes an important aspect while performing surgery for acute type A aortic dissection. Core cooling of the patient up to 16°C is necessary to reduce the metabolic activity of all the organs including brain during total circulatory arrest. Patients temperature can be monitored from various sites such as nasopharyngeal, jugular bulb, esophageal, rectal, arterial inflow, urinary bladder, skin, and tympanic membrane. Most of these sites do not reflect true brain temperature. Out of all these sites, jugular bulb temperature correlates best with cerebral temperature, but it is very invasive and not routinely used. In a study published by Kaukuntla et al, 9 the authors concluded that nasopharyngeal temperature monitoring as an easy and safe method for measuring brain temperature.
Cerebral blood flow and metabolism: The incidence of neurological compromise happening during the surgical procedure is quoted between 5% and 20% and is associated with poor early and midterm outcomes.
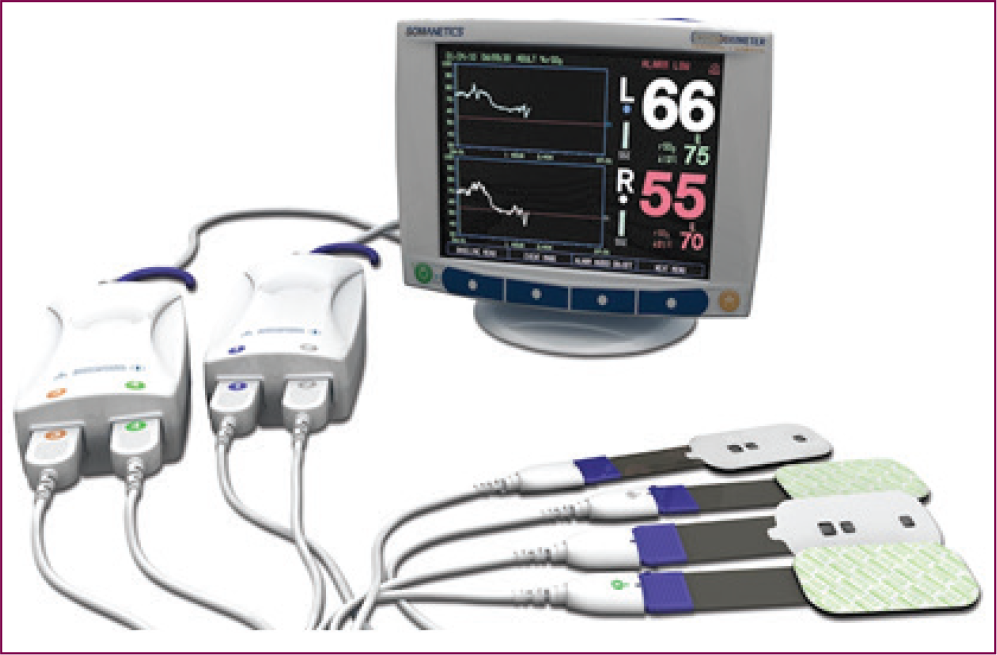
To avoid cerebral hypoxia during surgery, it is essential to monitor cerebral metabolism and blood flow. A number of gadgets are available for this purpose. These include transcranial doppler, EEG, near-infrared spectroscopy (NIRS), jugular venous oxygen saturation. We routinely use NIRS as a cerebral monitoring device in all these patients. NIRS is a simple and noninvasive way to monitor rCSo2—regional cerebral tissue oxygen saturation. The mean cerebral tissue oxygen saturation is around 60% to 70%. Any drop in the rCSo2 of more than 20% from the baseline during surgery can be associated with ischemic neurological injury. 10
Near-Infrared Spectroscopy With Adhesive Patches (NIRS).
