Abstract
Background:
Peribronchiolar metaplasia (PBM) is considered a reaction to injury characterized by the proliferation of bronchiolar epithelium into immediately adjacent alveolar walls. While an association of PBM with diffuse interstitial lung diseases has been recognized, the clinical significance of PBM remains uncertain.
Methods:
A cohort (n = 352) undergoing surgical resection of a lung nodule/mass in a rural area was retrospectively reviewed. Multivariate logistic regression analysis was performed to determine the association of PBM with clinical, physiological, radiographic, and histologic endpoints.
Results:
In the total study cohort, 9.1% were observed to have PBM as a histologic finding in resected lung tissue (n = 32). All but one of these patients with PBM were ever-smokers with a median of 42 pack years. Clinical COPD was diagnosed in two-thirds of patients with PBM. Comorbid gastroesophageal reflux disease (GERD) was significantly associated with PBM. All patients with PBM demonstrated radiologic and histologic evidence of emphysema. Measures of pulmonary function were not impacted by PBM. Mortality was not associated with the histologic observation of PBM. In a logistic regression model, centrilobular-ground glass opacity interstitial lung abnormality and traction bronchiectasis on the CT scan of the chest and histologic evidence of fibrosis, desquamative interstitial pneumonia and anthracosis all strongly predicted PBM in the cohort.
Conclusion:
A constellation of radiologic and histologic smoking-related abnormalities predicted PBM in study cohort. This confirms a co-existence of lung tissue responses to smoking including PBM, emphysema, and fibrosis. Acknowledging the physiologically “silent” nature of small airway dysfunction on pulmonary function testing, our findings support PBM as a histologic marker of small-airway injury associated with cigarette smoking.
Keywords
Introduction
Injuries of the lung can include a peribronchiolar metaplasia (PBM) of the alveolar epithelium with the observation of simple columnar and cuboidal (“low columnar”) bronchiole epithelium located in immediately adjacent alveolar walls. Such proliferation of bronchiolar epithelium on the alveolar surface was initially referred to as Lambertosis as it was assumed that it followed a conveyance of cells from the bronchioles to the alveoli via the canals of Lambert. 1 PBM is now considered a nonspecific reaction to a variety of stimuli including tobacco smoke, air pollutants (eg, diesel exhaust and ozone), microbes, and gastric content aspiration.2 -5 It is often an incidental finding in patients with a diverse number of interstitial lung diseases (ILD) including respiratory bronchiolitis-interstitial lung disease (RB-ILD), desquamative interstitial pneumonia (DIP), non-specific interstitial pneumonia (NSIP), usual interstitial pneumonia (UIP), hypersensitivity pneumonia (HP), and rheumatoid arthritis.6 -9 PBM has also been noted as the only histologic finding in a small number of patients with ILD and identified as a separate entity of bronchiolocentric interstitial pneumonia (BCIP) and airway-centered interstitial fibrosis (ACIF).10 -13
Despite these recognized associations, the clinical significance of PBM remains uncertain. Accordingly, the current study delineates correlations of PBM with clinical, physiological, radiographic, and histological endpoints in a cohort of heavy smokers from a rural area.
Materials and Methods

Study approach for creating groups with and without histologic finding of peribronchiolar metaplasia (PBM).
Results
Of the 392 patients who underwent surgical resection for lung nodules/masses, 40 patients (10.2%) were excluded due to 1) lack of a satisfactory quality CT scan of the chest or lung tissue and 2) presence of non-lung cancer related metastatic lesions (Figure 1). The final cohort included 352 patients (Figure 1). The prevalence of PBM in the study cohort was 9.1% (32/352 patients). The remaining 320 patients (90.9%) constituted the comparison group without PBM (Figure 1).
