Abstract
Objective:
The B-type natriuretic peptide (BNP) levels could predict future cardiovascular events in congestive heart failure patients. Most studies have correlated basal BNP levels to long-term outcomes. Limited data exist on the prognostic significance of 1-month postdischarge BNP levels after acute heart failure.
Methods:
Consecutive patients admitted for worsening heart failure were enrolled. BNP was measured at admission, predischarge and at 1-month following discharge. Patients were followed for 1 year for end points of death and rehospitalization.
Results:
A total of 150 patients (mean age 60.8 + 13.8 years) were included in the heart failure study. 81 (54%) patients had acute heart failure secondary to acute coronary syndrome, while the rest (46%) had acute decompensation of chronic heart failure irrespective of etiology. Mean ejection fraction was 28.6 + 8.9%. 14 patients expired during hospitalization. BNP at admission was an important predictor of in hospital mortality (P value = .003). Following discharge, 7 events (3 deaths and 4 rehospitalizations) occurred over next 1 month. 1-month outcome was predicted by baseline BNP (P value = .01) as well as discharge BNP value (P value = .001). A total of 55 events (26 rehospitalization and 29 deaths) occurred at follow-up of 1 year. Age > 50years, ejection fraction at baseline and all time sequential BNP levels (at admission, discharge, as well as 1 month) were univariate predictors of death and rehospitalization at 1 year. The BNP at 1 month had best discriminative power and remained the lone significant predictor in the multivariate analysis (P = < .001).
Conclusions:
1-month postdischarge BNP level is a useful prognostic factor that predicts mortality and rehospitalization at 1-year follow-up, in patients admitted with heart failure, and helps in identifying patients who need more intensive drug treatment and closer follow-up.
Introduction
B-type natriuretic peptide (BNP) is a cardiac neurohormone secreted from the membrane granules in the cardiac ventricles in response to ventricular volume expansion and pressure overload. BNP levels are raised in many cardiac and noncardiac conditions.1-4
Hospitalization for decompensated congestive heart failure (CHF) carries a poor vital prognosis, with frequent subsequent readmissions.5-9 It has recently been shown that the outcome among high-risk patients with decompensated heart failure can be improved by treatment intensification and regular follow up.10, 11 There is currently however no established criterion or score which could predict postdischarge adverse outcome in near future. BNP represents a reliable risk marker for diagnosis, risk-stratification, and prognostication in patients with chronic heart failure.12-17 The American College of Cardiology/American Heart Association Heart Failure Guidelines from 2013 have given a class I recommendation for the measurement of BNP or N-terminal pro-BNP for establishing prognosis and disease severity in patients with acutely decompensated heart failure.18 However, most of the heart failure studies have evaluated only the baseline BNP levels.15, 19, 20 There are only a few studies evaluating predischarge and follow-up BNP levels. A prospective study with serial BNP measurements is lacking from our part of the world. We aimed to test the prognostic significance of 1-month postdischarge BNP levels to clinical outcomes in patients admitted with heart failure and establish a cut off BNP level which could be a marker for poor prognosis at 1 year.
Material and Methods
This was a single center, prospective, nonrandomized, observational study conducted in a tertiary care government hospital in India from August 2015 to December 2017. Consecutive patients admitted for heart failure from August 2015 to December 2016 were enrolled. Patients were classified as having heart failure according to the Framingham criteria, 21 namely the presence of 2 major criteria or 1 major criterion + 2 minor criteria. Only patients with New York heart association (NYHA) functional class III or IV were included. Patients who were enrolled included either acute coronary syndrome presenting with heart failure or patients of chronic heart failure of any etiology who were hospitalized for worsening heart failure. Chronic heart failure patients included patients with idiopathic dilated cardiomyopathy, ischemic dilated cardiomyopathy, hypertensive heart disease, or restrictive cardiomyopathy. Dilated cardiomyopathy was diagnosed when there was no obvious cause for ventricular dysfunction with dilated left ventricle. Ischemic cardiomyopathy patients had either a history of myocardial infarction or severe coronary artery disease on angiogram with presence of left ventricular dysfunction. Restrictive cardiomyopathy was characterized by restrictive filling with normal or near normal left ventricular systolic function. Patients with moderate to severe primary valve disease, poor adherence to therapy, sepsis, cirrhosis, morbid obesity, pregnancy, and critically ill patients with reduced life expectancy were excluded from the study. Informed consent was taken from the patients before enrollment in the study.
Patients’ data were collected in a prestructured case recording pro forma. Clinical and demographic data of patients were recorded. Baseline biochemical parameters such as serum creatinine, hemogram, blood sugar, and electrolytes were noted. Echocardiography was done in order to assess the left ventricular end-systolic diameter (LVESD), left ventricular end-diastolic diameter (LVEDD), and left ventricular ejection fraction (LVEF). Echocardiography was performed with a Vivid 7 computed sonography system (GE Medical Systems, Waukesha, Wisconsin, USA). LVEF was calculated with a modified Simpson’s method using biplane apical (4-chamber) views. All the echocardiographic examinations were performed by expert operators blinded to the results of BNP assay.
Time Sequence of BNP Analysis
Serum BNP levels were measured at the time of admission, hospital discharge and at 1-month postdischarge. First sample (BNP1) was collected at admission in most of the patients. Few patients for whom it was not collected at admission, it was done within 24 hrs. Patients who were discharged successfully after stabilization underwent BNP measurement at the time of discharge (BNP2). For patients who were event free at 1 month, third BNP measurement (BNP3) was done at first visit which was scheduled at 1 month after discharge. To ensure 1-month visit, telephonic contact was made in the preceding week.
