Abstract
Background
Communicating and disseminating individual results to research participants is an ethical imperative, however, this practice is still unusual. In the present study, we investigated two delivery formats of individual results to older participants on the perspectives of understanding (main outcome), satisfaction, and short-term psychological impact.
Methods
This Study Within A Trial (SWAT) is a randomized, single-blinded (outcome assessors), parallel-group intervention hosted by “Hypertension Approaches in the Elderly: a Lifestyle study” multicenter, two-arm, randomized trial (HAEL Study). Participants who entered the HAEL Study in July 2019 or after were eligible. Randomization was generated by computer and allocation concealment by an independent investigator. The delivery of individual results was carried out in individual or group meetings between December 2019 and September 2020 at a Clinical Research Center. Outcomes were assessed by an unvalidated questionnaire on a 5-point Likert scale and multiple choice questions.
Results
Of the 20 participants who agreed to participate in the SWAT, 10 from the individual format and 7 from the group format, with a mean age of 68 years old, were evaluated through per-protocol analysis. Most participants showed good understanding of their results in both delivery formats - individual 70% (7/10) and group 71% (5/7) (p=1.00). Satisfaction with the results delivery format was reported in both groups, with moderate negative emotional impact. Any research-related physical harms were not identified.
Conclusion
Both formats for delivering individual results generated adequate understanding and satisfaction with low negative emotional impact to a partial sample of older participants in the HAEL Study.
Introduction
Communication is a pivotal process in clinical research. Notably, trial participants are stakeholders before, during and after an interventional study, which demands adequate communication exchanges on pre-trial clarifications and proper guidance when they have engaged in the research. In this regard, the dissemination of both aggregate findings (i.e. findings disclosing the participants’ characteristics and study outcomes) and individual results (i.e. data from exams, evaluations, and tests performed during the study) are of particular importance to respect the participants’ rights to receive their own data and value their key role in science.1–5 Although there are guiding principles for returning individual results3,6 to research participants, more clarity is needed regarding the format of study reports and their contents.7,8
Among potential barriers for proper trial dissemination for research participants, points of concern include insufficient budgetary planning, misinterpretation of findings, and possible negative emotional impact among participants.1,7,8 Nonetheless, the Patient and the Public Involvement from the planning to the dissemination of research2,9–11 can minimize such barriers and facilitate participant-researcher communication, creating strategies that enable better understanding of scientific data.11–13 Additionally, such practices may promote greater satisfaction and engagement of participants with the studies, enabling conscious decision-making in relation to their own health condition and necessary care.1,11,14–16
Surveys show that most participants (∼90%) declare interest in knowing aggregated or individual results from the studies they participated in,4,15,17 albeit the practice of dissemination of such data is still uncommon among researchers.18–20 Among approximately 140 health researchers surveyed in 2017, 25% reported disseminating the results verbally to the community and only 8% reported having the intention to return results to participants and a specific plan to do so. 20 Out of the 3381 research participants registered in a health research database, 33% reported receiving results from the studies in which they participated, whereas 52% had no opportunity to request their results. 19 Furthermore, the research on individual results dissemination has been explored in genetic and oncology studies 4 , however, scant data are available from other clinical populations. Because research data in clinical trials pertain to the participants, who should be understood as active research actors that use their time and put themselves at risk to contribute to scientific knowledge, we highlight the ethical need of making their individual results available. Beyond the feedback of their own data, such information may enable choices regarding their health and substantiate potential further involvement in research.
Previous research has indicated that older participants preferred to receive written and verbal health information 21 and research results in the format of a letter or face-to-face meetings.12,15,22 However, the implementation of these strategies for dissemination results is not clear in this population profile, so uncertainty as to the type of information, delivery format and communication style for dissemination of individual results prevails. Importantly, there is a growing interest in more transparency in exercise sciences23,24 and adequate tailoring of disclosure of results to participants could improve the perception of participants regarding the trial experience and physical activity engagement. Therefore, we conducted a study following the Study Within A Trial (SWAT) methods with the aim of evaluating two delivery formats of individual results to older participants of a physical activity program of the study entitled “Hypertension Approaches in the Elderly: a Lifestyle study” (The HAEL Study) on the perspectives of understanding (main outcome), satisfaction, and short-term psychological impact. The present study was exploratory.
