Abstract
Chest pain is a common clinical symptom, especially in patients with sickle cell anemia. Owing to the vast differential diagnosis, investigation can be challenging. Typical chest pain (angina) is often attributed to obstructive coronary artery disease, with patients being referred for exercise stress tests for confirmation.
Microvascular angina is emerging as a cause of significant morbidity worldwide, clinically identical to obstructive coronary artery disease. Clinical assessment and exercise stress testing often give the same results, making differentiation difficult. Coronary angiography however shows either normal coronary arteries or a nonobstructive lesion. An incomplete understanding of the etiopathogenesis of microvascular angina may result in missed diagnosis and suboptimal treatment in some cases.
Coronary microvascular dysfunction plays a key role in this phenomenon, driven by chronic inflammation, thrombotic microangiopathy, and endothelial dysfunction. This article reports the case of a young man with chest pain, linking the highlights of the pathophysiology of microvascular disease in sickle cell anemia to microvascular angina.
Keywords
Introduction
The exercise treadmill test (ETT) is a useful and cost-effective screening tool in the evaluation of obstructive coronary artery disease (CAD), especially in developing countries without easy access to advanced cardiac imaging modalities.1, 2 Its diagnostic accuracy is improved by applying the Duke Treadmill Score (DTS) to predict the likelihood of significant obstruction and mortality risk if no intervention is made.1, 3 Positive tests require invasive coronary angiograms to be performed to identify culprit lesions and to perform angioplasty where necessary. Over the past 2 decades, however, there have been increasing reports of patients with positive ETTs whose invasive coronary angiograms show no evidence of obstructive CAD, with a worldwide prevalence ranging from 10% to 50% of angina patients (depending on the population being assessed).4-8 These patients fall under the diagnosis of ischemia with nonobstructive coronary artery disease (INOCA), with the 2 main subgroups being microvascular angina (MVA) and vasospastic angina.4-6, 8 This is an important clinical entity because the morbidity, risk of major adverse cardiovascular events, and financial implications are comparable to what occurs in patients with obstructive CAD.6-9
Case Presentation
This is the case of a 28-year-old male with sickle cell anemia who was referred for an ETT on account of a year’s history of recurrent crushing exertional chest pain relieved by rest. He was an attendant at the Sickle Cell Clinic, adherent on his prescribed doses of hydroxyurea and folic acid, and had a steady state hemoglobin level of 7 to 8 g/dL. Lipid profile showed a low-density lipoprotein level of 1.8 mmol/L. His echocardiogram showed a mildly dilated left ventricle. All other echocardiographic features were normal.
In stage 2 of the ETT, he complained of crushing central chest pain radiating to his left shoulder at 74% of the maximum predicted heart rate. The echocardiogram tracing showed 3 to 4 mm ST segment depressions in inferior and anterolateral leads. He had achieved 4.6 metabolic equivalents of tasks. He was given 2 puffs of sublingual nitroglycerin and recovery stage monitoring commenced. After 6 minutes of rest, pain had reduced significantly in severity (though still present) and ST segment depressions had improved to 2 mm below baseline. He had a DTS of –23.8.
On account of the positive ETT, he was referred for coronary angiogram which showed a 40% stenosis of the proximal left anterior descending coronary artery. He was consequently commenced on bisoprolol, candesartan, and atorvastatin, and on subsequent clinic reviews, chest pain severity had improved significantly.
Discussion
Obstructive CAD has been defined by various authors as arterial stenosis of more than 50%, while others used a cut-off of 70%. 6 In either case, this patient does not fall in that category as he had a 40% coronary artery stenosis. The most objective means of distinguishing between obstructive and nonobstructive lesions is by measurement of the fractional flow reserve distal to the stenosing lesion.
