Abstract
Background
Providing accessible information during informed consent practice helps people make important decisions about research participation. The aim of this review is to critically appraise the current Australian clinical research guidelines to assess their guidance on delivering accessible informed consent processes for people with vision impairment.
Methods
An archival search of the Overton database was conducted for policy documents and guidelines pertaining to informed consent practice in Australia. This was cross checked with the Australian Research Council’s online catalogue of national codes and guidelines. Content analysis was performed using an implementation science framework to code the actors, actions, context, target and time (AACTT) elements of informed consent practice. The AACTT framework enables a detailed specification of the behaviours targeted for championing or change.
Results
Ten policy documents specific to conducting consent procedures for clinical research or healthcare participation in Australia were identified and included in this review. Five of the 10 guidelines provided sufficient detail for researchers to know what actions to perform to enable inclusive consent practice and mutual engagement with people with vision impairment. However, they varied in their content describing how and when to perform them. Two of the key reference documents for clinical research conduct in Australia entirely lacked recommendations for accessible informed consent practice.
Conclusions
Australian research policy and practice documents offer some direction in helping researchers to deliver accessible informed consent practice. However, guidelines produced by peak disability bodies and patient advocacy groups offer more direction on how to engage people with vision impairment and offer useful templates for producing accessible print materials.
Keywords
Background
Accessible communication and information provision by researchers is paramount to supporting an equitable approach to informed consent practice. Informed consent is achieved through a process of timely communication, discussion and shared decision making.1,2 Informed consent embodies a person’s voluntary decision to be involved in a research study/trial based on knowledge and a good understanding of what is involved. 3 Traditionally this process involves research candidates being provided study information in a written document that explains the fundamental elements of the study, including the benefits and risks of taking part, and the requirements such as time commitment. Referred to as a Participant Information and Consent Form or PICF in Australia (also called a Plain Language Statement or PLS), this document serves to outline and meet the Australian code for ethical and responsible conduct for obtaining informed consent. 4 The document must be easy to read and understand, along with meeting the target group’s health literacy level.
Meeting the target’s group health literacy level when writing a PICF/PLS is of particular importance given that 44% of Australians have low literacy, equating to 8 years or less of schooling.5–7 In recognition, the National Statement on Ethical Conduct in Human Research mandates that PICF/PLS documents are “grade 8 level” or literacy level 2 or below to encourage engagement and understandability.8–10 Having information that is in plain language, and in Easy-Read and accessible format is also important for information accessibility. Plain language uses everyday words and clear active sentences with Easy Read extending accessibility by presenting short sentences (i.e. under 16 words per sentence) accompanied by a meaningful picture for each main point. 11
In Australia, 1 in 5 people (20%) have a disability and around 1 in 17 people (6%) have vision impairment. 12 Individuals with vision impairment, encompassing both low vision and blindness, require printed information in accessible formats to reduce the challenge and visual demand from trying to read small print. Without due consideration of vision ability, there is the chance that inaccessible information could contribute to difficulties reading print, a cause for print disability. Furthermore, if information providers do not offer accessible information options, they may potentially be denying a person access to that information.13–15 In the context of clinical research, this has implications for determining whether someone is adequately informed and has been given an equitable process for receiving information and making decisions as appropriate. Signing a consent document or agreement may serve as evidence of consent but may not adequately capture the process nor capacity of the person responsible for providing consent. Providing information that is accessible to people with low vision or blindness means meeting both visual and print accessibility needs through all communications.
Rationale
The literature around information accessibility practices taken during informed consent procedures involving people with vision impairment is sparse. 13 A small number of studies have reported the perceived benefits of having printed materials in larger font within select health care 16 and research settings. 17 Few studies also report the issues faced if printed materials are not accessible for people with vision impairment such as incorrect medication adherence.18,19 These studies do not include investigation of informed consent processes, in either medical or research settings, involving researchers (those conducting or facilitating informed consent procedures i.e., investigators and study coordinators) and prospective research participants. However, a recent study has indicated that people with vision impairment find accessing information about research in video and audio formats helpful. 20 This study helps to build an understanding of the information needs of people with vision impairment for research participation. Broadly, presenting information in audio-visual and digital formats has been shown to aid understanding and decision making during the informed consent process.21,22 Yet despite this evidence, there remains limited implementation and routine adoption of alternative information formats, fueling a renewed call to increase inclusive informed consent strategies, particularly for people with disability including those with low vision or blindness.23–26
The benefits of increasing access, equity and inclusion in health research are clear, with diversity encouraging applicability of results across all of society and having positive health and economic impacts for society over time.27,28 Given the paucity of evidence in the literature around what inclusive communication and consent practices are being used by researchers to support information accessibility for people with vision impairment, an investigation is warranted. 13 Furthermore, the strategies and interventions being used by researchers to support communication practices within clinical research practices are largely under-reported in countries including Australia 29 and the United Kingdom. 30 It is likely that researchers are tailoring their informed consent practices to meet participant needs, but the question arises, do they have the knowledge, skills and tools to be able to confidently tailor their practice? Are there sufficient guidelines, policies and resources available to assist researchers with adapting their practice as required?