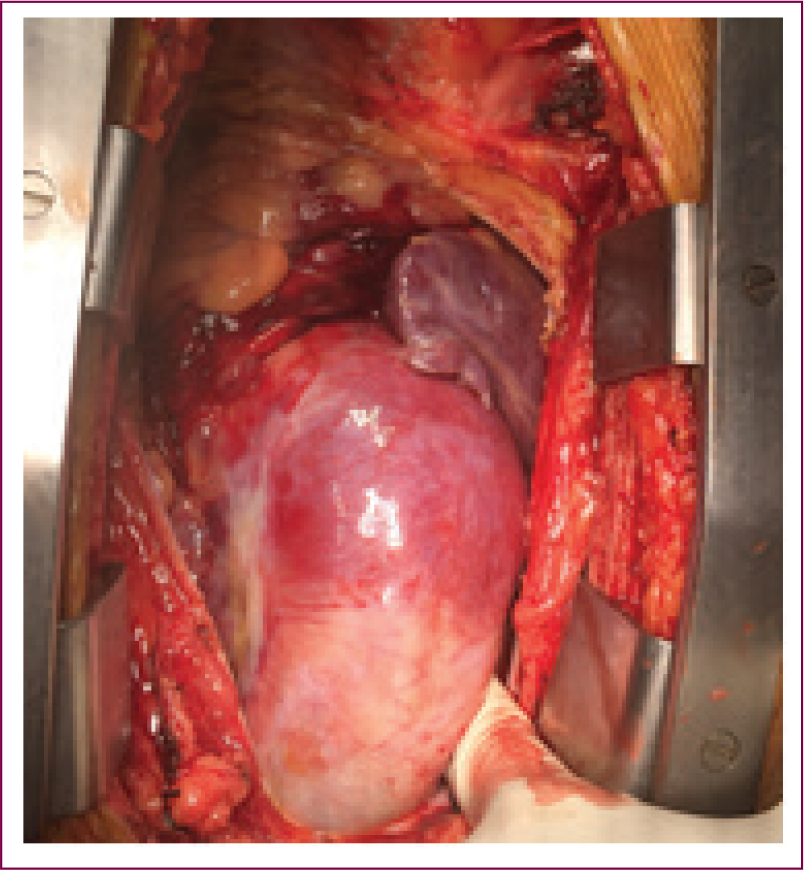
Transected Ascending Aorta Showing True and False Lumen.
Ascending Aorta Showing Intramural Hematoma.
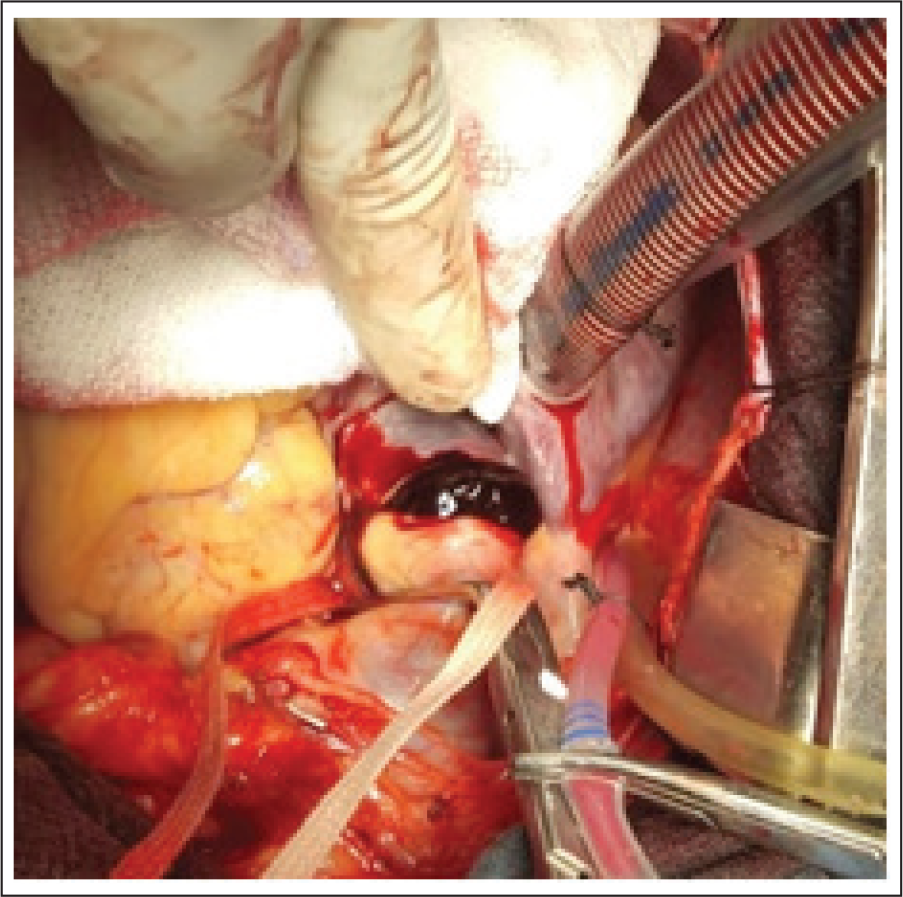
As the core cooling progresses, patient’s heart will fibrillate. Aorta is then cross-clamped at mid-ascending aortic level. Although in the past there has been reluctance to cross-clamp dissected ascending aorta, over the past decade no aortic disruption has resulted from cross-clamping, even in patients with connective tissue disorders. Clearly the cross-clamp should be placed well below the innominate artery take off. After the ascending aorta is cross-clamped without malperfusion, it is divided at the level of right pulmonary artery and cold blood cardioplegia is given via direct coronary osteal cannulation.
