Abstract
Atrial fibrillation (AF), the most common arrhythmia encountered worldwide, is associated with significant morbidity. The three important considerations with regard to AF management are stroke prevention, rate control, and rhythm control, with the latter two overlapping to some extent. While antiarrhythmic drugs have had limited success as a rhythm control strategy, being limited by side effects and proarrhythmia, catheter ablation has emerged as a potentially better alternative. Current ablation techniques afford good success for paroxysmal AF, especially when done in experienced centers; however, further improvements and innovations are required to improve results for more persistent forms of AF. The current review critically summarizes the present strategies employed for rhythm control in AF and briefly outlines some of the newer developments in this field.
Introduction
Atrial fibrillation (AF) is the most common arrhythmia encountered in clinical cardiology practice. It is estimated that approximately 33 million individuals are afflicted with AF worldwide, 1 although robust estimates, especially from the developing world, are lacking. AF can not only cause significant symptoms and reduced quality of life but it has also been shown to result in adverse cardiovascular outcomes including cardiovascular death and heart failure,2, 3 AF is also a major risk factor for ischemic stroke of the cardioembolic type with resultant major disability,4, 5 It is generally classified as paroxysmal (lasting or therapeutically terminated < 7 days), persistent (lasting > 7 days; long standing persistent lasting > 1 year), and permanent (AF clinically accepted where attempts to convert to sinus rhythm are no longer pursued). The management of patients with AF involves treating the arrhythmia itself and mitigating the stroke risk associated with AF via anticoagulation in appropriate candidates. The 2 broad approaches adopted for dealing with AF per se are rate control and rhythm control.
Rate Control Versus Rhythm Control in AF
AF results in irregular, fast ventricular rates which is responsible for symptoms as well as potentially adverse effects on cardiac function. To this, one needs to add the loss of atrial contribution to cardiac filling as well as adverse remodeling in the atrium and ventricle over time. 6 Deleterious atrial remodeling spans the spectrum from tachycardia-induced atrial remodeling (due to which “AF begets AF”) to a primary cardiomyopathic process—fibrotic atrial cardiomyopathy—wherein, AF is the result rather than the cause of this cardiomyopathy. The hallmark is atrial fibrosis which produces varied manifestations, including AF, atrial tachycardia, and sinus node dysfunction. 7 The strategy of rate control aims to only reduce ventricular rates resulting from AF to ameliorate the symptoms, while the rhythm control strategy aims to abolish AF, and restore and maintain sinus rhythm. The landmark Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) trial, comparing these 2 strategies, failed to show a significant benefit for the rhythm control strategy over rate control. 8 However, subsequent subanalyses have suggested that although sinus rhythm is beneficial, this is offset both by adverse effects associated with antiarrhythmic drugs (AADs) used for the purpose as well as by the relatively poor efficacy of drugs in maintaining sinus rhythm in a good proportion of patients,9, 10 In discussing the need for, as well as strategies for rhythm control in AF, it is useful to separately consider acute and chronic rhythm control.
Acute Rhythm Control
This refers to the situation where the patient presents with an acute episode of AF, most often symptomatic with rapid ventricular rates. The key elements to consider in deciding on acute rhythm control are the severity of symptoms, hemodynamic stability, and duration of AF. Recent-onset AF with lesser degree of structural heart disease is more likely to convert to sinus rhythm as compared to long-standing AF with greater underlying substrate. However, a recent study suggests that a routine strategy of converting all recent-onset AF may not be necessary, as a good proportion of AF may spontaneously revert within 48 h. 11 Hence, decision-making should take into consideration the overall clinical picture. Potential precipitating factors, such as electrolyte imbalance, infection, cardiac ischemia etc., should always be looked for and corrected. The AF duration is also important for the assessment of acute stroke risk, which is often overlooked. Conversion of AF to sinus rhythm, either by pharmacologic means or direct current (DC) cardioversion, is associated with enhanced stroke risk in the peri-conversion period. This is especially true if the AF has lasted longer than 48 h, but even shorter durations of AF, such as 12 h, carry some risk of stroke. 12 Hence, in AF of > 48 h or unknown duration, adequate anticoagulation for at least 3 weeks should be provided before attempting cardioversion. However, if there is a need to convert the rhythm straightaway, an alternate strategy is to perform a transesophageal echocardiography (TEE) to rule out the presence of a left atrial clot before proceeding with cardioversion. 13 Only in an absolute emergency, where rapid AF is causing hemodynamic compromise, should one proceed with prompt electrical cardioversion even without TEE. The institution of parenteral anticoagulation in the form of unfractionated or low-molecular-weight heparin should always be done early prior to conversion. Furthermore, anticoagulation needs to be continued for a minimum of 4 weeks after cardioversion of AF to account for atrial stunning post restoration of sinus rhythm. Further, long-term continuation of anticoagulation after that is based on individual assessment of stroke risk through well-established risk scores such as the CHADS-VASc score.14, 15
Strategies for Acute Rhythm Control
The 2 methods that can be used to achieve rhythm control in the acute setting are pharmacological conversion via use of AADs and electrical cardioversion.