End Points and Follow-Up
Patients were followed for 1 year for end points of death and/or rehospitalization. Follow-up was done with out-patient department (OPD) visits at first month, 3 months, 6 months, and 1 year. Patients who did not report were telephonically contacted and enquired about any clinical event.
BNP Measurement Methodology
For each BNP measurement, 5 ml of whole blood was collected into tubes containing potassium ethylenediaminetetraacetic acid (1 mg/ml blood) and processed within 4 hours of sample collection. BNP was measured using the Triage BNP test (Alere, San-Diego, California, USA). The Triage BNP test is a fluorescence immunoassay for the quantitative determination of BNP in whole blood and plasma specimens. In the analysis, the concentrations of BNP are directly proportional to the fluorescence detected by an Alere Triage Meter (Alere San Diego, Inc. California, United States of America).
Statistical Analysis
Data analysis was performed using Statistical Package for Social Sciences (SPSS 17.0) software (SPSS Inc., Chicago, IL, USA). Data were expressed as means +/– standard deviation. Comparisons between 2 independent groups was done using independent samples t test or analysis of variance test for continuous variables, and paired-samples t test for differences between means of 2 related groups. Chi-square test was used to analyze differences between categorical data. The predictive value of BNP for detecting events was assessed using regression analysis. Kaplan Meier analysis was used to see the effect of various factors on survival and the log-rank test was used to compare the incidence rate of combined events of death and readmission among the BNP ranges. To compare the predictive values of the parameters, receiver-operating characteristics and their area under the curve were constructed. The best prognostic cutoff value for a combined event was defined as that which had the best compromise between sensitivity and specificity for predicting readmission or sudden death.
