Abstract
Background and Aims
Poor retention of trial participants is common and can result in significant methodological, statistical, ethical, and financial challenges. To improve trial efficiency, we aimed to assess the extent to which commonly used strategies to retain participants within trials are supported by evidence for their effectiveness.
Method
A systematic methodological review was carried out to identify commonly used retention strategies in National Institute for Health and Care Research (NIHR) Health Technology Assessment (HTA) trials (January 2020–June 2022). Strategies were then mapped to evidence for their effectiveness from the most recent Cochrane retention review (published 2021), and a future Study Within A Trial (SWAT) priority list was created.
Results
Amongst 80 trials, the most frequently reported retention strategies were: flexibility with data collection method/location (53%); participant diaries (38%); use of routine data (29%); PPI input (26%); telephone reminders for participants (26%); postal reminders for participants (25%); monitoring approaches (21%); offering flexibility with timing of data collection (20%); pre-paid return postage (18%); prioritising collection of key outcomes (15%); and participant newsletters (15%). Out of the 56 identified strategies, mostly no, very low or low evidence for their effectiveness was identified (64%; 14%; 13% respectively).
Discussion and Conclusions
Commonly used retention strategies are lacking good quality evidence for their effectiveness. The findings support the need for more SWATs and help identify priority areas for future SWAT research. These priorities could be used with other priority lists to inform future SWAT conduct.
Keywords
Introduction
Background and rationale
Randomised Controlled Trials (RCTs) are considered the gold standard for clinical research due to their ability to reduce bias and confounding variables. 1 The random allocation of participants to study arms is essential to eliminate selection bias by balancing known and unknown participant characteristics between groups. Attrition bias, a form of selection bias, occurs when there is a difference in the characteristics of patients who are lost to follow-up by initial group allocation. Additionally, if participants withdraw before data collection time points, are lost to follow up, or provide incomplete patient-reported outcomes this results in missing data, which can question the trial’s external validity. 2 Poor retention will also reduce the trial’s statistical power, reducing the chance of a true effect being identified.
A review of National Institute for Health and Care Research (NIHR), Health Technology Assessment (HTA) funded trials found the median attrition rate to be 12%. 3 This is higher than the 5% participant loss to follow-up which has been suggested to result in low risk of bias, and anything over 20% may seriously threaten the trial’s internal validity. 4 A survey by Kearney et al. 5 found that 78% of Chief Investigators (CIs) of NIHR HTA funded trials, recognised the challenges of participant retention at the beginning of the trial and implemented strategies within the trial design to overcome this. Adjusting the sample size in anticipation of missing data was also highlighted as common practice. Whilst this method maintains study power, it does not address any bias on outcomes caused by attrition. In addition, attrition is responsible for research waste. A recent study estimated the average cost per participant in trials funded by the NIHR HTA programme to be £2987. 6 If retention rates were routinely higher than they are now, then the average trial could be smaller, and its costs would be reduced.
The strongest evidence for the effectiveness of retention strategies mainly comes from randomised trials of different retention methods which are often referred to as a Study Within A Trial (SWAT). These are self-contained studies embedded within a host trial, which evaluate an aspect of trial methodology. 7 Evidence suggests that more randomised retention SWATs are needed. A recent Cochrane review evaluated 52 retention strategies and found none to be supported by Grading of Recommendations, Assessment, Development and Evaluation (GRADE) high-certainty evidence, and most retention interventions had only been evaluated in just one study. 8
This review reports retention strategies that are used within recently published NIHR HTA trials, examines what justification exists for choice of retention strategy, and determines the extent that these strategies are supported by robust evidence for their effectiveness. Previous research has reported commonly used retention strategies by surveying UK CTUs 5 but no published reviews have established current retention practice through reporting the strategies used in recently published NIHR HTA trials. Trials funded by the HTA programme have been chosen to examine current retention practice in this review for several reasons. Regardless of the trial’s outcome, virtually all trials funded by this programme are required to publish a report, and as these reports are extensive, more retention information is expected to be ascertained compared with trials published in traditional peer reviewed journals. No reviews have been identified that map retention strategies used in these trials, to evidence for their effectiveness. This review aims to address the current gap in the literature by assessing what strategies are actually used in practice to reduce attrition and if these are grounded in evidence.