Electrical Cardioversion
Electrical cardioversion by means of DC synchronized shock has a higher efficacy than drugs and is also immediate. Hence, it is the preferred strategy whenever prompt reversion to sinus rhythm is needed such as in cases of hypotension or severe heart failure related to rapid ventricular rates. Synchronization with the QRS is important to avoid triggering ventricular fibrillation. Success of electrical cardioversion depends on the amount of electrical current effectively delivered to the myocardium, an important determinant of which is the transthoracic impedance (TTI). TTI, in turn depends on many factors such as body weight, surface area, chest wall thickness and fat, paddle orientation, prior shocks given, etc. Hence, the energy required for defibrillation can differ from case to case. Biphasic shock waveforms are generally more effective than monophasic waveforms for defibrillation, with a lower energy requirement. 16 Repetitive (stacked) shocks have been said to be useful by lowering TTI, but there is limited evidence for improved efficacy. Either paddles or patches can be used for defibrillation; while patches are more convenient, paddles show some superiority due to lower TTI and more uniform contact. Cardioversion success rate is lower in obese patients as they have greater chest circumference, thickness, more intrathoracic as well as epicardial fat. While there is no uniform standard, cardioversion is initially attempted with energy of 50 to 100 J. If unsuccessful, escalating energies can be used. Most available biphasic defibrillators have a maximum energy of 200 J, but 360 J biphasic shock may succeed when lower energies have failed and can be used if available. Other strategies for failed DC version include different paddle orientation such as anteroposterior (instead of usual anteroapical) or manual pressure augmentation over the patches with a gloved hand which can be performed safely,17, 18 Finally, facilitated cardioversion after loading with AADs, such as amiodarone and vernakalant, may achieve success where simple cardioversion fails,19, 20 Although theoretically DC cardioversion can result in other serious arrhythmias such as ventricular tachycardia (VT) or fibrillation (VF), these are rare and generally occur in the setting of dyselectrolytemia or other drug toxicity, with electrical cardioversion overall displaying good safety. 14 However, DC cardioversion does require sedation/anesthesia with a small attendant risk of related complications.
Pharmacologic Cardioversion
Acute pharmacologic cardioversion is accomplished through the use of intravenous antiarrhythmic agents in the emergency room or intensive care unit setting. All antiarrhythmic agents have the potential for proarrhythmia, and therefore, continuous monitoring of the rhythm and ready availability of defibrillation/resuscitation equipment is a necessity. The agents demonstrated to have efficacy for conversion include class I and class III AADs.
Class I AADs used for acute conversion of AF are flecainide and propafenone, which are typically used in a dosage of 2 mg/kg body weight, given as an infusion for 10 to 20 min. Class I drugs should not be used in the setting of significant structural heart disease such as old myocardial infarction (scar), left ventricular (LV) dysfunction, or LV hypertrophy due to risk of ventricular proarrhythmia. Varying efficacy rates between 50% and 90% have been seen for conversion to sinus rhythm. It has been suggested that flecainide may be more efficacious than propafenone or amiodarone in the acute setting. 21 Intravenous flecainide and propafenone are currently unavailable in India. Another approach would be to load the drugs orally which has been shown to be efficacious, though with a longer time to conversion. 22
Amiodarone, though conventionally classified as a class III agent, has multiple mechanisms of action and has broad efficacy against several arrhythmias. It is generally given as a slow bolus of 150 mg followed by an infusion protocol over the next 24 h. A meta-analysis of studies showed about 75% efficacy rate for AF conversion in 24 h with amiodarone. 23 Amiodarone has comparable overall efficacy with class Ic drugs; however, it is slower acting and conversion often occurs in a delayed fashion. 24 Unlike class I drugs, amiodarone can be used in the presence of structural heart disease and LV dysfunction. Oral amiodarone is even more slow-acting and has low efficacy for immediate conversion, though small studies have suggested higher conversion rates with very high doses (25-30 mg/kg).25 Another class III agent recently made available in India is ibutilide, which has a faster onset of action, with most reversions occurring within 30 to 60 min. The usual dose is 1 mg (0.01 mg/kg if weight < 60 kg) given as an infusion over 10 min, with a repeat dose after a 10-to-20-min interval if sinus rhythm is not achieved. Ibutilide has a 50% to 60% efficacy rate for conversion of new-onset AF and an even better success rate in case of atrial flutter. Ibutilide can cause prolongation of the QT interval and carries a small risk of triggering polymorphic VT. 26 Hence, rhythm monitoring for at least 6 h post administration is essential. Ibutilide infusion should be stopped even before completion of dose if sinus rhythm is achieved or if nonsustained or sustained VT occurs. Preloading with intravenous magnesium prior to ibutilide is recommended to reduce VT risk as well as improve conversion success rate. 27 In addition, combination therapy of ibutilide with other agents has been described to improve success rates. Fragakis et al described that pretreatment with esmolol improved ibutilide conversion rates and also reduced QT prolongation and VT risk. 28 In other studies, addition of propafenone or use of ibutilide in patients already on amiodarone therapy has been reported to improve efficacy,29, 30 Yet another agent vernakalant, demonstrated to have reasonable efficacy for converting recent-onset AF, is currently unavailable in India. 31
Chronic Rhythm Control
Which Atrial Fibrillation Patients Need Rhythm Control?
Many patients with AF can have significant symptoms related to the arrhythmia despite rate control and can hence benefit from a rhythm control strategy aimed at chronic maintenance of sinus rhythm (Figure 1). Current guidelines advocate rhythm control only for relief of AF-related symptoms and improvement of quality of life. 15 Although long proposed, there is no concrete evidence to date that chronic rhythm control improves hard cardiovascular outcomes such as cardiovascular death or stroke. Although the AFFIRM trial showed no overall benefit for rhythm control strategy, some points are noteworthy. The patients studied were relatively older (mean age about 70 years), with comorbidities, had left atrial enlargement (65%), many of them had a relatively low AF burden (where rhythm control is less likely to show benefit), and finally anticoagulation was prematurely stopped in many. Rhythm control is especially likely to be beneficial in younger patients with no comorbidities, recent-onset AF, early in the course of disease with normal left atrium (LA) size. Patient preference for a rhythm control strategy is also important to consider in decision-making. It is also the only strategy when rate control has failed. The advent of catheter ablation for AF has especially reignited this debate as maintenance of sinus rhythm by catheter ablation and avoidance of chronic AAD therapy might prevent the offsetting of the benefits of sinus rhythm by AAD adverse effects (as in the AFFIRM trial). However, evidence to back this premise is still lacking as discussed later below.