Objectives
To address this evidence gap, it is important to firstly understand if researchers know what actions to take, how to take them and in what circumstances. This review will answer these questions and use an implementation science approach to demonstrate any knowledge and practice gaps in Australia. The action, actor, context, target and time (AACTT) implementation framework offers a systematic approach for clarifying “who” should be doing “what” with “whom” in “what” setting and “when”. 31 The AACTT framework has been reported as an effective method to review practice standards in the Australian healthcare setting.32,33 Globally, it has also been used widely in evaluating staff behaviours in hospital, research and community health settings.34–36 Previous studies using the AACTT framework have suggested that it is particularly useful in interrogating clinical practice guidelines to ensure there is enough clarity about what behaviours should be championed or highlighted for change.37–39
In Australia, Good Clinical Practice certification involves training across government endorsed guidelines and recommendations for ethical and professional research conduct. It is imperative that these documents adequately educate staff on how to deliver an optimal informed consent procedure to suit the information and communication needs of prospective participants. There is a need to identify how the current national guidelines for ethical conduct in Australia educate and inform researchers on inclusive consent practices. Therefore, this scoping review aimed to critically appraise the current Australian clinical research guidelines to assess their guidance for delivering accessible informed consent processes for people with vision impairment. The AACTT framework will be used to explore and appraise what researchers (actors) are recommended to do (actions) to support accessible communication practices with people who have vision impairment (target) during the informed consent process (context, time).
Methods
Design
This scoping review is the first phase of an implementation development project (Open Science Framework (OSF) registration identifier: DOI 10.17605/OSF.IO/B4ZWA) that aims to identify methods to enhance information access during the informed consent process for clinical research. The review employed a quantitative policy record audit and documentary analysis (Part 1) followed by a deductive content analysis and appraisal of included documents using the AACTT framework (Part 2). The research team conducting this review consisted of three authors (FOH, MK and LA) who are clinician researchers that engage with people with various levels of vision impairment and those with dual vision-hearing loss. One author (DF) is an access and inclusion specialist, one author (CO) works in health professional training and library services, and the two remaining authors are researchers with special interest in implementation, evaluation and participant engagement (TS, CP). Two authors have lived experience of disability, involving vision or hearing. Reporting of this scoping review has been guided by the PRISMA extension for scoping reviews (PRISMA-ScR) checklist. 40
Part 1: Review of informed consent policies and guidelines
Database search
The first step involved conducting a database and online catalogue search using archival research methods.41,42 This meant applying methods to facilitate the investigation of policy documents and records specific to an informed consent procedure. The Overton database, hosted by the University of Melbourne, was used to conduct the database search. We selected the Overton database as it is the largest searchable grey literature and policy document database. It covers diverse regions and coverage of relevant document types including guidelines, white papers, and publications from government, non-government organizations and think tanks. There is a growing body of evidence to support the utilization of the Overton database to identify relevant policy documents and grey literature.43–46 The search strategy was developed by two authors (FOH, CO) with key search terms and Boolean operators being “informed consent” or “research” and “Australian policy” and “disability” (see Supplement 1 for database search strategy).
