Abstract
Background
There is no consistent approach in assessing swallowing function both in clinical or research settings. However, its significance in ensuring proper nutrition, growth/ development, and overall quality of life is undeniable. We aim to introduce a novel methodology that enhances swallowing function as an outcome measure in clinical and rare disease research and demonstrate its efficacy in Niemann-Pick Type C1 (NPC1).
Methods
We reviewed commonly implemented qualitative and quantitative swallowing assessments in current clinical practice, including patient/proxy/clinician reports, clinical swallowing evaluation, videofluoroscopic swallow assessment (VFSS), and post-VFSS interpretive measures [American Speech Hearing Association National Outcomes Measures Scale (ASHA-NOMS) and National Institutes of Health penetration and aspiration scale (NIH-PAS)]. Data analysis involved descriptive statistics, longitudinal statistical modeling to account for NPC1-specific covariates, and Kappa weighting correlations to determine inter-rater reliability.
Results
Our NPC1 cohort (n = 120) underwent baseline and longitudinal evaluations, (n = 269 VFSSs). We identified three statistically significant (p < 0.05) NPC1-specific variables associated with post-VFSS interpretive measures, providing insights into disease progression and treatment effects. Inter-rater reliability correlations for ASHA-NOMS and NIH-PAS demonstrated strong agreement between two speech pathologists [ASHA-NOMS: 0.76 (95% CI 0.70-0.83); NIH-PAS: 0.88 (95% CI 0.82- 0.93)]. VFSS mean radiation dosage was calculated (n = 129) and fell within the acceptable range declared by the American College of Radiology (263.79 ± 147.44 cGy*cm2).
Conclusions
This comprehensive methodology successfully documented functional swallowing status with dietary modifications and aspiration risk in a rare disease cohort (NPC1). Additionally, we effectively employed this methodology to support swallowing function as a research endpoint, specifically for phenotyping and developing therapeutic interventions for rare diseases.
Keywords
Background
Dysphagia
Swallowing dysfunction, clinically referred to as dysphagia, affects a significant number of individuals in the United States, with approximately 569,000 children aged 3-17 years and 25% of adults being affected.1,2 When dysphagia occurs with various physiological impairments throughout the oral cavity, pharynx, and gastrointestinal tract, it can allow the entry of unwanted material into the upper respiratory system. This can result in either laryngeal penetration or aspiration. Laryngeal penetration is characterized by the entry of any material into the upper laryngeal vestibule up to the true vocal folds, frequently observed clinically as a wet vocal quality or cough.3,4 On the other hand, aspiration refers to the entry of secretions, liquid, or food into the airway below the level of the true vocal folds. 3 Clinical signs of aspiration consist of volitional coughing or throat-clearing to expel aspirated material. Of further concern is silent aspiration, which occurs without any obvious sensory or motor response. Unfortunately, due to the intricate anatomical and physiological complexities involved in swallowing, there is no single comprehensive assessment available. Therefore, healthcare providers must choose the most appropriate assessment based on a patient’s report or symptoms. Oftentimes, the focus is directed towards the oropharyngeal phases of swallowing function, given its highly coordinated neuromuscular and autonomic system control, where dysfunction can lead to adverse sequelae.
Overview of swallowing assessments
Over the course of the last half century, the assessment and treatment of oropharyngeal swallowing dysfunction has fallen under the purview of a speech-language pathologist (SLP), with approximately 92% of SLPs serving the dysphagic population. 5 Additionally, it has been reported that 38% of all SLP encounters are related to swallowing and feeding disorders. 6 Assessments which attempt to evaluate dysphagia usually begin with screening for possible signs and symptoms of swallowing dysfunction followed with a clinical swallow evaluation (CSE) — an observational evaluation of swallowing function considering a patient’s past medical history and neurocognition. If needed, instrumental assessments are conducted to further delineate swallowing impairments and determine overall swallowing safety for eating and drinking.
The CSE, recognized as the “bedside swallow” clinically, provides the opportunity to assess swallowing function with various food and liquid textures for any individual. This assessment is usually the preliminary tool prior to more invasive instrumental evaluations such as Fiberoptic Endoscopic Evaluation of Swallowing (FEES) and Videofluoroscopic Swallowing Studies (VFSS). The CSE can identify risk factors for dysphagia and aspiration (e.g., coughing, choking, or changes in vocal quality) and assist in identifying possible physiological impairments (e.g., one’s ability to manipulate the various textures, timeliness in swallow response). 7 Additionally, it can aid in recognizing safety and efficiency of swallowing, determining the need for further instrumental assessment, and implementing interdisciplinary referrals to other professionals (e.g., gastroenterology, otolaryngology, pulmonary, nutrition). Advantages of the CSE include a non-invasive direct swallowing observation with various textures. However, limitations of this evaluation include non-standardization, inconsistent qualitative and quantitative interpretations amongst clinicians, and overlooked sequalae from physiological impairments. Reports have revealed that CSE alone underestimate the frequency of aspiration in patients with neurological dysfunction.8,9 This further aligns with approximately 50% of dysphagic patients identified via CSE who further underwent VFSS and were noted to have aspiration — undetected in the CSE alone due to absence of sensory motor responses associated with silent aspiration. 8
For the purposes of obtaining a comprehensive dysphagia diagnosis, instrumental assessments viewing anatomical and physiological elements including temporal and biomechanical correlates are utilized. Two instrumental studies frequently utilized clinically for oropharyngeal dysphagia include FEES and VFSS. FEES, often performed as a combined otolaryngology and speech pathology evaluation, utilizes a trans nasal endoscopy to obtain real time visualization with a 3-dimension anatomical view of the pharynx and larynx immediately prior to the swallow and immediately following the swallow. The benefit of this evaluation is that it provides a clear picture of the anatomy and visualization of saliva aspiration, allowing providers to guide clinical management. Despite clear visualization of the pharynx prior to the swallow, the actual swallow is not seen due to the physiological soft palate closure causing a “white out”. Therefore, food and liquid aspiration cannot be visualized and is inferred based on residual material following the swallow or ejection of material from the trachea. Additionally, other challenges include a lack of quantitative and qualitative standardization across practitioners in food and liquid consumption, variable analysis, and mild discomfort complaints and noncooperation among patients.