Demographic variables were not different between the groups with PBM and without PBM (Table 1). Ninety percent of the study cohort was ever-smoker with mean pack years of 45.4 ± 34.3 and median pack years of 42 (IQR 35.7). This reflects the distinct, heavy smoking habit in the Appalachian region. One patient with PBM had significant passive smoke exposure whereas remaining 31 were ever-smokers. Approximately two-thirds of the entire study cohort (61.4%) carried a clinical diagnosis of chronic obstructive pulmonary disease (COPD), whereas only a minority of patients were diagnosed to have ILD (n = 5, 1.5%); all 5 patients with a history of ILD were in the group without PBM. There were no significant differences between the groups with PBM and without PBM in smoking behavior, COPD, and occupational exposure history. Comorbidities were not significantly different for PBM group except gastro-esophageal reflux disease (GERD, 59.4%vs 38.7%, P = .036). A baseline O2 requirement was documented in 14.5% of all patients, predominantly due to a clinical COPD diagnosis, with no difference between the groups (Table 1). Finally, mortality did not differ between those patients with PBM and those without PBM (15.6% vs 10.9%, P = .387) (Table 1).
Characteristics of groups of patients with and without histologic finding of peribronchiolar metaplasia (PBM) in the cohort (n = 352).

Abbreviations: CAD, coronary artery disease; COPD, chronic obstructive lung disease; DLCO, diffusion capacity for carbon monoxide; DM, diabetes mellitus; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; GERD, gastro-esophageal reflux disease; ILA, interstitial lung abnormalities; ILD, interstitial lung disease; OSA, obstructive sleep apnea; RV, residual volume; TLC, total lung capacity; VTE, venous thromboembolism.
Pre-surgical PFTs were performed for most patients in the cohort (n = 336, 95.4%). Corresponding to the diagnoses of COPD, the study cohort showed an obstructive impairment with a mean FEV1/FVC ratio of 67.74 ± 12.17, air trapping with a mean percent predicted RV of 141.13 ± 51.41, and a mildly decreased percent predicted DLCO of 69.06 ± 21.95. None of the indices of pulmonary function showed a statistically significant difference between the groups with and without PBM.
Radiographic emphysema was extremely frequent in the study cohort (70.4%). The group with PBM showed a significantly higher prevalence of any form of emphysema relative to the group without PBM (100%vs 67.5%, P < .0001). Additionally, the group with PBM had a higher prevalence of both CL and PS emphysema (90.6%vs 59.4%, P < .0004 and 43.7% vs 25.6%, P = .036, respectively). A detailed description of the prevalence of subclinical ILA and ILD patterns in the cohort has been reported. 22 A combination of ILA and ILD patterns were observed in a greater proportion of the group with PBM compared to the group without PBM (87.5%vs 49.4%, P = 0). The PBM group demonstrated a higher prevalence of CL-GGO ILA pattern (18.7%vs 6.8%, P = .031) and any ILD patterns (43.7%vs 15%, P = .002). Lastly, traction bronchiectasis on the CT scan was more common in the group with PBM (28.1%vs 13.1% P = .032) (Table 2).
Radiographic and histopathologic features of groups with and without histologic peribronchiolar metaplasia (PBM) in the cohort (n = 352).

Abbreviations: DIP, desquamative interstitial pneumonia; DIPNECH, diffuse idiopathic pulmonary neuroendocrine hyperplasia; GGO, ground glass opacity; HP, hypersensitivity pneumonitis; ILA, interstitial lung abnormalities; ILD, interstitial lung disease; OP, organizing pneumonia; RB, respiratory bronchiolitis.
Any fibrosis included fibrosis, fibroblastic foci, subpleural fibrosis, and architectural distortion.
Miscellaneous findings for PBM group (n = 6) includes FB giant cell reaction, cholesterol cliff (n = 1), follicular bronchiolitis (n = 1) and vascular medial hypertrophy (n = 1), calcification or metaplastic bone formation (n = 3). Miscellaneous findings for non-PBM group (n = 23) includes chronic inflammation (n = 4), silicotic nodule (n = 3), consolidation/necrosis (n = 5), pleural plaque (n = 4), lymphocytic interstitial pneumonia (n = 2), foreign body giant cell reactions (n = 1) DIPNECH (n = 1), adenomatous hyperplasia (n = 1), and bronchiectasis (n = 1), calcification or metaplastic bone formation (n = 1), and carcinoid (n = 1).