Methods
Study design and host trial
The SWAT was framed as a randomized, single-blinded (outcome assessors), parallel group intervention hosted by a multicenter, two-arm, randomized trial entitled “The HAEL Study” (NCT03264443). The host trial lasted from September 2017 to August 2020, aiming to assess the efficacy of a 12-weeks combined exercise training program (supervised aerobic exercise and resistance training) compared with a health education program to reduce blood pressure in older adults with hypertension. More details on the trial setting, assessments, study sites, and interventions are available in the HAEL Study protocol. 25 The present study was registered on the Northern Ireland Hub for Trials Methodology Research SWAT Repository (SWAT122) and its protocol is openly available at the Open Science Framework (OSF) (https://osf.io/cb6jx/).
Participants
For this SWAT, we invited all participants who entered the HAEL Study in July 2019 or after, and who had reached a minimum frequency of 25% in the intervention sessions of the host trial, which translates into 3 sessions in the health education program or 9 sessions in the combined exercise training program at the end of the 12 weeks.
Randomization and allocation concealment
The participants were allocated to delivering results in group or individual format based on computer-generated random numbers (random.org), with a 1:1 ratio, stratified by group on the host trial and with permuted blocks of random sizes. Allocation concealment was implemented by an independent investigator (DU) not involved with the intervention and data collection. Due to the nature of the interventions, neither the investigator who delivered the individual results nor the participants were blinded. Blinding was implemented for outcome assessors and data analysts of primary and secondary outcomes.
Setting
The data were collected at the Clinical Research Center/Hospital de Clínicas de Porto Alegre (Porto Alegre, RS, Brazil), from December 2019 to September 2020.
Ethics approval and consent to participate
The research was approved by the Ethics Committee for Research with Human Subjects of the Hospital de Clínicas de Porto Alegre (CAAE: 20,688,919.0.0000.5327). The procedures were guided by the Declaration of Helsinki and resolution no. 466/2012 of the National Health Council. All patients provided written voluntary informed consent.
Interventions
The delivery of individual results to the participants of the HAEL Study was carried out either by an individual approach or group meetings. Both delivery approaches occurred in person and were conducted by the same investigator (ATDN), graduated in Physical Therapy. In both delivery formats, a printed report was given to participants, displaying an initial welcome message followed by the individual results of blood chemistry, body composition, functional and strength performances, office blood pressure, cardiopulmonary exercise test, and ambulatory blood pressure monitoring associated with reference values available in the literature.
The individual format was carried out at a visit set to last up to 15 minutes. In this session, the investigator handed the report to the participant and read the results together with him/her, clarifying any doubts that arised.
The group-based format (one investigator with 3–4 participants) was carried out with the delivery of the printed report so that the participants could follow their information individually. This intervention was guided by the presentation of slides set to last up to 15 minutes, in which the researcher only instructed the participants by presenting the order of the data delivered and informing the reference values considered normal within each age group, thus maintaining the confidentiality of the participants with group delivery. After explaining the standardized structure and displayed variables, the participants had another 15 minutes to ask questions and clear up their doubts, totaling up to 30 minutes of the visit. In comparison to the individual approach, this group visit was set to be longer due to possible interaction between participants based on individual and peer questions.
The standardized report is openly available at the study materials repository (https://osf.io/5kdqg/).
Measures and outcomes
The SWAT participants initially answered a self-administered questionnaire with 7 questions on sociodemographic characteristics and 3 opens questions regarding expectations upon receiving the results. Afterwards, the Montreal Cognitive Assessment (MoCA) test was applied to assess mild cognitive impairment. 26 This tool consists of 11 questions measuring cognitive domains through several tasks: short-term memory (delayed recall), visuospatial abilities (clock-drawing task and a three-dimensional cube copy), executive functions (trail-making test, phonemic verbal fluency, verbal abstraction), attention, concentration and working memory (sustained attention task, a serial subtraction task, and digits forward), language (nomination, sentence repetition), and orientation to time and space. The application time is approximately 10–15 minutes, and the test final score was determined by the sum of different cognitive domains. The score range varies from 0 to 30, whereas a final score of 26 or higher is considered as “normal”, the scores of 25 or below are considered to be indicative of possible “mild cognitive impairment”. To counterbalance the effect of lower educational levels, 1 point was added to the final score of those individuals with ≤12 years of education. 26 This instrument was used as a control variable for possible confounding on the main outcome. The application was conducted by a physiotherapist trained and certified in MoCA administration investigator (ATDN), and the score analyzed together with a second researcher.