The VFSS, referred to as the modified barium swallow (MBS) study, is considered the gold standard of oropharyngeal swallowing assessments and is performed in partnership between speech-language pathology and radiology. It assesses swallow function and safety with various liquid and food textures using fluoroscopic imaging. While the advantages of VFSS are that it visualizes all phases of swallowing in real time with minimal radiation exposure, the challenges include dislike of barium taste, occasional positional difficulty, and inability to image infants who exclusively breastfeed. Further limitations consist of food and liquid quantity standardization as well as methodology and analysis variability amongst practitioners.
Despite limitations and challenges noted with swallowing assessments, documentation of oropharyngeal swallowing remains a worthwhile endpoint for clinical research. Swallowing outcomes are routinely applied to clinical trials studying head and neck cancers, strokes, and Parkinson disease to document disease progression and treatment effects.10,11,12 However, due to clinical variations among institutions when it comes to swallowing assessments and interpretations for safe swallowing function, there is limited utilization of swallowing function as an endpoint in rare disease research. About 45% of all rare diseases and 90% of pediatric rare diseases have major neurological effects that can impact safe swallowing function, further underscoring the value of swallowing function in rare disease research. 13 Specifically, dysphagia affects 55% to 80% of patients with Neimann-Pick type C1 (NPC1), a rare autosomal recessive lysosomal disorder characterized by highly variable clinical presentations and currently lacking FDA-approved treatments. 14 Off-label use of miglustat, a glucosylceramide synthase inhibitor, has shown promise in stabilizing swallowing function in NPC1.15,16
The United States Food and Drug Administration (FDA) has acknowledged the significance of swallowing function as a potential outcome in clinical trials, particularly when combined with disease natural history studies, patient-reported outcomes, and physiological correlates.
17
With this in mind, we propose a novel approach to evaluate swallowing as a functional measure, applicable to both research and clinical practice. This manuscript aims to: 1. Describe a unique combination of swallowing assessment tools applicable to both research and clinical settings. 2. Demonstrate its successful application in rare disease research.
Methodology
Swallowing assessments