Histologically, primary lung malignancy was noted in 92% of resected nodules/masses in the study cohort. Proportion of patients with various subtypes of lung cancer and benign finding were not different between the groups with PBM and without PBM. There were several smoking-associated pathologic findings observed in greater frequency in the group with PBM including emphysema (100%vs 48.7%%, P < .0001), any pulmonary fibrosis (56.2%vs 13.1%, P < .0001), DIP (12.5%vs 2.8%, P = .022), anthracosis (78.1%vs 32.2%, P < .0001) and honeycomb changes (15.6%vs 0.9%, P < .0001). While organizing pneumonia was solely present in non-PBM group (0 vs 6.5%, P = .238), respiratory bronchiolitis was noted in both groups without significant difference (15.6%vs 12.5%, P = .581) (Table 2). CT chest images and corresponding pathologic findings of a representative cases with PBM were displayed in Figures 2 and 3, respectively.

Pre-operative CT chest images of a 67-year-old former smoker (50 pack years) female who underwent left upper lobectomy for suspicious lung nodule. Findings displayed include significant non-dependent findings of: (a) centrilobular emphysema, (b) traction bronchiectasis, (c) subpleural reticulation, and (d) centrilobular ground glass opacities (all represented with white arrows).
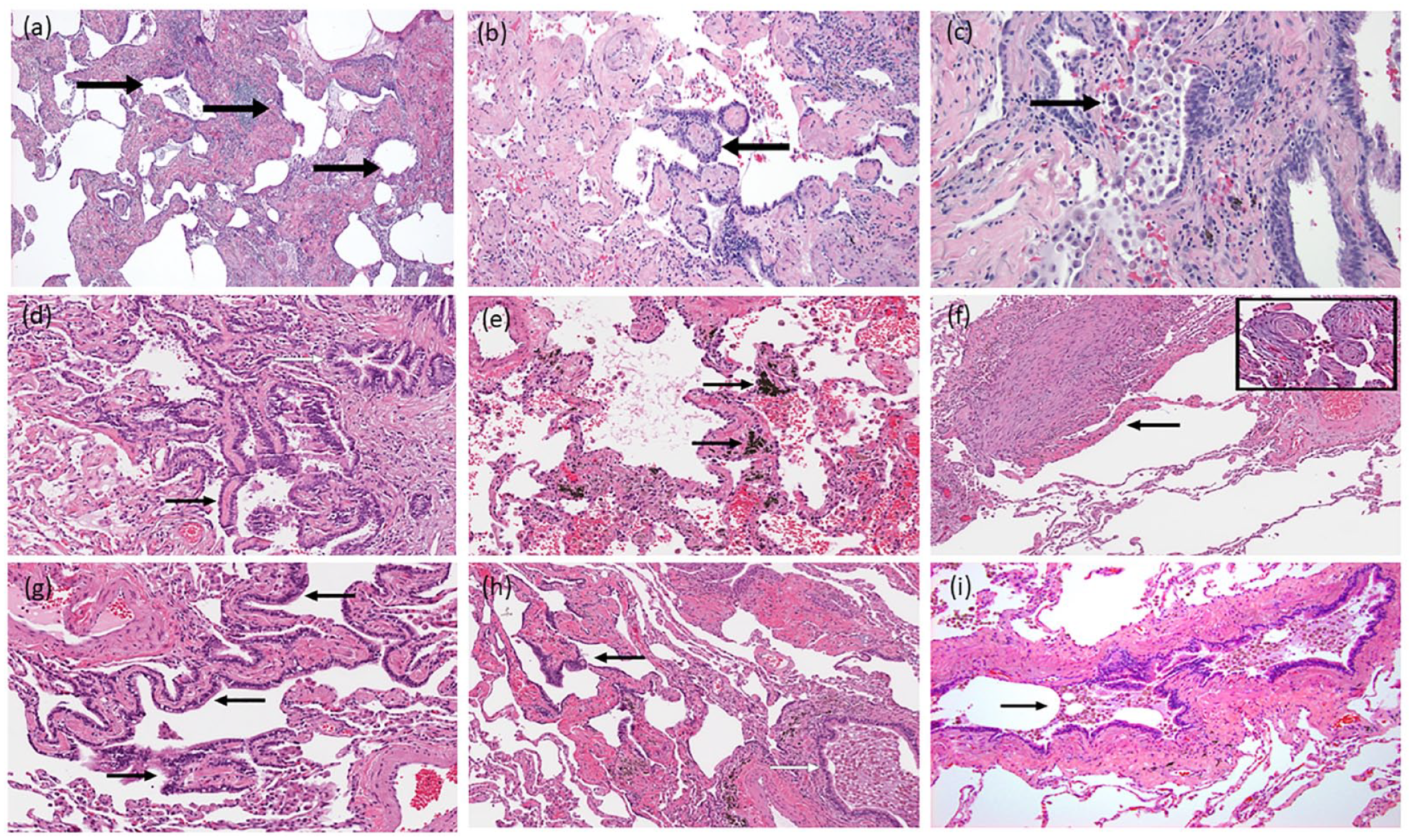
Peribronchiolar metaplasia and cohabitant spectrum of pulmonary pathology. Case #1. 67-year-old former smoker (50 pack-year) female with invasive lung adenocarcinoma. Non-neoplastic lung showed multifocal peribronchiolar metaplasia, honeycombing and DIP-like changes. (a) Microscopic honeycombing showing dense fibrosis, loss of lung architecture and cyst-like spaces (black arrows) lined by metaplastic bronchiolar epithelium (40×), (b) Respiratory-type epithelium lining alveolar walls with septal fibrosis (black arrow), characteristic of peribronchiolar metaplasia (100×), (c) Accumulations of numerous alveolar macrophages (black arrow), in addition to interstitial fibrosis and mild chronic inflammation, suggests a desquamative interstitial pneumonia (DIP) patter (200×). Case #2. 