Results
A total of 150 patients were enrolled after exclusion. Baseline characteristics are mentioned in Table 1. Mean age was 60.8 ± 13.9 years. Males constituted 80% (n = 120) of the study population. Acute coronary syndrome with heart failure was the etiology in 82 (54.6%) patients. Rest 68 (45.4%) were chronic heart failure patients admitted with worsening heart failure. Out of the chronic heart failure patients, 36 (24% of total) had ischemic dilated cardiomyopathy and 25(16.6%) had idiopathic dilated cardiomyopathy. Rest were of restrictive cardiomyopathy (3 patients), hypertensive heart disease (3 patients), and right-sided cardiomyopathy (1 patient). Overall, 78% had ischemic etiology of heart failure and rest were nonischemic. 62 (41%) patients had diabetes (DM) and 62(41%) had hypertension (HTN). Mean ejection fraction (EF) of study population was 28.6 ± 8.9%. Mean end systolic diameter was 44.7 ± 9.9 mm and mean diastolic diameter was 58.5 ± 8.3 mm. Mean serum Creatinine was 1.5 ± 0.55 mg%. Mean hemoglobin was 12.1 ± 2.5 g/dL, and mean random sugar level at admission was 164 ± 110 mg/dL. The number of deaths and rehospitalization during various time intervals of follow-up have been depicted in Figure 1.
Baseline Characteristics of Patients
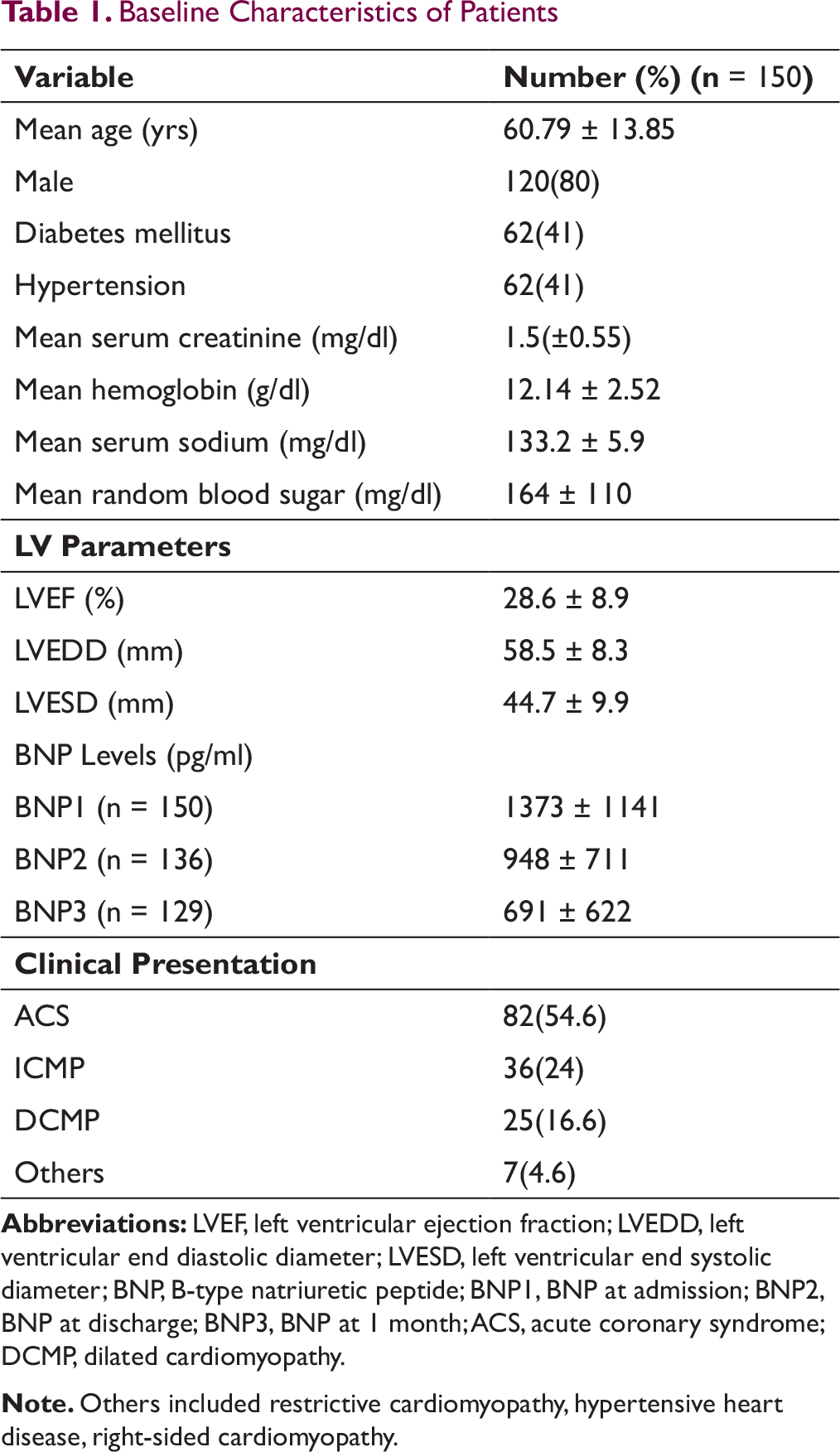
Graph Depicting Clinical Events of Total Deaths and Rehospitalizations
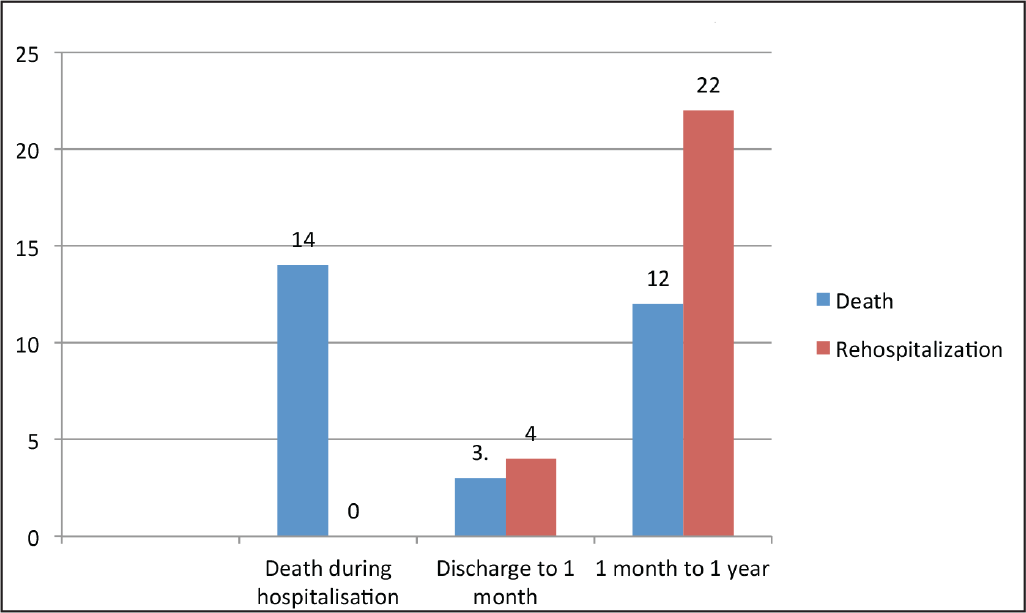
Clinical Events on Follow-Up
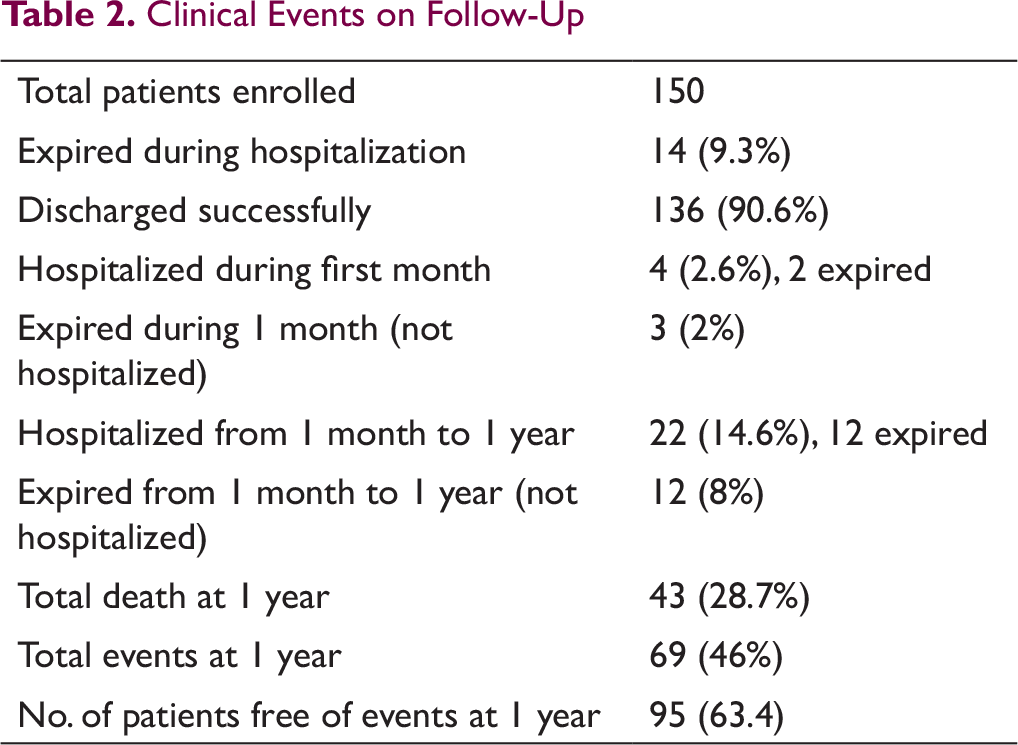
In-Hospital Outcome
Mean BNP at admission was 1373 ± 1141 pg/mL. 14 (9.3%) patients expired during index hospitalization (Table 2). The admission value of BNP was a strong predictor of in hospital mortality (P = .003). Other baseline variable such as age (P = .6), DM (P = .1), HTN (P = .3), serum creatinine (P = .7), LVEDD (P = .6), LVESD (P = .7), and LVEF (P = .2) did not affect the in hospital mortality (Table 3).
