Abstract
Advanced Non-Small Cell Lung Carcinoma (NSCLC) patients with ROS1 gene rearrangement have shown significant therapeutic responses to tyrosine kinase inhibitors approved by the US Food and Drug Administration, with approximately 40 fusion partners documented in the existing literature. Our report highlights a novel fusion partner of ROS1 that has demonstrated a conclusive response to the current standard of treatment.
Keywords
Introduction
ROS1 fusion rearrangement is a well-known genetic alteration in non-small cell lung cancer (NSCLC) that affects roughly 2.8% of NSCLC cases. 1 This alteration triggers a constant activation of tyrosine kinase activity, which, in turn, activates downstream signaling pathways, such as JAK/STAT, MAPK/ERK, and PI3K/AKT, leading to uncontrolled cell growth and survival advantages. The FDA has approved Ceritinib, Crizotinib, and Entrectinib as first-line therapies and Lorlatinib and Entrectinib as subsequent therapy for NSCLC patients with ROS1 fusion.2-4 Over 40 ROS1 partners have been discovered using next-generation sequencing. 5 While patients with ROS1 rearrangement show a robust response to the drugs above, there may be variations in treatment outcomes depending on the specific fusion partners and breakpoints present in each patient. 6
Next-generation sequencing has made it possible to identify novel fusion partners. However, evaluating the therapeutic responsiveness of these rearrangements with existing tyrosine kinase inhibitors is essential. This study has identified an actionable new ROS1 fusion partner. The awareness of this new fusion rearrangement that responds to the current standard of care is an addition to the knowledge base. It may help include the appropriate primer sets in real-time polymerase chain reaction kits and next-generation mRNA-based panels.
Description
A middle-aged man has been experiencing chest and back discomfort for the past month. During a routine chest X-ray, a uniform opacity was discovered. A subsequent chest CT scan revealed a soft tissue lesion with spiculated margins, bronchus cut off, and lytic destruction of the second to fourth ribs. A PET-CT scan then showed an active mass in the anterior segment of the right upper lobe of the lung that had infiltrated the lateral chest wall, eroding the second, third, and fourth ribs, along with an enlarged lymph node in the right hilar area. Additionally, a brain MRI indicated a tumor in the right parietal and left occipital lobes, which is likely metastatic. Following a CT-guided lung biopsy, it was determined that non-small cell lung cancer was present with dual immunopositivity for TTF-1 and p40 in nonoverlapping areas. Given the conflicting immunohistochemistry, a Non-Small Cell Carcinoma-Not Otherwise Specified (NSCC-NOS) diagnosis was made, commenting that Adenosquamous carcinoma cannot be excluded. (Figures 1 and 2)