Rhythm Control and Anticoagulation
Another popular misconception is that rhythm control can be used to avoid anticoagulation. This is lacking in scientific basis and data from AF trials have not demonstrated that successful rhythm control significantly reduces stroke risk making it safe to discontinue anticoagulation.8, 32, 33 The reasons for this are probably multifactorial. First, confirming that an individual is 100% AF free after institution of a rhythm control strategy is problematic, as short lived, asymptomatic AF episodes are difficult to detect in the absence of continuous monitoring. Such silent episodes can nevertheless pose a risk of stroke. 34 In patients with implanted cardiac devices such as pacemakers and continuous monitoring, even episodes of AF lasting few minutes to hours have been linked to stroke risk. 35 Hence, an apparently successful rhythm control strategy, where a patient experiences major relief from symptomatic AF episodes, may still have a significant residual stroke risk through such silent episodes. This is especially true with regard to drug therapy, where effective maintenance of sinus rhythm may be < 50%.8, 36 However, even with respect to catheter ablation for AF, pooled data from randomized trials have not shown a significant reduction in stroke risk compared to nonablated subjects. 37 In a 2013 meta-analysis of AF ablation studies, late recurrences were seen in up to 20% of cases even after multiple procedures. 38 Second, the AF–stroke relationship is complex, with the classical view of blood stasis in the left atrium due to AF alone, probably reflecting an oversimplified view. The lack of a clear temporal relationship between AF episodes and stroke reflects this. AF is, to some extent, a marker of adverse cardiovascular substrate with associated atrial cardiomyopathy, atherosclerosis, endothelial dysfunction, etc., all contributing to stroke risk, independent of the arrhythmia as such. 39 Therefore, guidelines recommend continuation of anticoagulation solely based on assessment of individual stroke and bleeding risk using standardized scores established for this purpose, irrespective of a rhythm control strategy. 15 Hence, at present, treating clinicians should not recommend AADs or catheter ablation for AF for the purpose of discontinuing anticoagulation. Ongoing studies such as the OCEAN trial hope to further address the question of whether a long-term truly AF-free period can reduce stroke risk adequately enough to make eventual discontinuation of anticoagulation feasible. 40
Strategies for Chronic Rhythm Control
The 2 major strategies for chronic rhythm control are AADs and catheter ablation. Before going into these, it is important to address underlying risk factors which initiate and perpetuate AF.
Management of Risk Factors: Underlying Substrate
Unlike many other supraventricular arrhythmias, AF is often a systemic disease, precipitated and maintained by risk processes which are common to other cardiovascular pathologies as well. Most important among these are systemic hypertension, obesity, and obstructive sleep apnea (OSA). 41 These factors result in adverse effects such as LV hypertrophy, diastolic dysfunction, left atrial enlargement and fibrosis, which in turn serve as substrate for AF. Fixation on AADs/catheter ablation often leads to lack of attention to adequate control of underlying risk factors, which can significantly impede a successful rhythm control strategy. 42 Targeting the underlying processes which result in AF has been referred to as “upstream therapy” which helps both prevent AF as well as improve success of rhythm control strategies in established AF.
Recent work has especially focused on obesity and OSA in relation to AF. OSA is associated with adverse atrial remodeling and increases AF recurrences. 43 Continuous positive airway pressure therapy can effectively ameliorate OSA symptoms and also reduce AF recurrences. 44 Similarly, lifestyle interventions aimed at weight reduction and optimal control of other cardiovascular risk factors are very important and have been shown to reduce AF burden and improve maintenance of sinus rhythm,45, 46 In the latest American College of Cardiology/American Heart Association guidelines, risk factor modification has been accorded a class I recommendation 15 ; however, most of the data supporting this recommendation comes from a single center. While good control of hypertension is in itself beneficial in reducing AF, angiotensin-converting enzyme inhibitors and angiotensin receptor blockers have been proposed to have specific benefits in this regard and it is reasonable to prefer these drugs for hypertension management in the setting of concomitant AF. 47
Anti-arrhythmic Drugs
Similar to acute rhythm control, AADs, mainly belonging to Class I and Class III, can be used to reduce AF recurrences and maintain sinus rhythm on a long-term basis. The choice and dosing of AADs depends on the extent of structural heart disease, and frequency and severity of AF.
The class I agents propafenone and flecainide are available in India in oral formulation and can be used for chronic rhythm control. They have reasonable efficacy in maintaining sinus rhythm33, 48, 49 and can be preferred as first-line agents, given the lack of significant long-term toxicity, such as in case of amiodarone. However, class I agents are contraindicated for use in the presence of significant structural heart disease, including significant coronary artery disease, LV hypertrophy, and LV systolic dysfunction, due to risk of ventricular proarrhythmia,50, 51 The other potential concern with class I agents is that they may slow the arrhythmia cycle length, thereby converting AF to slower atrial flutter, with possibility of 1:1 rapid conduction to the ventricle. This can result in paradoxically faster heart rates, worsening of symptoms, and hemodynamic compromise. Hence, it is prudent to co-prescribe atrioventricular nodal blocking agents such as beta blockers with class I AADs. 52
Class III AADs for treating AF include amiodarone, sotalol, and dofetilide; the last one being unavailable in India. Another agent, dronedarone was shown to be effective, but was withdrawn due to potential for hepatic toxicity, although later analyses suggest that this risk may not be excessive. 53 Amiodarone has better efficacy in maintaining sinus rhythm compared to the class I agents, but has several potential toxicities in the long run, including thyroid, pulmonary, and ocular toxicity. 54 Sotalol can cause QT prolongation, with a small risk for torsades-de-pointes; hence, initiation of therapy should be preferably done in inpatient setting with monitoring of the QT interval.
