Abstract
Objective
To describe the roles that pairwise randomisation has to prevent selection bias and allow a common outcome measurement point when timing of treatment is uncertain.
Results
When recruitment of participants allows two or more randomisations to occur simultaneously, then pairwise randomisation allows site stratification but prevents site predictability. For evaluations of treatments where timing of treatment is uncertain (e.g. elective surgery), pairwise randomisation allows a common post-treatment evaluation time point. In waiting list designs where length of treatment varies by patient, pairwise randomisation allows differential follow-up across pairs.
Conclusion
Since pairwise randomisation was described in 2004, to allow unpredictable site stratification, it has been rarely used. However, it can address other methodological issues and should be considered more widely.
Background
The randomised controlled trial (RCT) is, in principle, the most robust method for assessing effectiveness. 1 There are numerous methods of allocating participants into treatment groups in an RCT all with advantages and disadvantages. Simple randomisation (akin to tossing a coin or rolling a die) has some perceived disadvantages including, for small sample sizes where the method can result in significant numerical imbalances between groups. Whilst numerical imbalance is of little consequence for large trials (n > 100), important subgroups within a large trial might still be imbalanced, which may reduce the efficiency of the trial analysis. Furthermore, there is a risk of a large string of the same allocation by chance. Consequently, if the trial is recruiting slowly, there may be chance chronological imbalances if there is a large string of allocations, over time, favouring one group. Therefore, most clinical trials use some form of restricted allocation, such as stratified block randomisation, to reduce the risk of numerical, temporal and covariate imbalances. 2 Nevertheless, there are disadvantages to virtually any form of restricted randomisation. The main problem of restricted randomisation, especially using blocking, the most popular allocation method, is predictability, which increases the risk of allocation subversion. 2
In this paper, we will describe a rarely used randomisation method that solves a number of problems with ‘standard’ randomisation methods.
Pairwise randomisation
Pairwise randomisation was first described by Daniels et al. 3 It has been used, occasionally, in a clinical trial. 4 The idea is relatively simple. Instead of randomising individuals one at a time, the researcher or clinician recruits groups of patients, and once at least two (for a 1:1 randomisation ratio for a two armed trial) are consented, they are entered into the randomisation programme to be randomised to a treatment group. The programme automatically allocates one of the pair to the intervention group and the other to the control. For multi-armed trials of three groups or more, more participants need to be recruited before randomisation can take place. The original rationale for the development of this method was to reduce the risk of prediction of future allocations when randomisation is stratified by the site or centre recruiting the participant. Site or centre stratification is very common in multi-site RCTs. There are several reasons for why site stratification is attractive. Site characteristics, such as surgical expertise, might influence outcomes, so it makes sense to ensure that randomisation is balanced across sites. Site stratification may also be useful for logistical reasons if therapies need to be scheduled (e.g. number of surgical slots) in advance. The downside is that site stratification with blocking can lead to highly predictable randomisation, which means that future allocations can be subverted and the integrity of the trial undermined. 5 There is a significant amount of evidence that subversion of randomisation does occur with case studies6,7 and statistical evidence of the problem. 8 Daniels et al. described pairwise randomisation as a solution to this problem, as subversion is not possible because both participants need to be entered into the programme before allocation takes place. Thus, pairwise randomisation leads to site stratification without the predictability of conventional stratified block randomisation. Whilst pairwise randomisation was described to deal with the problem of site-specific subversion, we feel that it has other potential advantages.
Common timing of outcome assessment
Usually in RCTs, we follow up all participants for a fixed period of time after the date of randomisation. However, there may be instances where we might want to vary the length of follow-up after randomisation depending on when the patient is treated. For example, let us consider an RCT of elective surgery versus conservative care where we might be interested in knowing the outcome of surgical patients within a fixed time after surgery. Unless surgery (in the intervention arm) is performed at a set time after randomisation, this cannot be achieved by following up all participants at a fixed point after randomisation. For instance, if we wanted to compare the outcomes of patients 12 weeks after surgery with patients who had received non-surgical care and we knew that surgery would be performed within seven days of randomisation, then we could measure outcomes in both groups at 13 weeks post-randomisation and be confident that most patients would be assessed close to 12 weeks after the surgical patients received their surgery. However, if there is variation in waiting times between and even within centres, which often happens, then a fixed time for all patients after randomisation will mean that a proportion of patients’ surgery will have been more than one week after randomisation so that the gap between surgery and follow-up would be less than 12 weeks. This might, then, underestimate the value of a surgical intervention, at 12 weeks, compared with conservative treatment. Therefore, ideally, we would like to follow-up the surgical participant and a paired control participant 12 weeks after the surgical procedure. Whilst it is simple enough to ‘start the clock’ to follow-up surgical patients 12 weeks after surgery, the problem arises as to which control should be followed up to match the surgical patient. Unless this is done in advance, before randomisation, we cannot exclude the possibility that selection bias is introduced by non-random selection of a control participant. However, using pairwise randomisation allows us to identify before the point of randomisation, unlike other randomisation methods, a control participant that can be followed up at the same time point as the intervention patient and this avoids the potential of biased selection. In Figure 1, we show how this may occur. We have six patients that are randomised to surgery or conservative care using pairwise randomisation. After randomisation, the three surgical patients are eventually given surgery six, eight and nine weeks post randomisation. If we were to offer a fixed time assessment of outcome after randomisation, such as 12 weeks, then the outcomes would be assessed at six, four and three weeks after the operation. However, because of pairwise randomisation, we can offer a fixed 12-week assessment post-surgery with variable follow-up post-randomisation (Figure 1). Therefore, any delays in surgery would not lead to an underestimate of the effects of surgery on quality of life. For this use, we do not actually need to undertake site stratification, we can take pairs across the trial, and this may be particularly useful if site recruitment is not sufficiently swift to allow a centre pair to be formed within an appropriate time.
