Abstract
Background
Randomised controlled trials (RCTs) often struggle with various aspects of participant recruitment, including engaging clinicians to recruit effectively, and subsequently fail to reach their target sample size. Studies evaluating interventions to improve recruitment aimed specifically at recruiters to the trial are limited in number. The RCTs embedded into the World Hip Trauma Evaluation (WHiTE) cohort study use Trainee Principal Investigators (TPIs) to help manage and drive recruitment at trial sites. No formalised training or support is provided by central trials units to the TPIs. Additionally, trial recruiters receive a generic automated email confirming randomisation to the trial with no other communication to influence or incentivise their behaviour to further recruit. The primary aim of this factorial trial was to evaluate the effectiveness of an educational intervention to TPIs and a positive reinforcement intervention via an email (digital) nudge on increasing recruitment. Secondary aims included feasibility of implementing the interventions and surveying TPIs on the educational package quality of content, delivery and ongoing support.
Design
This was a multicentre, open, cluster, 2x2 factorial RCT embedded in the WHiTE 8 COPAL RCT, in which research sites were randomised 1:1:1:1 to receive the enhanced TPI package, the digital nudge intervention, both, or neither.
Results
1215 patients were recruited to the WHiTE 8 COPAL trial across 20 sites during the SWAT between August 2018 and March 2019. There was a statistically significant interaction between the interventions (IRR 2.09, 95% CI 1.64 to 2.68, p < 0.001). There was a statistically significant benefit on recruitment (IRR 1.23 95% 1.09 to 1.40, p=0.001) from utilizing an enhanced TPI education intervention. The digital nudge intervention had no significant impact on recruitment (IRR 0.89 95% CI 0.79 to 1.01, p=0.07). Within enhanced TPI package sites, the digital nudge had a beneficial effect, while in the standard practice TPI sites it had a detrimental effect. Feasibility analysis showed the median time to site digital nudge and enhanced TPI set up were one day and 17 days, respectively. 353 digital nudges were created taking an average of 12 min to construct, log the activity and then disseminate to recruiters. Median induction time for enhanced TPI was 32 min and 100% of the groups were extremely satisfied with the induction content, delivery and ongoing support.
Discussion
An education and support programme targeted at surgical TPIs involving a digital education package, 1:1 telephone induction and subsequent support package was effective in increasing recruitment in the first 6 months of trial commencement. There was no evidence for the effectiveness of the digital nudge intervention in isolation, although our results show that when combined with an education programme, it leads to enhanced effectiveness of that programme.
Keywords
Background
Randomised controlled trials (RCTs) are considered the gold standard when evaluating the efficacy and effectiveness of healthcare interventions. There are an increasing number of UK pragmatic orthopaedic randomised controlled trials being conducted investigating treatment options with the aim of determining the most effective and cost-efficient choice for patients and the National Health Service (NHS), respectively.1–4
In contrast, there is still only a limited evidence base regarding optimal trial design and trial processes. One method of increasing the evidence base is by embedding a self-contained study in a host trial for the purpose of evaluating or creating new methods of conducting trial processes. This embedded methodology is known as a ‘study within a trial’ (SWAT).5,6
Randomised controlled trials often struggle with various aspects of participant recruitment, including engaging clinicians to get involved effectively in recruitment, and subsequently fail to reach their target sample size. Many trials are forced to extend trial timelines or, due to insufficient funding, either revise their sample size downwards or close trials prematurely.7–11 Improving recruitment to RCTs is therefore a significant area for efficiency gains. Randomised and quasi-randomised trials that have targeted methods of improving recruitment to RCTs have been evaluated in a Cochrane systematic review. 12 There were limited studies of interventions directed towards healthcare professionals and other persons involved in recruiting participants to clinical trials, thus highlighting a need to evaluate strategies directed towards this cohort.
Clinical trials often recruit participants from multiple research sites. Whilst the trial is overseen by a single (or occasionally two) Chief Investigator (CI), each site has a delegated Principal Investigator (PI), whole role it is to take responsibility for the research activities relating to the trial at that site. The NIHR have seen the benefit of this role and have recently launched the NIHR associate PI programme to formalise this role. 13 Trainee (or Associate) Principal Investigators (TPIs) at a research site can work alongside, and gain experience from, the site PI. Typical responsibilities of a TPI include co-ordination of, and engagement in, the recruitment of patients to the trial at that site. There are no RCTs assessing the effect of a TPI on recruitment rate to a trial. In addition, the evaluation of this TPI role from a trainee perspective has not been undertaken.