Pill-in-the-Pocket Approach
A strategy for patients with infrequent, but symptomatic AF episodes is the so-called pill-in-the-pocket approach, where the patient self-doses with a loading dose of flecainide (typically 200-300 mg) or propafenone (typically 450-600 mg) at the onset of a perceived AF episode. This has been shown to be a safe and effective approach with > 90% success rates and further avoids unnecessary daily AAD therapy in the long run. 55
Overall, AADs may work well for chronic rhythm control in a subset of patients, but are hampered by limited efficacy and side effects. Pharmacogenetically directed therapy of specific agents may help select patients better and achieve better efficacy with AADs in the future. 56
Catheter Ablation for Atrial Fibrillation
The seminal discovery by Haissaguerre et al on the role of ectopic foci in pulmonary veins (PV) triggering AF opened the doors for catheter ablation targeting such foci. 57 The field of AF ablation has expanded since then to incorporate not only ablation of AF triggers, but also various methods of ablating the atrial substrate which maintains AF.
Who Is the Right Candidate for AF Ablation?
Ablation has been shown to be superior to AADs for maintenance of sinus rhythm in several studies. Success rates for achievement of sinus rhythm at 1 year have ranged from 70% to 80%, especially for paroxysmal AF,58, 59 Persistent and long-standing persistent AF have lower success rates. The longer the duration of AF, the more structural changes are likely to happen to the atrium, making both achievement and maintenance of sinus rhythm more difficult. A meta-analysis of trials combining all forms of AF showed success rates of 50% to 60% for catheter ablation. Therefore, recurrences after ablation are common, requiring second or sometimes multiple procedures. ,60
Since ablation is theoretically a better method of eliminating AF without adverse effects related to AADs, it has been hoped that it could potentially improve hard cardiovascular outcomes, such as mortality and stroke, beyond relieving AF-related symptoms. Observational, nonrandomized studies have suggested such a potential benefit; however, these are subject to unmeasured confounders and bias,37, 61 The randomized, controlled, CABANA trial, comparing catheter AF ablation with drug therapy did not show an improvement in a composite outcome of cardiovascular death, stroke, cardiac arrest, or major bleeding with ablation. AF recurrence and attendant hospitalization was less frequent, as expected, with the ablation group. 62 Similarly, quality of life measures related to superior relief of AF-related symptoms are reported to be better with ablation,63, 64 Overall, currently there is no concrete evidence that AF ablation improves hard cardiovascular outcomes, although there is low recurrence compared to drugs and quality of life is improved. Further ongoing trials such as the EAST-AFNET-4 trial may bring greater clarity in this regard. 65
Special Situation: AF Ablation in Heart Failure With LV Systolic Dysfunction
AF and heart failure often coexist and together it is a challenging combination to manage. AF can worsen LV systolic function and heart failure, and restoration of sinus rhythm may be particularly beneficial in this setting. Several small studies have reported an improvement of LV function as well as reduction in heart failure symptoms with AF ablation in the setting of reduced ejection fraction,66, 67 Two large randomized trials, namely the AATAC trial and the CASTLE-HF trial, demonstrated improvements in LV function and a reduction in mortality with AF ablation in patients with heart failure,68, 69 Similarly, the CAMERA-MRI study showed greater improvement in LV ejection fraction with catheter ablation compared to rate control alone, particularly in the absence of LV scar. 70 Current guidelines, however, accord only a class IIb recommendation for AF ablation as a means to reduce mortality in heart failure patients, in part, due to concerns regarding the comparator arm and dropouts in the CASTLE-HF trial,15, 71 No such data is available for AF ablation in heart failure with preserved ejection fraction.