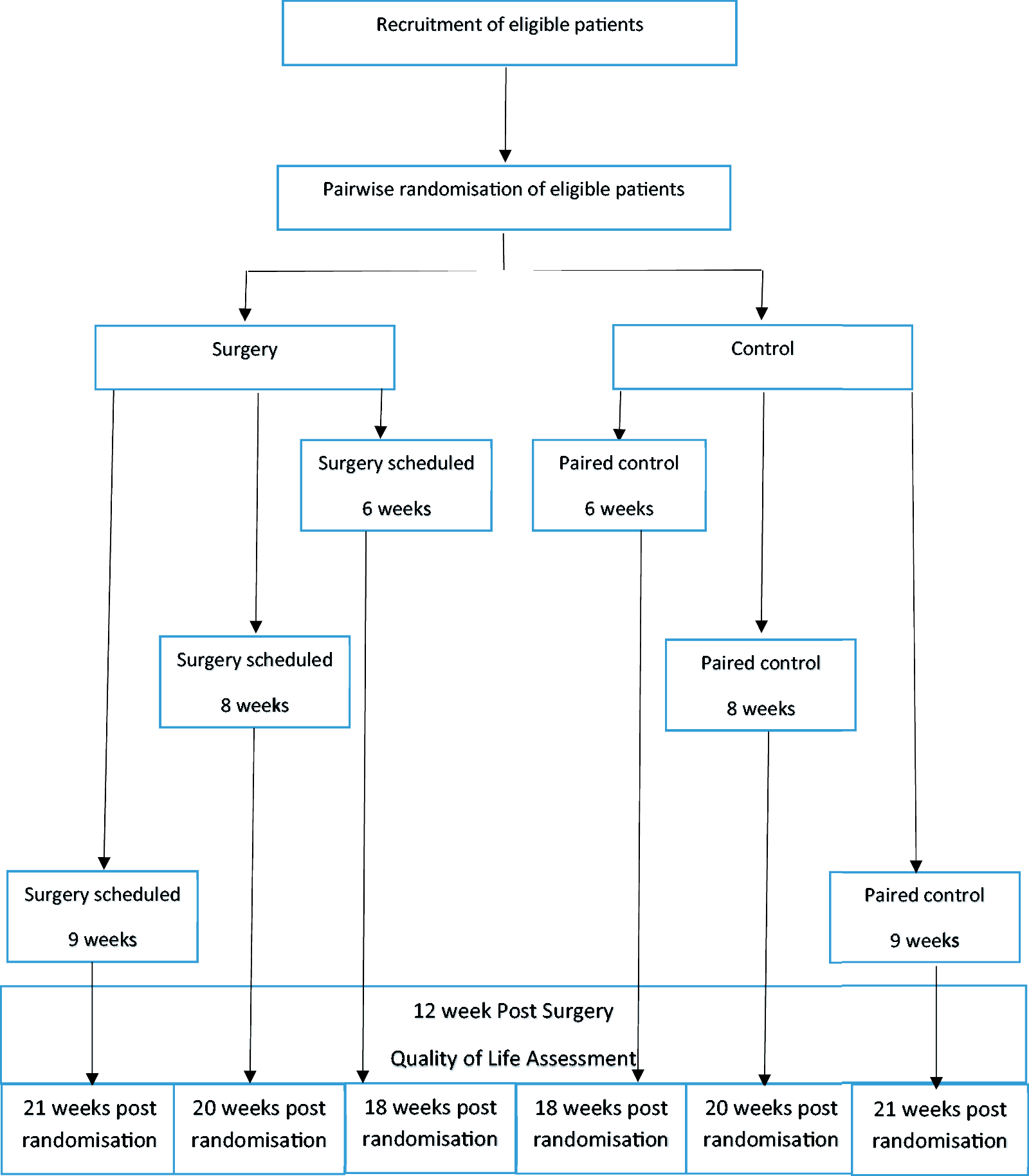
Study design of a hypothetical trial of a pre-surgical exercise intervention.
