Abstract
Aim/objective:
Mucosal neuroma (MN) is a benign peripheral nerve tumour that usually occurs as a symptom of multiple endocrine neoplasia type 2B (MEN2B) (MEN2B-associated MN). MN that is not associated with MEN2B is termed solitary mucosal neuroma (SMN). We present a case of SMN in the upper gingiva and examine previously reported cases.
Methods:
The study included nine patients (eight from seven articles and one from this study) with SMN. Age of onset, sex, disease duration, tumour site, clinical findings including subjective symptoms, diagnostic method, treatment, prognosis and relationship with MEN2B were investigated in these nine patients.
Results and conclusion:
SMN is thought to be caused by the overproduction of nerve growth factors due to trauma or chronic continuous stimulation, whereas MEN2B-associated MN is caused by GNDF-induced hyperplasia. Considering the pathogenesis of their development, both diseases exhibit hyperplasia of the nerve fibre bundles and have the same histological type of hypertrophy. MNs (SMN and MEN2B-induced MN) must be differentiated from benign peripheral neurogenic tumours. Morphological recognition and immunohistopathological analyses make this differentiation possible.
Keywords
Introduction
Mucosal neuroma (MN) is a benign peripheral nerve tumour histopathologically characterised by irregular tortuous nerve bundles in the endoneurium, scattered Schwann cells in the subepithelial layer, and marked thickening of the perineurium. MN usually occurs as a symptom of multiple endocrine neoplasia type 2B (MEN2B) 1 and refers to MEN2B-associated benign peripheral nerve tumours (MEN2B-associated MN). MN occurring independently and not associated with MEN2B is termed a solitary mucosal neuroma (SMN). Only a few cases of SMN have been reported, and no case series have been documented. In this study, we experienced an SMN in the upper gingiva and conducted a case series study of previously reported cases.
Patients and Methods
The study included nine patients (eight from seven articles2 -8 and one from this study) with SMN. The review period was between 1985 and 2021, and searches were conducted using the keyword “mucosal neuroma” in PubMed and the Japan Medical Abstract Society (JMAS) database. Age of onset, sex, disease duration, site, clinical findings including subjective symptoms, diagnostic method, treatment, prognosis and relationship with MEN2B were investigated in these nine patients.
Results
The backgrounds of the nine patients (four men and five women) with SMN are shown in Table 1. The disease duration ranged from 14 to 73 years. In seven patients, SMN developed in the head, neck and facial regions (eyelid conjunctiva, tongue, lower lip, maxillary gingiva, hard palate and larynx). In all patients, the tumour surface was covered with normal mucosa and was polyp-like or nodular. Subjective symptoms were painless nodules or masses in three patients (patients 1, 4 and 8) and pain in four (patients 2, 3, 7 and 9). Subjective pain was not mentioned in the reports of patients 5 and 6. Five patients (patients 1, 2, 4, 5 and 6) were only observed after diagnosis by biopsy, and four patients were treated with surgical extirpation, including excisional biopsy (patients 3, 7, 8 and 9). Patients 3 and 7 were observed after biopsy but were moved to resection due to tumour growth and pain. None of the patients who underwent surgery had recurrence, including those in our study (patient 9, 11-year follow-up after extirpation). Five of the nine patients were diagnosed based on morphological and immunohistochemical findings. Histopathologically, all patients showed nodular lesions without a capsule. The lesions consisted of a hypertrophic perineurium of different sizes and shapes in the subepithelial layer and proliferation of normal-form nerve fibres with many axons and Schwann cell proliferation. Immunostaining confirmed S-100 expression in Schwann cells and epithelial membrane antigen (EMA) expression in the perineurium in patients 2, 3, 7, 8 and 9. Other antibodies distinguished SMNs from peripheral neurogenic tumours in patients 3 and 7. In four cases, a search was performed for RET gene mutations (patients 4, 5 and 6) or RET protein expression by immunostaining (patient 9) to identify MEN2B-associated MN.
Patients.

Case Presentation (Patient 9)
A 37-year-old woman presented to the Department of Oral and Maxillofacial Surgery at Gunma University Hospital in February 2012. A well-demarcated 20 × 15-mm bone-like hard tumour with normal mucosa was observed on the palatal gingiva of the right maxillary second molar (Figure 1). Family and medical histories did not include any notable items. Computed tomography (CT) showed no abnormal bone resorption in the tumour area. The clinical diagnosis was a benign upper gingival tumour.