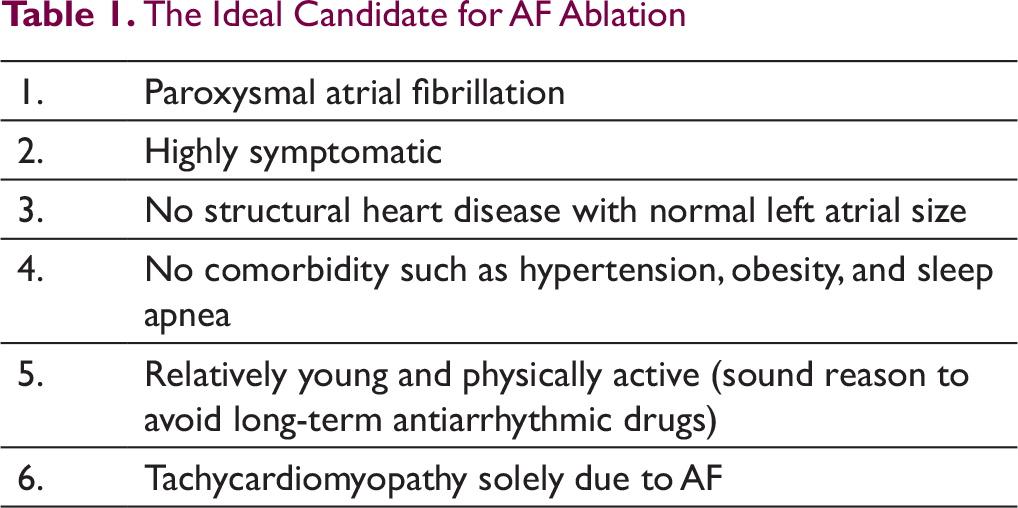
In summary, AF ablation is at present mainly advocated for symptomatic patients with AF who are refractory or intolerant to at least one AAD. 15 It is reasonable as a first-line alternative for selected patients, namely, younger patients with highly symptomatic, paroxysmal AF without structural heart disease and normal LA size, who may not wish to take long-term AADs and in whom success of AF ablation is also likely to be high (Table 1). 72 In patients with the so-called tachy-brady syndrome where atrial arrhythmias (including AF) can alternate with episodes of bradycardia, ablation may be considered upfront, as giving AADs risks worsening bradycardia and syncope. Also in highly active subjects such as athletes, use of AADs leads to heart rate reduction that may lessen exercise capacity and hence ablation can be considered as first option in such cases. Finally, ablation may improve cardiovascular outcomes in select patients with heart failure with reduced LV systolic function, especially if the LV dysfunction is believed to be due to AF-induced tachycardiomyopathy. Although formal guidelines do not recommend AF ablation in asymptomatic individuals, a 2017 Heart Rhythm Society expert consensus statement suggested that ablation may be considered in select asymptomatic patients with paroxysmal or persistent AF when performed by an experienced operator following a detailed discussion of the risks and benefits. This recommendation was made before the publication of the CABANA trial, based on the possibility of a potential benefit toward stroke or mortality reduction. 73
The Ideal Candidate for AF Ablation
Special Situation: Rheumatic Heart Disease
In the Indian context, AF in patients with rheumatic heart disease is an important issue to consider, which has not been well studied overall, as rheumatic heart disease is not prevalent in the West. Rheumatic mitral stenosis is associated with LA dilatation and fibrosis. As such rheumatic heart disease-associated AF is more difficult to treat in view of the significant substrate. In a small study, Vora et al reported about 70% success in maintaining SR with AADs with improvement in exercise capacity and quality of life; however, > 70% of patients had undergone valvular intervention. 74 Indeed performing early balloon mitral valvotomy may be important as rheumatic mitral stenosis-induced atrial stretch leads to significant adverse electrophysiologic remodeling in the LA which can be reversed by balloon mitral valvotomy. 75 Limited studies have suggested improved success when either AAD or catheter ablation is combined with a strategy of balloon mitral valvotomy.76, 77
Techniques in AF Ablation
AF ablation is mostly performed within the LA and techniques can be broadly divided into ablation of triggers which initiate AF (PV and non-PV foci) and ablation of atrial substrate which maintains AF. While the former is always done, substrate ablation is generally reserved for more persistent forms of AF and its added utility remains unclear.
Imaging in AF Ablation
Pre- and periprocedure imaging has an important role in AF ablation. Computed tomographic scan of the LA with high resolution is commonly performed prior, both to look for LA clot and have foreknowledge of the PV anatomy. Usually, during ablation, a 3-dimensional shell of the LA is created, aided by electroanatomic mapping systems and merged with the preprocedural computed tomographic scan to have optimal anatomic guidance for LA ablation. Intraprocedural echocardiography, either using TEE or intracardiac echocardiography, is often used which is again useful to rule out LA thrombus just before the procedure, to guide atrial septal puncture for LA access and for early detection of any intraprocedural pericardial effusion due to chamber perforation. Intracardiac echocardiography is almost universally used in the USA to avoid the need for full general anesthesia and endotracheal intubation needed for the use of TEE; however, it is considerably more expensive than TEE. Performance of AF ablation with near zero fluoroscopy using exclusive intracardiac echocardiography guidance has also been described to avoid radiation. 