Review objectives
1. To identify retention strategies that have been used in NIHR HTA trials published from January 2020 to June 2022. 2. To map strategies that are currently being used against evidence for their effectiveness from the Cochrane retention review.
8
3. To create a priority list for future retention SWATS, using insight from PRIORITY 2’s important unanswered retention questions.
9
4. To understand trial teams’ justification for using certain strategies.
Methods
A protocol for this review was registered prospectively on the Open Science Framework, 10 The review was reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement where appropriate. 11
Eligibility criteria
Due to logistics, and the desire to include recently conducted RCTs, only trials published within the last 30 months were considered for inclusion. This review was interested in retention strategies used across all clinical areas and patient types, so there were no exclusion criteria on these factors. The inclusion and exclusion criteria are summarised below. Cluster trial designs were excluded as randomisation is at a site-level, and not patient-level so retention strategies may be different compared with non-clustered designs.
Inclusion criteria
• Trials published in the NIHR HTA Journals Library from January 2020 to June 2022 • Patient-level, parallel randomised trials in any clinical area
Exclusion criteria
• Cluster trial designs • Feasibility studies • External pilot studies • Cross-over trials • Phase I and Phase II trials • N-of-1 trials
Search strategy
Due to the broad inclusion criteria, this review did not require an extensive or highly sensitive search strategy. ‘RCT’ was entered into the search bar within the NIHR HTA Journals Library, and filters were applied to identify trials published in 2020, 2021, and up to June of 2022.
Data management
Systematic review software Covidence, 12 was used to manage search results, independently screen studies and resolve any disagreements. 13 This enabled efficient use of time and reduced the risk of human error. EndNote, 14 was used to manage bibliographic references. 15
Study selection
All search results were screened by two reviewers independently (RW and AP), in line with the recommended gold standard method. 16 Titles and abstracts were screened first in accordance with the eligibility criteria. A third researcher (DT) was on hand to resolve disagreements if needed. Agreement was reached for inclusion/exclusion of all studies after reviewing titles and abstracts, and so full text assessment of studies took place simultaneously with data extraction.
Data extraction
A data extraction form was created in Microsoft Excel, 17 and initially piloted with 13 eligible trials from 2022. Following the pilot, any necessary changes were made to the form. If data were missing, the published trial protocol was searched on Google Scholar,18,19 to identify if the missing data item was recorded in the protocol. If the required information was still not located, the full trial protocol was searched via the NIHR webpage, which typically publishes the protocol of all NIHR funded trials. The second reviewer (AP) checked extraction accuracy for the first 16 (20%) studies by cross-checking each extracted data item. For the remaining studies, the second reviewer carried out spot checks for accuracy, and helped resolve any data items that had been queried by the lead reviewer (RW). Any data items that remained missing for eligible trials were marked as “NR” (not reported).
Data items
The data items that were extracted are included in Supplemental_material_1 along with details of any additional resources that were required to extract the data item, and where necessary, precise definitions of the data items.
Retention strategies were extracted when they were specifically reported as retention strategies by the trial, and in cases where a method was reported that was assumed by the lead reviewer that it would affect retention. Therefore, judgements had to be made, based on the available evidence reported in the HTA reports and trial protocols.
Data analysis
Characteristics of the included studies were narratively synthesised, and key comparative details tabulated. Retention characteristics of the included trials were also narratively synthesised, including detail on trials that included SWATs and if this related to retention; trials that included internal pilot studies with retention criteria; commonly reported reasons for missing data; and attrition summaries.
Retention strategies were identified for each trial and categorised, using guidance from the ORRCA retention research domains. 20 Strategies were then ranked according to their frequency of use, and all strategies were mapped to the results of the Cochrane retention review by Gillies et al. 8 for evidence of their effectiveness (which is the most up to date systematic review of SWAT retention evidence). From this point onwards, this review is referred to as the Cochrane retention review. If for example a trial reported using multiple reminders of the same method (e.g., two postal reminders) this counted as one ‘occurrence’ of the strategy. This logic was consistently applied. A short narrative overview of retention strategy characteristics within the included trials was produced.