The generally accepted repair of Acute type A Aortic dissection involves:
Ascending aortic replacement with aortic valve resuspension (for correction of aortic valve regurgitation) or Aortic root replacement (where in the ascending aorta and aortic valve are excised and replaced with a valve conduit along with reimplantation of coronary buttons). With or without arch replacement depending upon the presence or absence of intimal tears in the arch of aorta.
The distal anastomosis is usually performed in an open manner after the cross-clamp is removed under hypothermic circulatory arrest (HCA). However, HCA allows only limited time for distal aortic anastomosis. In order to extend the potential safety duration of circulatory arrest, adjunctive techniques have been developed. For a long time, retrograde cerebral perfusion (RCP) through superior vena cava was used as an adjunctive technique during HCA for improved cerebral protection. The advantages of RCP are cerebral perfusion, cooling of brain and washing out of metabolites and embolic debris. Disadvantages in this technique includes increased risk of cerebral edema and inadequate neuroprotection.
The adjunctive technique most often used now a days is selective antegrade cerebral perfusion. In this method, we selectively cannulate and perfuse one or both carotid arteries antegradely. 11
Once the cardioplegia is given, the aortic root is evaluated to see if it is repairable. The prerequisites for aortic root repair are (1) there should not be any intimal tear in the aortic root, (2) aortic leaflets should be normal, and (3) aortic root should not be dilated. Mild dilatation of sinotubular junction up to 30 mm in maximum diameter can be tolerated with an adequate repair. If the above requirements are met with, the aortic leaflets are resuspended with commissural sutures and the entire ascending aorta is replaced with a Dacron graft.
If the aortic root is severely aneurysmal or if there are intimal tears in the root or if the aortic leaflets are damaged, then usually aortic root replacement is performed. A mechanical valved Dacron conduit is a good option in most of the patients who are less than 60 years old. A biological valved conduit should be considered in patients more than 60 years old. Although valve sparing root operation is an option, it should be reserved for patients less than 50 years old and with normal leaflets. 12