The assessment of report understanding, satisfaction with dissemination format, and short-term psychological impact outcomes was performed using a non-validated self-administered questionnaire, which was based on previous questionnaires.11,12,16,19,27,28 This questionnaire included 14 items in 5-point Likert scale and additional 6 multiple choice questions that were applied to assess the understanding of data after the intervention as well as comparison of some results (e.g. cholesterol levels) in relation to reference values (https://osf.io/qykmp/).
Primary outcome
The prespecified primary outcome in this study was the performance in the five multiple-choice (single answer) questions measuring participants' understanding in relation to their own data. The items of questionnaire considered for evaluation of the understanding domain were related to the following variables: (i) cholesterol; (ii) body mass index; (iii) functional tests battery; (iv) blood pressure; (v) cardiorespiratory capacity. We considered it as an “adequate understanding” when the participant achieved four to five questions answered correctly, whereas zero to three questions answered correctly were considered as “inadequate understanding”.
Secondary outcome
The domains related to the satisfaction with results delivery and psychological impact were assessed using Likert scale questions. The satisfaction was assessed considering: (i) object; (ii) quality; and (iii) effect of delivery (questions 2–9). Psychological impact was assessed considering: (i) level of concern; (ii) level of anxiety; (iii) fearful feelings; and (iv) feelings of sadness (questions 12–15).
Three remaining items (questions 10, 11 and 19) of the questionnaire were analyzed separately and not within the domains. These questions were related to the recommendation to participate in studies such as the HAEL Study, general self-assessed understanding of the individual report, and interpretation of blood pressure values after the trial. As we did not present the normative values of blood pressure immediately before or during the disclosure of results, the blood pressure levels considered as “adequate” were solely based on a subjective analysis from the participants. Therefore, we disregarded this item to compute “correct questions” within the understanding domain.
Sample size
The SWAT started when the HAEL Study was already underway, therefore no formal sample size calculation was performed, which is possible under the SWAT methodology. 29 Thus, from the sample calculation of the host trial of 184 participants, we anticipated that 50 participants still needed to be recruited to complete the sample and receive the individual results. For this reason, this was the estimated sample size for entry into the SWAT. The projected sample size was not reached mostly because there was an early closure of the HAEL Study due to the COVID-19 pandemic.
Statistical analyses
Data were analysed using descriptive and inferential statistics. The normality of the data distribution was assessed using the Shapiro Wilk test. The difference between delivery formats and understanding was assessed using Fisher’s exact test. Spearman’s correlation was used to examine the association between the level of understanding and the MoCA instrument, and the association between the level of education and the MoCA was verified by Pearson Chi-Square. Continuous data are presented as mean ± standard deviation and categorical data as absolute and relative frequencies. The analyses were performed per protocol using the PASW Statistics for Windows software (Version 18.0 Chicago: SPSS Inc). The level of significance was set at 5%.
Changes to the planned protocol
Some protocol changes should be mentioned. First, we planned the group format meeting to take place with 4–6 participants. However, due to necessary schedule arrangements, most group meetings occurred with 3–4 participants (as described in the intervention item). Second, given the context of the pandemic, the delivery of results from 3 participants who were allocated to receive in group format was modified for individual delivery, thus these data were not considered in the statistical analysis. Third, our data analysis plan was modified. In evaluating the understanding domain outcome, we modified the score analysis that ranged from 0 to 5 points. To make the score more readily interpretable, we simply inverted the numerical scale of a given individual result, displaying a score of lower scores (zero to three) as an “inadequate understanding”, and increasing score (up to 5) as a more adequate understanding. In addition, changes were made to the statistical tests due to the small sample size, the low frequency of responses made it impossible to carry out the tests suggested a priori.