Other information sources
Grey literature was searched from Australian Government websites. Peak professional bodies known to produce national clinical research guidelines in Australia were searched for recommendations regarding informed consent practice and/or responsible research conduct. This was cross checked with reference to the Australian Research Council’s online catalogue of national codes and guidelines in Australia. 47 These were the National Health and Medical Research Council’s National Statement on the Ethical Conduct in Human Research,8,10 The Australian Code for Responsible Conduct of Research 4 and the Australian Health Practitioner Regulation Agency (Ahpra) codes of conduct for both doctors and health professionals.48,49 Additionally, the National Safety and Quality Health Service (NSQHS) Standards 50 report and the related Australian Charter of Health Care Rights 51 were reviewed for content specific to informed consent practices. The Australian Commission on Safety and Quality in Health Care online website was also searched for ancestral resources and guides. 52
Eligibility criteria
Documents were included for review and analysis if they provided information pertaining to conducting an informed consent process in research and/or healthcare settings in Australia. Documents were excluded if they were aimed at isolated targets such as hospital staff working at specific hospitals, or members of a State-based medical profession. In other words, they needed to be national guidelines and recommendations applicable across clinical research settings and professions.
Part 2: Critical appraisal framework
Appraisal of individual sources of evidence
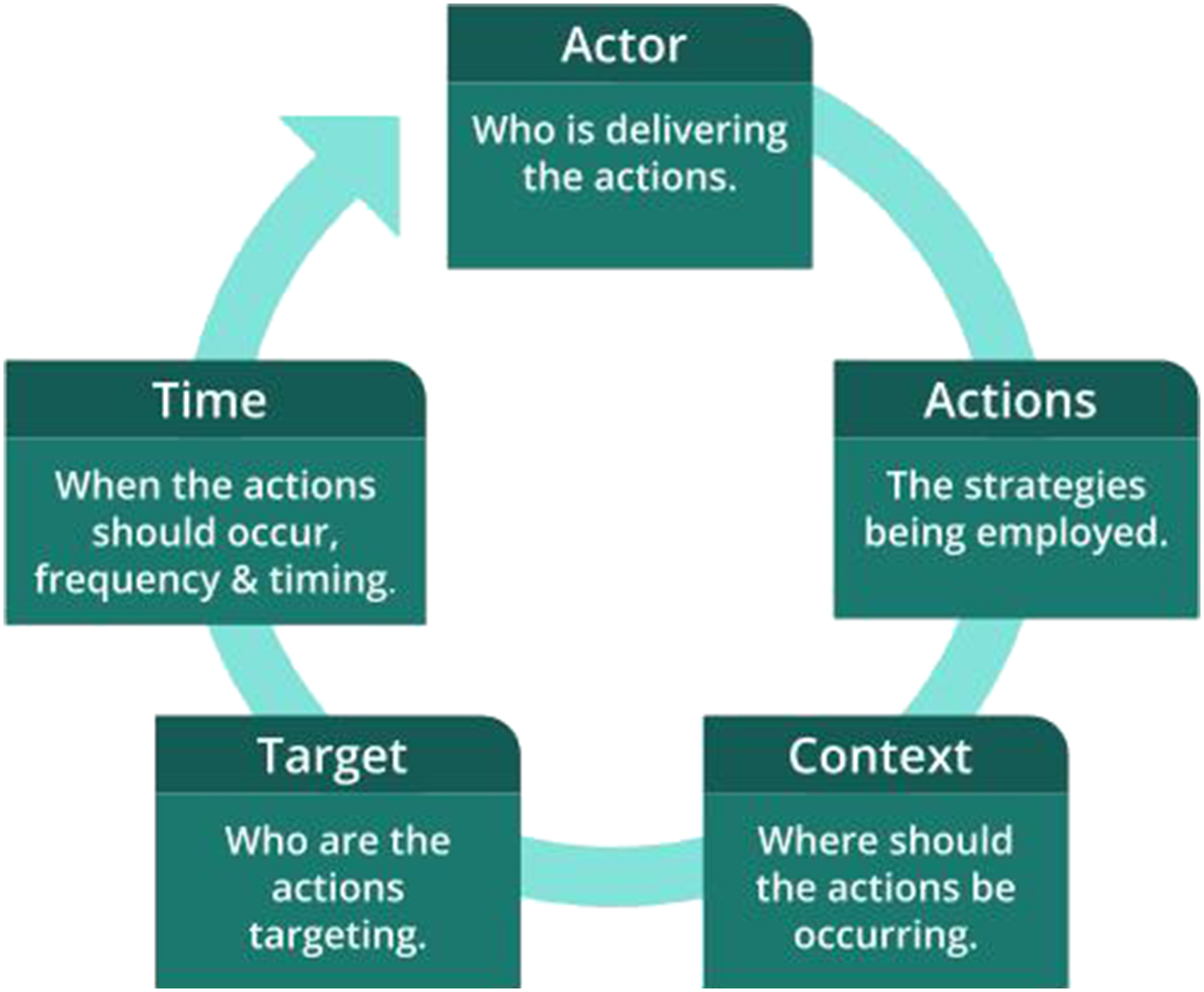
The review used the AACTT framework to explore local polices and clinical guidelines. The AACTT coding framework was developed by author FOH and reviewed by all authors (see Figure 1 and Supplement File 2).
