Abstract
Background:
Chronic gastritis (CG) is a very common disease. More than half of the worldwide population suffers from symptoms of CG. This disease has received great attention since the discovery of H. pylori as the most important cause of CG. Symptoms experienced by patients with CG are attributed to H. pylori-induced inflammatory reactions. Heparanase (HPSE) is a mammalian β-endoglucoronidase. In inflammation; HPSE degrades and remodels the extracellular matrix’s heparan sulfate polysaccharide chains liberating heparan sulfate-bound cytokines and chemokines, HPSE also facilitates movement of inflammatory cells.
Aims:
This study aimed to detect the function of HPSE in CG by correlating levels of HPSE expression with histopathological features of CG, including H. pylori infection, acute and chronic inflammatory cells, mucosal atrophic and/or metaplastic features.
Methods:
Ninety-five upper endoscopic-guided gastric punch biopsies were enrolled in this study. From each specimen, formalin-fixed and paraffin-embedded tissue blocks were prepared. Tissue sections were stained by Hematoxylin and eosin, Giemsa, and anti-heparanase antibody.
Results:
HPSE expression was statistically associated with H. pylori infection (P-value < .000), and intensity of chronic lymphocytic inflammatory infiltrate in the gastric mucosal tissues (P = .004). High levels of HPSE expression were also related to the presence of neutrophils in the gastric surface epithelium and lamina propria (P-value < .009).
Conclusions:
HPSE expression was upregulated in H. pylori-associated chronic gastritis. Thus, future therapeutic agents that could specifically inhibit HPSE enzyme activity, may aid in the reduction of sequelae of H. pylori infection.
Keywords
Introduction
Chronic gastritis (CG) is a global disease. The prevalence of CG is not strictly determined. However, it is worth to be mentioned that the prevalence is increasing with advancing age. 1 The 2 primary varieties of CG are type A, or immune (less common), and type B, or non-immune gastritis (more common). Both types of CG have rather identical histological characteristics. However, type A commonly involves the funding portion of the stomach in a diffuse manner while it leaves the antrum. As denoted by its name, type A is associated with the production of auto-antibodies against parietal cells. On the other hand; type B or non-immune CG starts initially in the antrum and proceeds toward the fundus and cardia. 2 The pathogenesis of type B became clear after the identification of the etiological role of Helicobacter pylori (H. pylori) in CG. H. pylori is a spiral, microaerophilic, gram-negative bacterium. 3 Previous studies showed that more than 50% of the worldwide population is infected by H. pylori. 4 Infection by H. pylori produces a significant inflammatory response and sustained recruitment of inflammatory cells in the gastric mucosa, resulting in chronic inflammation and progressive damage to the gastric epithelium. 5 H. pylori colonizes the gastric mucosa in different patterns; H. pylori may float freely in the gastric mucous, shows adhesions to the gastric surface epithelium, or colonizes intracellularly. 6 The latter pattern of H. pylori colonization has the greatest damaging effect on the gastric surface epithelial cells, ranging from apical mucous depletion to surface erosions and ulcerations. 7 Antimicrobial therapy is typically required for H. pylori-associated CG to eradicate the causative organism and subsequently stop the inflammatory processes and the symptoms of gastritis. However, many individuals were unable to completely eradicate their H. pylori infections. Previous research demonstrated a vicious cycle between the inflammatory cells in the gastric mucosa and the gastric epithelial cells that are infected with H. pylori. 8 Heparanase (HPSE) is a mammalian β-endoglucoronidase, its gene is located on chromosome 4. HPSE is released as a proenzyme that undergoes a proteolytic cleavage into active HPSE of 50 kDa, and 8 kDa polypeptides. 9 HPSE plays an essential function in multiple inflammatory reactions by changing the physical and chemical features of the extracellular matrix’s (ECM) heparan sulfate polysaccharide chains. These HPSE-induced modifications of the ECM result in the liberation of multiple cytokines and chemokines which are anchored to the ECM. In addition, HPSE facilitates the migration and subsequent migration of inflammatory cells toward the inflammatory sites. 10 3 inhibitors of the enzyme HPSE have been investigated in early-stage clinical studies as a potential therapeutic target. 11 Numerous investigations have shown that several inflammatory illnesses, including. inflammatory bowel diseases, 12 acute pancreatitis, 13 vasculitis, 14 and septic conditions 15 have up-regulated HPSE expression.