Predictors of In-Hospital Mortality
Outcome at 1 Month
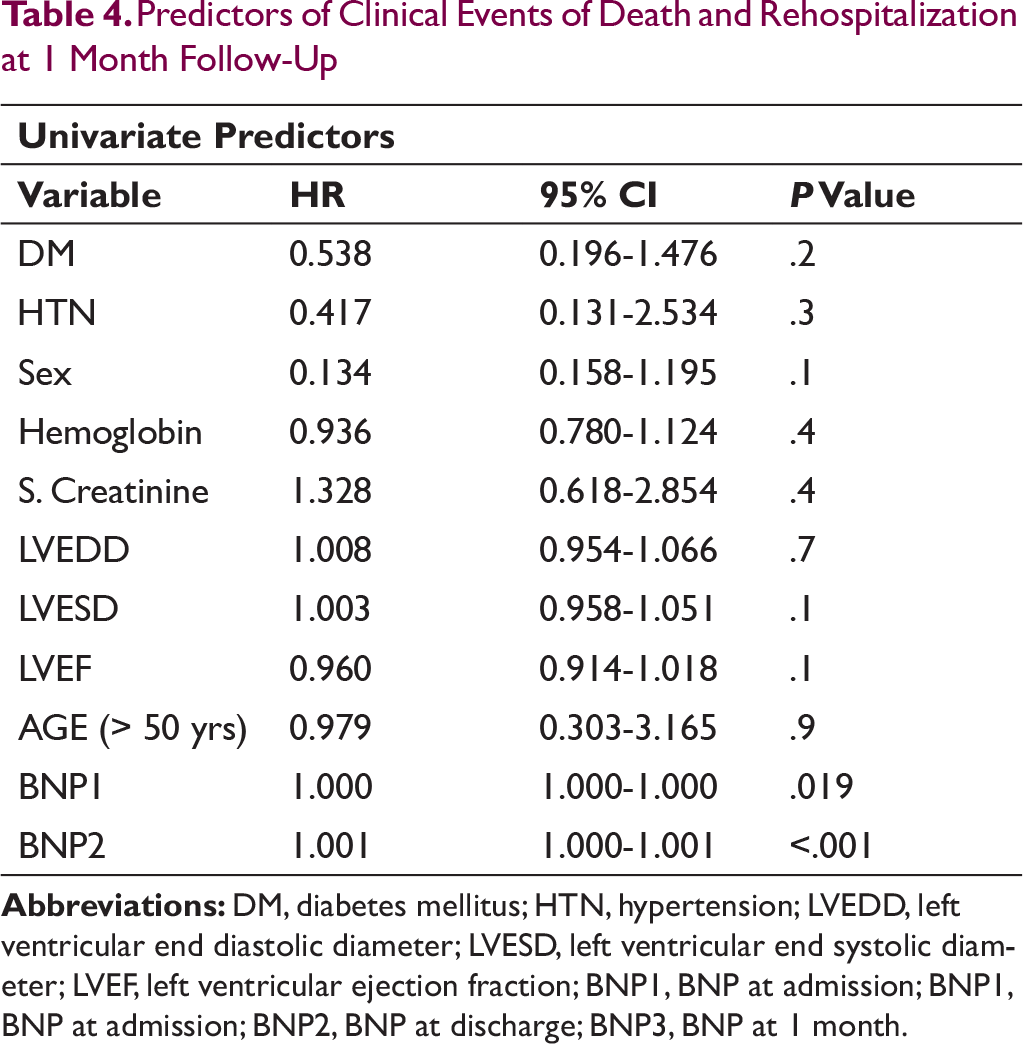
136 (90.6%) patients were discharged after stabilization. Mean discharge BNP was 948 ± 711 pg/mL. Following discharge, a total of 7 events (3 deaths and 4 rehospitalization) occurred in the next 1 month. Out of 4 hospitalized patients, 2 patients expired during hospitalization. A total of 23 events occurred at the end of 1 month. Event free survival at 1 month related to both BNP at admission (P = .01) and BNP at discharge (P ≤.001). However, there was not significant relation of other baseline variables such as sex (P = .1), DM (P = .2), HTN (P = .13), creatinine (P = .4), LVEDD (P = .7), LVESD (P = .1), and LVEF (P = .1), on the 1-month outcome (Table 4).
Predictors of Clinical Events of Death and Rehospitalization at 1 Month Follow-Up
Outcome at 1-Year Follow-Up
129 (86% of total 150 enrolled) patients were free of adverse events at 1 month. Mean BNP at 1 month was 691 ± 622 pg/mL. From 1-month to 1-year follow-up, 12 (8%) deaths occurred. 22 (14.6%) patients were rehospitalized out of which 12 expired. Total 43 (28.7%) deaths and 69 (46%) events occurred at 1 year. 95 (63.3%) patients were free of clinical events and 5 (3.3%) patients were lost to follow-up after 1-month visit.
Event-free survival at 1 year was independent of sex (P = .1), diabetic status (P = .4), hypertension (P = .2), serum creatinine (P = .3), LVEDD (P = .1), or LVESD (P = .1; Table 5).