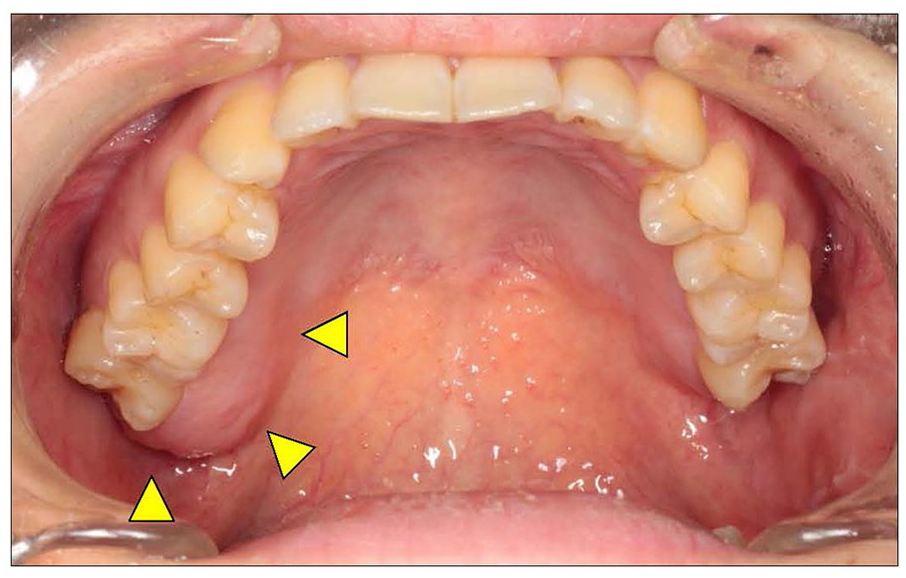
Intraoral findings.
A benign peripheral neurogenic tumour was histopathologically diagnosed after biopsy. The tumour was extirpated subperiosteally under general anaesthesia on December 21, 2012, with a horizontal safety margin of 3 mm due to the absence of a capsule. The slightly rough bone surface was scraped off. The wound was covered with a polyglycolic acid sheet and fibrin glue.
Haematoxylin and eosin-stained specimens of the removed tumour showed enlarged nerve bundles running irregularly without a capsule in the subepithelial tissue. The nuclei had no fenestrations (Figure 2a and b). Immunostaining revealed that the tumour cells were S-100 positive (Figure 3a-A), suggesting the presence of Schwann cells. EMA was expressed in the perineurium (Figure 3a-B). Neurofilament (NF)-positive fibres were considered axons (Figure 3b). The rate of MIB-1 positivity was <1% (Figure 3c). Based on these pathological findings, the patient was diagnosed with MN.

Histopathological findings.
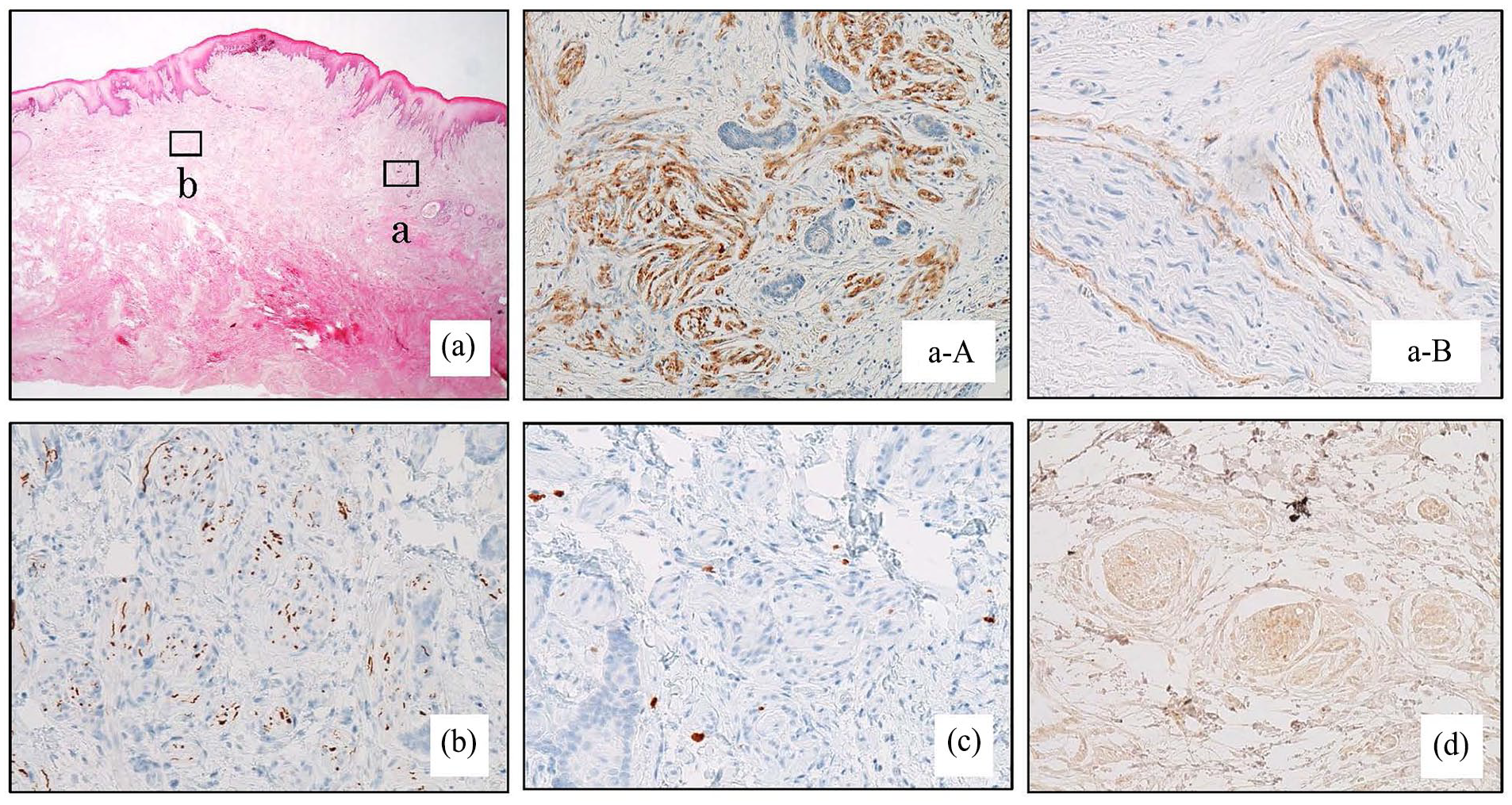
Immunohistochemical findings.
The patient was referred to the Department of Endocrinology and Metabolism to investigate the possibility of MEN2B. Medullary thyroid cancer was excluded because no abnormal thyroid findings were observed on cervical ultrasound, and markers for medullary thyroid cancer (carcinoembryonic antigen and calcitonin) were within reference limits. Abdominal computed tomography (CT) revealed no adrenal or other tumours or gastrointestinal neuromas and no excess catecholamine secretion, ruling out pheochromocytoma. RET is usually expressed in MEN2B-associated MN because of a