Aim of the Work
The goal of our study is to highlight the possible enhancing function of heparanase (HPSE) in chronic gastritis (CG) by correlating the different levels of HPSE expression with different histological parameters of CG as the presence of H. pylori infection, chronic lymphocytic infiltrate, gastric mucosal atrophy, intestinal metaplasia and presence of neutrophils, in order to develop a novel modality in the management of CG.
Materials and Methods
Clinical data and specimen collection
The current cross-sectional study enrolled 95 patients who complained of upper gastrointestinal symptoms including (hematemesis, dyspepsia, anemia, persistent epigastric pain, and/or vomiting). Patients were admitted to the Department of Tropical Medicine and Gastroenterology, Sohag University Hospital, from September 2021 to March 2022. Patients underwent upper endoscopic intervention and upper endoscopic-guided random gastric punch biopsies were obtained. Gastric biopsies were sent to Pathology Laboratory, Sohag University Hospital. Each specimen was preserved in formalin and labeled with the patient’s name, age, and sex. From the specimens, tissue blocks that had been formalin-fixed and paraffin-embedded were obtained. From each block, three 4 µm tissue sections were obtained to be stained by Hematoxylin and Eosin (H&E) stain, Giemsa stain, and the third section was stained by anti-human heparanase by using an immunohistochemical approach. H&E-stained sections were used to confirm the diagnosis of gastritis, determine degrees of gastric atrophy and chronic inflammatory infiltrate and establish the presence or absence of intestinal metaplasia and acute inflammatory cell infiltrate. Giemsa stain was used to detect H. pylori infection. Stained tissue sections were captured by Olympus EP50 5 megapixel microscope camera. Inclusion criteria included patients with clinical symptoms and upper endoscopic findings of gastritis. Exclusion criteria were patients who received antibiotics to eradicate H. pylori, patients with insufficient clinical data, and patients with gastric carcinoma. All patients gave their written informed consents. The study was approved by the Committee of Medical Ethics at Sohag Faculty of Medicine, Sohag University (Registration number: Soh-Med-21-09-43). The study was registered in Clinical Trials.gov PRS (Clinical Trials.gov ID: NCT05073614).
Immunohistochemical staining of Heparanase
Tissue blocks that had been formalin-fixed and paraffin-embedded were prepared from the upper endoscopic-guided gastric punch biopsies. These blocks were sectioned into 4 μm-thick sections. Deparaffinization was achieved by putting the sections in hot and then cold xylene, for 10 minutes each. Rehydration of the sections was done by inserting them in descending grades of alcohol, then they were put in distilled water for 10 minutes. Tissue sections were incubated in 3% H2O2 for 10 minutes at room temperature to inhibit endogenous peroxidase activity. Antigen retrieval was performed by using 0.01 mmol/L Citrate buffer fluid at 92°C for 20 minutes. A polyclonal rabbit IgG antibody produced against a recombinant protein of human Heparanase 1 was used to incubate the primary antibody on the sections for an overnight period at room temperature (Gene ID: 10855, Catalog Number: NBP3-03846-20ul, Concentrated form of 20l, NOVUS biological, USA). Tissue sections were incubated in streptavidin-biotin for 10 minutes, then they were immersed in diaminobenzidine (DAB) for 12 minutes at room temperature (ScyTek, P.O. Box 3286-Logan, Utah 84323, USA). To avoid excessive hematoxylin staining, which could obscure heparanase immunostaining, tissue sections were quickly washed in tap water after being submerged in Harris’ Hematoxylin for 15 seconds. Dehydration of the stained sections was performed by using ascending grades of alcohol. Lastly, xylene was used to clear the sections.
Evaluation of Heparanase immunostaining
Heparanase (HPSE) expression was detected as a brownish, granular, cytoplasmic stain (Figure 1). An established immunoreactivity grading system (IRS) was used to evaluate the immune-stained sections. The IRS was based on 2 basic components. Firstly, we calculated the proportion of positive cells and they were classified into; 0 (⩽5% positive cells), 1 (6%-25% positive cells), 2 (26%-50% positive cells), 3 (51%-75% positive cells), and 4 (⩾76% positive cells). Secondly, the intensity of HPSE expression was graded as 0 (negative expression), 1 (weak expression), 2 (moderate expression), and 3 (strong expression). The final score was calculated by multiplying the proportion of positive cells with the intensity of HPSE expression. The obtained IRS was divided into 4 expression categories: low (1-4), moderate (6,8), and high levels of expression (9,12). 16

Immunohistochemical expression of Heparanase in gastric glands as brownish cytoplasmic granules (anti-Heparanase immunohistochemical staining, X; 200).