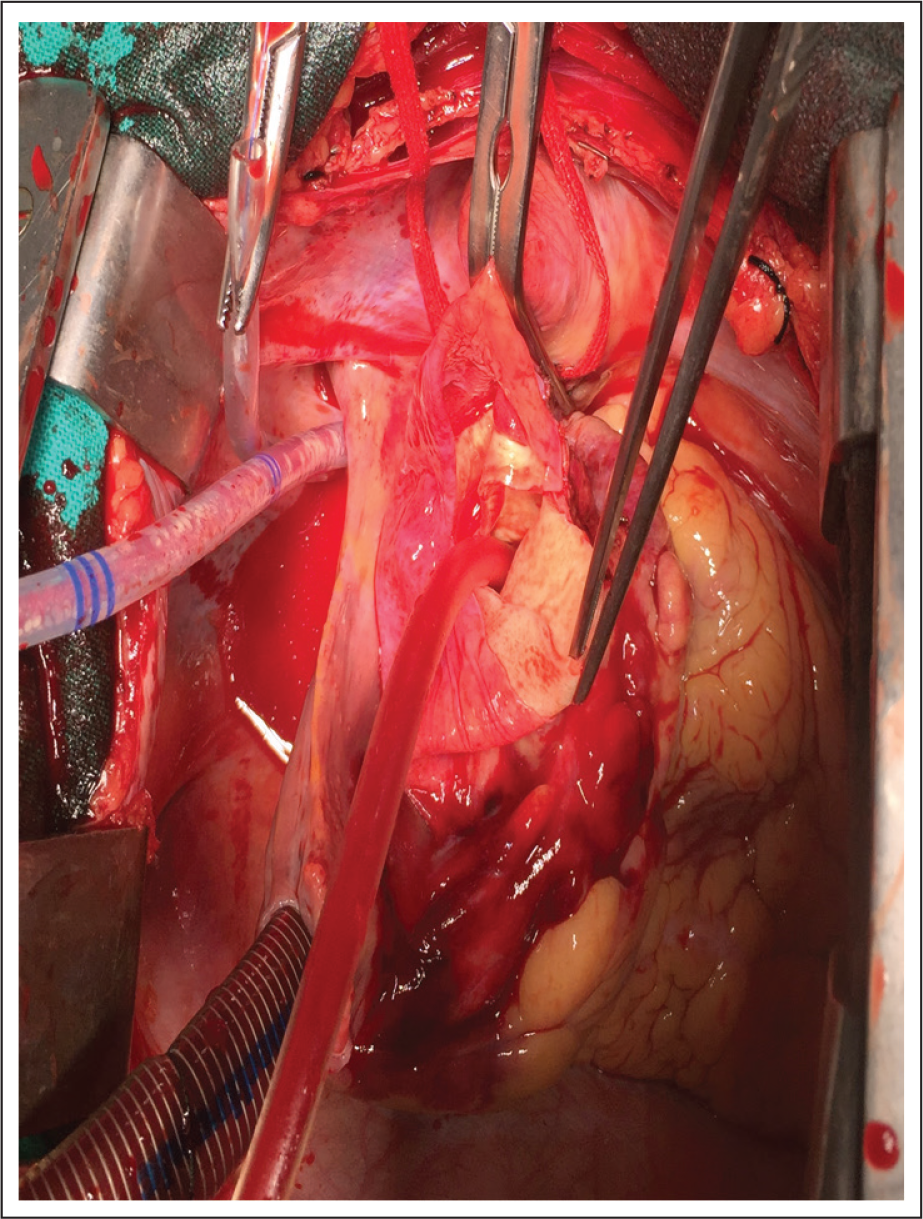
Performing Aortic Root Replacement.
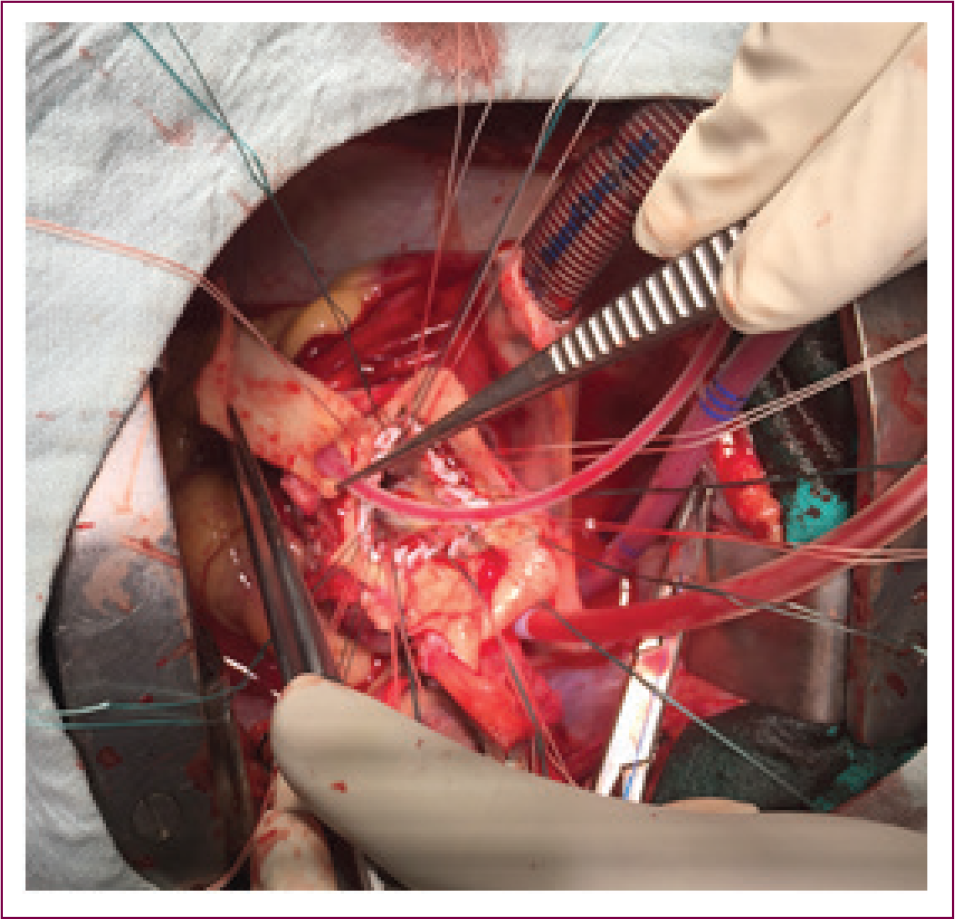
Completed Aortic Root Replacement and Distal Aortic Anastomosis.
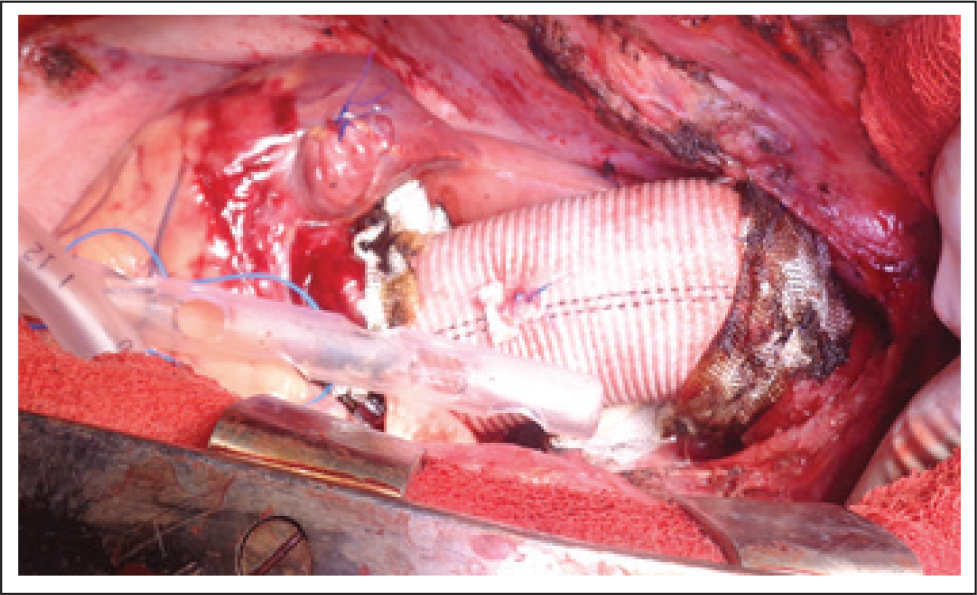
Usually adequate cooling will be reached before completion of root replacement. Attention is directed toward distal aorta. Total circulatory arrest is instituted and antegrade cerebral perfusion is commenced at a rate of 10 to 15 mL/kg/min. Then the rest of ascending aorta is debrided. Majority of the undersurface of the aortic arch is excised. A thin teflon felt strip is placed within the false lumen area in the arch. Again, the primary goal here is to prevent arch malperfusion with a secondary goal to create a layer that would hold sutures well for aortic arch reconstruction. A Dacron graft is then appropriately beveled and sewn to the reinforced arch tissue. Occasionally the aortic arch disruption is so severe that a total arch replacement is necessary (elephant trunk or frozen elephant trunk (FET) can be used if there is involvement of descending thoracic aorta).