Results
Of the 50 participants estimated to enter the host trial from July 2019, only 33 participants entered and 24 completed the activities before lockdown due to the COVID-19 pandemic. Of these, 20 agreed to participate in the SWAT (Figure 1). Two meetings were held for group delivery, both planned with 4 participants, however, one participant did not show up, which resulted in 3 participants for one session of the group deliveries. In additionally, because of the trial termination related to the COVID-19 pandemic, two participants randomized to the group-based format could not have their group sessions scheduled, resulting in individual delivery of results. Therefore, following the per-protocol analysis, the data from these 3 participants were not included in analyses, and we considered only data from 17 participants. Flowchart SWAT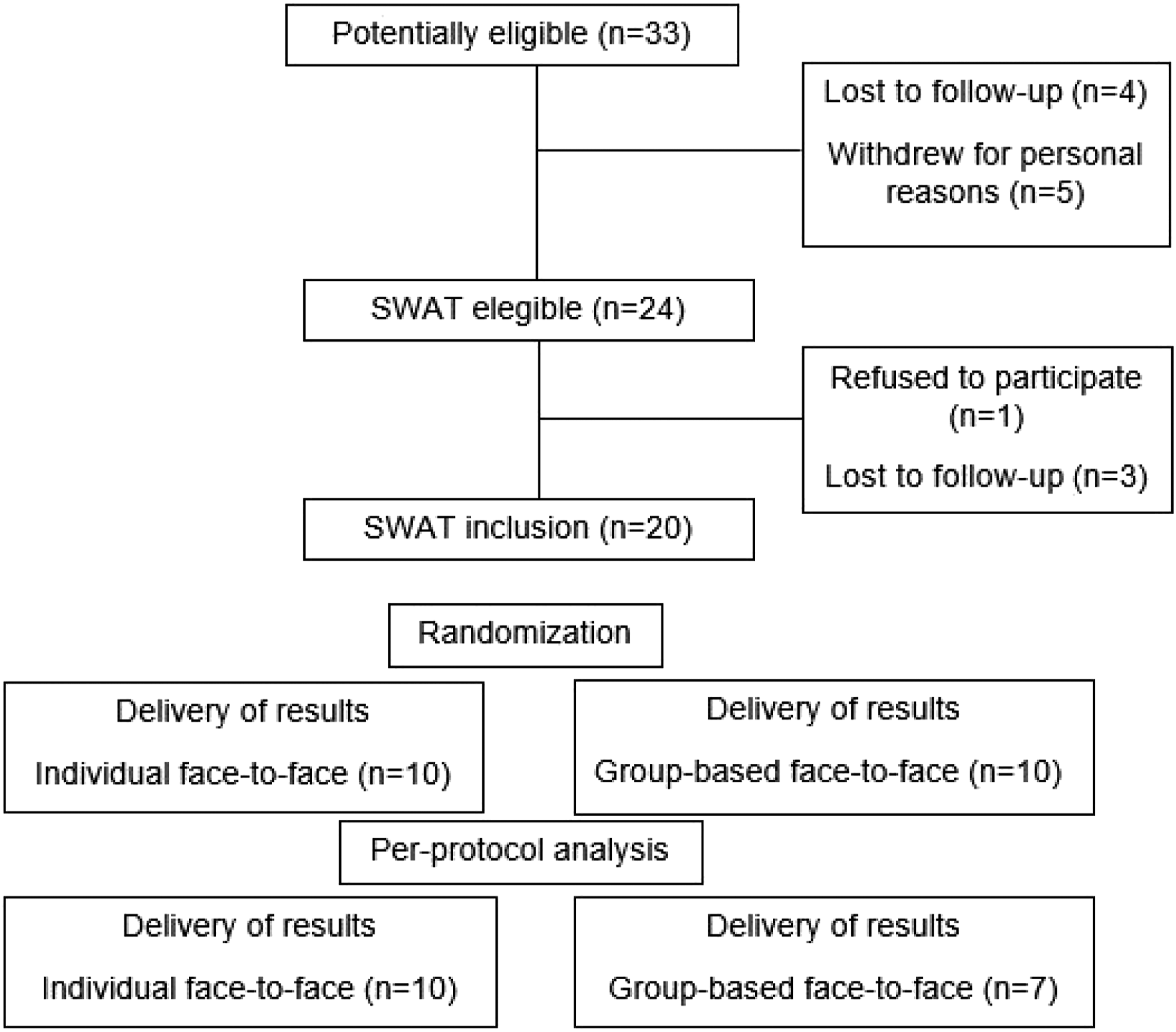
Characteristics of SWAT participants.