Statistical analysis
The Statistical Package for the Social Science (SPSS) version 16 was used to interoperate the information. The mean, standard deviation, minimum, maximum, and median were used to present quantitative data. Frequencies, percentages, and Fisher’s Exact test were used for qualitative data.
Results
Patients’ characteristics
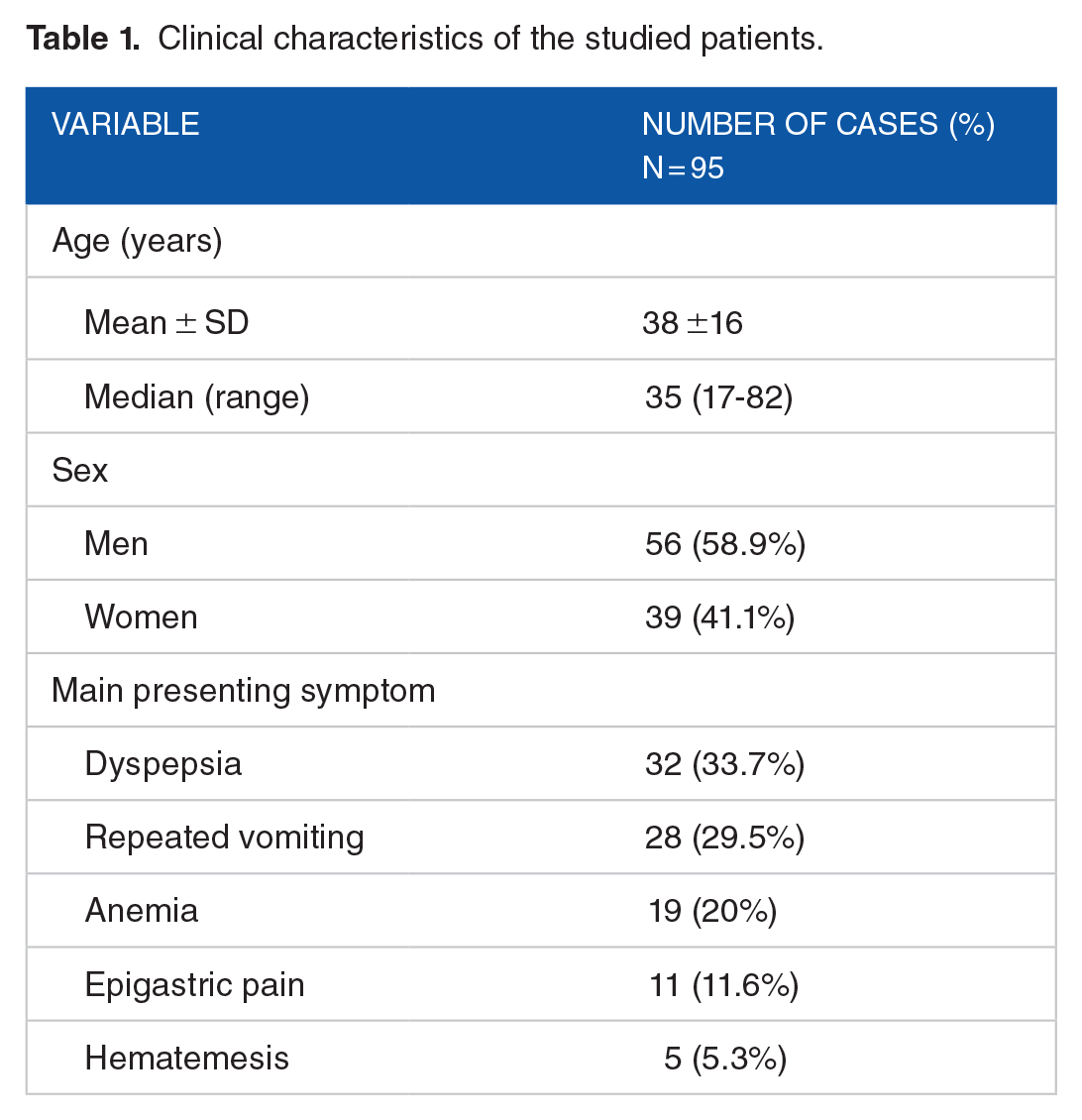
The current study enrolled 95 patients, all of them suffered from upper gastrointestinal upsets. The studied patients were composed of 56 men and 39 women. Their ages ranged from 17 to 82 years old, with a mean of age 38 and a median was 35 years old.