45-year-old ex-smoker (15 pack-year) female with invasive lung adenocarcinoma. Non-neoplastic lung showed multifocal peribronchiolar metaplasia, anthracosis, emphysema and fibroblastic foci. (d) Focus of metaplastic bronchiolar epithelium (black arrow) showing typical predilection to involve alveolar walls immediately adjacent to a bronchiole (white arrow). The alveolar septa are mildly fibrotic (200×), (e) Foci of anthracosis present throughout the lobectomy specimen (black arrows) in addition to dilated air spaces with associated fibrotic changes consistent with emphysema (200×), (f) Emphysematous lung showing fibroblastic foci within airspaces (black arrow) (100×). These microscopic nodules were scattered haphazardly within the parenchyma distant from the tumor and often exhibited a “young” basophilic appearance (see inset image) (400×). Case #3. 58-year-old current smoker (58 pack-year) male with squamous cell cancer. Non-neoplastic lung showed multifocal peribronchiolar metaplasia, anthracosis, emphysema and respiratory bronchiolitis-interstitial lung disease. (g) Bronchiolar mucosa extending along thickened alveolar septa, consistent with a focus of peribronchiolar metaplasia (black arrows) (200×), (h) Distorted, emphysematous lung with fibrotic alveolar walls, a focus of peribronchiolar metaplasia (black arrow) and a nearby bronchiole filled with mucus and macrophages (white arrow). Patchy deposits of anthracotic pigment are also present (100×), (i) Another bronchiole showing collections of pigmented macrophages within the airway lumen (black arrow) as well as in the surrounding alveolar parenchyma, characteristic features in the spectrum of respiratory bronchiolitis-interstitial lung disease (100×).
A logistic regression model was developed to predict PBM based on relevant clinical, physiologic, radiologic and histologic endpoints observed in the cohort (Table 3). Considering mutually existing prevalence of both radiologic and histologic emphysema in all PBM patients, they were excluded from the model. Radiologic findings of CL-GGO ILA and TB were highly predictive of PBM (odds ratio (OR) 5.72, 95% CI [1.53-21.32] and OR 3.45, 95% CI [1.21-9.79], respectively). Histologic findings of fibrosis, DIP and anthracosis were significantly associated with PBM (OR 5.83, 95% CI [2.17-15.68], OR 13.60, 95% CI [2.74-67.52] and OR 6.66, 95% CI [2.50-17.70], respectively). A radiographic ILD pattern and a histologic finding of honeycomb changes trended toward significance.