78 Another described role of imaging is to quantify LA fibrosis (which portends a poorer prognosis) using contrast-enhanced magnetic resonance imaging. The Delayed Enhanced MRI Determinant of Successful Radiofrequency Catheter Ablation of Atrial Fibrillation (DECAAF) study revealed that extent of atrial fibrosis detected by magnetic resonance imaging was independently linked to recurrence of AF. 79
Ablation of Trigger: Pulmonary Vein Isolation and Non-PV Sources
Pulmonary vein isolation (PVI) remains the cornerstone of AF ablation (Figure 2). With the discovery of ectopic PV foci triggering AF, initial strategies involved limited radiofrequency (RF) ablation within the PVs to eliminate such foci. 58 However, since ablation within the veins could result in PV stenosis, isolation of the entire PV from the LA using wide antral circumferential ablation became the method of choice, encompassing all 4 PVs to take care of potential new sources developing in the future.80, 81
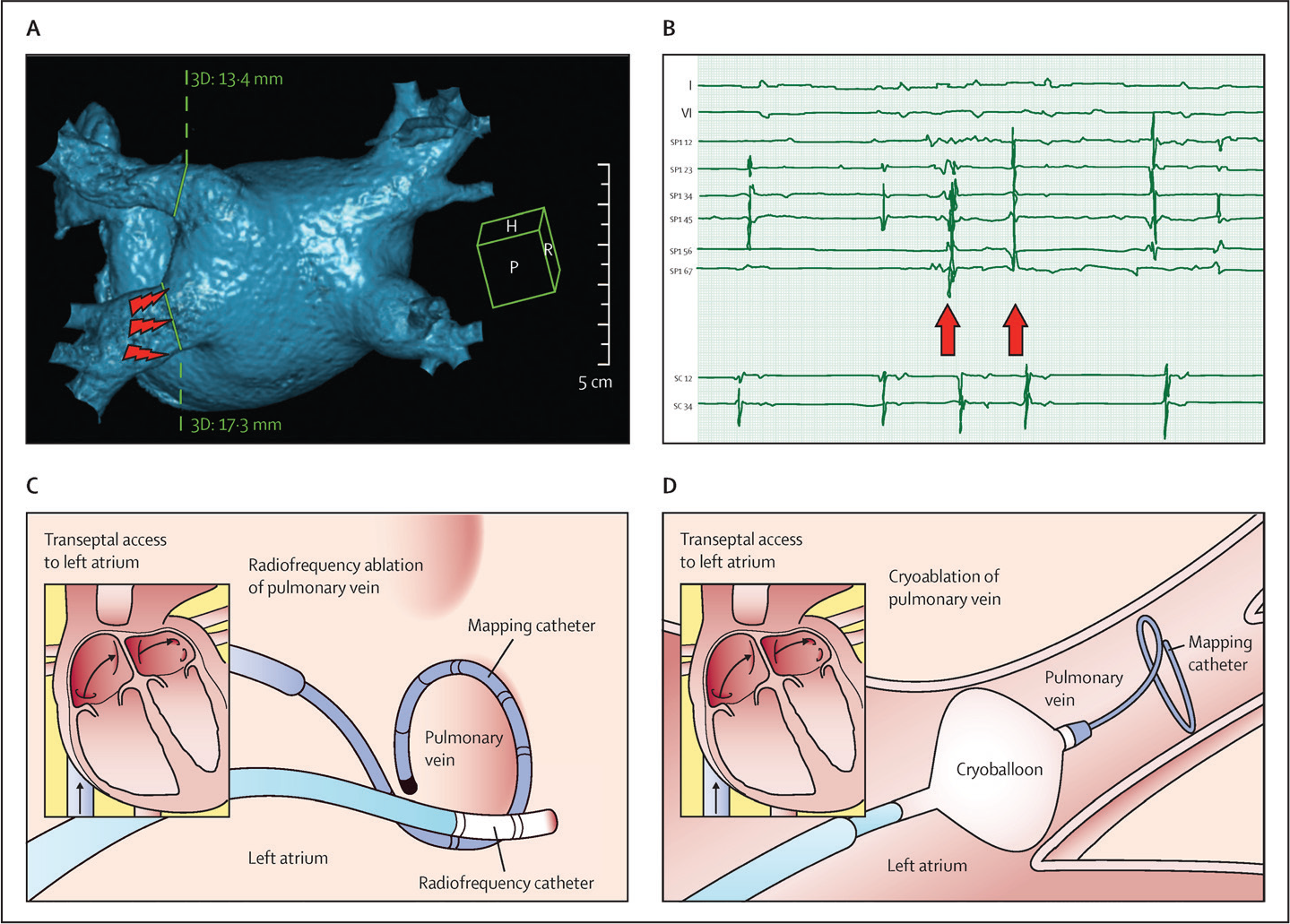
PVI is commonly done using point-by-point ablation around the PV ostia using RF energy; although, some newer techniques, especially the cryoballoon (see later), have emerged. It is important to achieve reliable and durable PVI, as the most common reason for AF recurrence post ablation is PV electrical reconnection. 82 Durable PVI depends in turn on making effective transmural (spanning entire myocardial thickness) lesions. Studies have shown that lack of adequate tissue contact can be an important reason for not achieving a good lesion. Hence, catheters incorporating contact force information using pressure sensors at the tip are widely used now and these have been reported to improve success rates. 83 A systematic protocol (CLOSE protocol) aiming for close, contiguous lesions ( < 6 mm interlesion distance) with adequate power, duration, and contact force (ablation index) has been described to further improve ablation success. 84
The acute procedural end point of PVI is complete isolation of all 4 PVs. A circular multi-electrode catheter is commonly used at the PV ostium (Figure 2) to record the PV signals. Disappearance of these signals (“silent” PV) or their dissociation from the LA signals is the end point for PVI. For the latter end point, it is usual to test for bidirectional block between the PV and LA by pacing maneuvers. Intravenous adenosine testing to bring out any dormant residual connections between PV and LA, with further ablation to abolish such residual connections, has been shown to improve success. 85
In a small proportion of patients, AF is triggered by ectopic activity from non-PV sources. Such sources include the superior vena cava (SVC), coronary sinus, ligament of Marshall, Crista terminalis, and the posterior left atrial wall. 86 When AF does not terminate or recurs despite properly confirmed PVI, extra-PV triggers should be sought and ablated. Isoproterenol infusion helps bring out ectopic activity from extra-PV triggers. 87 The SVC is a common source of extra-PV triggers and systematic isolation of the SVC has been shown to be beneficial, especially in paroxysmal AF; however, SVC isolation may not be possible to achieve in all patients due to the proximity of the right phrenic nerve and sinoatrial node. 88
Ablation of Atrial Substrate