According to their main presenting symptoms; 32 patients suffered from dyspepsia and 28 patients suffered from repeated vomiting. Anemia, epigastric pain, and hematemesis were the main complaints in 19, 11, and 5 patients, respectively (Table 1).
Clinical characteristics of the studied patients.
Hematoxylin and Eosin evaluation of the studied patients
Gastric mucosal atrophy, as detected by edema of the lamina propria and separation of the gastric glands, was histologically confirmed in 71 patients. Intestinal metaplasia, either in the form of a change of the gastric epithelium to another epithelium that is morphologically identical to the absorptive epithelium of the intestine or the appearance of goblet cells, was detected in 17 patients. All the enrolled patients showed chronic lymphoplasmacytic inflammatory infiltrate in their H&E stained sections, but with variable intensity, 12 patients had severe lymphoplasmacytic inflammatory infiltrate with the frequent formation of lymphoid follicles. Neutrophil infiltrate of the lamina propria with or without attacking the gastric faveolar epithelium and gastric glands was detected in 72 patients (Figure 2).

(A) Atrophy of the gastric faveolar epithelium and glands (H&E staining, X; 40). (B and C) Appearance of goblet cells in the surface epithelium (H&E staining, X; 100& 200, respectively). (D and E) The lamina propria showed chronic lymphocytic infiltrate with the formation of lymphoid follicles (H&E staining, X; 100, each). (F) Neutrophils in the lamina propria and inside the gastric glands (H&E staining, X; 200).
Giemsa evaluation of the studied patients
Giemsa stain was used to detect H. pylori infection of the studied gastric specimens. H. pylori infection was detected in 81 patients (Figure 3). The histological characteristics of the studied patients were summarized in (Table 2).

(A and B) Colonies of H. pylori in the gastric glands (Giemsa staining, X, 1000).
Histopathological assessment of the studied population.

Immunohistochemical evaluation of HPSE.
Heparanase (HPSE) was detected in all of the enrolled tissue specimens with variable staining intensities (Figure 4). The expression levels of HPSE were low in 6 cases, and moderate in 20 cases, and a high level of expression was detected in 69 patients.

(A and B) moderate and high immunohistochemical expressions of Heparanase in chronic gastritis (anti-Heparanase immunohistochemical staining, X; 200).
There was a statistically significant association between the intensity of HPSE expression and H. pylori infection, 60% and 94.2% of cases that showed moderate and high levels of HPSE expression, respectively, showed colonies of H. pylori in their Giemsa-stained slides (P < .000).
The severity of lymphocytic infiltrate and formation of lymphoid follicles were associated with an increase in the staining intensity of HPSE, 91.7% of cases showed severe lymphocytic infiltrate with the formation of lymphoid follicles, and 81.5% of cases showed moderate lymphocytic infiltrate, were associated with high levels of HPSE expression (P = .004) as demonstrated in Table 3.
Correlation between HPSE expression and different studied histological parameters (Number of cases = 95).

P-value was calculated by Fisher’s Exact test.
Significant.
Another statistically significant positive association was detected between HPSE expression and the intensity of neutrophil infiltrate (activity), 72 out of 95 patients showed both neutrophil infiltrate in their H&E-stained slides and HPSE expression with variable staining intensities (P = .009) as shown in Table 4.
Levels of HPSE expression about the intensity of neutrophilic infiltrate in the studied cases (Number of active cases = 72).

The P-value was calculated by Fisher’s Exact test.
Significant.