MVA, otherwise called cardiac syndrome X, is a diagnosis of exclusion that has been attributed to coronary microvascular dysfunction (CMD) by many studies.4-12 Contributing factors include endothelial dysfunction, inflammation, atherosclerosis, sluggish coronary blood flow, and increased sympathetic output. It has been found that patients with endothelial dysfunction have reduced levels of endothelium-derived nitric oxide (NO) with elevated levels of endothelin-1 (ET-1), resulting in impaired local vasodilatation at capillary beds.5, 11, 13 The depletion of local NO also favors platelet aggregation and the expression of E-selectin and vascular cell adhesion molecule-1 (VCAM-1),9, 11, 13 which are both vital inflammatory mediators. Majidinia et al found that MVA patients had elevated levels of ET-1, VCAM-1, and E-selectin at rest compared to controls. 11 Chronic inflammatory conditions like rheumatoid arthritis and systemic lupus erythematosus have also been associated with CMD. 8
Sickle cell anemia is an inherited hemoglobinopathy due to the presence of large amounts hemoglobin S in red blood cells and has a complex role to play in the pathophysiology of MVA (Fig. 1). This hemoglobin defect results in reduced life spans of RBCs due to increased hemolysis, reduced solubility, and the tendency to polymerize and occlude vessels periodically, under certain conditions. 13 Hemolysis releases free hemoglobin into circulation, which reacts with NO to produce nitrate. This therefore depletes endothelial NO, offsetting the balance in favor of ET-1 and inducing vasoconstriction and a proinflammatory state. It is also involved in the generation of reactive oxygen species which contribute to the pathology.13, 14 Chronic hemolysis results in an increase in plasma viscosity, 13 resulting in sluggish blood flow that has a role to play in microvascular dysfunction.7, 13 Patients with low steady state hemoglobin levels undergo a significant amount of hemolysis compared to the normal population, meaning they have a greater exposure to these proinflammatory molecules. 13
A study also identified sickle cell anemia as a significant cause of secondary thrombotic microangiopathy. 15 This occurs due to an upregulation of cell adhesion molecules resulting in occlusion at vascular beds, causing ischemia, necrosis, and continuous inflammation that heals by fibrosis.12, 13-15 This alters both anatomical and physiological characteristics of the affected organs. The microvascular effects of sickle cell disease are well documented in causing retinopathy, nephropathy, neuropathy, and pulmonary disease. Diastolic dysfunction has also been reported in patients with sickle cell, 13 occurring due to fibrotic changes from persistent inflammation.15, 18 However, its contribution to ischemic heart disease due to its effect on coronary microcirculation does not seem to have been investigated extensively. Bakeer observed that mice with sickle cell disease developed more cardiac problems than healthy controls and other anemic mice. 14 This suggests that the pathophysiology underlying sickle cell disease has a more profound role to play in cardiac complications than the resultant chronic anemia. Although anemia could play a role in this patient’s exertional chest pain, it is unlikely as his hemoglobin level was within his steady state values, to which he has acclimatized.