For each strategy, the following details from the Cochrane retention review were reported: • If any relevant evidence existed • GRADE certainty of evidence (very low, low, moderate, high) • The intervention and comparator for the evidence • The Risk Difference (RD) • 95% Confidence Intervals • Number of studies included in the meta-analysis • The “conclusion” regarding the strategy’s effectiveness
For each strategy, evidence was sought that compared the intervention to usual follow-up. If such evidence did not exist this was indicated by “no evidence.” If additional evidence was available (for example evidence for telephone reminder vs usual follow up, and evidence for telephone reminders vs postal reminders) these other comparators were reported. If evidence was only available as a comparator against another strategy (e.g., the only evidence for telephone reminders was telephone reminders vs postal reminders, “no evidence” was reported, but this further evidence was discussed.
Strategies were then prioritised for future SWAT research, based on their frequency of use, the available evidence for their effectiveness, and the degree that it was felt they aligned with the PRIORITY 2 important unanswered retention research questions. 9 With insight from these prioritisation factors, a list of priority SWAT retention research topics was created.
To map the identified retention strategies to the PRIORITY 2 questions, the reviewers compared each identified strategy to the list of top 10 PRIORITY 2 questions. If the reviewers decided the PRIORTY 2 question aligned with the identified strategy in the review, the PRIORTY 2 question number(s) was recorded next to the identified retention strategy. For example, any strategies that were aimed at encouraging participants to complete follow up (e.g. reminders, pre-paid postal strategies, newsletters) would be deemed aligned with the PRIORTY 2 question: “What are the best ways to encourage trial participants to complete the tasks required by the trial?.”
Results
Included studies
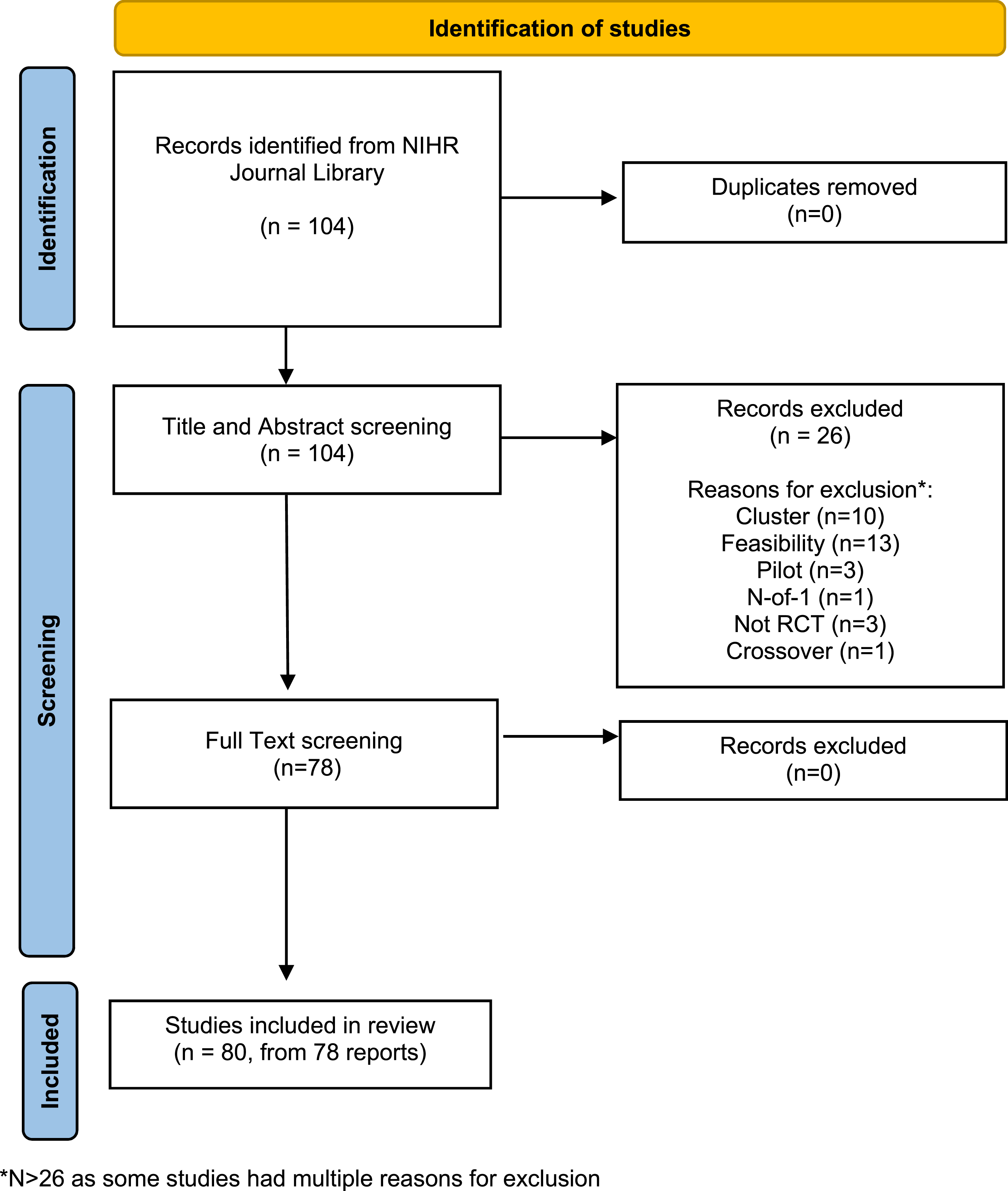
Between January 2020 and June 2022, 104 records were identified when ‘RCT’ was applied as a search term. No duplicate records were identified. Following independent screening with the eligibility criteria, two discrepancies between reviewers arose, which were discussed and resolved. A total of 26 records were excluded at the title and abstract screening stage, and no further studies were excluded during full text assessment. Some studies had multiple reasons for exclusion, but the main reasons were cluster ( PRISMA flow diagram (adapted from Page et al.,
11
).
Characteristics of included studies
Characteristics of included trials.
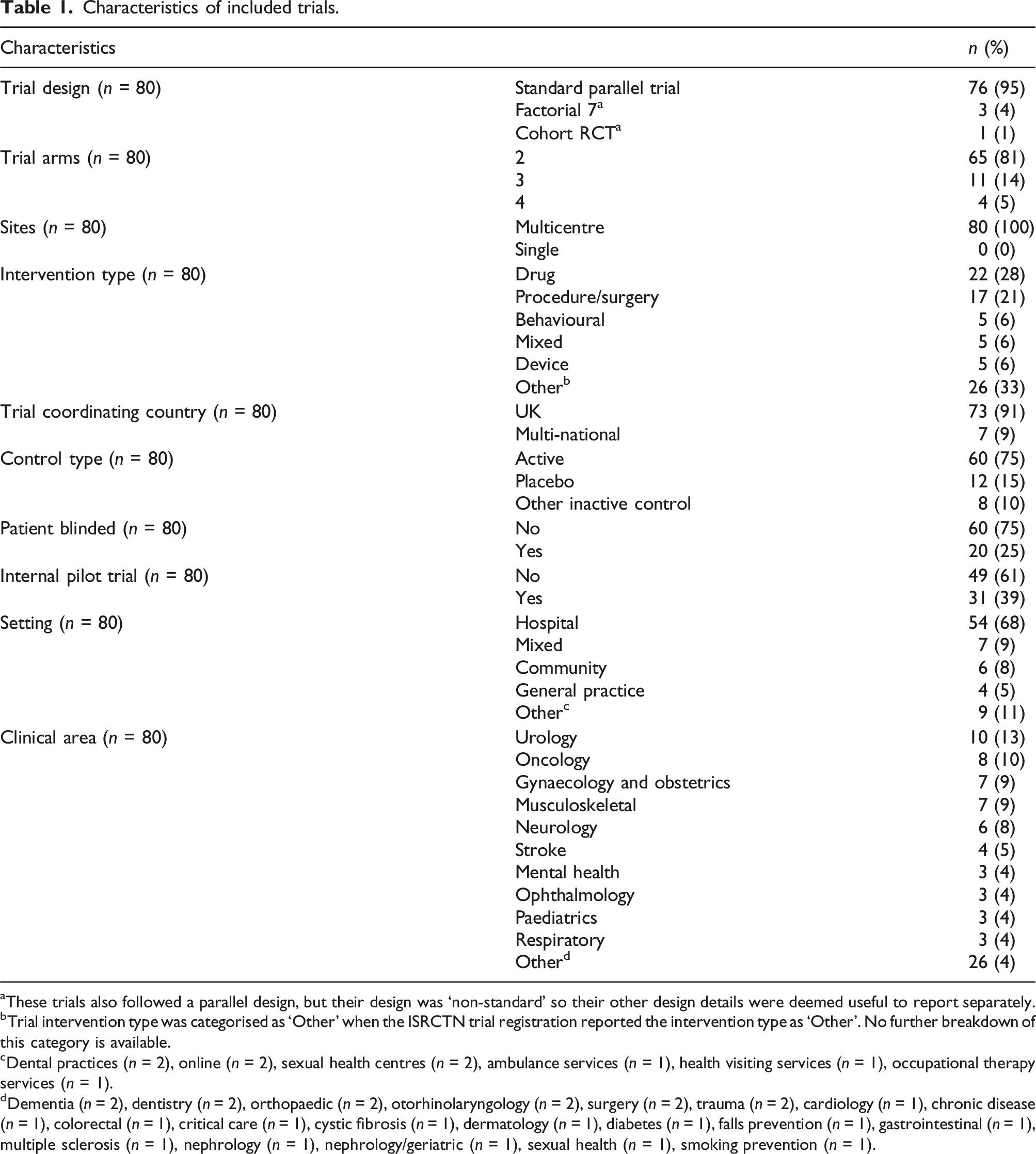
aThese trials also followed a parallel design, but their design was ‘non-standard’ so their other design details were deemed useful to report separately.