The World Hip Trauma Evaluation (WHiTE) is an initiative to rapidly and efficiently investigate interventions to improve the outcomes of patients requiring hip fracture surgery. 14 All hip fracture patients being treated in participating centres are approached for consent to be enrolled in the WHiTE cohort, which collects standardised outcome data from participants. This cohort is a valuable tool in which to embed RCTs to evaluate novel treatment options for hip fracture patients. Trainee Principal Investigators, recruited and managed by the local PI, are being employed in some of the RCTs embedded in the WHiTE cohort. 14 A TPI manual is provided by the management team based at Oxford Trauma and Emergency Care, University of Oxford with no further education or support. There is, therefore, the potential to create an enhanced support package for TPIs consisting of formal initial education and ongoing assistance to enhance their knowledge and confidence in undertaking the role. A systematic review of training programmes for recruiters to RCTs found that these programmes were well received and increased recruiters’ self-confidence. 15 There was no definitive conclusion on the impact on recruitment rate and the need for further research in the area, ideally in a randomised evaluation, was highlighted.
An additional method of improving recruitment rates may be through the use of ‘nudging’. Fundamentally, this is a way of influencing an individual’s behaviour through an intervention without limiting their choice. This concept is used extensively in marketing, economics and healthcare promotion.16,17 Digital nudging is used regularly in RCTs, for example, emails, recruitment league tables circulated to recruiting sites and encouragement emails; however, there is limited formal assessment regarding the effect of nudging interventions targeted at recruiters on recruitment rates. In the WHiTE trials, an automated non-specific (generic and non-personalised) email is sent to the research staff at the recruiting centre after each patient randomisation. An additional email sent to the randomising clinician in a timely manner and incorporating features such as personalisation, appreciation for recruitment and praise may positively reinforce the behaviour of recruiting to a trial. Personalised emails to the recruiting clinician have not been evaluated using an RCT; however, there is evidence that personalised study invites improve patient recruitment in breast cancer survivors 18 and also in invitations for survey research.19,20 In this paper, we describe a 2x2 factorial, randomised trial evaluating both enhanced training and support for the TPI and personalised, digital nudging to recruiting clinicians to improve recruitment rates.