Waiting list design
Pairwise randomisation could also be used in the case of a ‘waiting list design’ where the intervention is offered to the control participants after intervention participants have received the intervention and all participants have been followed up. For example, a trial by Ronaldson et al. used a waiting list design to assess whether having a lung health check among smokers led to a reduction in self-reported smoking. 9 In this trial, the waiting list varied from two to six months. Because the trial recruited participants from general practices (GP), which governed the waiting list length, the GP was included in the statistical model to account for variation in follow-up time. However, such an approach may not be possible if follow-up times are not dependent on cluster membership or there is variation of follow-up times within a cluster. In these instances, pairwise randomisation can deal with this potential problem by design. This would allow us to have a variable waiting list time, which we could exploit in an analysis to assess the effect of different follow-up periods on outcome.
Note that using pairwise randomisation is not the same as a ‘matched’ pairs design where pairs of participants are matched on some baseline characteristic (e.g. age, gender) and then are randomised. In pairwise randomisation, we do not match the patients in terms of their characteristics except that they are both available at a similar time for the intervention.
Analysis
The use of pairwise randomisation does not lead to the same statistical limitations as a ‘matched’ pairs design. Pairwise randomisation is more analogous to blocking but without the same prediction concerns. Both participants are equally likely to be randomly allocated to the trial arms (i.e. both participants have 50% chance of being selected to be randomly allocated to the intervention group in a two-arm trial); consequently, foreknowledge of treatment allocations is prevented. If simple randomisation is used to allocate the randomly chosen individual of the pair to treatment groups, then assuming time homogeneity, there are no additional impacts on the analysis. If there are time trends or differences amongst strata, then the intrablock (or intrapair) correlation is expected to be positive, and the analysis should account for the blocking. 10 In addition, if stratification or minimisation is used to allocate the randomly chosen individual of the pair, then the analysis should reflect the additional restriction on the randomisation.11,12 Indeed, pairwise randomisation can be combined with other allocation methods such as minimisation 4 – although this could introduce allocation predictability.
Discussion
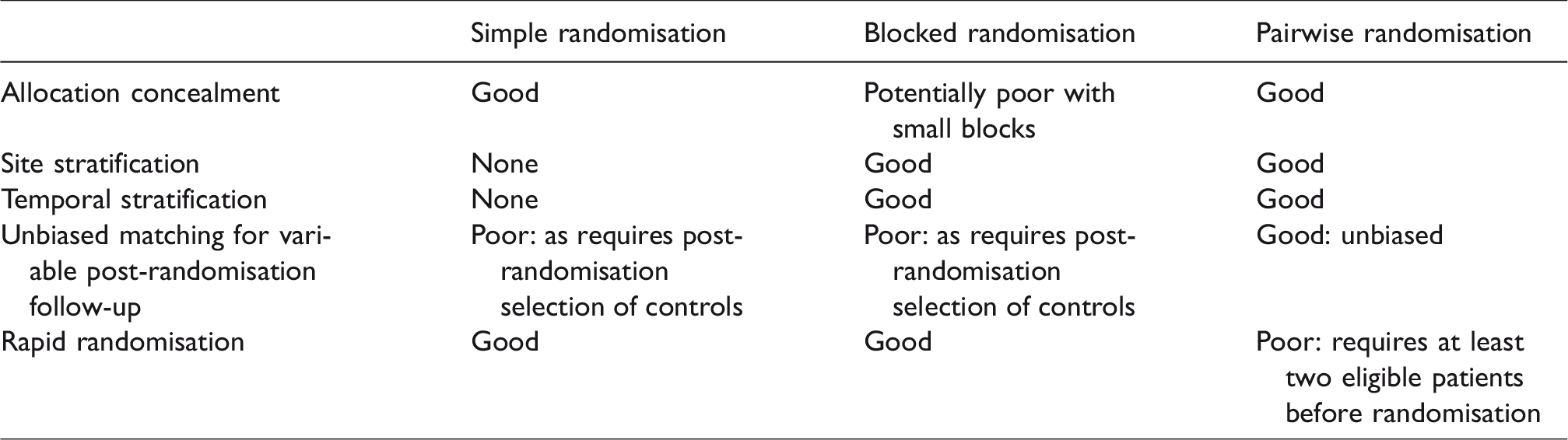
We have described the situations where pairwise randomisation might be usefully applied. In Table 1, we summarise some of the advantages and disadvantages of pairwise versus blocked and simple randomisation. Although pairwise randomisation was described nearly 15 years ago, it has rarely been used in practice. It does have some drawbacks, which may partly explain this: the main one being there needs to be sufficient recruitment to ensure that a minimum of two participants can be assembled in a short space of time if treatment is relatively urgent. Where treatment timing, such as elective surgery, is less urgent, then pairwise randomisation might be a solution to either maintaining centre stratification without risk of subversion bias and/or ensuring a common post treatment analysis. Pairwise allocation does not allow predictability, as both participants are entered into a computer program before randomisation and only when they are both present will allocation occur. We believe, therefore, pairwise randomisation represents a useful randomisation option for trials where recruitment allows multiple participants to be randomised simultaneously, when site stratification is required, and where there may be a variable post-randomisation follow-up point.
Key elements of pairwise randomisation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