Swallowing Questionnaire. This 43-question survey, completed by patients, caregivers, or guardians, assesses swallowing complaints. Results guide the speech-language pathologist in determining the need for additional evaluations to ensure safe swallowing and assess aspiration risk.

The VFSS began with a verbal pre-procedural huddle between the SLP and radiology staff reviewing a patient’s history, developmental levels, behavioral issues, dietary constraints/restrictions and objectives for assessment. Adaptations were made to accommodate individuals with allergy restrictions by providing alternatives such as applesauce and gluten-free cereal bars as needed. Food preparation with the various textures (liquid, pureed, and solid) along with suitable seating, if one was unable to sit independently, were determined by the SLP. Swallows were initiated with the patient seated comfortably, their back against the back of the x-ray swallowing chairs, and imaging conducted in the lateral plane. For liquid textures, E-Z Paque single barium contrast was mixed with 2-3 teaspoons of chocolate syrup and placed in a 60 cubic centimeters (cc) catheter tip syringe. 18 For pureed textures, Varibar pudding paste (2 teaspoons) was mixed with pudding/applesauce and placed in a 30 cc syringe. 19 Syringe type varied to account for anticipated volume utilized in swallowing trials and ease in texture specific-viscosity delivery. For solid textures, 1-2 cc of Varibar pudding/barium mixture was topped on 0.06 g of a graham cracker equivalent to ¼ of a single graham cracker.
Swallowing studies imaging was captured with the Siemens Fluoroscopy Imagining LUMINOS dRF Max 20 and recorded at a rate of 30 digital pulses per second from (1) the base of the orbits, (2) the nasal turbinate’s superiorly through the first tracheal rings anteriorly and (3) the upper esophageal sphincter posteriorly (Supplemental A). Vertical and horizontal collimation was applied by the x-ray technologist to reduce a patient’s radiation exposure. Clinicians followed the As Low As Reasonably Achievable (ALARA) principle for all VFSS procedures. 21 Acknowledging the radiation exposure associated with VFSS, radiation time and radiation dose area product (DAP) were calculated by the radiology staff for a randomized subset of patients and included in our analysis. Digital recordings and storage were obtained via the Pentax Medical swallowing workstations. 22
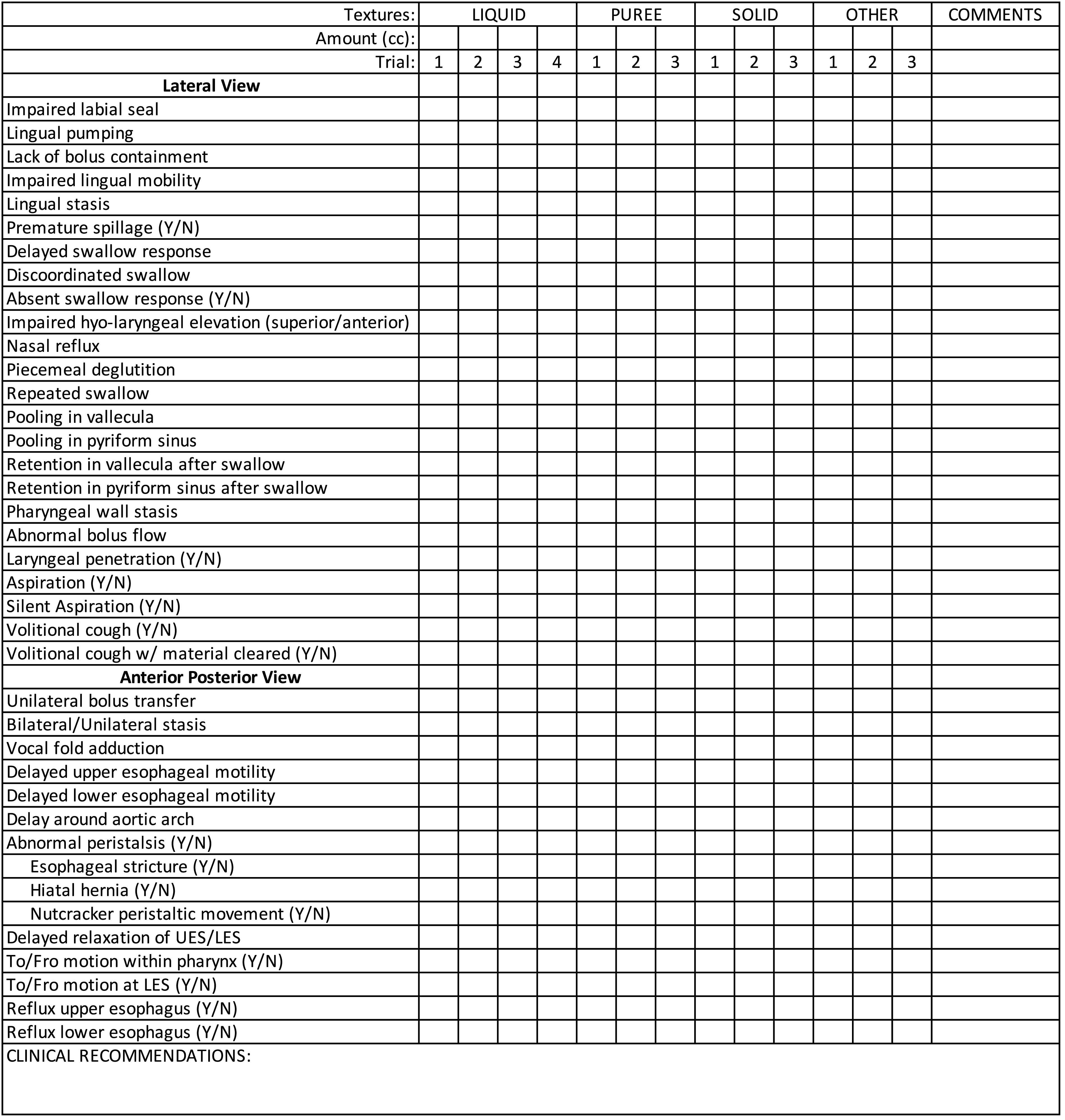
Videofluoroscopic swallow study impairment levels. The speech-language pathologist documented each swallow's physiologic impairment and severity using 39 defined impairment levels.
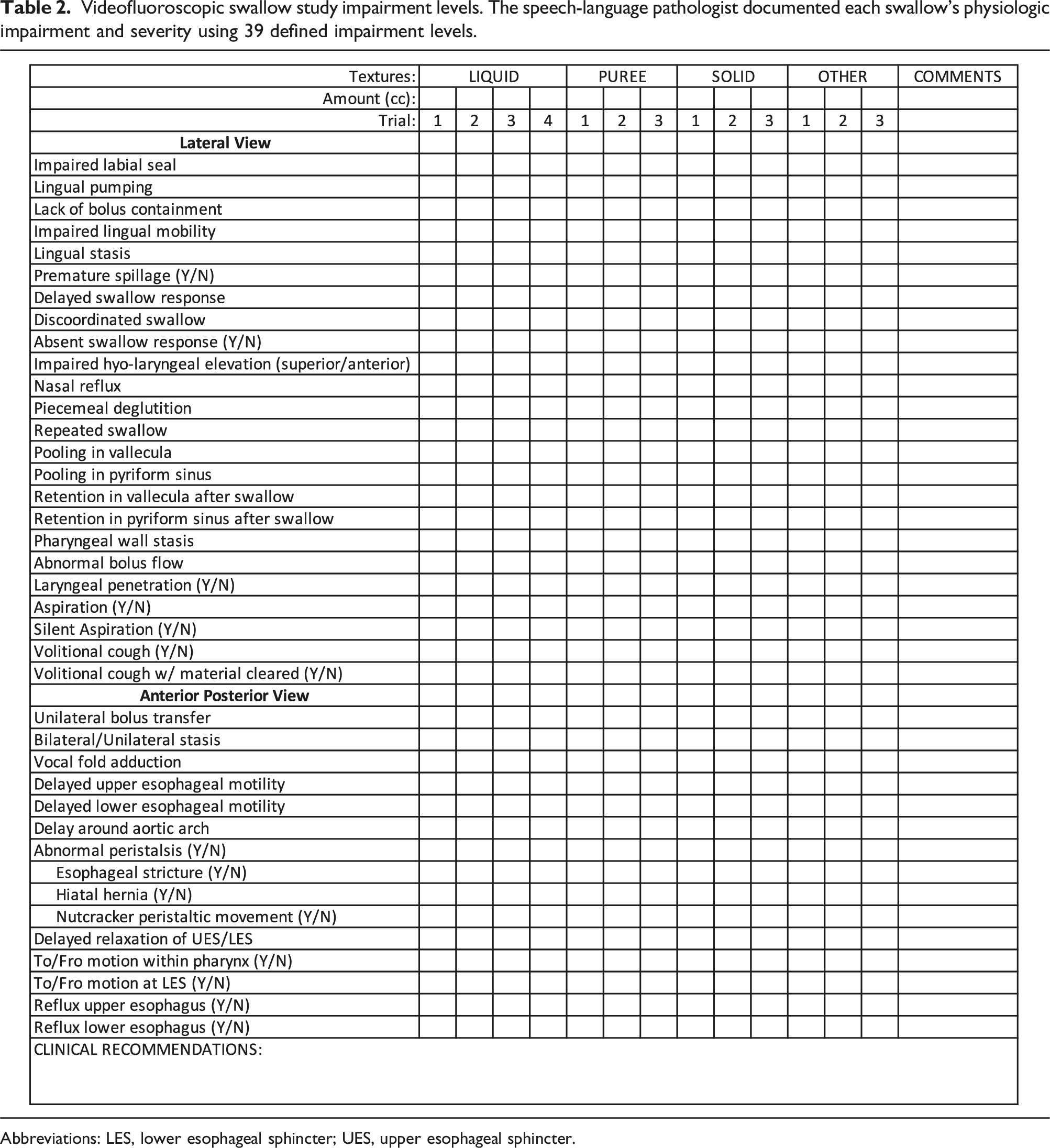
Abbreviations: LES, lower esophageal sphincter; UES, upper esophageal sphincter.