It shows a tumor arranged in a solid manner (A) with polygonal tumor cells with abundant pink cytoplasm (B), vesicular nucleus, and prominent nucleoli (C, D).
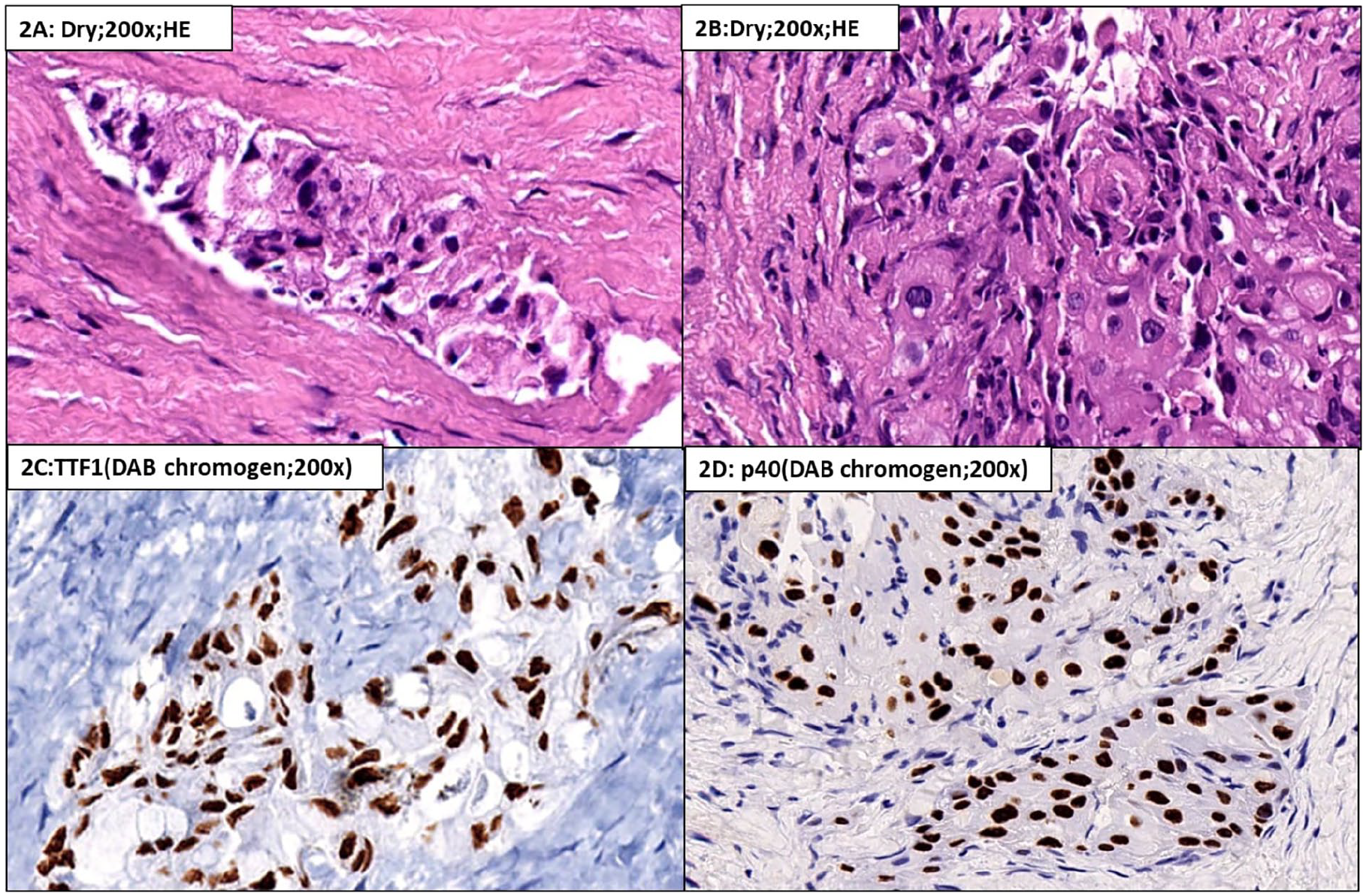
It shows a solid tumor with bubbly cytoplasm (A) to amorphous eosinophilic cytoplasm with evidence of keratinization (B). The tumor cells showing foamy cytoplasm are positive for TTF-1 (C), and those with squamous morphology are positive for p40 (D).
A Gemcitabine and carboplatin-based palliative chemotherapy were started, and Next-generation testing was requested to search for actionable biomarkers. Next-generation sequencing (NGS) was performed utilizing the Oncomine focus assay panel, interrogating 52 genes for single nucleotide variations and fusion rearrangements. The test returned a novel oncogenic VCL(16): ROS1(35) fusion with 378 fusion reads of high mapping quality (Figure 3) that exceed the 250 reads required as evidence for a non-targeted fusion that is not overtly included in the Oncomine™ panel. The NGS analysis was negative for EGFR, MET, KRAS, BRAF, HER2 driver mutation, or any other fusion rearrangement.

Integrated genome viewer showed a non-targeted VCL-ROS1 fusion transcript (chr10:75865111-chr6:117642557). Note the perfect quality of alignment around the fusion site.
Following the identification of a novel fusion partner, immunohistochemistry (IHC) was utilized to test for the overexpression of ROS1 protein. The results, displayed in Figure 4A, showed strong immunopositivity. To confirm these findings further, Fluorescent-in-situ-hybridization (FISH) was performed using ZytoVision, Germany’s ZytoLight® SPEC ROS1 Dual Color Break Apart Probe. The FISH results demonstrated gene rearrangement in 80% of the cells, with 1 fused and isolated green signal detected (Figure 4B).
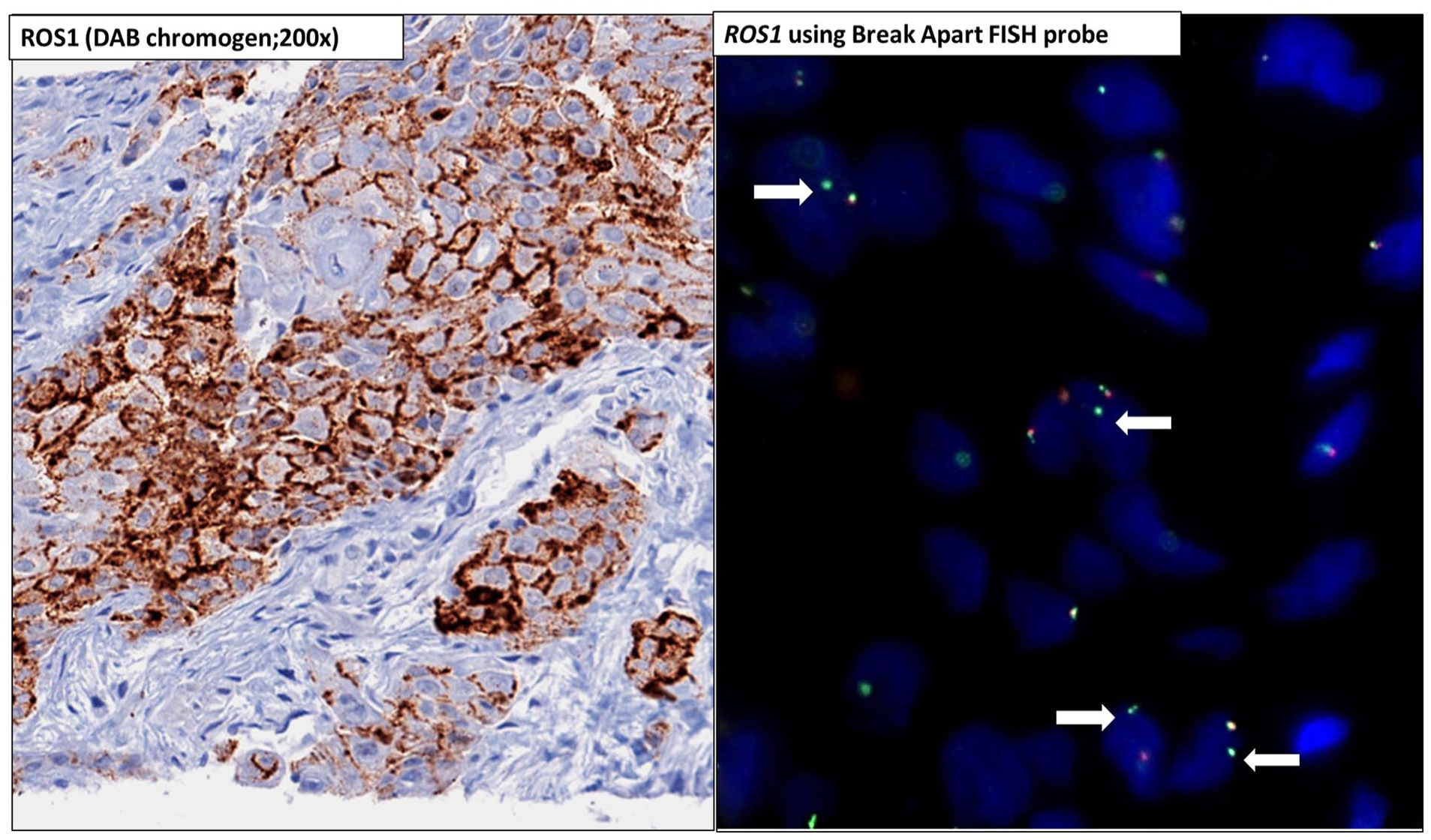
It shows ROS1 protein expression in tumor cells on immunohistochemistry (left panel), which was orthogonally confirmed on FISH (right panel). A break-apart FISH image shows gene rearrangement (highlighted by arrows) with 1 fused and 1 isolated green signal.
After 2 cycles of palliative chemotherapy, the patient was started on Crizotinib. PET-CT and MRI-brain, after 3 months, showed a partial response to treatment that has now continued for 6 months of therapy (RECIST 1.1). The patient developed oligo progression after 1 year, for which he received Stereotactic body radiation therapy (SBRT) and maintained a stable disease status after that (RECIST 1.1). The patient is on crizotinib and has regular follow-ups (Figure 5).