Methods
This SWAT investigated two different methods of enhancing recruitment: introducing a TPI with an enhanced training and support package to a site, and personalised, digital nudge to healthcare professionals involved in patient recruitment. The SWAT was implemented in a large, UK, multicentre orthopaedic RCT, the WHiTE 8 COPAL trial (ISRCTN15606075).
The primary aim is to: • Assess the effectiveness of an enhanced TPI package, and of a digital nudge, on the total number of patients recruited to the WHiTE 8 COPAL trial in the first 6 months of recruitment at a site.
The secondary aims are to: • Determine the time taken to implement each intervention from the time recruitment commences at the site. • Compare the randomisation rate of eligible participants in each of the intervention groups. • Gain feedback on the trainee perspective of the TPI role via a survey. • Determine the time needed to conduct the 1:1 educational training session for TPIs. • Determine the required time and method of additional contact for peer support of the TPIs.
Design
This was a multicentre, cluster, 2 × 2 factorial RCT embedded in the WHiTE 8 COPAL RCT, in which research sites were randomised 1:1:1:1 to receive the enhanced TPI package, the digital nudge intervention, both, or neither. This is an open trial and participating sites, the data analyst and trial team were not blind to allocation. The first site was randomised into the SWAT on 22/08/2018 and follow-up was completed on 20/09/2019. The trial was approved by the NHS Wales Research Ethics Committee and York University Research Governance committee and reported in accordance with the trial protocol and Consolidated Standards of Reporting Trials (CONSORT) statement (Figure 1). Consolidated standards of reporting trials flowchart.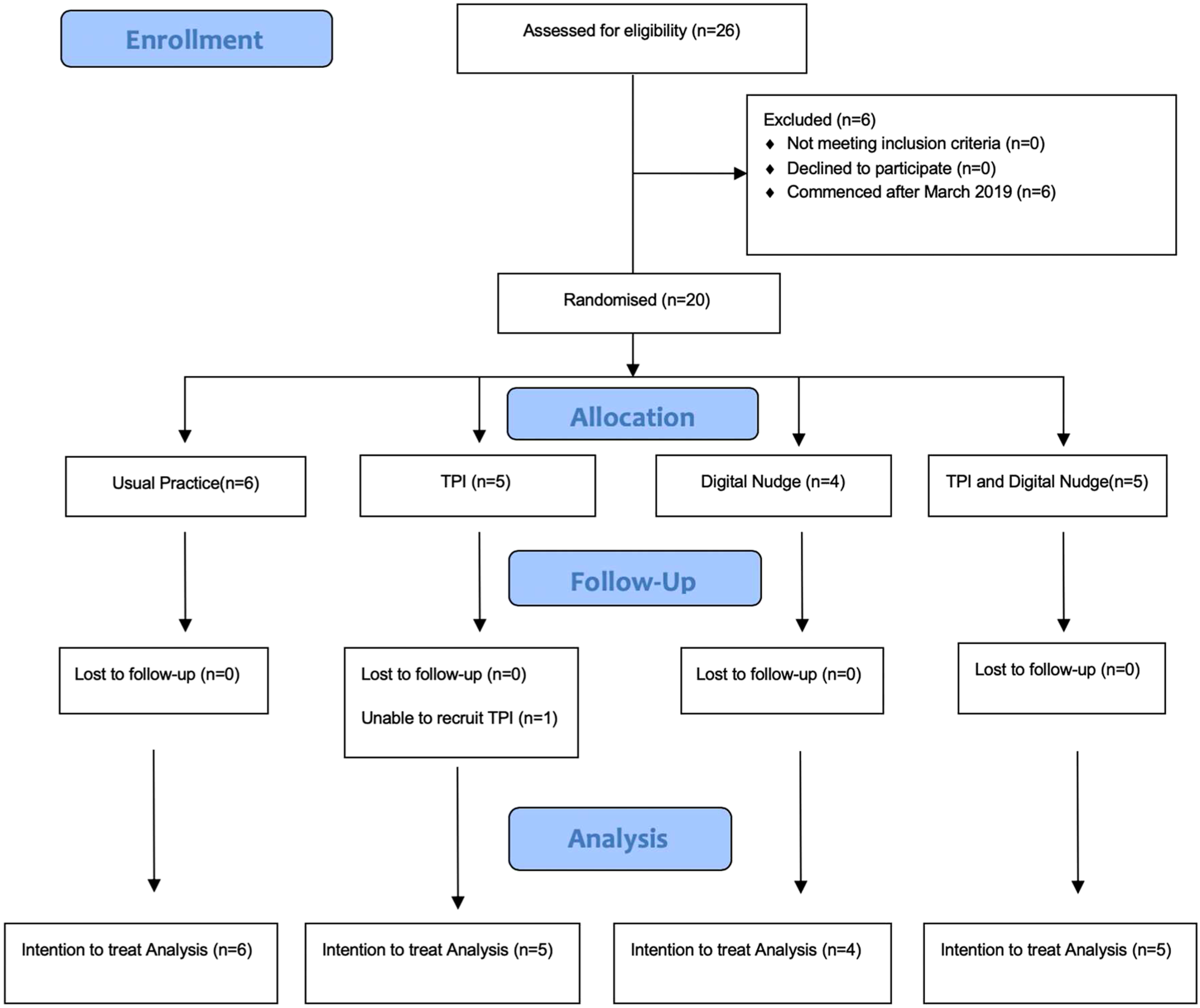
The first 20 WHiTE centres recruiting to the WHiTE 8 COPAL trial were included. The recruitment centre where the CI for the factorial SWAT (author NRA) was based was excluded to prevent bias.
Study within a trial interventions
EnTraP intervention summary.
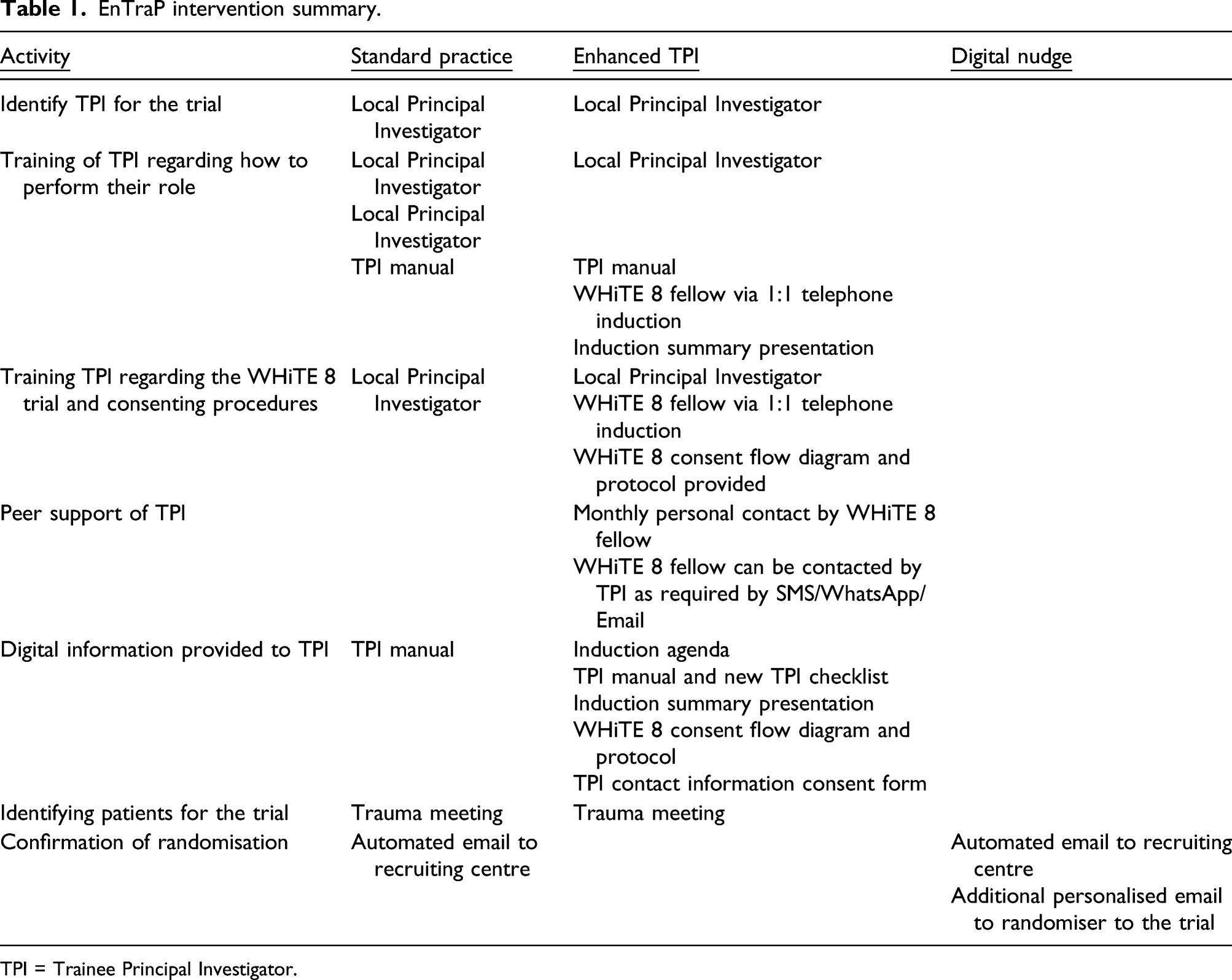
TPI = Trainee Principal Investigator.