Logistic regression model showing predictors of peribronchiolar metaplasia (PBM) in the cohort (n = 352).
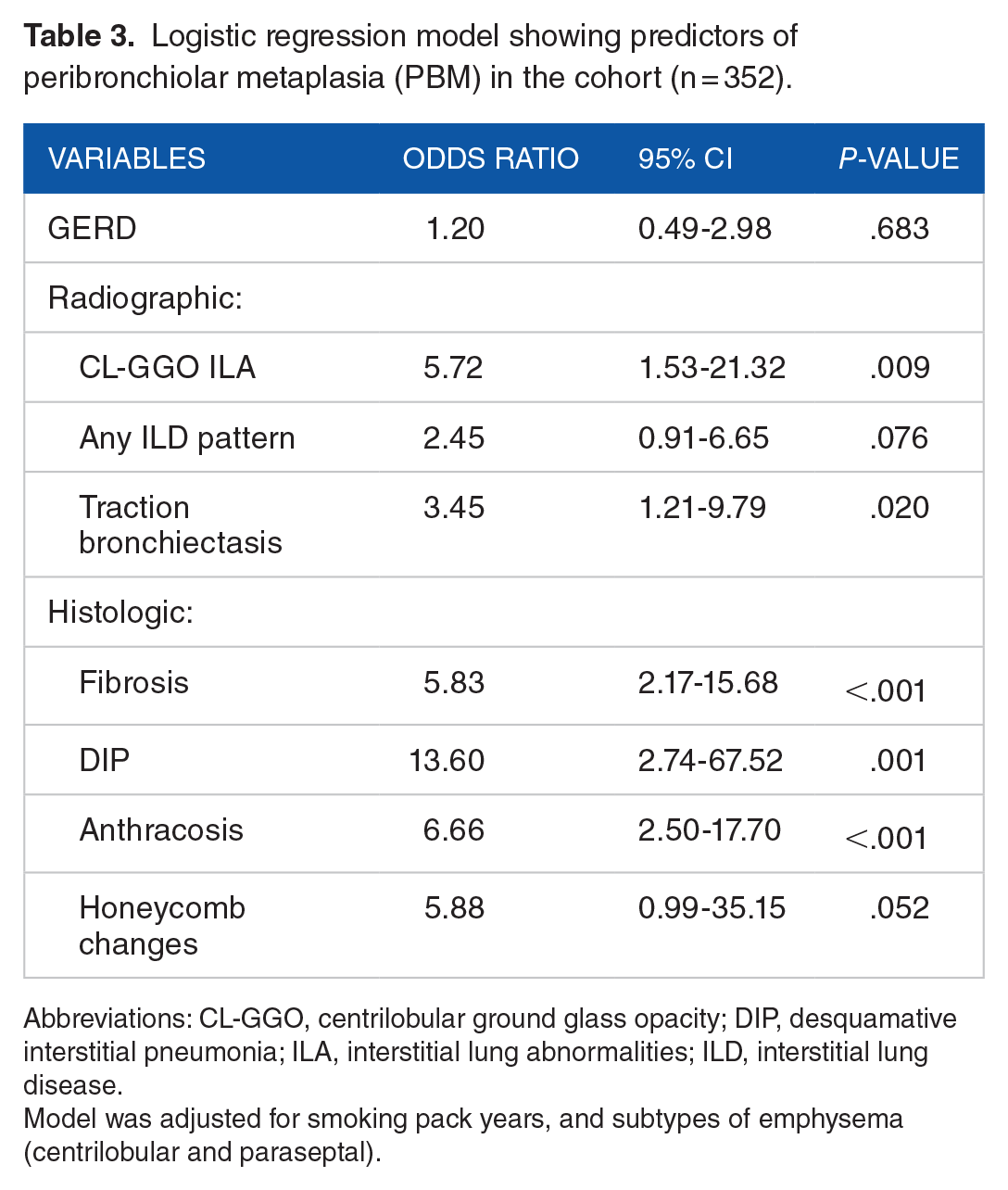
Abbreviations: CL-GGO, centrilobular ground glass opacity; DIP, desquamative interstitial pneumonia; ILA, interstitial lung abnormalities; ILD, interstitial lung disease.