bTrial intervention type was categorised as ‘Other’ when the ISRCTN trial registration reported the intervention type as ‘Other’. No further breakdown of this category is available.
cDental practices (
dDementia (
Summary of retention-associated activity in the included trials
Attrition and reasons for missing data
The majority of included trials adjusted their sample size calculation to account for expected participant attrition (89%,
Of the included trials, the most common reasons for missing outcome data, were due to participant withdrawal, participants being lost to follow-up, and participant death. Reasons for participant withdrawal were varied, and often were not reported. Of those that reported participant withdrawal reasons, common reasons were due to the treatment allocation or perceived effectiveness of treatment; the burden of data collection; and personal reasons such as the participant moving away or becoming ill. It was not possible to report this data quantitively due to inconsistency of reporting.
SWATs and internal pilot studies
Just under one third of the trials included a SWAT (30%,
Of the 31 trials that included pilot studies, roughly one third (
Identified retention strategies with mapping
Amongst the included trials, 56 retention strategies were identified, with 409 total occurrences. Most trials used several retention strategies, with the mean number of retention strategies identified per trial as 5 (IQR 3:7). Two trials had no retention strategies identified26,27 and the maximum strategies identified per trial was 18. 28
Identified retention strategies with mapping, ranked by frequency of use.
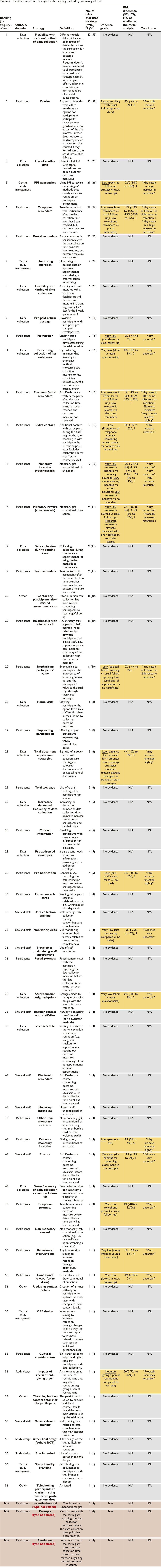
To note, within the ORRCA ‘participant domain’ category, there were 11 occurrences where not enough detail on the strategy was provided to adequately classify the strategy. Three categories were created and are included at the bottom of the table for reference, but these categories were not included in the ranking, mapping, or prioritisation exercise. These were: reminders-type not stated (
Commonly used strategies amongst the included trials, generally were participant or data-collection orientated. The most frequently occurring strategy was offering flexibility with data collection method/location, which was used by over half the included trials (
The following results are reported for strategies that were used by greater than 10% of trials, refer to Table 2 for the remaining strategies. Rankings refer to the commonality of the strategy amongst the included trials.
Postal reminders for participants ranked 6th (
SWAT prioritisation
Top-10 most frequently occurring retention strategies, with mapping to evidence, PRIORITY 2 questions, and example SWAT research questions.