Images A, B, C, D (fused axial PET-CT) and E, F, G, H (maximum intensity projection, MIP) show right lung upper lobe mass with chest wall infiltration with a partial response to treatment (b), oligo progression after 1 year (c) and stable disease after SBRT (d).
Discussion
To our knowledge, the fusion of VCL::ROS1 has not yet been reported in NSCLC. This case report presents a unique instance of ROS1 fusion in a smoker with NSCC-NOS, with suspected adenosquamous lung carcinoma. Our patient’s fusion breakpoint occurred at exon 16 of the VCL gene and exon 35 of ROS1. The ROS1 rearrangement is typically observed in non-smokers with adenocarcinoma morphology. However, our patient, a smoker, brought home the importance of performing biomarker testing in all non-small cell lung carcinoma cases, regardless of smoking status.
Several ROS1 fusion partners have been identified, and the downstream oncogenic pathway may vary depending on the fusion partner. Patients with ROS1 rearrangement have a median response duration of 17.6 months with Crizotinib. Still, some may show primary resistance or only respond for a few months due to the acquisition of early resistance. 6 Evaluating Crizotinib response in ROS1 rearranged NSCLC with different fusion partners can provide insights into the heterogeneity of treatment response.
This study reports a novel ROS1 fusion in NSCLC, which has responded well to Crizotinib treatment. The patient has shown partial response for the past 6 months since starting Crizotinib therapy and is currently under follow-up. Long-term follow-up of the patient is planned to monitor the effectiveness of treatment over time. The Institutional Review Board approved the study via RGCIRC/IRB-BHR/Res/SCM/53/2022/74. Informed consent was obtained from the subjects included in the study.
Footnotes
Author Contributions
AM, HD, SN, UB, MG, MKP, DK, SM, AS: Substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data. HD and AM: Drafting the article and revising it critically for important intellectual content. HD and AM: Final approval of the version to be published.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The Institutional Review Board approved the study via RGCIRC/IRB-BHR/Res/SCM/53/2022/74.
Informed Consent
Informed consent was obtained from the subject included in the case report.
Consent for Publication
Consent for publication was obtained for the individual person’s data included in the manuscript.