Model was adjusted for smoking pack years, and subtypes of emphysema (centrilobular and paraseptal).
Discussion
In our study cohort, histologic evidence of PBM was observed in 9.1% of lung specimens. Despite considerable interest, it has not been feasible to determine the prevalence of PBM in the general population due to prior descriptions being predominantly included in case reports and series.7,10 -13,23 Previously, patients undergoing lobectomy for malignant neoplasms have demonstrated peribronchiolar fibrosis (PBF) and metaplasia in approximately one-fifth of patients (17.4%-24.2%) approximating the result in this study.24,25
Clinical characterization of the study cohort revealed enormous cigarette smoking histories and a high prevalence of co-morbidity related to such smoking. However, the prevalence of co-morbidities was not significantly different for the group with PBM relative to the group without PBM except for GERD. The increased prevalence of GERD in the group with PBM may reflect either a differential impact of smoking or an independent effect of GERD on PBM. Physiologically, PBM showed no specific relationship with any measure of pulmonary function. This is identical to other small airway diseases with a lack of pulmonary function changes challenging their detection. 26
Regarding the various radiographic and histologic endpoints investigated in this study, significant associations of PBM with evidence of emphysema were demonstrated using both. Cigarette smoking induced inflammation is widely reported in COPD, however, post-inflammation repair and/or response by lung tissue is not well-defined. Previous investigation on the small airways involvement among emphysematous patients has frequently revealed goblet cell metaplasia and not PBM. 27 Systematic evaluation of lung tissue in COPD patients has demonstrated that diffuse parenchymal epithelial metaplasia can be seen multifocally in association with goblet cell hypertrophy and inflammation occurring in response to cigarette smoking. 28 Moreover, these metaplastic cells exhibit surfactant and mucin production, suggestive of a primitive epithelial phenotype and potentially a consequence of injury to the cigarette smoke with aberrant repair. 28 Mutual co-existence of PBM and emphysema observed in our study support further examination of this relationship in an attempt to understand the chronology of events.
In contrast to the relationship between PBM and emphysema, the association of PBM with fibrotic injuries of the lung observed in this investigation has been reported previously. There were a greater number of radiologic findings of fibrosis (ILD and TB) and histologic endpoints of fibrosis (as any fibrosis, DIP, and honeycombing) in the group with PBM relative to the group without PBM in our study supporting a relationship between PBM and lung fibrosis. Patients with isolated PBM have been demonstrated to have a variety of radiographic findings including mosaic attenuation, lobular air trapping, subpleural fibrosis, septal thickening, and centrilobular ground glass nodules.7,23 Other radiologic endpoints of fibrosis (CL-GGO and TB) were predictive of PBM in our cohort. Histologic evidence of fibrosis associated with PBM can include patterns of RB-ILD, DIP, NSIP, UIP, hypersensitivity pneumonia (HP), BCIP, and ACIF.6 -13 Histology of lung tissue from patients with lung fibrosis can identify disease as: (1) primary bronchiolar disorders, (2) interstitial patterns with prominent bronchiolar involvement, and (3) bronchiolar pathology in association with large airway diseases. 29 While the role of an injury to the alveolar and adjacent interstitial regions in fibrosis is acknowledged, there is also a recognition of a possible contribution of small airways in the pathogenesis of fibrotic interstitial pneumonias.30,31 Such a role has been proposed for PBM in the predominantly bronchiolocentric pathology of chronic hypersensitivity pneumonitis. 32 Bronchiolar epithelial cells, rather than type II alveolar cells, were demonstrated to be the major source of epithelial renewal and proliferation in areas of severely fibrotic lung tissue. 33 Investigation has identified a differential pro-fibrotic response of small airways relative to lung parenchyma in response to cigarette smoke exposure. 34 Detailed morphological evaluation of both IPF and NSIP lung tissue showed increased inflammation in both bronchioles and peribronchiolar tissue as PBM and/or PBF supporting an active involvement of small airways in fibrotic lung diseases. 35 Emerging histologic evidence supports small airway remodeling as a possible contributory event toward fibrosis occurring in both ILAs and ILDs.