The patient being discussed had symptoms of typical angina with a positive ETT and a nonobstructive CAD on angiography, which are basic features needed for the diagnosis of INOCA to be made.4-6, 8, 12 The pain of MVA is described to be generally less responsive to nitrates4, 5 than that of obstructive CAD. It has been suggested that microvascular dysfunction is not limited to specific vascular beds. This means that in the presence of MVA due to CMD, other organs should be assessed for microvascular disease 10 and vice versa. In this context, angina could then be seen as a symptom of a systemic disorder, as opposed to an isolated cardiac diagnosis. 10
Exercise stress testing is unable to distinguish between the MVA and CAD, 5 as seen in this patient who had a DTS of –23.8 which suggested he was a high-risk CAD patient requiring revascularization. 3 On stress imaging tests, patients with MVA have been found to have essentially normal tests, as opposed to patients with epicardial CAD who show overt regional wall motion abnormalities on stress imaging. 5
Pathophysiology of MVA in Sickle Cell Anemia
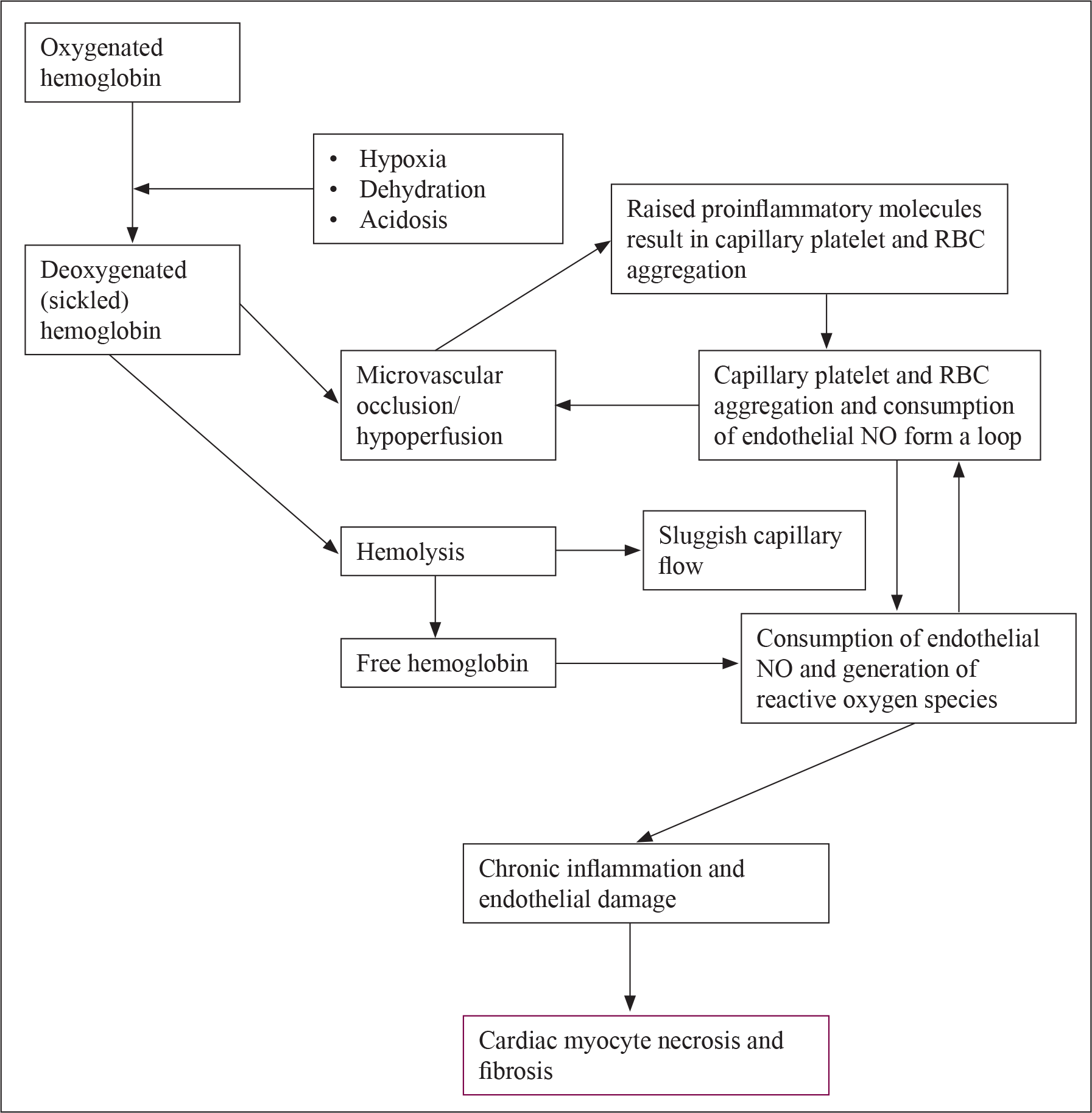
A conclusive diagnosis of MVA requires further testing, including microcirculatory evaluation using cardiac magnetic resonance imaging, assessment of coronary flow reserve, intravascular ultrasound scanning, and assessment of coronary vascular reactivity using pharmacological probes among others.4-6, 13 These tests are however not readily available in many centers.
Unfortunately, many physicians do not recognize this clinical entity and may even proceed to discharge patients from their care once a nonobstructive CAD is observed on coronary angiogram. 6 This results in patients visiting multiple health facilities seeking help, causing financial strain.6, 8, 10 Furthermore, they suffer the effects of associated morbidity because they may not receive treatment. Although there are no specific guidelines on the management of MVA, 6 treatment is essentially as for obstructive CAD.
While beta blockers like metoprolol are known to be useful because of their effect on lowering the heart rate and consequently reducing myocardial oxygen demand. Majidinia et al observed that they help to replenish fallen levels of NO by increasing production and bioavailability, and reducing degradation. 11 This addresses a key component of CMD and will help significantly with clinical symptoms. The role of atherosclerosis 9 also indicates the use of statins in management.4-6, 8 There is evidence of endothelial conversion of angiotensin-1 to angiotensin-2, 11 causing a powerful vasoconstriction. For this reason, angiotensin converting enzyme inhibitors are helpful.4-6, 8
Conclusion
MVA, and by extension INOCA, is a diagnosis that physicians need to know about and seek out actively. It is important to look for causes of microvascular dysfunction actively and treat them aggressively as they are at a high risk of major adverse cardiovascular events. The evidence of microvascular disease in one vascular bed should necessitate a full assessment to pick up disease in other organ systems. Sickle cell disease is a key cause of microvascular dysfunction and thrombotic microangiopathy, especially in tropical climates, predisposing to MVA. MVA should therefore be considered in the differential diagnosis for chest pain (especially effort-dependent chest pain) in patients with sickle cell disease.
Footnotes
Declaration of Conflicting Interests
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