31
The coding framework was based on its use in a previous study exploring oral health policies and implementation of practices to support oral hygiene care.
32
In the context of the informed consent process, the actor is the individual/s performing the consent procedure, and the actions or behaviours are the strategies used by these individual/s to ensure that information presented is accessible. Context may include settings such as online, face to face or over the phone and may also include physical settings such as within a research clinic. Target is the individual/s for whom the actor performs the action, and includes all parties involved. Time is the period or point in the informed consent process where the actions are performed. The Actor, Actions, Context, Target and Time (AACTT) framework.
31
To assist the appraisal of included documents, a data extraction template form, in Microsoft excel format, was developed by one reviewer (FOH) and included document title, year of publication, selected text, text reference details such as recommendation number and page number, and AACTT ratings. Two authors (FOH and DF) independently performed data extraction and each document was then rated using a three-tier rating scale (0, 1, 2) for each of the five elements of the AACTT framework. 31 Discrepancies were discussed with the involvement of a third author (MK) until an agreement was reached. As an example, for actions, a score of 2 was obtained if a list of actions were stated that referred to providing accessible information to participants. The actions needed to describe not just what to provide, but how to present accessible information to participants (i.e., what researchers need to do to ensure that their consent practice aligns with the behaviours that support an equitable and inclusive practice).
Deductive content analysis was conducted to identify principles to guide clear behaviours that supported adapting communications and information provision with people that have vision impairment. The purpose of this step of the review was to identify instructional guidance that helped inform researchers on planning and preparing accessible communications and information to support their informed consent practice.
The behaviours that support an inclusive process during the informed consent process for people with vision impairment were deemed as follows: • Assessment of capacity to see and be able to read written information. • Assessment of information needs specific to vision and individual preferences for communication. • Interventions that support adapting the way information is provided or received i.e. provision of information in multiple accessible formats such as enlarged font version of printed materials. • Interventions that support research staff training and education in communication practices and/or consent practices. • Interventions that support shared decision making, agency, engagement and/or empowerment in the informed consent process.
The scope of the informed consent behaviours was agreed on by all authors who collectively have a research focus on equity and inclusion in the informed consent procedure for diverse populations.
Results
Characteristics and results of individual sources of evidence
Overview of the document details included in the review.

National Health and Medical Research Council, NHMRC; Australian Research Council, ARC; Research Development Impact, RDI; International Conference on Harmonisation's Good Clinical Practice guidelines, ICH-GCP; Therapeutic Goods Association of Australia, TGA.
Analysis of adaptive and accessible practices for engaging people with vision impairment during an informed consent process using the AACTT framework (listed in publication order by year).

*The Australian Clinical Trials Handbook (2021) is a related document but not specifically included in this table as it does not mention informed consent practice (and was therefore excluded). Similarly, 14 other full texts were reviewed but excluded as they did not meet the eligibility criteria, namely did not mention informed consent processes.
**ICH, International Council for Harmonisation of technical Requirements for pharmaceuticals for human use. TGA, Therapeutic Goods Association. Footnote: To ensure that Australia meets international guidelines, the Therapeutic Goods Association (TGA) annotated the Guideline for Good Clinical Practice to ensure it reflected Australia’s specific regulatory and governance requirements. 28 Whilst not developed for Australia, the International Council for Harmonization of technical requirements for pharmaceuticals for human use (ICH) Good Clinical Practice 9 guide was included in this review. Rather than having a separate document, the TGA annotated step version 4, published in 2016 and this has been referred to as the E6 (R2) TGA annotated version. A more recent version of the ICH-GCP guide was identified in the search and was published on 06 January 2025, referred to as E6 R3 9 However, the TGA had not annotated this latest amendment at the time of this review (both R2 and R3 are included and appraised in Supplemental File 2).