Pictorial Representation of Different Surgical Options Used During Treatment of Type A Aortic Dissection. (A, B, C) Resuspension of Aortic Valve, Aortic Root Reinforcement With Teflon Felt. (C, D) Hemiarch Replacement. (E, F) Total Arch Replacement Along With Frozen Elephant Trunk.
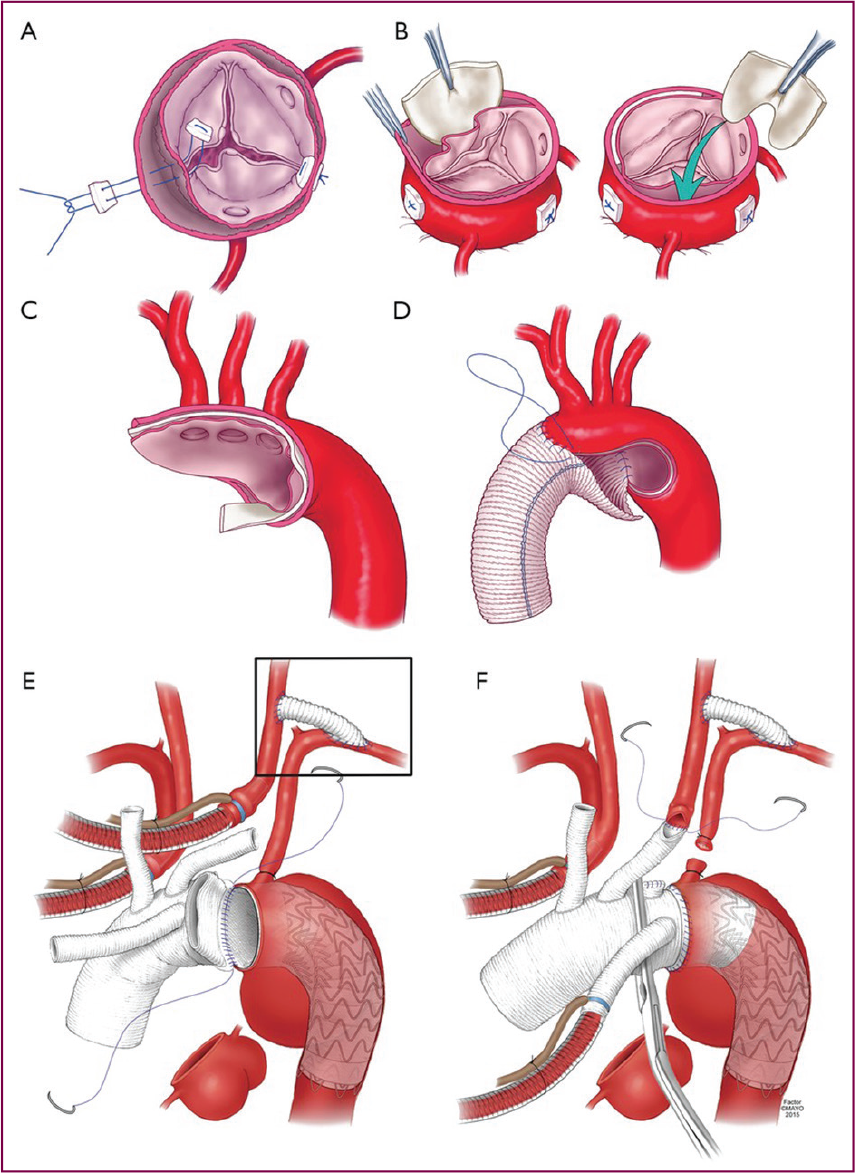
Arch replacement along with antegrade stent graft placement into the descending aorta is known as FET technique. It is a hybrid surgical approach used in cases of complex type A aortic dissection. Indications are as follows: (1) When the primary tear is located in the transverse arch or proximal descending thoracic aorta. (2) Dilated arch and descending thoracic aorta (size >50mm). (3) Severe dissection involving the arch vessels or when there is extensive intimal intussusception. FET obliterates the false lumen at the proximal descending thoracic aorta and also covers secondary entry tears located in proximal descending thoracic aorta.