Swallow assessment functional interpretations (ASHA-NOMS & NIH-PAS)
Baseline characteristics and swallowing outcomes for Niemann-Pick disease type C1 cohort. Median baseline scores for swallowing outcomes were within normal ranges for the overall cohort. Table adapted from, 25 with permission.
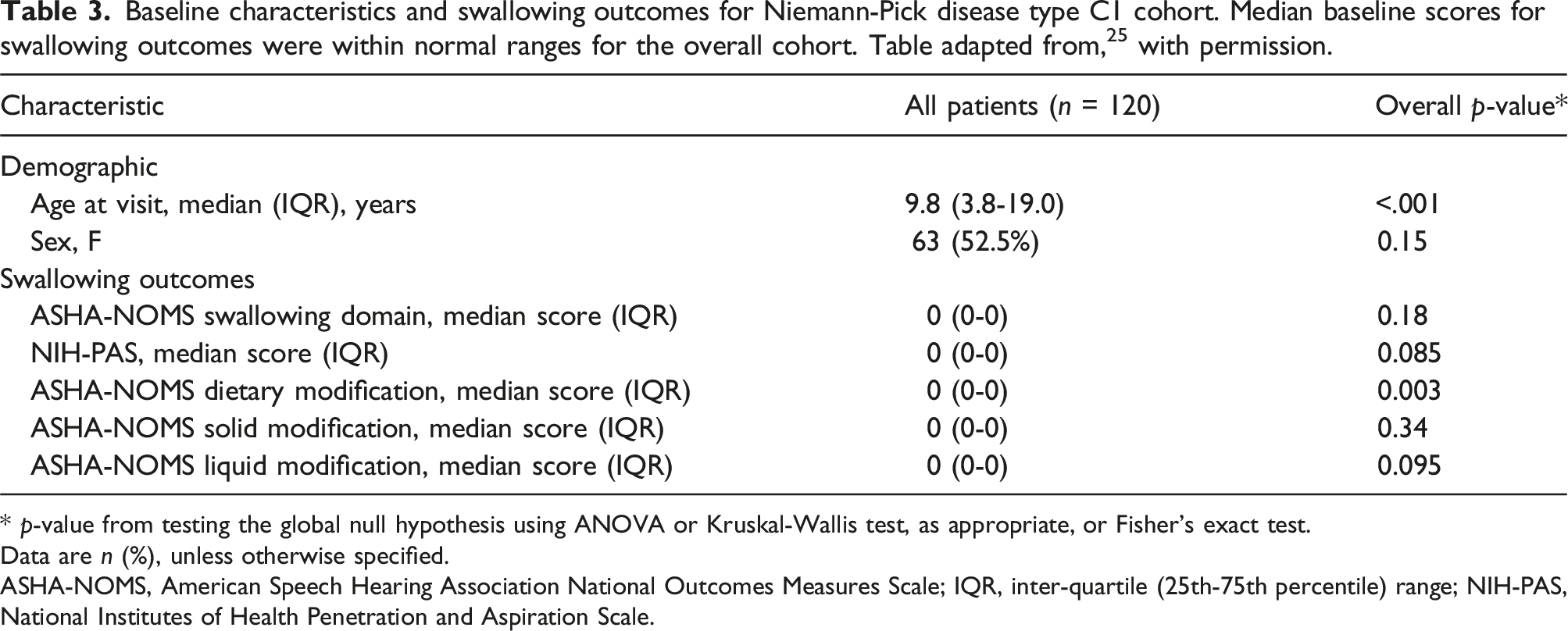
* p-value from testing the global null hypothesis using ANOVA or Kruskal-Wallis test, as appropriate, or Fisher's exact test.
Data are n (%), unless otherwise specified.
ASHA-NOMS, American Speech Hearing Association National Outcomes Measures Scale; IQR, inter-quartile (25th-75th percentile) range; NIH-PAS, National Institutes of Health Penetration and Aspiration Scale.
Speech assessments
To complete the methodology’s comprehensive assessment of swallowing, oral sensory motor function and dysarthria speech parameters were evaluated for each patient visit (Supplemental B).
Rare disease population
Between 1997 and 2019, 120 patients with a confirmed diagnosis of NPC1 were enrolled under an NIH IRB-approved NPC1 natural history study (NCT00344331), and classified into four groupings as done in other NPC studies [Early Childhood-Onset (ECO, onset <6 years); Late Childhood-Onset (LCO, onset ≥6 years to <15 years); Adult Onset (AO, onset ≥15 years); no neurological onset].26,27 Patients or guardians provided consent/assent for enrollment. The aim of the NIH study was to monitor patients over their disease course while documenting potential NPC1-specific biochemical and clinical markers. Collection of biospecimen samples and evaluations completed at each visit included history, physical, speech, and swallowing. From these evaluations, NIH study duration, neurological history, seizure history, and miglustat use were used as disease characteristics.
NPC1-specific characteristics
The NPC1 Neurological Severity Score (NSS) is a clinical outcome measure developed to characterize and quantify disease progression in nine functional domains including swallowing, ambulation, cognition, fine motor, hearing, memory, seizures, and speech. 28 We utilized the NSS to characterize potential biomarkers and as an outcome measure to monitor disease progression. The NSS swallow domain score was extracted from the NPC natural history data set (NCT00344331) as described by Yanjanin et al. 28 Its use offered a distinctive insight into daily functioning that may not be readily observed in a clinical setting, allowing for a better understanding of the individual’s functional status. Therefore, its value as a descriptive quantifier was investigated with the interpretive outcomes from VFSS.