From highest to lowest rated documents, the Research Development Impact Network, “Research for All: Making Development Research Inclusive for People with Disabilities” report (referred to as ‘Research for All report’ herein) was the highest scoring and obtained a rating of “2” across all categories. 54 This was followed by the Joint Commission report on Informed Consent Safety Actions (2022) which obtained a rating “2” on four of the five AACTT framework categories (excluding the category actor, rated ‘1’). 55 The third leading document to score a “2” across two AACTT categories (for context and target) was the National Statement on Ethical Conduct in Human Research (2023) (referred to as the ‘National Statement’ herein). 56 The AACTT framework category that had the highest frequency of rating “2” across the included documents was the “actions” category, namely four out of 10 included documents. Conversely, the “context” category had the lowest ratings overall with five documents scoring “0” for this element, namely five out of 10 included documents. Refer to Supplemental file 2 for full description of the AACTT framework for each category for each included document.
Key examples of top rated AACTT framework components across policy and guideline documents.

Discussion
Summary of key findings
This review performed a critical appraisal of Australian policy and research practice guidelines using an implementation science framework. The AACTT framework was applied in this review to help define what professional behaviours are needed to deliver an inclusive informed consent process with people who have vision impairment. This review included 10 primary documents that pertain to national policy and practice for conducting an informed consent process. Five reference guides (half) specifically mentioned how to tailor information and ‘provide information in ways that meet the needs’ of participants with vision impairment. Overall, there was a lack of specificity, particularly in relation to who should be delivering the informed consent process, at what time and in what context within the included documents.
Referring to the AACTT framework can help identify the gaps in understanding and inform areas for more focused education and support. 59 We found that there needs to be greater specificity around the ‘actions’ that the ‘actors’ need to take to enhance accessibility during informed consent practices for people with vision impairment. There is under-representation of information specific to people with vision impairment across the documents included in this review. However, there is a trend toward greater breadth and focus of information around accessibility principles in the more recently published documents. For example, the National Statement 2025 update has an entire chapter that describes engaging with people with disability (Chapter 5). This observation is in parallel with the leading international guideline, ICH-GCP, adopted in January this year. Specific to all prospective participants in research, the ICH-GCP guideline states that “varied approaches such as images, videos and interactive methods may be used in the informed consent process” (Rec 2.8.1). The international guideline is more specific than Australia’s National Statement for outlining accessible actions for consent practice. Currently, researchers would need to look outside the Australian Code, National Charter for Healthcare Rights and Australia’s professional best practice guides (Aphra guides) for information specific to ‘how’ to adapt communications and information provision for people with vision impairment.
How do researchers tailor their practice to meet the needs of people with vision impairment?
This review has identified other guides from the disability sector that provide explicit direction for researchers on how to produce accessible information. Specifically, the Good Clinical Practice Guide for engaging with people with disability and the RDI Network ‘Research for All’ report both provide consent form templates, digital and print templates and tips for engaging with people with vision impairment (see resources and templates throughout reference 58 and 54 such as Appendixes 4–6). Additionally, the Blind Citizens Health Healthcare Policy also provides specific guidance on creating accessible information and communication for people who are vision impaired, as well as recommendations for how to tailor and ensure the environment also meets the needs of people. 57 These documents highlight inclusive practices that involve adopting multimedia approaches, including using audio formats, which is a key method that have been shown to be positively viewed by people with vision impairment undergoing an informed consent procedure.13,21 A range of tools are now readily available, and the number is increasing with progression in generative artificial intelligence which has the potential to assist both the researcher and the participant with readability of information and methods for communication. 60 Starting with a well written accessible print document, such as the PICF, can help prepare the script for an audio narration. However, implementing audio formats, and any multimedia information consent supplements, requires additional resources, infrastructure and time. 21 This necessitates early planning, budget and identification of people who have the necessary skills and experience to assist with producing these formats.
Remaining practice gaps and areas for future progress
For researchers to know how to provide information in varied accessible formats, there needs to be support for skills, knowledge and capacity building. Implementation strategies that could assist with this include, but are not limited to, dedicated funding for accessibility actions, creating a learning collaborative, recruiting and cultivating relationships with partners in the implementation effort (i.e., digital accessibility experts, consumers), develop and share educational materials, build and share high-quality multimedia information templates across networks, and mandate practice change. 61 Knowing what supports are available, and how to access and offer them, is a vital part of the responsible research conduct framework that is currently missing. Given the reliance on researchers to augment their consent practice, there is a need to describe the actions (behaviours) as clearly as possible. This review highlights the need to clarify the roles and responsibilities of the people involved, the organization and the professional sector.