Predictors of Death and Rehospitalization at 1-Year Follow-Up
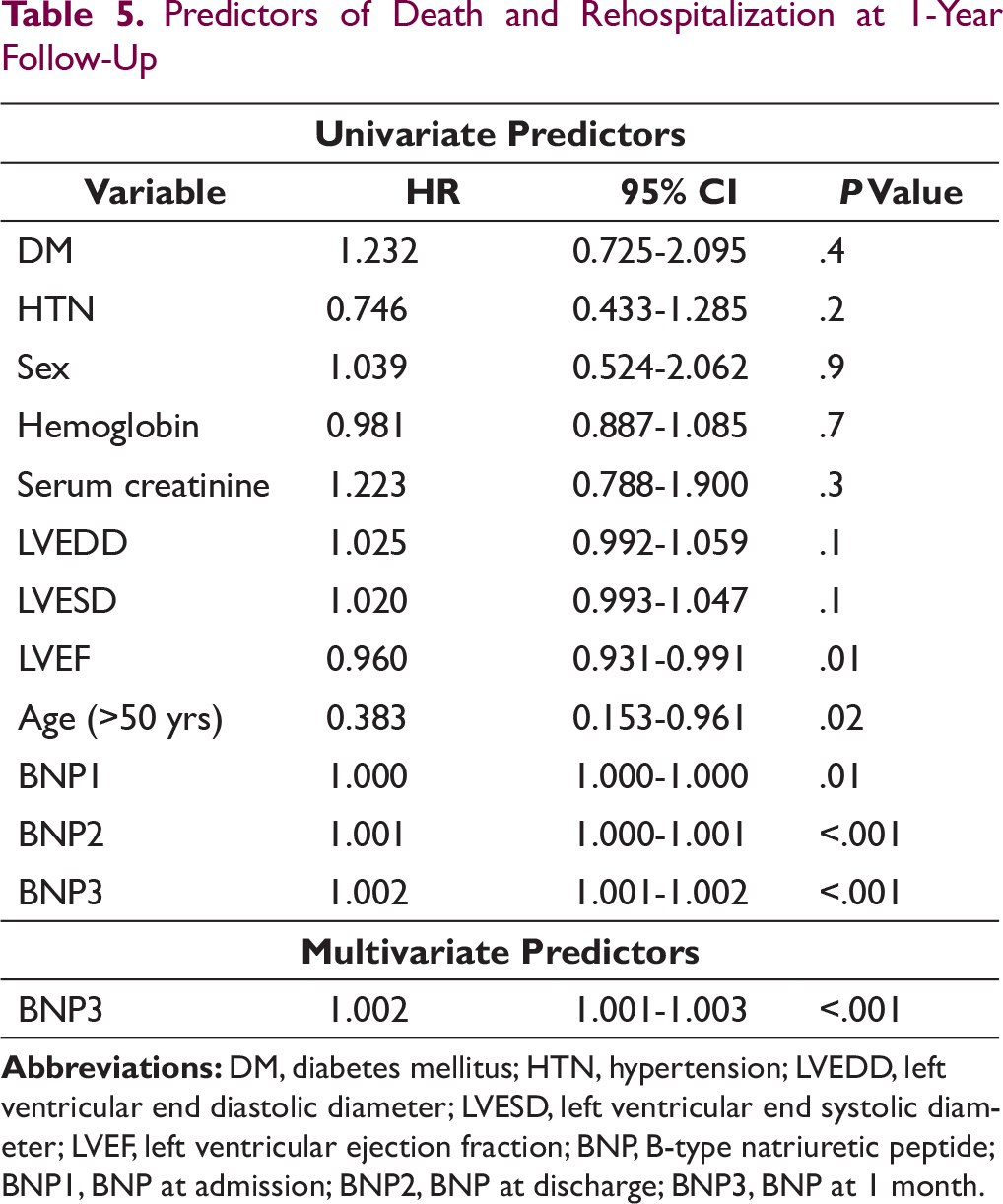
Age > 50 years (P = .02), baseline EF (P = .01), and BNP were important predictors of event-free survival at 1 year. The mean age for patients who had event was 62.6 ± 14.6 yrs. For patients with event-free survival, the mean age was 59.8 ± 13.4 yrs. The mean survival time for age group < 50 yrs was 306 days versus 250 days for age > 50 yrs (log rank test P = .04).
Among various echocardiographic parameters, only baseline left ventricular EF was predictor of clinical events at 1 year. The mean EF for the patients who were event-free was 30.0 ± 8% compared to 26.3 ± 9% for the patients who had event (P = .01).
All BNP values were important predictors of event-free survival at 1 year. BNP at admission was an important predictor of survival (P = .01). Admission BNP value of 1115 pg/mL had a sensitivity of 67% and specificity of 67% to predict clinical event at 1 year (Figure 2a).
BNP at discharge also had impact on event-free survival (P < .001) at 1 year. Patients who had event-free survival had a mean discharge BNP of 792 ± 663 pg/mL compared to mean BNP of 1308 ± 696 pg/mL for patients who had event. Thus, the patients who responded well to treatment had a significant fall in BNP at discharge and did well thereafter. Discharge BNP value of 813 pg/mL had a sensitivity of 73% and specificity of 65% to predict clinical event at 1 year (Figure 2b).
Similarly, BNP at 1 month also had a significant effect on event-free survival (P < .001). Patients who had event-free survival had a mean BNP at 1 month of 468 ± 428 pg/mL as compared to mean BNP of 1289 ± 669 pg/mL for patients who had event. BNP (1 month) levels of 431 pg/mL had 97% sensitivity and 60% specificity for clinical events of death and hospitalization at 1-year follow-up (Figure 2c).