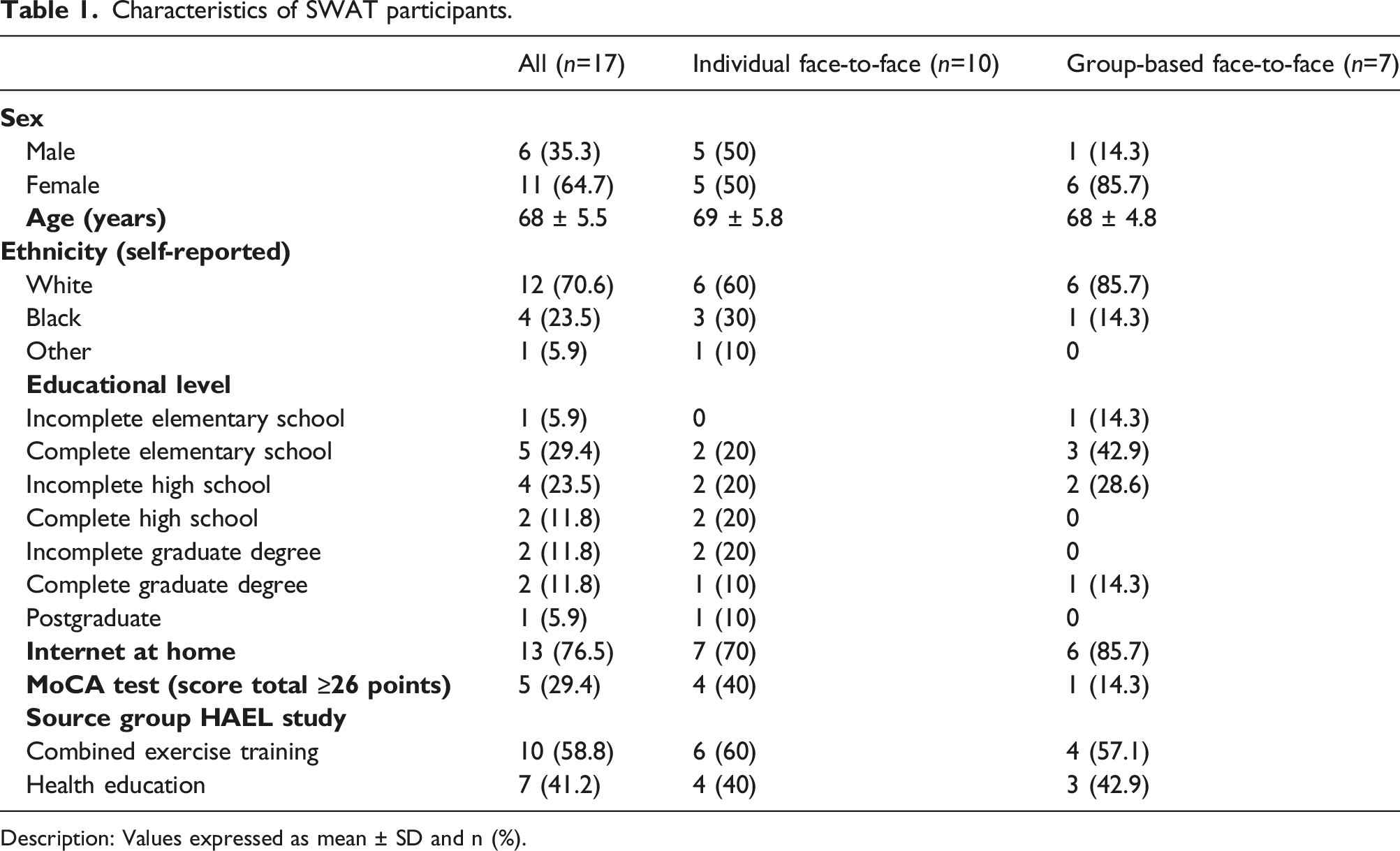
Description: Values expressed as mean ± SD and n (%).