point mutation caused by the translocation of the RET gene. However, the patient was diagnosed with negative results (Figure 3d). Based on these findings, MEN2B was excluded, and the gingival lesion was identified as an SMN. No recurrence has been observed, and the patient has been doing well for 12 years after surgery.
Discussion
MN commonly presents as a symptom accompanying MEN2B. We encountered an extremely rare case of SMN and performed a case series study based on our case and previously reported cases.
MEN, an inherited autosomal dominant disease characterised by tumours in multiple endocrine organs, is classified into types 1, 2A and 2B, and oral MN is a symptom of MEN2B (MEN2B-associated MN).1,9 RET is a tyrosine kinase receptor that binds to the extracellular signals of glial cell line-derived neurotrophic factor (GDNF). In humans, it is encoded by RET gene. 10 MEN2B occurs due to gain-of-function mutations in the RET gene. 11 GNDF acts as a nutritional factor in various neurones. 12 The GNDF-bound GNDF family receptor α−1 interacts with RET, activating RET. Overactivity of RET due to gain-of-function mutations in the RET gene may also lead to overproduction of GNDF, which may be associated with the formation of MEN2-associated MN. In contrast, SMN is caused by the overproduction of nerve growth factors due to trauma or chronic continuous stimulation, and its developmental mechanism and pathophysiology differ from those of MEN2B-associated MN. However, considering the pathophysiology of development, SMN and MEN2B-associated MN are both formed as hyperplastic nerve fibre bundles and have the same histopathological features as dose hypertrophy.