Standard practice for TPIs
Trainee Principal Investigators were not mandated but were recommended by the trial management team at site initiation visits to the participating site. A TPI manual was made available with specific information regarding the role but no further involvement thereafter.
Standard practice following successful randomisation of a participant
An automated email was generated to local research teams after each successful patient randomisation via an online randomisation portal. There were monthly email updates to local research teams regarding trial processes and progress. The usual incentive to randomisers is acknowledgement as a collaborator in the WHiTE 8 COPAL trial publication and trainee orthopaedic surgeons, in addition, receive evidence of randomisation through certification.
Enhanced TPI package
This was a complex intervention involving education, support and supplementary information. A 1:1 telephone training session by the WHiTE 8 surgical research fellow (CI for the SWAT) was conducted along with monthly communication regarding progress and problems via direct messaging/email and/or phone calls if required. A comprehensive package of supplementary information was also provided via email prior to commencing their role.
Digital nudging
A personalised email nudge expressing a combination of appreciation and encouragement from the WHiTE 8 COPAL Research Fellow was sent each time a healthcare professional randomised a participant to the trial. These were to be sent to the randomiser within 72 h; where a clinician recruited multiple patients in the period, only one nudge was sent referring to the number recruited in the period.
Outcome assessment
The primary outcome measure was the total number of patients randomised, from each site, in their first 6 months of recruitment to the WHiTE 8 COPAL trial. These data are collected by the trial management team on a monthly basis.
Site setup details including activation date, date of first patient recruited and dates of implementation of each SWAT intervention were recorded on an Excel spreadsheet. This allowed for calculation of time taken to implement each intervention from centres commencing recruitment. Conversion rate from screened population was collected monthly from the main trial database.
The trainee perspective of their role was collected through a TPI Qualtrics survey at the end of the SWAT trial period sent via email. Responses were based on a 5-point Likert scale ranging from ‘not very satisfied’ to ‘extremely satisfied’. The research fellow maintained a time log for delivering the TPI education intervention and a log of communication for peer support during the period of the SWAT.
Randomisation
Baseline data for sites involved in EnTraP.
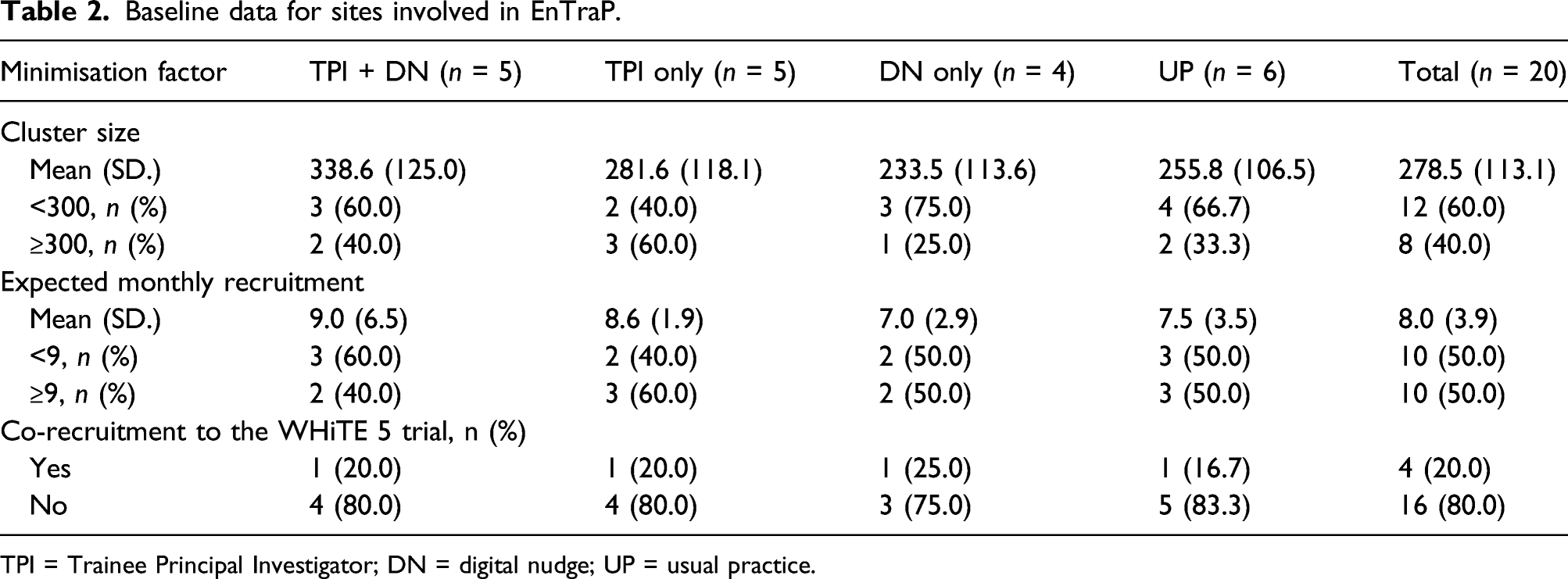
TPI = Trainee Principal Investigator; DN = digital nudge; UP = usual practice.