ROC Curve for Different BNP Values
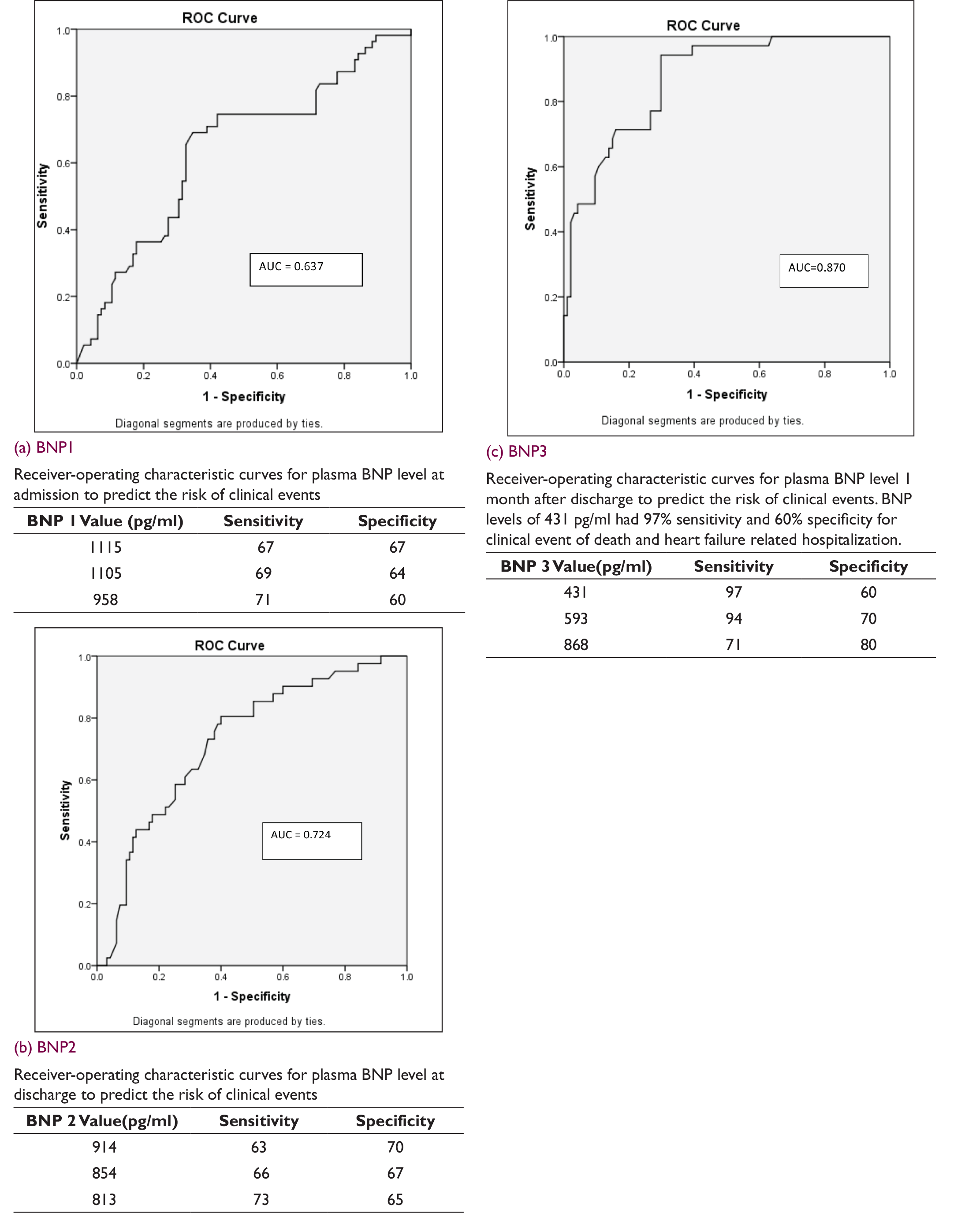
On multivariate analysis, BNP at 1 month remained the lone significant (P < .001) variable, while other 2 BNP values (admission as well as discharge), age and EF were insignificant. The receiver operator curve, shown in Figure 2c, illustrates the sensitivity and specificity of BNP measurements at 1 month after discharge in discriminating patients who will have clinical event in 1 year. The area under the curve was 0.87 for BNP at 1 month, indicating good discriminatory power.
There was no difference in survival between ischemic and nonischemic dilated cardiomyopathy group as predicted by Cox regression analysis (P = .7).