Statistics
The data are presented using frequency (percentage), mean (± standard deviation), or median (inter-quartile range). Statistics related to baseline and longitudinal disease characteristics were completed as described in Solomon et al. (2022) to emphasize this methodology’s application to rare diseases. Two NIH SLPs evaluated 255 VFSSs from longitudinal visits within the NPC1 cohort, blinded to personally identifiable information, and inter-rater reliability was computed using weighted kappa statistics for ASHA-NOMS and NIH-PAS. Further correlations of ASHA-NOMS and NIH-PAS with a disease-specific severity scale (NSSswallow domain) were analyzed with Spearman’s rho. Average VFSS duration and radiation dosing were calculated for a subgroup of patients to ensure overall patient safety.
Results
To highlight the above methodology, NPC1 was utilized as the rare disease population to demonstrate the types of qualitative and quantitative data that can be obtained with the methodology described. This multifactorial approach was efficiently conducted within 90 min, encompassing the swallowing questionnaire, which participants completed without requiring clinician assistance. All patients included in our analysis were referred to the SLP team and underwent fluoroscopy based upon clinical identification of swallowing dysfunction indicated by swallowing questionnaire or CSE findings. The most frequently concerning swallowing complaints were the following: difficulty swallowing liquids, coughing when eating, episodes of choking, and difficulties swallowing solid textures (e.g., puree, raw vegetables, meats). These triggers resulted in a total of 269 VFSSs being completed for 120 patients at baseline and 57 longitudinally, with only three incomplete evaluations due to behavioral non-compliance. As in Solomon et al. in 2022, patients were followed with a median number of 4.0 visits (IQR: 3.0-6.0; range: 2.0-11.0) over a median of 3.3 years (1.3-4.4; 0.6-10.3). 25 We adapted the previously published demographic table in Solomon et al. (2022) to highlight pertinent findings needed to demonstrate this methodology’s utility. Each VFSS underwent impairment level documentation that revealed common physiological issues that included lack of bolus containment, premature spillage, discoordinated/delayed swallow, repeated swallows, pooling and retention in the vallecula and pyriform sinuses, aspiration, and silent aspiration.
Associations between swallowing outcomes and Niemann-Pick disease type C1 specific characteristics. Adapted from 25 with permission.
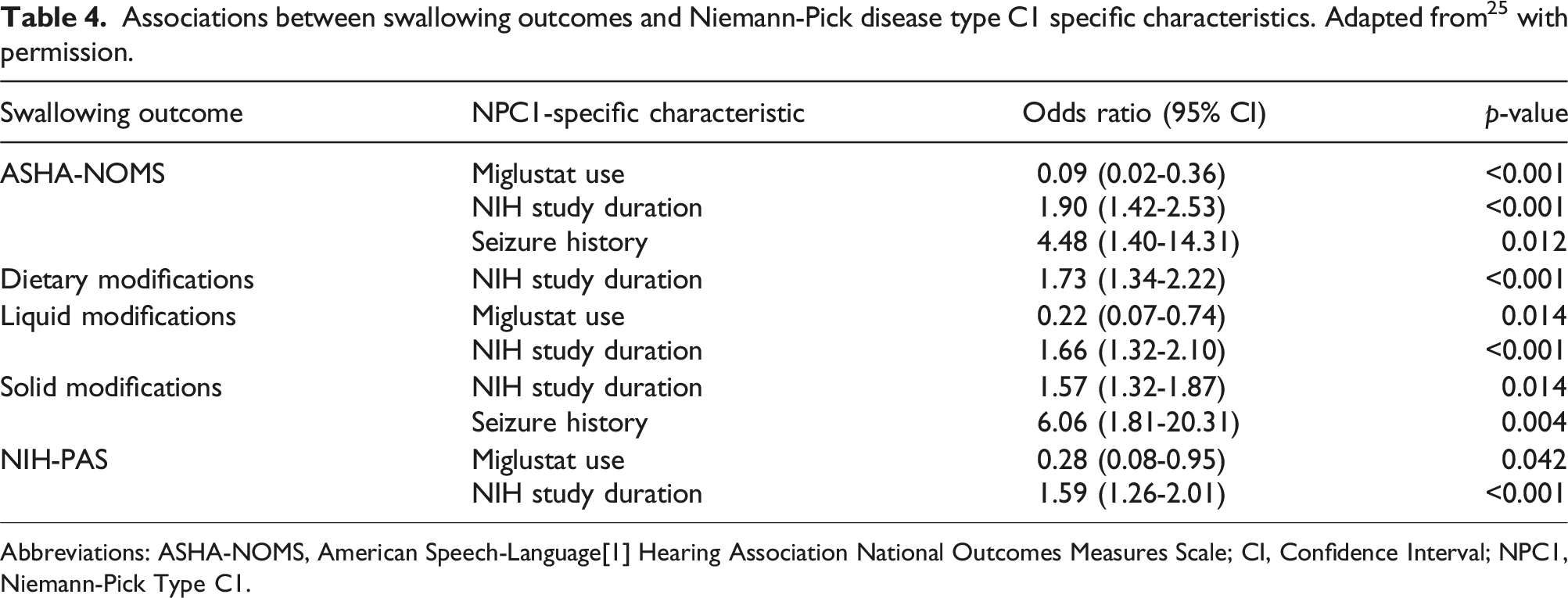
Abbreviations: ASHA-NOMS, American Speech-Language[1] Hearing Association National Outcomes Measures Scale; CI, Confidence Interval; NPC1, Niemann-Pick Type C1.
Additionally, baseline ASHA-NOMS and NIH-PAS scores were robustly correlated with the NSSswallow domain (ASHA-NOMS rs = 0.543, p < .001; NIH-PAS rs = 0.535, p < .001).