Approximately 29% of participants reached a score of ≥26 points in MoCA test; years of education were not associated with the MoCA final score (p=0.372).
In the pre-intervention questionnaire, most participants (15/17) expected to receive all their results after the study, and when asked what they expected to understand from the information made available, most reported (12/17) “knowing about my conditions and health status”. However, we highlight some answers that varied, such as: “depending on the results, make a change in the routine, in food, get out of a sedentary lifestyle to improve the quality of life”; “I hope to understand if I need to seek medical attention”; “I hope to have a better understanding of what physical exercise has provided to improve my health”; “General orientation for health and mainly to improve memory”, and “How to take better care of myself and my health”.
Understanding domain.
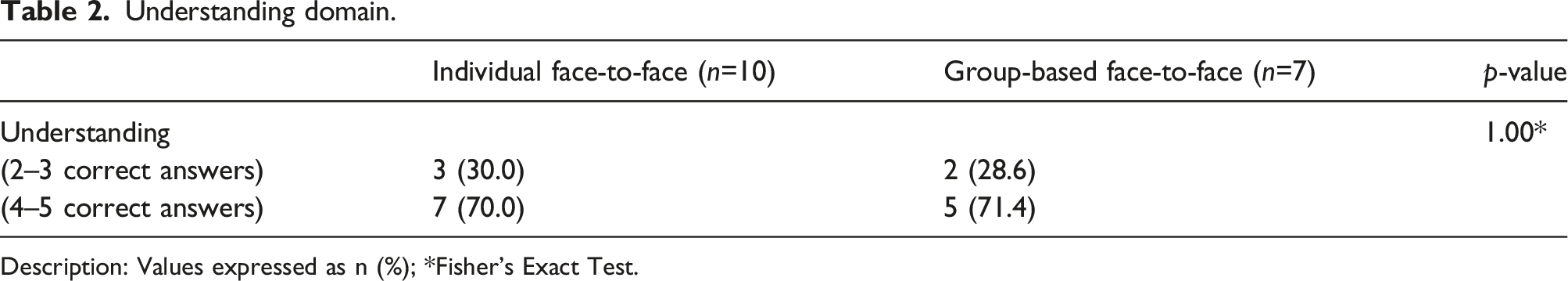
Description: Values expressed as n (%); *Fisher’s Exact Test.
In general, participants reported understanding the results that were presented to them (question 11). Of these, a total of 6 individuals indicated “I understood a lot” of which 4 received their report in the individual format and 2 in the group format. The other participants chose the option “I understood”. Regarding the participants' perception of their office blood pressure values after interventions in the HAEL Study, 15 participants considered their values as “good”, data ranging between 105/68 mmHg and 143/88 mmHg. Only one participant (from the group format) indicated to be “indifferent” to the blood pressure values (104/81 mmHg) and one (from the individual format) judged the values as “bad” (147/89 mmHg).
Satisfaction delivery of results.