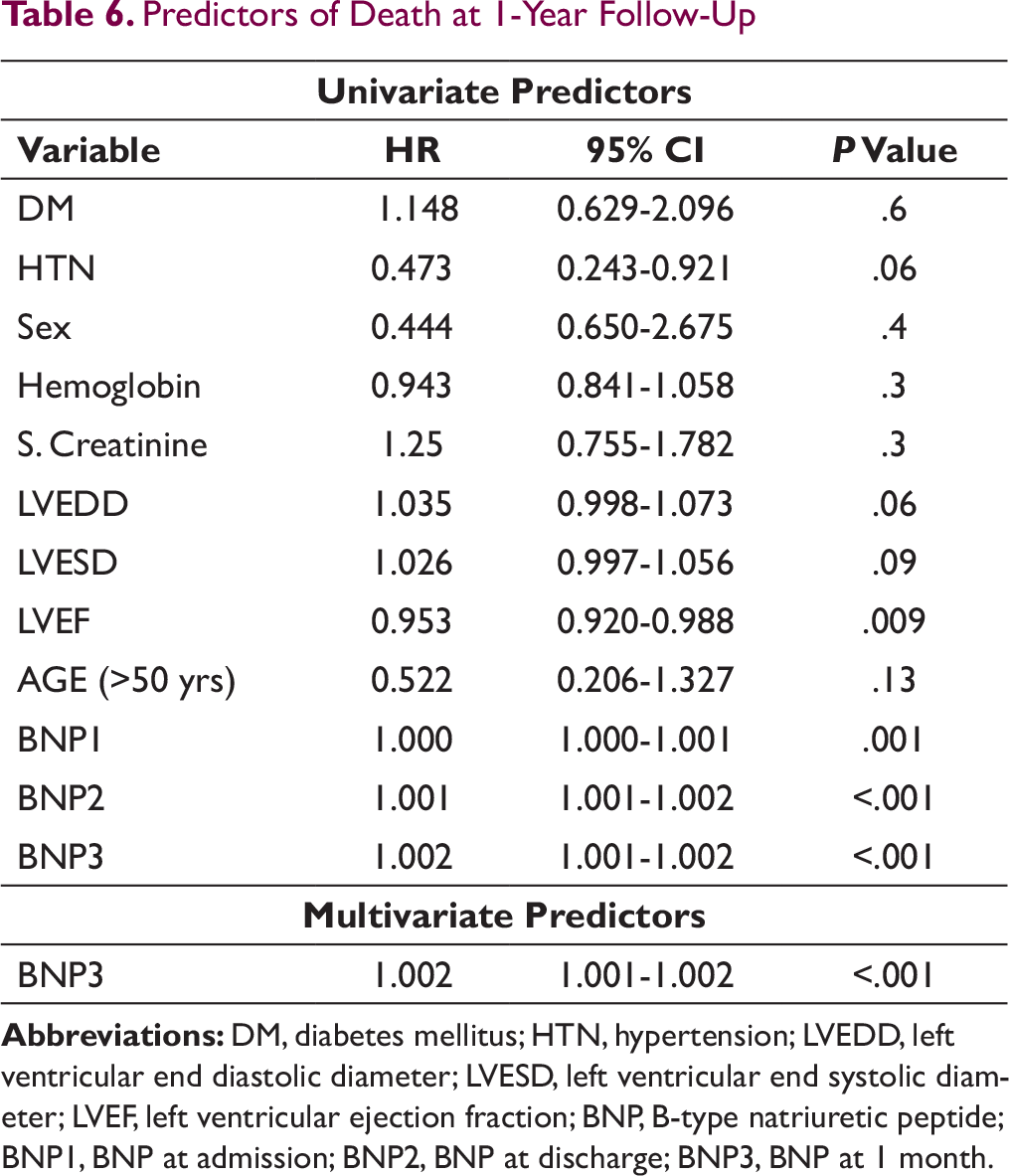
Predictors of Death at 1 Year
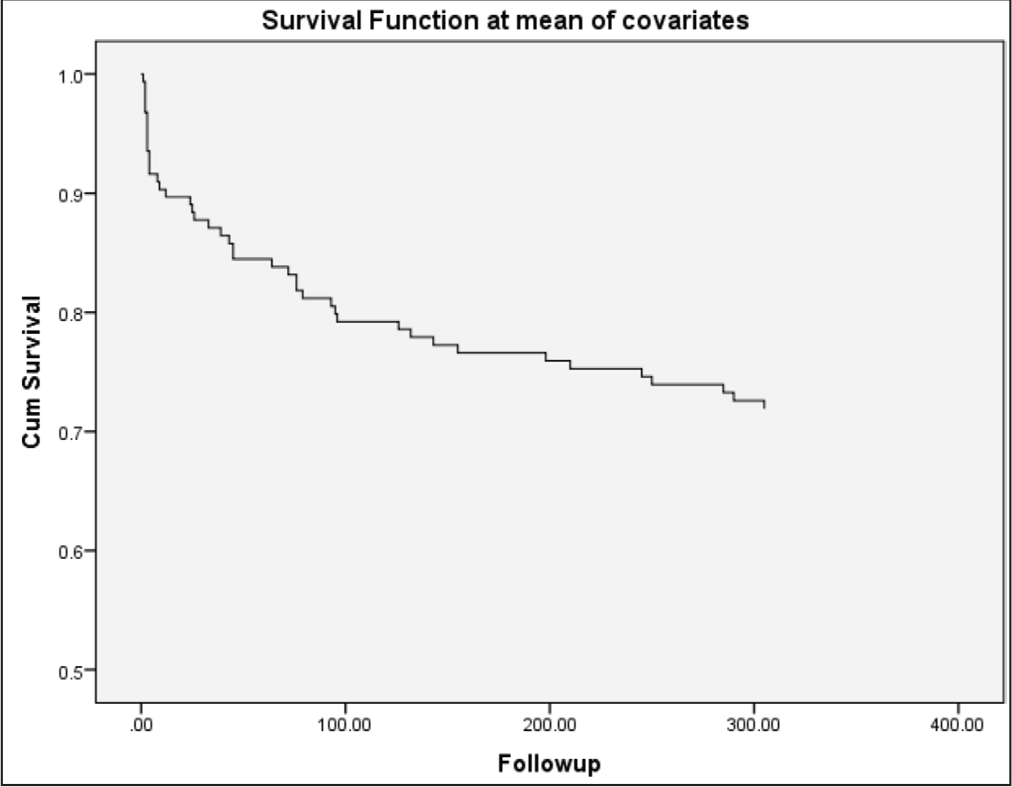
When end points of deaths were analyzed separately, it was found that baseline LVEF (P = .009) as well as BNP (admission BNP [P < .001], discharge BNP [P < .001], and 1-month BNP [P < 0.001]) were important predictors of death at 1 year in univariate analysis (Table 6). In multivariate analysis, only BNP at 1 month remained significant. Kaplan Meier analysis estimating survival at 1 year follow-up is depicted in Figure 3.