If researchers were adequately prepared with the tools and templates to deliver an equitable and inclusive informed consent process, research participants would be in a better position to access the information they need to make an informed choice about participating. This has been shown in studies adapting the informed consent process for people who are Deaf or Hard of Hearing62,63 and for those with Deafblindness. 64 Wittach et al (2023) present a list of ways to tailor the informed consent process when engaging persons with Deafblindness. 65 These include preparations and procedural adaptations (i.e., provision of accessible information ahead of first contact), preparations for making the consent form content more accessible (i.e., simplified language; have Braille, large print and video recorded sign-language versions available) and adapted consent format options (i.e., verbal audio recorded consent). This study reports suggestions that would be applicable to enhance inclusion and access for people with vision impairment, particularly useful given the limited number of empirical studies and evidence-base in this area. During the document search, other noteworthy resources were also found produced by peak disability organizations and patient advocacy groups including Vision Australia, Able Australia, Round Table and Blind Citizens Australia. Additionally, the recommendations highlighted in Australia’s Disability Strategy (2021–2031) also bridge knowledge gaps and advise on how to meet information needs. Collectively these documents also raise the importance of providing, as well as asking, individuals their information preferences early. There is merit in all informed consent interactions being based on meaningful, two-way, information exchange that foster shared decision and empowered consent.66,67
Strengths and limitations
The limitations of this review include having a specific focus on engaging with people with vision impairment. It is recognized that information access challenges can be experienced by people with other communication and information needs including those with cultural, linguistic and cognitive considerations therefore recommendations and guides need to address diversity. It is therefore recognized that national policy and practice guides do need to be broad to a certain degree, but specific enough that they refer researchers to quality resources for more information to be able to effectively tailor their informed consent approach. The review may spark concern that current guidelines are just simply ‘guidance’ and need not be so prescriptive to allow flexibility in informed consent practice. This review may be criticized for advocating more specificity around actions researchers should take for improving information access. However, some documents fall very short and are now outdated in terms of alignment with inclusive best practice. Another limitation is that the search for included documents was contained to one policy database, Overton, and Government websites. It is noted that institutions may have policies that relate to the conduct of informed consent procedures, but these may not be publicly available or available on the Overton database. A future search in this area could also use the Analysis, Policy and Observatory 68 database that searches policy documents in Australia, along with Policy Commons, 69 to include global policy documents.
Despite these limitations, this review helped to identify the evidence for policy-based knowledge gaps. In doing so it highlighted a list of resourceful documents that have specific information and tools that may assist researchers in finding information to assist in adopting accessibility practices. This work is important to identify the specific actions that support inclusive informed consent practice, ultimately to facilitate uptake of accessibility practices in the future.
Conclusions
Future updates to Australian policy, guidelines and codes of conduct are warranted and would benefit from including how the informed consent process should be adapted to include people with vision-related information access needs. If these suggestions are not incorporated, key policy and practice documents should direct researchers to reliable participant-centred resources, tools and templates to find this information. There is hope that providing clear, timely and multiple accessible communications and information becomes the foundation for good clinical practice, and empowered informed consent practice, in Australia in the future.
Supplemental Material
Supplemental Material - Australia’s best practice guidelines for information access in the informed consent process for people with vision impairment: A scoping review using the AACTT framework
Supplemental Material for Australia’s best practice guidelines for information access in the informed consent process for people with vision impairment: A scoping review using the AACTT framework by Fleur O’Hare, Camille Paynter, David Foran, Maria Kolic, Caroline Ondracek, Tessa Saunders, Lauren N. Ayton in Research Methods in Medicine & Health Sciences
Footnotes
Acknowledgments
Our work is a research partnership between researchers, consumers, community representatives (i.e., patient advocacy representatives, disability sector representatives) and experts in the areas of clinical practice, policy, access and inclusion. The Centre for Eye Research Australia wishes to acknowledge the support of the Victorian Government through its Operational Infrastructure Support Program (VIC, Australia).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Health and Medical Research Council (LNA is supported by grant #1195713), National Industry PhD Program (FOH is supported by scholarship #34984).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