One in every 4 adults living in West Virginia is a current smoker (with an equivalent gender distribution) and there is an even higher prevalence of smoking (34.8%) among young adults (25-34 years of age). 36 Smoking is a well-known risk factor for the development and progression of emphysema and fibrotic lung conditions as well as lung cancer. This investigation demonstrated an excessive burden of malignant and non-malignant chronic lung diseases which continues to afflict the local Appalachian population.22,37 Cigarette smoke-induced chronic inflammation promotes these tissue responses and post-inflammatory changes at the level of small airways is an area of ongoing research. It was anticipated that a diverse number of radiologic (emphysema, CL-GGO of ILA, ILD patterns, and TB) and histologic (emphysema, fibrosis, DIP, anthracosis, and honeycombing) findings would be observed in our PBM subgroup since they share the strong affiliation with cigarette smoking. Identical to a role of respiratory bronchiolitis as a histologic marker of active smoking, the results of this investigation support that PBM can function as a histologic marker of small airway remodeling in a heavily smoking cohort (Figure 4). 38 This characteristic pattern of PBM and PBF in association with a wide variety of pathology was suggested to represent a “tombstone” marking of cigarette smoke-induced damage at and around small airways. 39
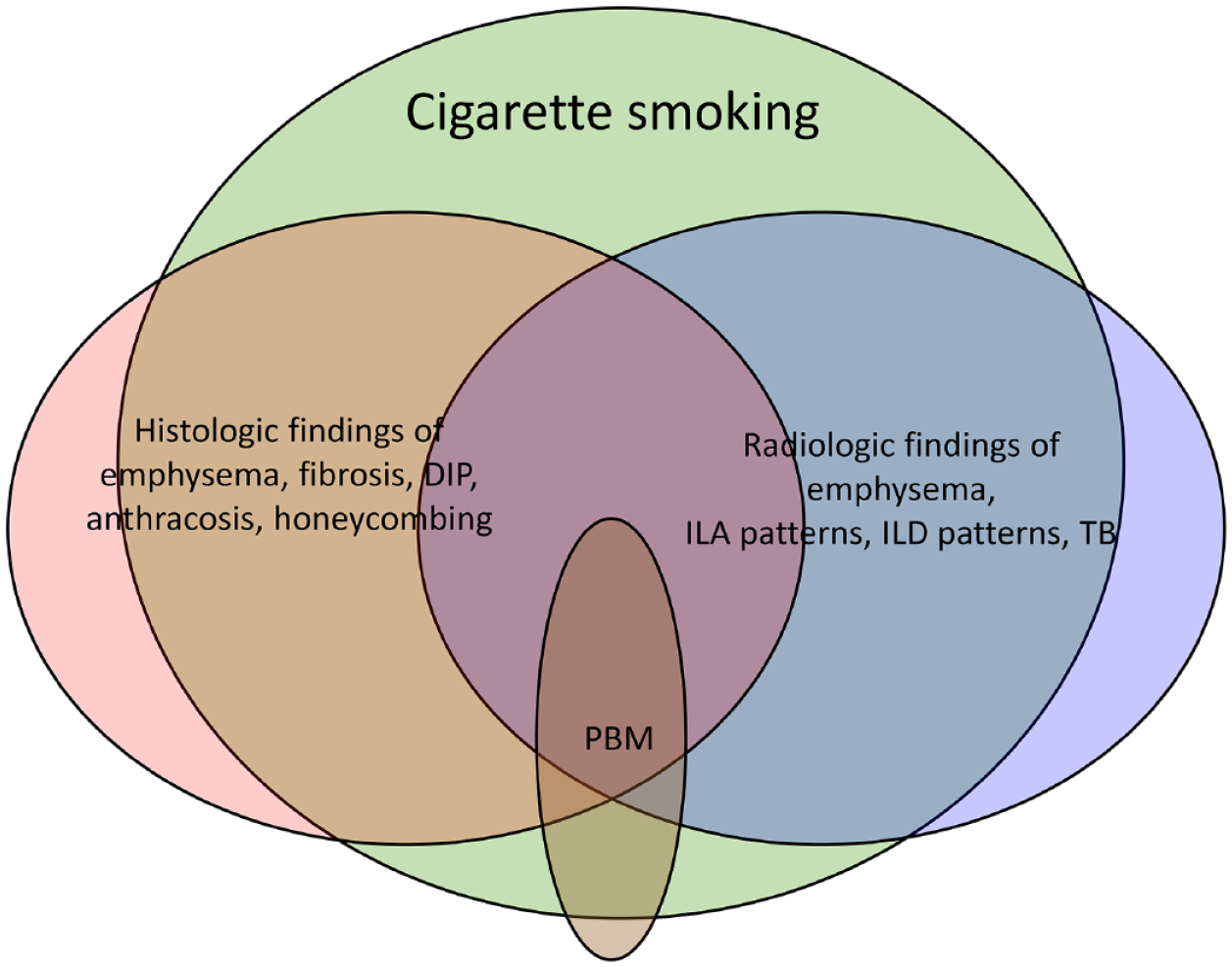
Diverse histologic (emphysema, fibrosis, desquamative interstitial pneumonia (DIP), anthracosis, and honeycombing) and radiologic (emphysema, interstitial lung abnormalities (ILA), interstitial lung disease (ILD) patterns, and traction bronchiectasis (TB) findings were seen in association with peribronchiolar metaplasia (PBM) in a heavily smoking rural cohort. Confluence of these findings can co-exist while sharing a risk factor of cigarette smoking. Accordingly, PBM can act as a histological marker of small airway remodeling process in response to cigarette smoking.