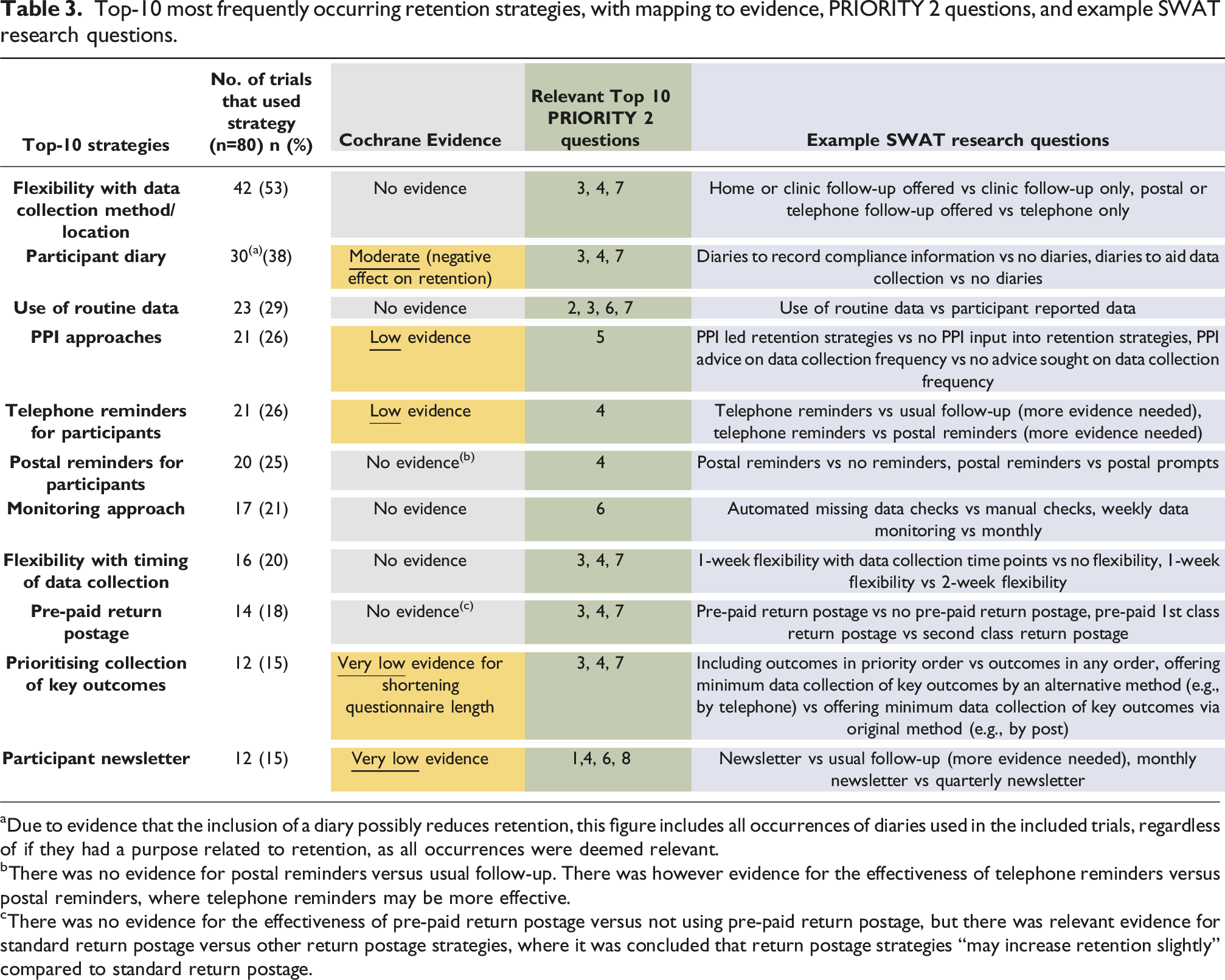
aDue to evidence that the inclusion of a diary possibly reduces retention, this figure includes all occurrences of diaries used in the included trials, regardless of if they had a purpose related to retention, as all occurrences were deemed relevant.
bThere was no evidence for postal reminders versus usual follow-up. There was however evidence for the effectiveness of telephone reminders versus postal reminders, where telephone reminders may be more effective.
cThere was no evidence for the effectiveness of pre-paid return postage versus not using pre-paid return postage, but there was relevant evidence for standard return postage versus other return postage strategies, where it was concluded that return postage strategies “may increase retention slightly” compared to standard return postage.