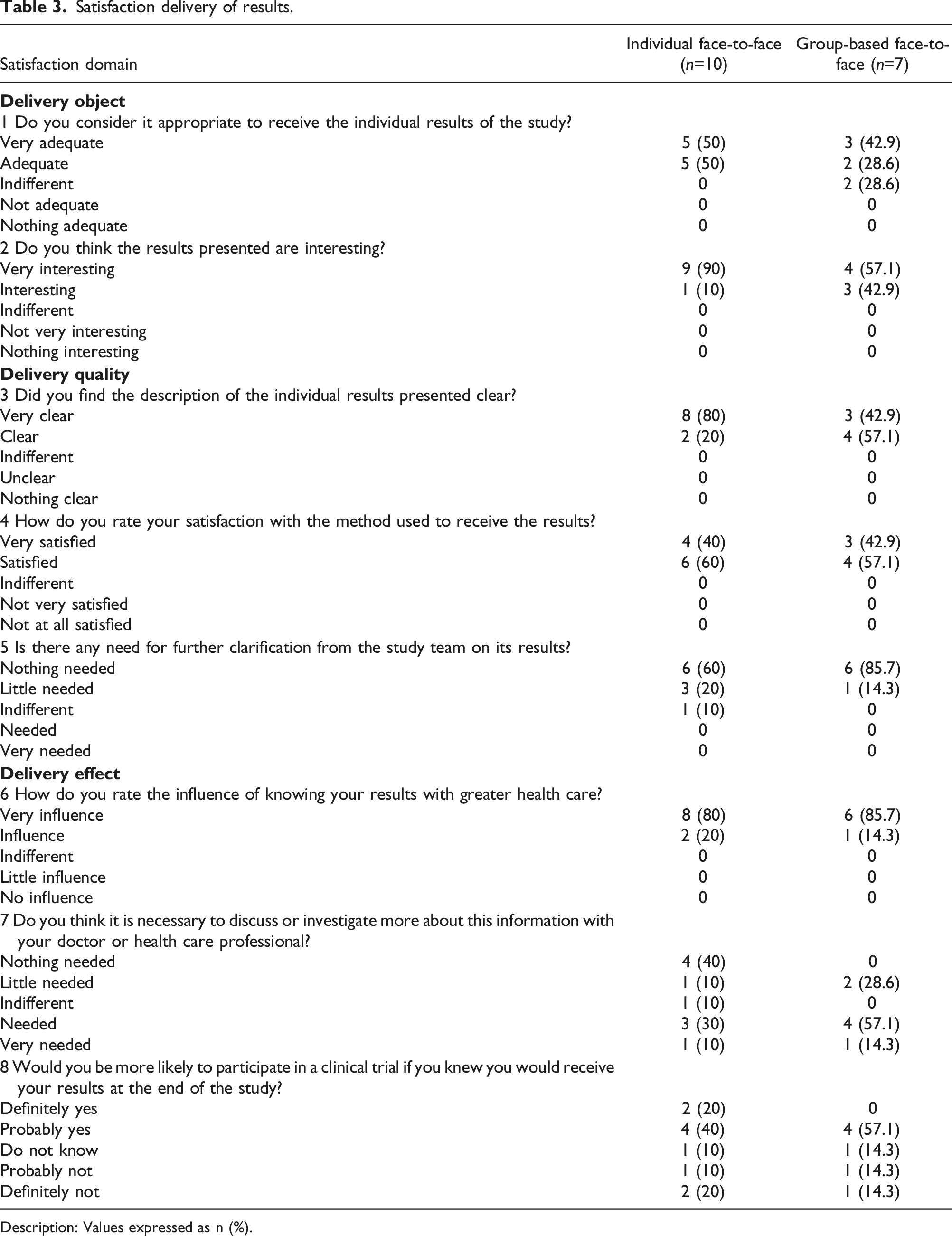
Description: Values expressed as n (%).
Psychological impact domain.

Description: Values expressed as n (%).
Harms
We did not identify any research-related physical harms or psychological discomfort, in addition to the outcomes assessed in the study.
Discussion
This SWAT identified that both delivery formats of individual results to older participants in the HAEL Study generated adequate understanding, satisfaction and moderate negative emotional impact. Our findings corroborate studies that evaluated the older participants' perspectives on satisfaction 16 and understanding 12 when receiving aggregated results of the study. Although there are recommendations and guides that mention the right of participants to receive their data and encourage researchers to disseminate aggregated and individual results,3,6,30 to our knowledge, this study is the pioneer in assessing the delivery of individual results in the physical activity field.