The retrospective approach employed in this study creates inherent limitations. Given consecutive enrollment of lung resection patients in the cohort during the study time frame, sample size calculation was not performed. The cohort was predominantly white, and the study was performed at a rural, quaternary medical center which may also limit generalization of study findings to different settings. The strengths of the study include the relatively large number of consecutively included patients undergoing surgical resection, detailed description of clinical and radiologic findings, and their correlation with histological features.
Conclusion
A cohort with heavy smoking undergoing surgical resection for suspected lung nodules/masses provided an opportunity to study the relationship between PBM and clinical, physiologic, radiographic, and histologic endpoints. There were associations of PBM with a constellation of various radiologic and histologic endpoints. The findings in the study may support the role of PBM as a histologic marker of small airway remodeling following exposure to cigarette smoke particles.
Footnotes
Funding:
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ Contributions
RGS and AJG conceptualized the study. RGS, VD, ZP, EA, and JV performed data collection, analysis and interpretation. RGS and VD were involved in drafting the manuscript whereas AJG, EA, JV and ZP provided critical review of the manuscript. All authors have read and approved the manuscript.
RGS takes primary responsibility for the accuracy of the findings presented.
Ethics Approval and Consent to Participate
The study protocol for retrospective review of electronic medical records was approved and “informed consent” was waived by the institutional review board of WVU (ID# 2010131995).
All ethical standards were adhered in accordance with the Declaration of Helsinki.
Consent for Publication
Not applicable.
Availability of Data and Material
All data generated or analyzed during this study are included in this published article.
RGS takes the responsibility of the content and accuracy of work presented in the manuscript.
Data Availability
This manuscript is not under consideration for publication at other journals.
A preprint version of preliminary data-analysis and interpretation is available on non-peer-reviewed platform of Research Square and can be accessed as “Peribronchiolar Metaplasia: A Cigarette Smoke- Induced Small Airway Injury Associated with Interstitial Lung Abnormality, Interstitial Lung Disease and Emphysema” (DOI: ![]() ).
).