PRIORITY 2 important unanswered questions ranking. 9

Given that there was poor quality evidence to support all the top-10 most frequently used strategies, and that all these strategies could be mapped to PRIORITY 2 questions (indicating some alignment in research priorities), these strategies were identified as a priority for further SWAT research. The exception to this logic concerns participant diaries, which had moderate certainty evidence that they reduced retention. This strategy is included as a SWAT priority topic as future SWAT research into diaries is considered a priority. Given their high frequency of use, increased evidence is needed on the certainty of their effect on retention (e.g., moving from moderate, to high evidence certainty), and research is needed to determine in what situations diaries are most likely to reduce retention. PRIORITY 2 question nine and 10 were unable to be matched to the top-10 frequently used strategies in this review. Example SWAT research questions are displayed next to each priority retention research topic.
Retention strategy justification
Approximately one third of the included trials (30%,
Discussion
This review found a continuing problem with attrition in HTA funded trials (January 2020–June 2022). A total of 80 trials were identified for inclusion, with a median size of 518 randomised participants. There was a mean attrition rate of 12%, which was slightly lower than the mean anticipated attrition rate of 16%. For the average trial of 518 participants, this difference of 4% may on average result in the recruitment of 21 more participants than is necessary (4% of 518 = 20.7). As cost per participant in NIHR trials was previously estimated to be £2987, 6 this may increase the costs by £62,727 (£2987 × 21) per trial. If this figure was applied across the 80 trials included in this review, the NIHR could potentially save over half a million pounds annually. In the future, if more effective interventions were implemented to reduce retention, then an even lower anticipated attrition rate could be built into sample size calculations, which would further drive down trial costs. In this report, the key attrition reduction strategies used by HTA trialists were identified and mapped against the best evidence from a recent Cochrane review of SWATs. The following section summarises and discusses the key findings.
Principal findings
This review found that the most used retention strategies (Table 4) have mostly low, very low, or no evidence in support of their effectiveness. Participant diaries were the second most common retention strategy (used by 38% of trials) but had moderate certainty evidence that they may reduce retention. As this was the only commonly used strategy that may be harming retention, this is a priority strategy for future SWAT research. Future SWAT research is also recommended for the other commonly used strategies which all need considerably more evidence to support their effectiveness. Example SWAT research questions are displayed next to each priority retention research topic in Table 4. Further transparency is needed surrounding why certain retention strategies are chosen by trial teams, as only approximately one third of the included trials reported some form of justification for their choice, with very few citing literature evidence. Justification for future use of participant diaries is particularly needed, due to the evidence that this strategy may reduce retention.
This review highlighted that only 9% of the included trials embedded a randomised retention SWAT into their design, implying this is not currently common practice. With the clear need to increase the evidence base for the effectiveness of retention strategies, and with randomised SWATs providing the best quality evidence for the effectiveness of such strategies, it is important that future trialists understand the benefits of embedding randomised retention SWATs into their trial design.
Out of the trials in this review that included an internal pilot, only approximately one third had retention criterion, to inform continuation to the main study. Based on this finding, it appears that retention may be commonly being deprioritised, compared with recruitment at this initial study stage. Considering the significant methodological, statistical, ethical, and financial challenges that poor retention can bring, early identification of poor retention is a must to enable appropriate counter strategies to be implemented. We hope that more triallists will consider the value of including retention criterion in the internal pilot.
Strengths
This is the first study that has examined current retention practice of trials published to the NIHR HTA journal library and has mapped strategies used in these trials to evidence for their effectiveness. Trials registered to the NIHR HTA library are required to publish a trial report so risk of publication bias in this review is low, and the detailed level of reporting required by the library ensures all relevant data is likely to have been captured. Retention strategy use has been established directly from the trial reports, which offers a more precise estimate compared to strategies trial teams may say are commonly used. Cochrane reviews are high quality, regularly updated, peer reviewed, systematic in nature, and aim to synthesise all relevant empirical evidence, so the mapping exercise in this review is highly likely to have encompassed most, if not all, available randomised SWAT evidence for each strategy.