As some authors argue that MNs should be considered hamartoma, ganglioma, or ganglioneuromatosis rather than neuroma, 9 we considered the similarities and differentiations between traumatic neuroma (TN)/amputation neuroma (AN) and MNs. TN/AN is a reactive lesion with non-neoplastic growth, and immunohistological findings are similar to MNs in that they are normal-form nerve bundles. In other words, S-100 (Schwann cells), EMA (perineurium cells) and NF (nerve fibres: axons) are positive (Table 2). The histological morphology in TN/AN also shows irregular proliferation of axons, Schwann cells, perineurium, fibrous connective tissue and especially mature normal-form nerve bundles (nerve fibre bundles), forming numerous nodules and myxomatous changes in the connective tissue of the stroma. 1 MNs have less Schwann cell proliferation than TN/AN, and the main pathological features are perineurial thickening and bundle tortuosity. These are the differentiating features between MNs and TN/AN. There is evidence of the aetiology of palisading encapsulated neuroma (PEN) that supports the role of trauma and is similar to MN and TN/AN in histopathological morphology and immunohistological findings. However, nuclear palisading and an EMA-positive capsule usually distinguish PEN from MN and TN/AN. Some PENs lack a nuclear palisading or show an unclear capsule and have recently been termed solitary circumscribed schwannomas. It should be noted that differentiation may be difficult in such cases.13 -15
Immuohistochemical features of benign peripheral neurogenic tumours.

Abbreviations: S-100, S-100 protein; NF, neurofilament; EMA, epithelial membrane antigen.
Mucosal neuroma has lower expression of S-100-positive Schwann cell than traumatic neuroma/amputation neuroma.
Palisaded encapsulated neuroma has high expression of S-100-positive Schwann cell than traumatic neuroma/amputation neuroma.
Expression in the perineurial cell.
Expression in the capsule.
Tumours with axons are NF-positive.
Usually negative. S-100-positive Schwann cells may be present in the centre of pseudo-onion bulbar structures, but in small numbers.
Usually negative. NF-positive neurofilament axons may be present in the centre of pseudo-onion bulbar structures, but in small numbers.
Besides TN/AN, MNs must be differentiated histopathologically from benign peripheral neurogenic tumours, such as schwannoma, neurofibroma and intraneural perineurium (IP) 1 (Table 2). Fundamentally, MNs lack a capsule, whereas schwannomas are encapsulated, and nuclear palisading is absent. Neurofibromas are similar to MNs in these features. Plexiform neurofibroma (PN), an atypical form of neurofibroma with no capsule, in which proliferation occurs within the nerve sheath and that has irregular tortuous nerve bundles, is often reported to have a histopathological morphology similar to that of MNs. 1 This tumour shows myxomatous metaplasia in the connective tissue of the stroma, and the tumour cells are S-100-positive because they are derived from Schwann and mesenchymal cells in the nerve sheath. The tumours with axons are NF-positive. However, perineurial cells are EMA-negative. These findings and those of fibrotic changes between the nerve fibres can be used to differentiate it from MNs. IP is derived from perineuronal cells and shows thickening of the perineurium, similar to MNs. This tumour is positive for S-100, EMA and NF; however, S-100 and NF are expressed less than in other peripheral nerve tumours, including MNs. A pseudo-onion bulb-like structure is often observed around the axon, allowing a morphological differential diagnosis from MNs.
Conclusions
SMN is thought to be caused by overproduction of nerve growth factors due to trauma or chronic continuous stimulation, whereas MEN2B-associated MN is caused by GNDF-induced hyperplasia. In both cases, considering the pathogenesis of the development, hyperplasia of the nerve fibre bundles appeared and was assumed to have the same histological type of hypertrophy. MNs (SMN and MEN2B-associated MN) must be differentiated from benign peripheral neurogenic tumours. Morphological recognition and immunohistochemical analyses make this differentiation possible.
Footnotes
Author Contributions
YT contributed to the conception and design, acquisition of data, analysis, and interpretation of data. TY, KS, TS, MO, AG and TM contributed to analysing the patient’s data/findings. MS carried out the immunohistochemical staining. SY conceived the study, participated in its design and coordination, and helped to draft the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
A case report is exempt from approval at our institution. The patient provided written informed consent.