Discussion
Chronic gastritis (CG) is a very common disease with an insidious onset and a chronic course. 1 It has been estimated that more than 50% of the worldwide population is suffering from symptoms of CG with variable severity. It is well established that CG may progress to peptic ulcer or primary gastric malignant neoplasms, through an ongoing chronic inflammation. 17
Most of the symptoms experienced by patients with CG are caused by H. pylori-induced inflammatory reactions. Infection by H. pylori produces a sustained inflammation of the gastric mucosa. Immediately after infection, H. pylori penetrates through the viscid mucous layer of the gastric mucosa. The bacteria multiply on the gastric surface epithelium and release chemical mediators which cause damage to the gastric faveolar epithelium and recruitment of neutrophils in the lamina propria and close to the surface epithelium. 18
The inflammatory response of H. pylori in chronic gastritis is augmented by the appearance of chronic inflammatory cells. Sustained recruitment of lymphocytes with subsequent formation of lymphoid follicles represents a constant feature of H. pylori-induced chronic gastritis. 19
As a result of a sustained inflammatory process, the gastric surface epithelium will undergo surface erosions. Regeneration of the surface epithelium may be accompanied by metaplastic changes such as intestinal metaplasia. 20 Sustained epithelial damage and recruitment of chronic inflammatory cells with the formation of frequent lymphoid follicles will produce gastric mucosal atrophy. 18
Heparanase (HPSE) is a mammalian β-endoglucoronidase, encoded by a gene located on chromosome 4. 9 Physiologically; HPSE is expressed in high levels in the human placenta, platelets, inflammatory cells as neutrophils, and mast cells, while it is undetectable in other human tissues. 21
Up-regulated expression of HPSE has been reported in many inflammatory conditions such as inflammatory bowel diseases, 12 hepatitis, 22 and rheumatoid arthritis. 23
Two observations highlighted the activating role of HPSE in inflammatory reactions; the first one came from the observation that there is a heparane-sulfate degrading activity in inflammatory cells as neutrophils, macrophages, and activated T-lymphocytes. The second clue of the inflammatory-mediating function of HPSE was suggested as inhibitors of HPSE enzyme activity had anti-inflammatory effects. 24
We aimed to evaluate the inflammatory-mediating role of HPSE in CG. This could be beneficial in developing future therapeutic agents that specifically block HPSE activity in order to relieve symptoms of CG and prevent occurrence of metaplastic and/or dysplastic lesions, before occurrence if invasive neoplasia.
In the current study, we evaluated the expression of HPSE in CG; we correlated the expression of HPSE with different parameters of CG, as gastric atrophy, chronic inflammation, neutrophil infiltrate, intestinal metaplasia and H. pylori infection. We believed that detection of HPSE in gastric tissues by immunohistochemical approach is better than its detection by serological approaches, as any increased expression of HPSE in gastric tissues would be related to CG.
A significant positive correlation was detected between H. pylori infection and enhanced HPSE expression. Tang et al evaluated the role of HPSE in H. pylori-induced chronic gastritis in animal models. They postulated that H. pylori up-regulate HPSE expression in the gastric epithelial cells of the tested mice. HPSE, in turn, modulates the ECM and facilitates the movement of neutrophils and macrophages toward the lamina propria and gastric surface epithelium. They suggested the existence of a vicious circle between H. pylori infection and overexpression of HPSE by gastric epithelial cells. 11
In the current study, high levels of HPSE expression showed a significant positive association with the severity of inflammatory cells in lamina propria and gastric faveolar epithelium. HPSE degrades heparan sulfate polysaccharide chains in the ECM and basement membrane. This facilitates the movement of immune cells. Also, HPSE liberates heparan sulfate-bound cytokines and chemokines, creating a concentration gradient that attracts immune cells to sites of inflammation. 25
HPSE expression didn’t show statistically-significant association with intestinal metaplasia or gastric mucosal atrophy, we recommend future use of larger sample size in which all parameters of CG are presented.
We postulated that termination of H. pylori-induced inflammatory reactions by blocking HPSE enzyme activity will improve symptoms of CG and it will decrease the incidence of peptic ulcers and gastric neoplasms that may complicate H. pylori-associated chronic gastritis.
Limitations of the Study
The study was a single center study in which the data were collected over 8 months without sample size calculation. Furthermore, all the studied gastric punch biopsies were randomly punched from endoscopically- suspected gastric mucosal lesions and submitted in a single patient’s name-labeled container without separating fundic, cardiac or pyloric samples.
Recommendations
Future studies should evaluate HPSE in separate gastric regions in order to verify and possible difference in HPSE expression in different gastric regions.
Conclusion
Heparanase expression is upregulated in H. pylori-associated chronic gastritis. So, developing new medications which can block heparanase enzyme activity may help in decreasing the incidence of metaplastic and /or neoplastic lesions which may complicate H. pylori-associated chronic gastritis.
Footnotes
Acknowledgements
We would like to thank all health care workers and our patients who help us in the collection of data.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SFMA and MHM: Conceptualization, formal analysis, and original draft writing; MMA: Resources provision, data curation management, and supervision; All authors: Methodology, review, and editing.
ClinicalTrials.gov Identifier
NCT05073614.
Ethical Consideration
The study protocol was accepted by the Ethical Committee of Scientific Research, Faculty of Medicine, Sohag University. Protocol registration number: Soh-Med-21-09-43.