After the hemiarch or total arch repair, the Dacron graft is directly cannulated, and CPB is reestablished. The proximal reconstruction is then sewn into the arch graft. Cardiac deairing maneuvers are performed and the heart is allowed to reperfuse. After complete rewarming, the patient is weaned off CPB. Early postpump is important to assess aortic valve insufficiency and ventricular function with TEE.
Postoperative Complications
According to the NORCAAD registry, 13 the common complications following surgery for acute type A aortic dissection include major bleeding (39%), stroke (20%), acute kidney injury requiring renal replacement therapy (12%), malperfusion (16%) (resulting in myocardial infarction, mesenteric ischemia, limb ischemia), prolonged ventilatory support(33%), and infections(2%-10%) of the patients.
Postoperative bleeding in these patients is due to pre-existing coagulopathy (secondary to tissue factor exposure in the false lumen), coupled with surgery, hypothermia, and poor-quality tissues. There is a progressive reduction in clotting factors, platelet function, and fibrinogen resulting in coagulation derangement similar to disseminated intravascular coagulation. The optimal approach to the management of coagulopathy consists of (1) use of thromboelastography to identify blood component deficiency, (2) aggressive goal directed replacement of fibrinogen, platelet, and factor deficiencies, and (3) use of fibrinolytic agents.
Prolonged duration of CPB and HCA contribute to being important risk factors for perioperative stroke. NORCAD registry shows that preoperative cerebral malperfusion is associated with a 3-fold increase in stroke rate.
Patients’ age, BMI >30 kg/m2, hypertension, prolonged CPB time, 14 multiple transfusions are independent risk factors for acute kidney injury and is associated with poorer 30-day survival.
Major predictors of 30-day mortality include cardiac or visceral malperfusion, postoperative stroke, acute kidney injury and massive bleeding. Short- and long-term survival following repair of acute type A aortic dissection has ranged between 52% and 95% at 1 year and 45% and 88% at 5 years. One recent study reported a 10-year survival of 55% and a 20-year survival of 30% after surgery. 15
Single Surgeon Experience
Between 2008 and 2020 a total of 60 cases underwent surgery for acute type A aortic dissection (performed by a single surgeon). Mean age of the patients was 48 +/- 21 years. In total, 80% were male and 11 patients had Marfan’s syndrome. Moderate to severe aortic regurgitation was present in 88% of the patients on presentation. Features of significant pericardial effusion/tamponade were present in 9 patients. The duration between the diagnosis and operation was less than 2 h in all the patients. Axillary artery cannulation was performed in 60% of patients and the remaining patients had femoral artery cannulation. Distal open anastomosis was done in all the patients. Cerebral protection was ensured by HCA along with antegrade cerebral perfusion, which was used in 55% and RCP was used in 45% of the patients. Concomitant procedures performed include mitral valve repair in 3 patients, mitral valve replacement in 1 patient, coronary artery bypass grafting in 3 patients, full arch replacement was performed in 2 patients. Postoperatively, significant neurological dysfunction was present in 2 patients, out of which 1 patient recovered completely within 6 weeks. Total arch replacement in aortic dissection patients was a significant predictor of poor neurological outcome. In hospital 30-day mortality was 3.38% (2 patients). The presence of low ejection fraction, post-operative renal failure, and the need for aortic arch replacement are significant predictors of mortality. Long-term survival in these patients was 96%, 72%, and 55% at 1, 5, and 10 years, respectively.
Follow-up
It is important to understand that long-term follow-up is essential in these patients after initial successful treatment. Aortic dissection is a chronic disease and patients can develop aneurysm, progression of dissection or aortic rupture in the distal residual aorta. So, it is mandatory to monitor all these patients with yearly CT aortogram. The predictors of progression of this disease include uncontrolled hypertension, dilated distal aorta and perfused false lumen. Effective B-blockade and control of hypertension is important to reduce these vascular complications. By and large about 30% of patients who underwent successful surgery for acute type A aortic dissection will have disease progression and will need further intervention in about 5 years.
Footnotes
Acknowledgment
Vikram, Vishnu, Srikanth, Vamshi, Haseena, Rajamma, Soundarya, Honey, Soniya, Aruna, Mounika, and Ridhima have contributed equally to this work.
Declaration of Conflicting Interests
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