Inter-rater reliability for ASHA-NOMS and NIH-PAS
Amongst 255 VFSSs obtained from a group of 54 NPC1 patients seen longitudinally, the inter-rater reliability was strong for both ASHA-NOMS [kw = 0.76 (95% CI 0.70-0.83)] and NIH-PAS [kw = 0.88 (95% CI 0.82-0.93)] (Figure 1). Inter-rater reliability ASHA-NOMS and NIH-PAS for All visits.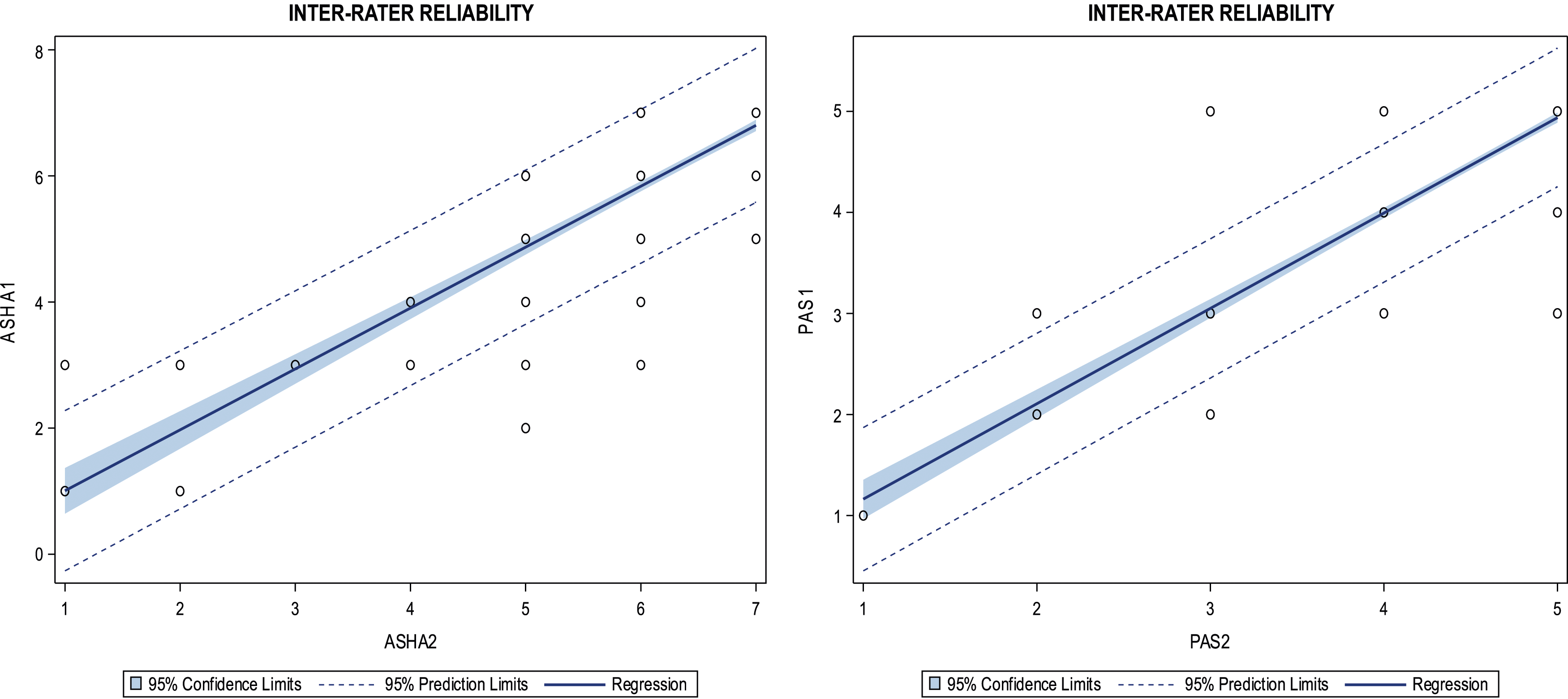
Radiation exposure
A subgroup of 129 VFSSs were randomly selected from the total collection of 269 VFSSs conducted as part of the NPC1 natural history study. Analysis of this subgroup revealed an average radiation fluoroscopy time of 3 min and 4.03 s ±1 min and 29.58 s. Additionally, the average radiation DAP was calculated to be 263.79 ± 147.44 centigray cm squared (cGy*cm2) equivilant to an effective dose of 0.0976 ± 0.0546 millisievert (mSv).
Discussion
Undoubtedly, the importance of swallowing function cannot be overstated when it comes to adequate nutrition, hydration, secretion management, and overall quality of life for individuals. Swallowing assessments are frequently employed in clinical settings to manage symptoms and assess treatment outcomes for common medical conditions. However, there are a lack of comprehensive prospective studies that utilize swallowing as an endpoint in rare disease research, hindering the ability to define an individual’s functional status. Moreover, the interpretation of swallowing assessments in research is often difficult due to inconsistencies in assessment tools and varying techniques among practitioners. Additionally, assessing individuals across different age groups presents its own set of challenges, leading to varying approaches and mixed results. This paper presents a systematic methodology applied to the NPC1 cohort, which supports its application clinically and promotes swallowing as an endpoint for research.
We developed a systematic approach by merging common clinical tools to facilitate the evaluation of swallowing function. This approach includes the utilization of the 43-item swallowing questionnaire and a patient-report measure that demarcates the frequency and severity of swallowing complaints. In addition to this, the clinician’s observation of swallowing function with the CSE contribute to formulating hypotheses regarding potential oropharyngeal dysfunction. While being descriptive in nature, this information holds great value as it takes into account the day-to-day changes in an individual’s swallowing ability, which may otherwise go unnoticed in VFSS. The combination of qualitative and quantitative tools employed in this study represents a widely recognized approach that effectively captures patient complaints, clinician observations, instrumental results, and interpretive findings (ASHA-NOMS and NIH-PAS). If VFSS is not performed, silent aspiration may go undetected due to the absence of overt signs, further justifying insurance coverage of the VFSS and providing valuable insights into an individual’s swallowing function. FEES was not utilized because it has limitations in capturing the full physiological process of swallowing, providing images only before and after the swallow.