Sample size
As in many SWATs, a power calculation was not undertaken as the number of participating sites was fixed and driven by the needs of the host trial. The first 20 WHiTE centres recruiting to the WHiTE 8 COPAL site were included in the SWAT. Further sites were not included due to time constraints (i.e. SWAT CI [lead author NRA] was returning to clinical practice and could no longer manage the SWAT).
Statistics
Analysis was conducted in STATA v15 on an intention-to-treat basis. Baseline data relating to the sites (including the minimisation factors) are summarised for the four groups as randomised. No formal statistical comparison of baseline data was undertaken.
The number of participants recruited per site was summarised. A Poisson regression model, containing the two interventions (Enhanced TPI and Digital Nudge) and the three minimisation factors (cluster size and expected number recruited per month were included in their continuous form) was undertaken. Adjusted incidence rate ratios (IRRs) and associated 95% confidence intervals (CIs) and p-values were obtained from this model. We undertook an interaction test between the two interventions.
Feasibility outcomes including time to commence intervention, time required to run the education intervention and communication time and methods used for the peer support aspect of the intervention were reported descriptively.
Results
Baseline
The first 20 sites recruiting to the WHiTE 8 COPAL trial opened between 16 August 2018 and 21 February 2019 and were randomised into the SWAT between 22 August 2018 and 20 March 2019, an average of 14.9 days (SD 17.0) after site activation. Six sites were randomised to usual practice, four to digital nudge only, and five each to TPI only and TPI plus digital nudge. The overall expected mean recruitment rate per site was 8.0 patients per month (SD 3.9) (Table 2). Mean cluster size was 278.5 (SD 113.1) and four sites were co-enrolled into the WHiTE 5 trial.
Primary outcome
Site recruitment as stratified by group randomisation.
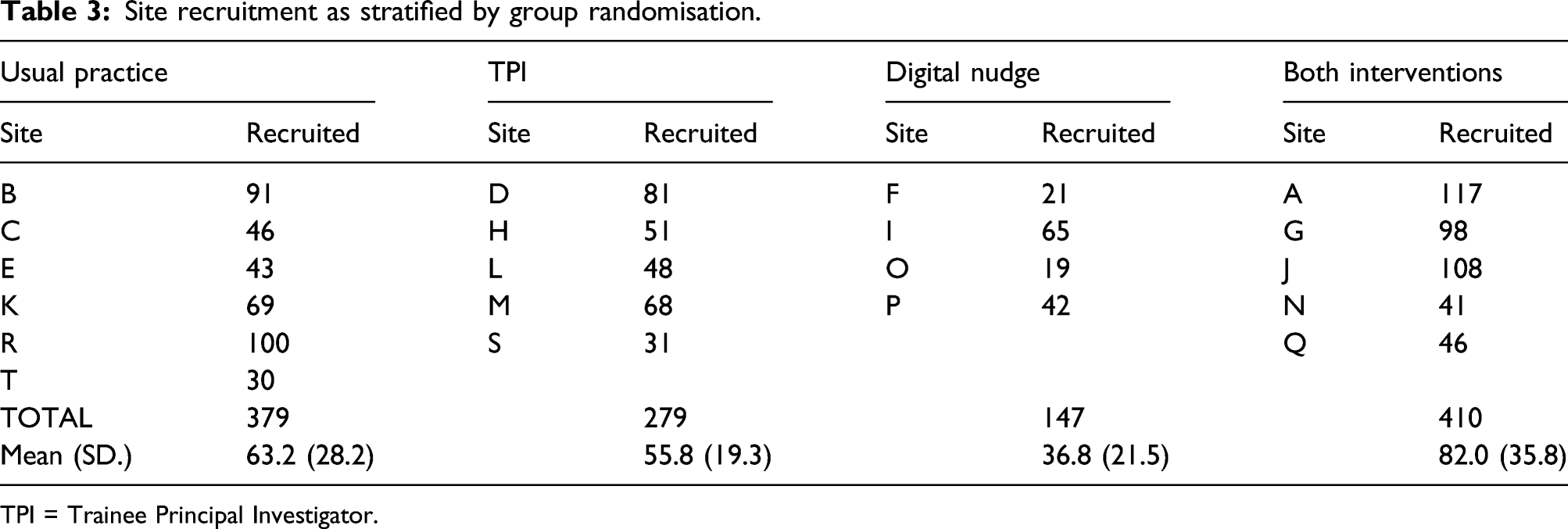
TPI = Trainee Principal Investigator.