While PVI is almost universally performed, several other adjunct ablation techniques have been described that aim at disrupting the atrial substrate which is responsible for maintaining and perpetuating AF. These techniques, in a sense, are similar in basis to the Cox-Maze surgery for AF, which, through multiple incisions (or more recently RF or cryo) in the atria, aims to produce lines of electrical interruption that would make it difficult for reentrant circuits to sustain. 89 Most strategies for persistent AF are empiric addition of lesions to PVI. The most commonly performed adjunct ablations are creation of ablation lines in the LA, cavotricuspid isthmus, and ablation of complex fractionated atrial electrograms within the LA. Examples of linear ablation lines in LA include roof line (connecting superior PVs), mitral isthmus line, and “box” isolation of LA posterior wall using RF lines connecting the PVI circles superiorly and inferiorly along the posterior wall. Some others have described ablating low voltage areas within the LA. 90 The LA appendage is a complex structure with important physiologic and electrical properties that has been implicated in the genesis/maintenance of AF. Electrical isolation of the LA appendage through catheter ablation, surgical excision, or percutaneous ligation has been reported to reduce AF recurrence. One concern with pure electrical isolation (by ablation) is risk of stroke due to stasis of blood and this approach needs further study. 91 While several groups claim improved success rates with their specific techniques of adjunct ablation, systematic evidence for an added benefit over and above PVI is lacking. 92 The STAR AF II trial, which randomized patients with persistent AF to PVI alone versus PVI plus additional substrate ablation, failed to show a significant difference in outcomes with added ablation. 93 Moreover, extensive ablation could also be potentially proarrhythmic, resulting in complex LA flutters due to gaps in ablation lines. Thus, ablation of persistent forms of AF with extensive substrate ablation strategies is plagued by both higher rates of AF recurrence and occurrence of new flutters which may require multiple follow-up procedures. 94
Another described approach has been ablation of so-called rotors driving AF, which suggests that AF may be maintained by localized rotors and focal impulses rather than completely random, chaotic reentrant fronts. A specialized mapping system developed to identify such rotors was reported to have good success initially 95 but could not be replicated in subsequent studies. 96
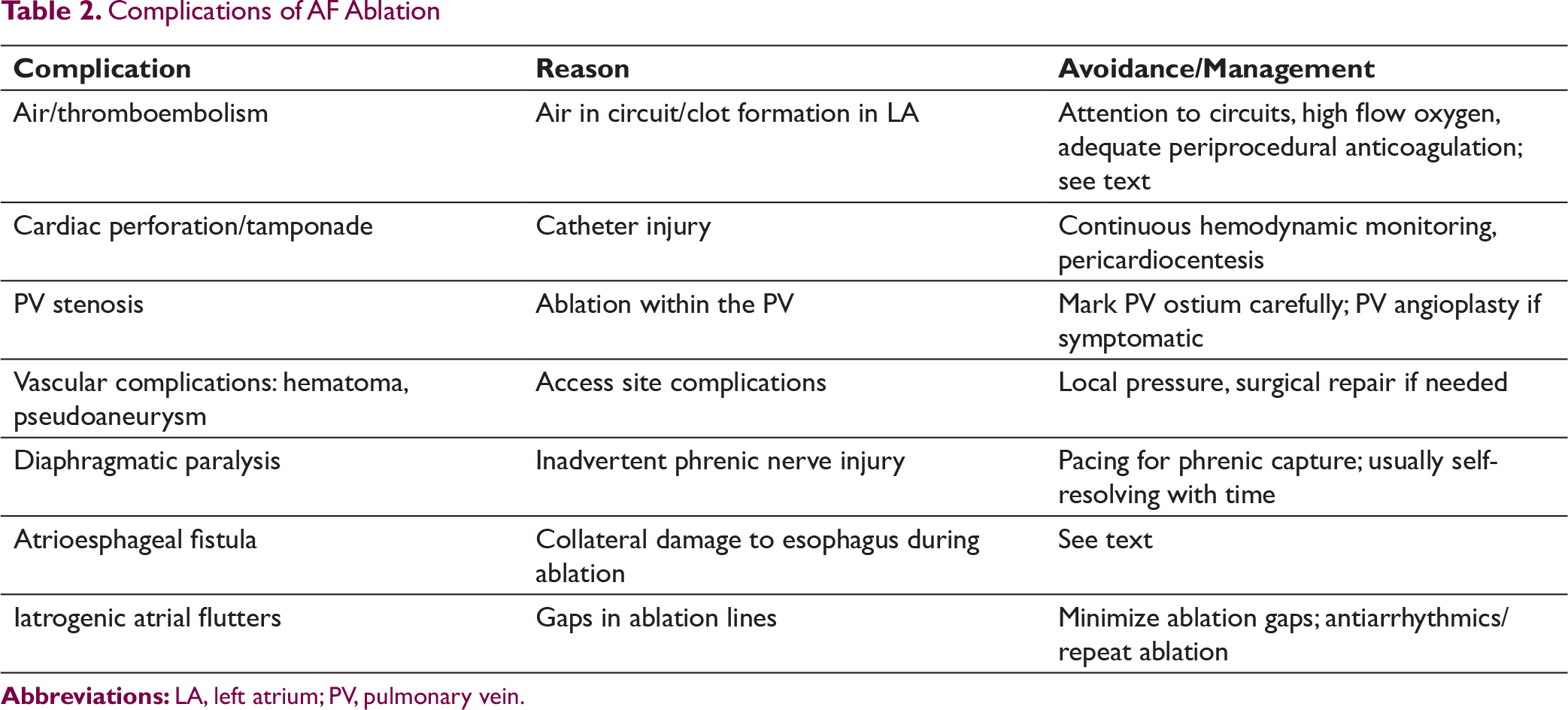
Complications of AF Ablation
It is important to bear in mind that AF ablation has a small, but nonnegligible risk (5% to 7%) of serious adverse events. Possible complications of AF ablation are listed in Table 2. Periprocedural embolic stroke is a major issue, which should be avoided by ensuring no LA/appendage clot by imaging, meticulous attention to intraprocedural anticoagulation (activated clotting time > 300 s), and using irrigated ablation, minimizing chances of charring at catheter tip. It is now recommended to perform AF ablation without interruption of oral anticoagulation, both vitamin K antagonists and novel oral anticoagulants. 75 Phrenic nerve injury can occur as the right phrenic nerve is anatomically close to SVC and right superior pulmonary vein, while left phrenic nerve is related to LA appendage. Prior to ablation in these areas, high-output pacing should be performed to look for diaphragmatic capture and if present, RF delivery should be avoided or energy reduced. Phrenic nerve injury tends to recover spontaneously in most cases. 97 Atrioesophageal fistula (due to the close relationship between the LA and esophagus) is a dreaded and largely lethal complication. Monitoring of esophageal luminal temperature with a probe, especially while ablating on the posterior LA wall, with discontinuation of ablation if there is ≥ 1°C temperature rise, is followed by many centers. Strategies to reduce esophageal thermal injury include esophageal deviation using preshaped deviators and luminal cooling methods.64, 98-100 The risk of complications could be potentially even higher with less experienced operators and centers. 101 Hence, a practical framework for decision-making regarding AF ablation has to take into account the issues of recurrence, procedural risk, cost, and balance it against the expected benefit from the procedure, which depends upon the extent of morbidity caused by AF in a given case.