Historically, impairment parameters have been provided by Logemann et al. and identified from VFSS during swallowing function evaluation. 29 However, the exact parameters used by each SLP and across institutions vary. Thus, we delineated 39 impairment parameters to determine a patient’s swallowing safety and aspirations risk. These parameters serve as a foundation for clinicians to make the necessary decisions regarding an individual’s oral intake and need for dietary modifications and/or clinical interventions. Impairment level documentation provides clinicians with physiological explanations for swallowing dysfunction. However, it is important to highlight the existence of another valuable tool, known as the MBS Impairment Profile (MBS-ImP), 30 which is a standardized impairment level profile currently available for assessing swallowing impairments. Regardless of the specific impairment level documentation used, the need to determine a patient’s oral intake and safety modifications is paramount. Although some studies have utilized Functional Oral Intake Scale (FIOS), ASHA-NOMS is preferred due to its inclusion of dietary modification such as liquid and solids. Even though the ASHA-NOMS domains and NIH-PAS are not standardized, they have been successfully applied as interpretative measures to document swallowing safety, diet modifications, and aspiration risk post-VFSS in research.15,25
While this precise methodology has yet to be employed by other clinical sites, it is currently being applied both clinically and in research at the NIH with other diseases. These diseases include GM1 gangliosidosis, Batten disease, Smith-Lemli-Opitz Syndrome, Parkinson’s disease, spinal muscular atrophy, and RASopathy syndromes.
Application to NPC1
Given the significant impact of dysphagia on NPC1 patients and their families, we utilized this cohort to demonstrate the successful application of our methodology. Beginning with the swallowing questionnaire, a variety of swallowing concerns were noted for the NPC1 cohort, reflecting the heterogeneity of clinical manifestations seen in NPC1. Each patient’s swallowing concern(s) aided in forming the initial hypothesis regarding swallowing dysfunction and prompted the need for VFSS.
From the impairment levels identified from each VFSS, we gained a greater physiological understanding of each patient’s swallowing. However, it is not currently standardized like the MBS-ImP, which requires extensive continued education training not offered in standard SLP training programs. Neither impairment level analyses fully capture and quantify an individual’s functional status. Therefore, ASHA-NOMS and NIH-PAS were incorporated and identified the need for dietary modifications, clinical management strategies, and the risk of aspiration within the NPC1 cohort, including the subgroups ECO, LCO, and AO. 25 These tools were selected due to the lack of quantitative interpretive outcome measures post-VFSS in the literature that specifically address an individual’s ability to eat and the risk of aspiration. The ASHA-NOMS swallow domain and dietary modification subdomains, commonly employed by SLPs to document treatment outcomes and quantify swallowing function, were instrumental in providing granularity by documenting dietary modifications, particularly for solids and liquid textures. Moreover, these domains contributed to patient and caregiver education and complemented initiatives focused on collaboration among professional groups internationally (e.g., International Dysphagia Diet Standardisation Initiative). The combined use of the quantative scores from ASHA-NOMS and NIH-PAS contributed to the decision for g-tube placement, further reinforcing the importance of employing interpretive measures following VFSS to determine safe swallowing outcomes in neuroprogressive and rare disease research. 25
Notably, ASHA-NOMS and NIH-PAS scores were successfully correlated with the NSS swallow domain, supporting the accuracy of swallowing functionality from patient/caregiver reports and clinician interpretation from VFSS. As this methodology is a novel approach in clinical research, our inter-rater reliability for ASHA-NOMS and NIH-PAS amongst a subgroup of 54 patients with longitudinal data demonstrated a strong correlation, consistent with the gold standard for inter-rater agreement greater than 75%. 31
To further emphasize this methodology’s application to rare disease research, we integrated NPC1-specific characteristics from the NIH NPC1 natural history study to illustrate how disease characteristics could be linked to functional status and treatment outcomes. Through this approach, we successfully identified three significant variables that showed variations across the swallowing outcomes. 25 Although not observed for all outcomes, we found that the use of miglustat was associated with a protective effect on swallowing function, while longer NIH study duration (a proxy of disease duration) and a history of seizures correlated with worsening swallowing function. 15 These findings highlight the potential use of our methodology in providing valuable insights into disease-specific characteristics that influence swallowing function and treatment outcomes in research.
Furthermore, within the NPC1 cohort for ASHA-NOMS and NIH-PAS, the probability of swallowing decline exhibited heterogeneity. Thus, we defined a clinical cut off score of less than 50% oral intake with the need for adjuvant nutrition and moderate aspiration to evaluate swallowing decline within the cohort and its subgroups. 25 This type of analyses offers valuable clinical insights (e.g., dietary modification, g-tube placement) and plays a significant role in advancing therapeutic development within the research field.
At the forefront of our research, ensuring patient safety remained paramount. Thus, we adhered to the guiding principle of ALARA to implement protective measures for radiation safety among the NPC1 cohort. We employed a standardized VFSS protocol with cognitive behavioral treatment (CBT)-related techniques to enhance patient compliance, and achieved an average acceptable radiation DAP.32,33,34 However, the large standard deviation may be potentially attributed to varying body mass in children’s growth and development. These results aim to alleviate common radiation concerns associated with VFSS examinations and underscore our commitment to safeguarding patients’ well-being throughout the research process. We acknowledge that fluoroscopic examinations, such as VFSS, are low-dose radiation procedures. Bonilha et al. (2019) reported an average adult radiation exposure of 0.27 mSv from MBS procedures, which is comparable to the “radiation exposure one would receive by living on Earth for 32 days”. 35 In our study, we observed a radiation exposure of 0.0976 ± 0.0546 mSv, further confirming the minimal radiation dose associated with VFSS. However, if any safety concerns persist, it is advisable to consult with the National Council on Radiation Protection and Measurements for further guidance.