From the primary Poisson regression model (no interaction term), the main effect of enhanced TPI intervention was a statistically significant benefit on recruitment (IRR 1.15 95% CI 1.02 to 1.29, p = 0.02).
The digital nudge intervention had no significant impact on recruitment (IRR 0.95 95% CI 0.85 to 1.07, p = 0.39).
In the Poisson model including an interaction between the two interventions, the main effect of the enhanced TPI intervention was IRR 1.23 (95% CI 1.09 to 1.40, p = 0.001) and of the digital nudge intervention was IRR 0.89 (95% CI 0.79 to 1.01, p = 0.07). There was a statistically significant interaction (IRR 2.09 95% CI 1.64 to 2.68, p < 0.001). There is a qualitative interaction in that the addition of the digital nudge is beneficial in the enhanced TPI sites (IRR 1.29, 95% CI 1.11 to 1.51, p = 0.001) but detrimental in the standard TPI sites (IRR 0.62, 95% CI 0.51 to 0.75, p < 0.001).
Secondary outcomes
From the 557 patients recruited at sites allocated to receive the digital nudge intervention, 353 nudges were created for the recruiters. Median time to first nudge from first randomisation at the site was one day (range 0–3). 224 (63.5%) of the nudges were for single randomisations, while 129 (36.5%) were for multiple randomisations conducted over a 72 h period (relating to 333 randomisations and mean 2.6 per nudge). Seven of the 353 nudges created (2.0%) were unable to be sent due to the lack of an email address despite two follow-up emails to local research teams. The average time to construct a nudge, log the activity and then disseminate was 12 min. 53 nudges (15.0%) were sent 72 h after randomisation. Of these late nudges, reasons for protocol deviations include CI on annual leave (n = 25, 47.2%); CI clinical commitments (n = 17, 32.1%); delay from local centres in retrieving email addresses (n = 7, 13.2%) and unknown (n = 4, 7.5%).
Incidence rate ratio between the intervention and participant recruitment to WHiTE 8 COPAL trial.
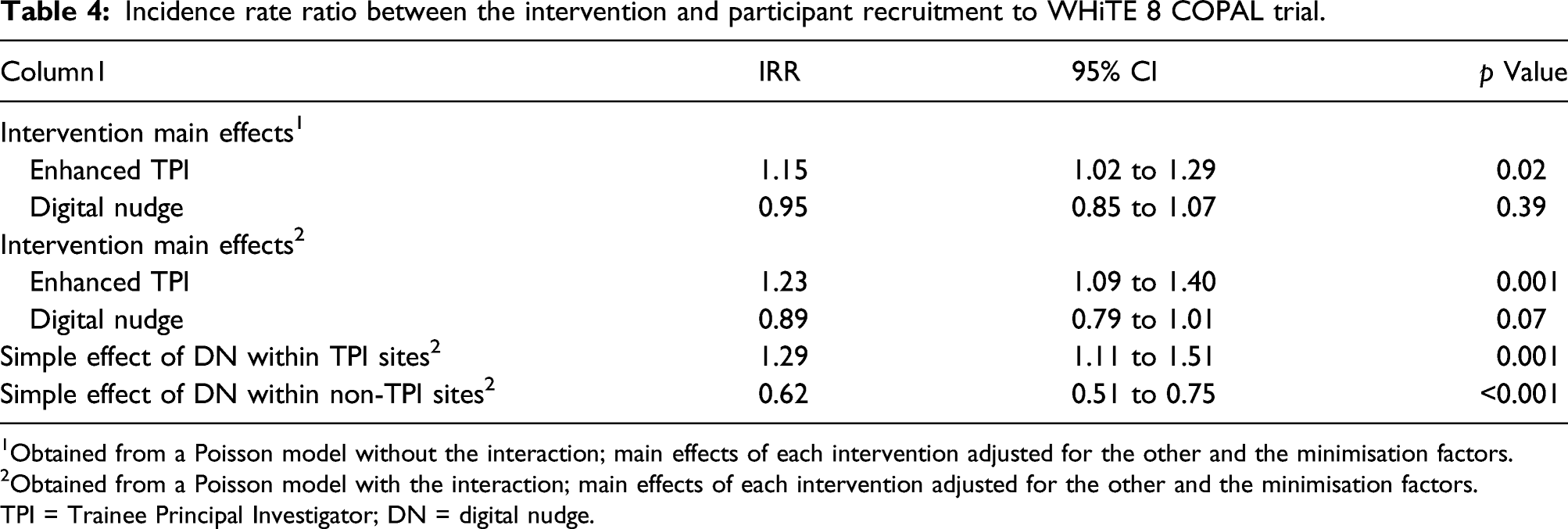
1Obtained from a Poisson model without the interaction; main effects of each intervention adjusted for the other and the minimisation factors.