The concern with the misinterpretation of results is identified as a barrier to dissemination, as well as possible emotional burden,1,4,8 which leads researchers to mention that the results should not always be shared with participants. 8 The decreased sensory-perspective capacities of the aging process, which affect the ability to receive and treat information from the environment,31,32 can also be considered a concern to dissemination of results to older participants together with the condition of arterial hypertension that contributes to cognitive decline.33,34 However, we observed that most participants (12/17) with scores for mild cognitive impairment (below 25 points in the MoCA Test) 26 showed good understanding (4 and 5 correct answers). The use of simple language, explanations about sharing numerical data and visual strategies are recommended to ensure understanding among participants and may have contributed to our findings.22,30 Moreover, we emphasize that the interpretation of a score below the cutoff point considered as “normal” in the MoCA does not necessarily have a direct impact on cognitive function, since factors such as stress, fatigue, emotional state, and educational level may influence the test performance.26,35
We underscore that the dissemination of the individual results is a right and desire expressed by research participants4,12 even when it offers a possible risk of emotional impact.4,36 As observed in our data, the majority of participants (15/17) considered “adequate” or “very adequate” to receive the individual results of the study, and all reported as “interesting” and “very interesting” the data presented in their reports. In addition, knowing about their own results generated “Much influence”/“Influence” with greater health care, and interest in investigating or discussing more with a health professional. These findings reflect satisfaction with the delivery object and related effects, facilitating further actions to health promotion in trial participants.1,4
Together with the participant’s interest, understanding and satisfaction in receiving research results, the potential emotional impact should be considered. We reason that most trials with physical activity interventions would present some outcomes highly valuable to be known by participants, with moderate odds for generating negative emotional impacts.
In addition, involving participants more actively in the planning of results delivery may enable clear and effective communication between participants and researchers, reducing possible barriers to dissemination and empowering the participants for decision-making in health.3,9,10,28
Strengths and limitations
This SWAT provides important information about the dissemination of individual results to older participants, however, some limitations should be considered. First, although the outcomes of understanding, satisfaction and psychological impact were comparable in the two delivery formats, the small sample size in our study likely reduces the generalization of findings. Second, the questionnaire developed to assess the outcomes of interest contained few questions and was not validated, which may impair the reliability of the findings. In addition, we emphasize that the answer option as “indifferent” may have impaired the participants' interpretation of some questions and, consequently, generated response bias. As we did not identify questionnaires for individual results delivery in the literature, we refer to articles that evaluated the aggregated results delivery and we refined and organized the questions into domains, which can contribute to future validation studies. Third, SWAT participants were older adults with hypertension. Thus, the layout, format, and language of the results delivery were based on information from the literature for this population profile, which limits generalization to other target audiences. Fourth, the involvement of participants in the construction of different delivery formats was not considered since the initial phase, which could have identified other preferences to dissemination as email, text message or video. Despite these limitations, the study findings do support the importance of returning participants’ results and exercising the participant-centered communication. This study is an initial but important step to encourage further research and practices aimed at greater transparency and accountability to trial participants, especially with a focus on effective and clear communication for older subjects or those with potential difficulties in understanding their data. Furthermore, we encourage researchers to replicate this SWAT proposal in other trials, aiming at the aggregation of various SWATs to future evidence syntheses.
Conclusion
Older participants showed adequate understanding and satisfaction in both formats for delivering individual results face-to-face meetings, and moderate negative emotional impact. From this study we suggest further SWAT research on the dissemination of individual results, mainly in the field of physical activity, based on patient and public involvement through focus groups, identifying preferences of delivery formats and evaluating the effects of this practice to research participants at advanced ages or with possible data assimilation difficulties.
Footnotes
Acknowledgements
The authors are grateful to all HAEL participants and to the roles of Anderson Donelli da Silveira, Akemy Kawany Neubert Kamitoyo, Bruna Góes Moraes and Raíssa Monteiro in supporting this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The SWAT or staff involved have been funded by the same institutions as the host study, as listed below. These funding bodies have no role in relation to study design, data collection, analysis and interpretation of data, or in writing the manuscript. This study was financed in part by: Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) – Finance Code 001; Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) [number 429,849/2016–8]; Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS); Fundo de Incentivo à Pesquisa e Eventos, Hospital de Clínicas de Porto Alegre (FIPE/HCPA); National Institute of Science and Technology for Health Technology Assessment (IATS/Brazil). ADN, LOP received doctoral funding support from the CAPES foundation. NLO and CEB received post-doctoral fellowship funding support from the IATS foundation. LPS received funding support from the IATS foundation. DU received research productivity grants and support from the CNPq foundation.
Trial registration
Northern Ireland Hub for Trials Methodology Research SWAT Repository (SWAT122). HAEL Study ClinicalTrials.gov (NCT03264443).