Considerations of the methodology
Clinical research often falls short due to the incorporation of multiple factors required for comprehensive evaluations. Similarly, this methodology required careful consideration of patient compliance in performing each swallowing assessment. In determining our VFSS protocol, we considered various barriers that may affect the successful completion of VFSS such as allergies, food intolerances, and dietary/religious restrictions. Therefore, alternatives matching the viscosity of the testing samples were individualized and modified prior to the swallow study. Additionally, individuals commonly complain of the unappealing taste of barium, hence the incorporation of chocolate syrup. For those who preferred alternatives to chocolate, either strawberry syrup or apple sauce were substituted. The methodology incorporates the standardization of bolus quantities by employing specific syringes for each texture. However, we recognize that this approach may not be feasible for individuals with developmental deficits. Therefore, as an alternative, samples were delivered using the utensil of choice for these patients. Furthermore, we screened developmental milestones and the ability to sit independently to determine if any seating modifications were necessary (e.g., safety belts, pediatric tumbleform chairs, wedges, towel rolls). This information was obtained from the patient’s history and communication during the pre-procedural huddle, along with their disease status. Additionally, the SLP employed a CBT patient-centered approach (e.g., verbal praise, tokens, utilization of electronic devices) to facilitate a patient’s participation, particularly for children and patients with cognitive deficits. Of note in the methodology, the SLP’s utilization of the same wording when prompting patients to consume the samples and perform the swallowing tasks, was helpful in a patient’s understanding of what was asked of them. We attribute the successful completion of 269 VFSSs to these considerations mentioned above.
While there are no specific guidelines regarding the optimal timing of a swallowing study, systematic standardization of an evaluation’s timing can offer advantages for both clinical and research purposes. In the case of the NPC1 cohort, we conducted all VFSSs in the early morning. This approach was adopted to minimize potential interference from other medical evaluations and to mitigate the impact of daily fatigue on a participant’s swallowing function. Furthermore, the successful scheduling of VFSS with other medical evaluations was crucial to avoid barium artifact interference and provide patients ample time for recovery between assessments (e.g., sedative procedures). We recognize that swallowing evaluations conducted in an institutional setting can evoke stress and anxiety among patients and their families, potentially impacting their compliance. However, our methodology incorporated multiple qualitative and qunatative measures to provide an accurate understanding of an individual’s swallowing function. Although our approach provides valuable insights, we recognize that it represents only a snapshot in time and may not capture all the meal-time nuances occurring at home. The inclusion of family-reported functional status (e.g., NSS) and the swallowing questionnaire offers an understanding of home issues, but one must also consider potential biases related to under-reporting and over-reporting of swallowing complaints by patients/caregivers. We attribute the high correlations observed between the NSS swallow domain and the interpretive swallowing measures (ASHA-NOMS and NIH-PAS) to the heightened awareness of NPC1 disease progression among parents.
Other factors to consider during VFSS are pharmacological interactions with swallowing function. This methodology revealed the protective effect of miglustat use on swallowing within the NPC1 cohort. 15 However, it is essential to account for the potential impact of other medications, such as psychiatric and anti-convulsant drugs, which may lead to misrepresentations of swallowing dysfunction. Therefore, these factors should be carefully considered both clinically and in research.
Furthermore, the use of VFSS in research is often subject to scrutiny due to concerns about radiation exposure and unknown risks, particularly in pediatric populations. However, in neuroprogressive diseases, repeated assessments with interpretive measures can be beneficial in reducing co-morbidity risks associated with dysphagia and establishing safe swallowing practices for oral gratification that may override these concerns.
Conclusion
By compiling multiple parameters and evaluations commonly used in swallowing assessments, we have successfully developed and demonstrated a comprehensive methodology to qualitatively and quantitatively assess swallowing function and determine functional status. To highlight its applicability in clinical and research settings, we utilized NPC1, a rare neurodegenerative disease, as our study population. Currently, interpretive measures that offer quantitative endpoints of swallowing function, particularly in documenting rare disease progression and treatment outcomes for research purposes are lacking. In this methodology, we successfully employed the ASHA-NOMS and NIH-PAS alongside swallowing assessments to provide multiple levels of detail necessary to determine swallowing safety, the need for dietary modifications, and aspiration risk. These functional endpoints can serve as valuable outcomes when associated with specific disease biomarkers to monitor disease progression and development of therapeutic interventions. This methodology also suggests the vital importance of documenting an individual’s swallowing function, regardless of its application in clinical, research, or financial settings, to maximize one’s quality of life.
Supplemental Material
Supplemental Material - Swallowing assessments: A unique methodology to enhance medical research
Supplemental Material for Swallowing assessments: A unique methodology to enhance medical research by Beth Solomon, Andrea Munoz, Ninet Sinaii, Hibaaq Mohamed, Monique King, An Dang Do, Nicole Farhat and Forbes Porter in Research Methods in Medicine & Health Sciences.
Supplemental Material
Supplemental Material - Swallowing assessments: A unique methodology to enhance medical research
Supplemental Material for Swallowing assessments: A unique methodology to enhance medical research by Beth Solomon, Andrea Munoz, Ninet Sinaii, Hibaaq Mohamed, Monique King, An Dang Do, Nicole Farhat and Forbes Porter in Research Methods in Medicine & Health Sciences.
Footnotes
Acknowledgments
Our profound gratitude to the NIH radiology staff, Michael Backman, Dr Barbara C. Sonies for their invaluable support in facilitating this methodology. We also extend thanks to the dedicated parents, guardians, and patients whose participation made this study possible and greatly contributed to its academic advancement.
Author contributions
BS was the main SLP that assessed swallowing in the NPC1 cohort and created the methodological procedures described in the manuscript. AM created the figures and tables as well as being a major contributor in writing this manuscript. NS performed all the statistical analyses included in this manuscript and wrote the corresponding methodology section. HM assisted with literature review and writing of the manuscript. MK was one of the SLPs that scored the videofluoroscopic swallow studies used to determine the inter-rater reliability. ADD and NF performed the history and physical evaluations for the NPC1 cohort. FDP is the principal investigator for the NPC1 NIH natural history study. All authors reviewed, provided edits, and approved the final manuscript version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIH funding sources provided salaries for intramural research professionals (ZIA HD008989).
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.