Limitations
Trial authors were not contacted for further information, so this review was unable to include data items that were not reported in the HTA report or published trial protocol. If retention strategies were used by trial teams but not clearly reported, then they may not have been captured in this review. The results of this review may not accurately reflect trials published outside of January 2020–June 2022. The HTA programme is publicly funded and funds high quality research that is mostly conducted by CTUs, and so findings may not represent commercially funded trials, international trials, or other non-CTU delivered trials. Using the ORRCA retention research domains to classify retention strategies could be seen as a limitation. Using this structured method to initially identify strategies may have resulted in different classifications and/or grouping of strategies, had the ORRCA domains not been used, which consequently may have affected the mapping process. This review has examined the retention strategies for a sample of large individually randomised controlled trials. We excluded pilot, feasibility and cluster designs as these are likely to use different retention strategies from the ‘standard’ trial design and their findings may not be generalisable to the majority of large individually randomised trials. However, it would be useful to review retention strategies for these designs, particularly for cluster trials. It is possible that relevant evidence may have been identified from the Cochrane review if strategies had been identified and grouped differently. For example, there was evidence in the Cochrane review for telephone versus postal follow-up, but neither telephone follow-up nor postal follow-up were identified as individual retention strategies in this review. Instead, offering flexibility with data collection location/method was identified as the strategy and consequently, there was no evidence in the Cochrane review to directly support this strategy. This review did not consider non-randomised SWAT evidence or cost-effectiveness evidence. The Cochrane retention review included SWATs up to January 2020, and so there may be new relevant evidence for strategies that was unable to be accessed in the mapping exercise in this review.
Comparisons to other studies
Some consensus has been reached on current retention practice and priority SWAT research topics between the results of this review and research by Kearney et al.
5
Both identify telephone reminders, flexibility of appointment times, pre-paid envelopes, and newsletter strategies as within the top-10 most commonly used retention strategies. Further, offering multiple methods of data collection was identified as the most frequently used strategy in this review, and was reported as the fourth most recommended strategy by CIs to mitigate missing data in Kearney’s research. In addition, just under half of this review’s recommended SWAT priority topics (
Bower et al. 30 identified six priority recruitment and retention interventions, with none matching the priorities discussed in this review. Differences in results may be explained as some of their identified intervention priorities had a definite focus on recruitment over retention (e.g., observing recruitment), research was conducted prior to the Cochrane retention review 8 used to inform this review, and different methods were used to establish current retention practice. Apart from the PRIORITY 2 findings, 9 which were used to inform this research, no further studies were identified that advised priorities for retention research.
Conclusion
This study systematically identified retention strategies from a sample of 80 trials published to the NIHR HTA library and mapped these strategies to evidence for their effectiveness from the Cochrane retention review. Of the 56 retention strategies identified, there were none that had high quality supporting evidence for their effectiveness, and amongst the Top-10 most frequently used strategies, mostly no evidence, low evidence, or very low evidence was identified. Diaries were the second most frequently used strategy, despite moderate certainty evidence of their negative impact on retention. Little justification for the choice of retention strategy used by trial teams was identified in the final report. This study supports the need for more SWATs and helps identify priority areas for future SWAT research. These priorities could be used with other priority lists to improve the efficiency in the conduct of further SWATs.
Supplemental Material
Supplemental Material - Are retention strategies used in NIHR HTA trials supported by evidence for their effectiveness? A systematic mapping review
Supplemental Material for Are retention strategies used in NIHR HTA trials supported by evidence for their effectiveness? A systematic mapping review by Rosalind Way, Adwoa Parker and David J Torgerson in Journal of Research Methods in Medicine & Health Sciences.
Supplemental Material
Supplemental Material - Are retention strategies used in NIHR HTA trials supported by evidence for their effectiveness? A systematic mapping review
Supplemental Material for Are retention strategies used in NIHR HTA trials supported by evidence for their effectiveness? A systematic mapping review by Rosalind Way, Adwoa Parker and David J Torgerson in Journal of Research Methods in Medicine & Health Sciences.
Supplemental Material
Supplemental Material - Are retention strategies used in NIHR HTA trials supported by evidence for their effectiveness? A systematic mapping review
Supplemental Material for Are retention strategies used in NIHR HTA trials supported by evidence for their effectiveness? A systematic mapping review by Rosalind Way, Adwoa Parker and David J Torgerson in Journal of Research Methods in Medicine & Health Sciences.
Footnotes
Acknowledgements
The Authors acknowledge the contributions of Laura Clark and Izzy Coleman who were involved in initial project conceptualisation discussions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. Author AP is funded by the National Institute for Health Research (NIHR) Advanced Fellowship (award reference: NIHR302256). The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