2Obtained from a Poisson model with the interaction; main effects of each intervention adjusted for the other and the minimisation factors.
TPI = Trainee Principal Investigator; DN = digital nudge.
Enhanced TPI follow log of follow-up.
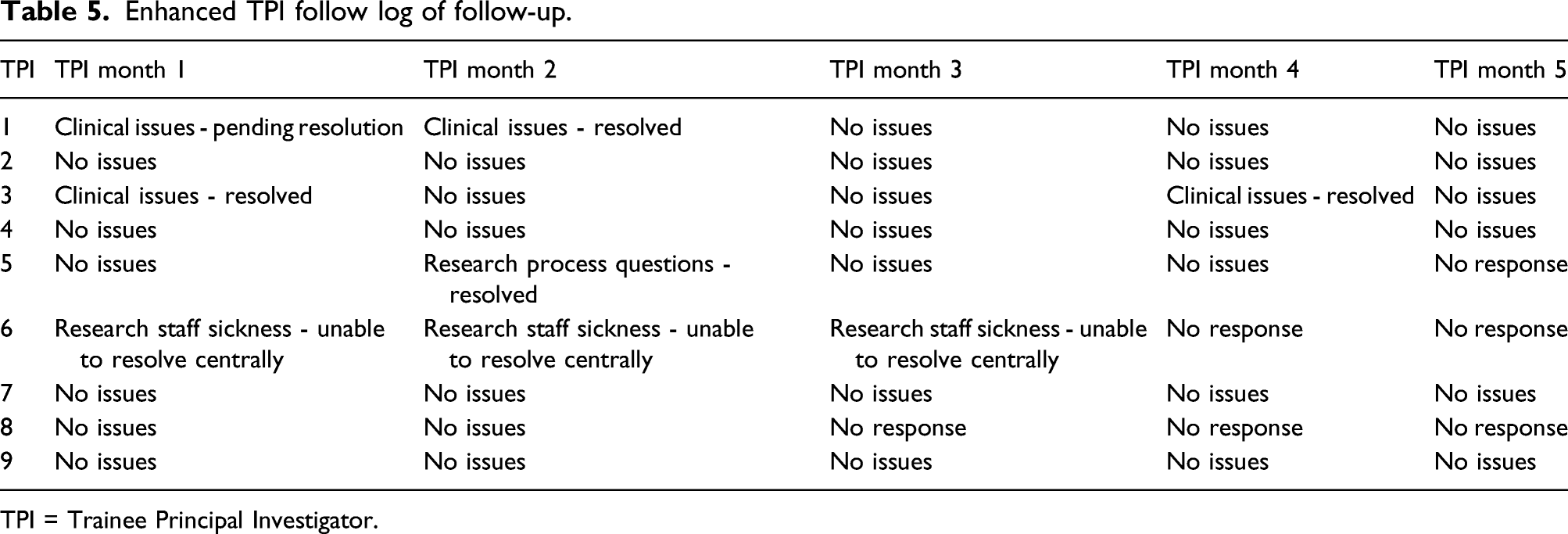
TPI = Trainee Principal Investigator.
Among the 20 centres running the WHiTE 8 trial during the intervention period, there were 17 TPIs identified from delegation logs. The response rate for the follow-up survey was 52.9%. Nine TPIs completed follow-up surveys of which seven had received the enhanced TPI intervention (77.7%). All TPIs were very satisfied or extremely satisfied with their inductions in explaining the purpose, consent, role of TPI and benefits of becoming a TPI. Amongst the enhanced TPI group, 100% of the responders were extremely satisfied with the induction process and felt ‘extremely supported’ with regards to monthly follow-up. Suggested improvements were generic UK TPI education workshops and e-learning modules to help reinforce and discuss key issues that arise across UK Orthopaedic trials.
The proportion of patients recruited as a percentage of the screened population were not analysed due to inadequate data retrieval from base site screening logs.
Discussion
This 2x2 factorial SWAT is the first randomised trial to investigate the effects of an enhanced TPI support package with or without the addition of a personalised digital nudge on recruitment rates; it was embedded within a large orthopaedic RCT. The combined use of enhanced TPI and digital nudging showed significant interaction (IRR 2.09 95% CI 1.64 to 2.68, p < 0.001) in this trial.