Complications of AF Ablation
Newer Technologies and Approaches in AF Ablation
Alternate energy sources, other than RF, have been explored for AF ablation. The most widely implemented so far in clinical practice has been cryoenergy, in the form of the cryoballoon (Figure 2). In this approach, a balloon is inflated flush against the PV ostia and freezing temperature delivered through the use of liquid nitrogen in the balloon. This freezes and ablates a circumferential zone of tissue around the PV, thus achieving a “one-shot” PV isolation compared to the more cumbersome point-by-point RF ablation. The cryoballoon can potentially simplify the procedure and reduce ablation time. Comparative studies show equivalent efficacy of the cryoballoon compared to traditional RF, with reduced procedural time and similar complication rates, except for right phrenic nerve palsy, which is reported to be higher with the cryoballoon, though mostly transient in nature.102, 103
Similarly, thermal balloons and laser balloons 104 have been used, as well as a circumferential RF catheter which can deliver RF at multiple points at once. Advanced imaging and remote navigation/robotic technologies are also being utilized; all the aforementioned techniques attempt to both improve efficacy as well as simplify the procedure and make it less labor-intensive. 105 The balloon-based and remote navigation ablation technologies are currently unavailable in India.
Another area of active research, not only for AF, but also for other arrhythmias as well, is the role of neuromodulation. It is well known that the autonomic nervous system has an important facilitatory role in the pathogenesis of various arrhythmias, and the use of sympathectomy to treat VTs is one example of a therapeutic application. 106 The LA is richly innervated with autonomic ganglia and considerable interest has focused on the role of modifying or ablating epicardial ganglionic plexi to treat AF. Interestingly, inadvertent achievement of axonal interruption of connections from ganglionic plexi to PVs, during PVI has been proposed to be at least partly contributory to the success of PVI as a means of AF ablation. 107 Stimulation of the vagus, using low-level tragus stimulation has been shown to suppress AF and reduce levels of inflammatory cytokines. 108
A newer and innovative approach is irreversible electroporation. This is the application of an electrical field, using DC energy, to create irreversible pores in cell membranes, thereby leading to cell death. Hence, electroporation relies on nonthermal damage unlike RF. In addition, the electrical threshold for electroporation has been seen to differ by cell type, thus opening up the promise of delivering tissue-specific ablation and avoiding collateral damage to close, crucial structures such as PVs, esophagus, and coronary arteries. Ongoing research in this field could open up new horizons in catheter ablation. 109
Lastly, mobile and wearable heart monitors including smartwatches, bands etc. are increasingly being studied. Mobile sensors capable of generating medical grade electrocardiograms have been developed. Use of such devices can greatly improve diagnostic capabilities and early detection of recurrent AF after ablation. 110
Surgical AF Ablation and Hybrid Strategies
As mentioned before, the traditional cut-and-sew Maze procedure uses multiple incisions within the atria, isolating the PVs, appendages, as well as creating additional block lines to the mitral annulus, coronary sinus etc., all of which make it difficult for reentrant circuits to sustain. While long-term success rates have been reported to be good, the traditional Maze procedure is complex with a significant risk of complications. Recently, RF energy, cryo probe, and even diathermy have been used as alternatives to surgical incisions, potentially simplifying the procedure. Surgical Maze is difficult to recommend as a standalone procedure, but should be strongly considered in patients with AF undergoing other cardiac surgery. 111 Newer minimally invasive approaches have been developed, using thoracoscopy and specially designed clamps which can provide single-shot PVI. In addition, hybrid approaches are also being tried that combine traditional endocardial catheter ablation with thoracoscopic minimally invasive surgical ablation. 112 The long-term efficacy and safety of such techniques need to be determined.
Finally, another “hybrid” strategy is to combine AF ablation with AADs. While traditionally, ablation for arrhythmias is performed with an expectation of stopping all antiarrhythmic medications post procedure, this may not always be optimal for a complex arrhythmia such as AF. Studies have suggested that continuing antiarrhythmics post ablation can lead to longer AF-free periods, reducing recurrence and need for repeat ablation. Moreover, drugs previously ineffective at maintaining sinus rhythm may be effective after modification of triggers and substrate by AF ablation. 113 Hence, such a hybrid strategy warrants consideration in some patients.
Conclusions and Future Directions
AF is a multifactorial arrhythmia encountered not only by cardiologists but by almost all physicians at some point. A rhythm control strategy aims to achieve and maintain sinus rhythm and is especially warranted in patients with significant symptoms or LV dysfunction. AF ablation, targeted at both triggers of AF as well as substrate modification, has emerged as a promising modality to achieve sinus rhythm, sans the adverse effects of AADs. However, concrete evidence for an improvement in hard outcomes via ablation, such as cardiovascular mortality and stroke, is lacking; and at present, ablation is mainly recommended for symptomatic patients who are refractory or intolerant to AADs. Significant rates of recurrence and periprocedural complications constitute important limitations of current AF ablation strategies. Newer modalities such as electroporation, neuromodulation, and hybrid surgical approaches may improve AF rhythm control strategies in the near future. Finally, the importance of adequate anticoagulation for stroke prevention, in at-risk patients, has to be always borne in mind, irrespective of the rhythm or rate control strategy used in AF.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