Predictors of Death at 1-Year Follow-Up
Kaplan Meier Analysis Estimating Survival at 1-Year Follow-Up
Discussion
Decompensated CHF patients have a high rate of adverse events and mortality. In-hospital mortality was 9% (14 out of 150) in our study which is equivalent to 8% events in a single center study published in 2004 by Logeart et al. 22 In-hospital mortality in the Trivandrum heart failure registry was 8.5%. 23 The postdischarge event rate (death and rehospitalization) at 1 year was 40% (55 out of 136) in our study which is still lesser than the event rate of 48% in study by Logeart et al (with follow-up of 6 months). 22 In our study, 1-month event rate was 14% (21 events) which is similar to 1-month event rate in Trivandrum registry (12.5% 1-month mortality).
In a study of 206 patients done by Feola et al published in 2011, 24 postdischarge event rate was 14.6%, but the patients enrolled were in NYHA classes II to IV and mean EF was 43.2%. In our study, only NYHA classes III and IV patients were included, with mean EF of 28.6%. In a study 25 by Cheng et al, death and 1-month readmission rate was 30.6%. Indeed, in-hospital mortality and readmission rates for patients with CHF were extremely high.26, 27 From these studies, it emerged that CHF patients discharged after an acute decompensation have high rate of adverse clinical outcome (death and hospital readmission).
In our study, 80% of patients were males and females were only 20%. Most of the other studies also have male preponderance. In a study by Koglin et al, 28 88.5% were males. In a study by Mototsugu Nishii et al, 29 71The outcome was not affected by the sex of the patient (log rank P = .795) in our study.
Various studies have been done to evaluate the role of BNP testing in CHF patients and its role as a prognostic marker has been proven by a few of them. Feola et al in their study on patients with CHF discharged after an acute decompensation with 6-month follow-up found that predischarge BNP (P = .001) were significantly associated with adverse mid-term outcome. The admission values of plasma BNP (P = .05) were not significantly correlated with clinical outcome. 24
Mototsugu Nishii et al in their study of 83 relatively stable low-risk patients showed that 6-month postdischarge BNP (hazard ratio 2.29; 95% confidence interval [1.42, 3.56]; P = .0005) is closely associated with long-term outcome (18 months follow-up); however, BNP at 3 months as well as at discharge are not predictive of it. 29
Logeart et al in their study found that the predischarge BNP assay was the most predictive parameter (area under the ROC curve = 0.83) for the event rate and also the lone significant variable in multivariate analysis (hazard ratio [HR] = 1.14). 22 Cheng et al in their study on 72 patients revealed that the last measured BNP was strongly associated with the combined end point and BNP discharge concentrations were strong predictors of subsequent readmission (area under the receiver operator curve of 0.73). 25 Ondrej Ludka et al in a study published in 2013 concluded that BNP levels on admission and at discharge were significant prognostic factor in patients hospitalized for acutely decompensated chronic systolic heart failure and that the BNP levels at discharge were a stronger predictor than those on admission. 30
So our study results are in parallel with the results of other studies evaluating role of BNP in heart failure patients. In univariate Cox analysis, all BNP measurements from admission to 1 month after discharge were significantly associated with the risk of death or readmission after discharge, but the postdischarge assay was the most valuable. After adjustment for BNP at admission and BNP at discharge (predictors of events in a univariate model), postdischarge BNP assay was strongly predictive of worse outcome; thus, it appears to be a simple and reliable test to identify the highest risk patients.
In our study, age was important predictor of outcome and patients aged > 50 years had poor outcome. This differs from the results of other studies. In a study by Logeart et al, age (mean age 69.4 + /–14.4) did not affect the outcome (P = .6). Cheng et al (mean age was 68 ± 1.6 years) also showed that survival was independent of age (P = .37). One important finding to note is that the mean age (60.8 ± 13.9 years) in our study was lesser than other 2 studies.
In our study, poor baseline EF was an important prognostic marker for survival (P = .011). In a study by Feola et al, EF was shown to be a strong predictor of outcome (P ≤ .01). But there was no effect of EF on survival in studies by Logeart et al (P = .6) and Cheng et al (P = .21).
The prognostic information of postdischarge BNP assay is greater than other common clinical variables, BNP during hospital stay, and echocardiographic parameters. High plasma levels of BNP have been identified as major predictors of progressive LV remodeling after acute myocardial infarction.31 In our study, patients with clinical events, in contrast to event-free patients, showed no significant decrease in BNP levels from the time of discharge to 1-month follow-up. In the event group, mean BNP at discharge was 1308 ± 696 pg/mL, whereas after 1 month, the mean BNP was 1289 ± 669 pg/mL. On the contrary, in the event-free group, the mean BNP at discharge was 792 ± 663 pg/mL, whereas after 1 month, the mean BNP decreased to 468 ± 428 pg/mL.
Our study further potentiates the role of BNP measurements on follow-up visits. Patients who were discharged after clinical stabilization have poor outcome if BNP levels persistently remain high. This group of patients probably reflects poor compliant to drugs or poor drug responsiveness. So these patients may be the target for intensification of drug treatment. Thus, BNP levels at follow-up visits may prove helpful in guiding therapy.
Limitation
The number of patients in our study is relatively small, and it was a single tertiary care center study. So, additional prospective multicenter studies will be necessary to test the therapeutic utility of postdischarge BNP assessment in heart failure patients.
Conclusion
BNP is a strong predictor of clinical outcome in heart failure patients. Serial BNP measurement is helpful in identifying patients who need more intensive drug treatment and close follow-up. Among patients admitted with heart failure, BNP level at 1-month was demonstrated to have prognostic significance with level > 431 pg/ml showing 97% sensitivity and 60% specificity for clinical events of death and hospitalization at 1-year follow-up. Thus, persistent high level of BNP at 1-month follow-up may indicate poor outcome and emphasize the need for intensive care in treatment of such patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