In both Poisson models (without and with intervention interaction), sites that received an enhanced TPI training and support package had a significantly increased rate of patient recruitment to the WHiTE 8 COPAL trial over 6 months (IRR 1.15, 95% 1.02 to 1.29, p=0.02 and IRR 1.23 (95% CI 1.09 to 1.40, p=0.001, respectively). There was excellent engagement with all aspects of the intervention by TPIs; 90.0% participated in the induction activity and 86.7% in the monthly follow-up communication indicating that participants were engaged in WHiTE 8 COPAL trial recruitment for the entire 6-month duration of the SWAT. The use of increased trial centre coordination through on-site visits has been shown not to impact patient recruitment 22 ; however, we have shown that we can deliver a similar educational package more conveniently to both trainer and trainee via off-site methods.
The follow-up questionnaire also highlighted no suggested areas for improvements in how the intervention was conducted. The monthly follow-up revealed five time points at which CI involvement was needed to address clinical and research issues. Although this represents only 11.1% of the follow-up points, all trainees felt ‘extremely supported’ and this may have contributed to the increased recruitment at these sites.
There was no significant difference in 6-month total recruitment at sites allocated to the digital nudge intervention in both Poisson models without or with the intervention interaction (IRR 0.95, 95% 0.85 to 1.07, p = 0.39 and IRR 0.89 (95% CI 0.79 to 1.01, p = 0.07, respectively). In terms of feasibility, the intervention had a median lag set up time of one day from first patient recruitment and delivery of the nudge averaged 12 min from construction to dissemination including logging the activity. One other study investigated an additional communication strategy directly to clinical sites compared to usual practice of little communication from central trial co-ordinators and found no difference in the recruitment rates, consistent with our results. 23
There was a qualitative interaction between the two interventions where the addition of the digital nudge was beneficial in the enhanced TPI sites (IRR 1.29, 95% CI 1.11 to 1.51, p = 0.001) but detrimental in the non-TPI sites (IRR 0.62, 95% CI 0.51 to 0.75, p < 0.001). This may be due to the combined promotion of the host trial at centres by having two interventions directed towards the same recruiting population.
The contact information available from delegation logs and local research nurse input was robust enough to ensure that 98% of nudges were disseminated to the respective trial recruiter. The protocol deviations for nudging beyond 72 h were relatively high at 15% but 79% (42/53 cases) were due to unavailability of the SWAT CI who delivered the intervention. Therefore, these deviations are potentially avoidable in future trials if the SWAT is conducted by the CTU team managing the host trial with multiple personnel.
The number of sites available to be randomised to interventions in this factorial trial was fixed and thus a low sample size was a limitation. However, 95% of sites randomised were able to run the interventions investigated, thus minimising any imprecision in the intention-to-treat analysis. The minimisation factors of cluster size and predicted recruitment could arguably closely correlate, thus being considered a single minimisation variable, that is, those centres with higher incidences of hip fractures per year may be better recruiters to this clinical trial due to increased opportunity for recruitment. However, from our experience from previous trials within the WHiTE cohort framework, there is no obvious relationship between the size of the recruitment population and performance of the recruiting site and thus having this variable as independent variables ensured balanced randomisation groups.
A weakness of this trial is that there was only one person involved in training and supporting the TPIs and they were also a surgical trainee; thus, there is a generalisation issue that others may not be sufficiently motivated or skilled to deliver the training and support or that TPIs may not respond as well to the interventions not being delivered by a peer. Consequently, this intervention ought to be replicated in further trials using different personnel to deliver the training and support.
These results are widely generalisable to UK multicentre surgical trials as the methodology of centralised randomisation and the use of TPIs is becoming the standard operating procedure. This should make the implementation of an Enhanced TPI support package and digital nudging relatively straightforward when designing RCTs. These interventions should be evaluated in further trials to achieve a greater sample size for meta-analysis. The costs associated with this intervention have not been formally investigated and a formal cost benefit evaluation would also be a valuable addition. A potential improvement would be assessing further time points to determine the length of time each of these interventions may have an effect for before recruitment fatigue.
Conclusion
An education and support programme targeted at surgical TPIs involving a digital education package, 1:1 telephone induction and subsequent support package was effective in increasing recruitment in the first 6 months of trial commencement. There was no evidence for the effectiveness of the digital nudge intervention in isolation; however, our results show that, when combined with an education programme, it leads to enhanced effectiveness of that programme.
Footnotes
Acknowledgement
Heraeus medical Gmbh
Individuals involved in the management of the WHiTE 8 trial at the University of Oxford: Miss Svetlana Milca, Ms Stephanie Wallis, Mr Rhys Painton, Mrs Amrita Athwal, Dr Juul Achten and Professor Matthew Costa.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Research ethics
Wales Research Ethics Committee five approved the WHITE eight COPAL trial on 3rd May 2018 (18/WA/0154) with subsequent approval for the SWAT via Amendment 07 Non-Substantial
Ethical approval
The University of York Health Sciences Ethics Committee has approved this study within a trial. Ethics Approval ID: HSRGC/2018/266/C
