Abstract
Purpose:
Mammary Paget’s disease (PD) in young women has seldom been reported. The aim of this study was to improve the knowledge of the clinicopathological characteristics in young patients with PD to provide a basis for the precise treatment of young patients.
Methods:
The medical records and pathological slides of 8 young patients (younger than 40 years old) with PD were reviewed. The data of 20 patients over 40 years old within the same period were used as controls.
Results:
The average age was 32.00 ± 3.96 years for the young patient group, with the youngest aged 27 years. The first symptom, physical examination, Paget cell morphology, and immunohistochemical marks were the same in different age groups. But young patients have varied tumor distribution patterns, fewer interstitial inflammatory cells, and advanced pathological local lymphatic metastasis than older patients in the same period.
Conclusions:
PD in young women has unique histopathological features. These manifestations seem to provide personalized treatment for PD treatment in young patients. More research is needed to clarify the significance of this research.
Introduction
Mammary Paget disease (MPD), also known as eczematoid carcinoma, is a special type of uncommon breast cancer. Its name was first reported by James Paget, which means that there are malignant glandular epithelial lesions in the skin of the nipple-areola complex.1 -4 It is observed in 0.5% to 5% of cases of breast carcinoma,5,6, and the prevalence of MPD was 0.091% in skin biopsy slides. 7 The median (range) age was 60 (20-90) years, 8 mainly occurring in patients over the age of 40, and is rarely found in young women. 9 The most common treatment is complete mastectomy and the 5-year overall survival rate of patients with MPD is greater than 80% but is lower among older and black people. Eight cases of PD younger than 40 years were reviewed in this paper, and we focused on the clinicopathological features and compared them with those of 20 PD patients aged older than 40 years within the same period.
Materials and Methods
Study population
Between May 2010 and May 2021, a total of 166 patients with Paget disease were admitted to the Pathology Department of Shandong Provincial Hospital and Chengdu Changjiang Hospital, confirmed by pathological examination. Of these, 96 cases occurred in the nipple area. The age distribution of these patients is shown in Figure 1. Of these patients, only 8 (8.3%) were under the age of 40, while the other 88 patients (91.7%) were 40 years of age or older. This study followed the principles outlined in the Helsinki Declaration and approved by the Institutional Review Board of Shandong Provincial Hospital (approval number: 202114200) and Chengdu Changjiang Hospital (approval number: 20210020). Informed consent was deemed unnecessary for this type of retrospective study by Shandong Provincial Hospital and Chengdu Changjiang Hospital. The medical records and pathological slides of 8 young patients with PD were reviewed. The metastasis and recurrence patterns of these patients were followed up. Twenty patients 40 years of age or older within the same period were selected randomly as controls.

Age histogram for the 96 patients with Paget disease of the Nipple Region (PDNR). Only 2 patients were in their 20s, and 6 patients were in their 30s. The age of peak incidence of PDNR was between the sixth and seventh decades of life, with a median age of 54 years.
Pathological studies
All 8 young cases and 20 elderly cases were discussed and confirmed by 2 senior pathologists. For cases that were difficult to diagnose, immunohistochemical or special staining was added to distinguish them. Immunohistochemistry (IHC) was performed with commercially available antibodies: CK7 (Clone No. OV-TL 12/30), CK8/18 (Clone No. TS1), ER (Clone No. bp6139), HMB45 (Clone No. HMB45), CK-Pan (Clone No. AE1/AE3), CEA (Clone No. cea31), HER-2 (Clone No. bp6020), GCDFP-15 (Clone No. ep1582y), CK5/6 (Clone No. D5/16b4), p63 (Clone No. 4a4), Ki67 (Clone No. MIB-1), PR (Clone No. bp6136), S-100 (Clone No. 4c4.9), CAM5.2 (Clone No. CAM5.2). And it was applied according to quality-controlled protocols that are regularly evaluated in round robin trials. The diagnostic criteria refer to relevant literature,9,10 excluding Paget-like melanoma in situ, squamous cell carcinoma in situ, Paget-like reticulohistiocytosis and other diseases. According to the distribution pattern of Paget cells proposed by Hoda et al, 11 patient tissue sections were evaluated. Parameters including age of onset, clinical manifestations, pathological characteristics, management, and outcome were summarized.
Statistical analysis
The Fisher’s exact test was used to compare age and lymphovascular involvement. The comparisons of other clinicopathological data and prognostic indicators between groups were performed using the rank test. P-values <0.05 were considered significant. All statistical analyses were performed using SPSS 18.0 software (SPSS Inc., Chicago, IL, USA).
Results
Clinical features
All patients were female. The median age for the entire study group was 54 years (range: 27-81 years). Only 8 patients were under the age of 40, and the average age was 32.00 ± 3.96 years, with the youngest aged 27 years. The clinicopathological data are summarized in Table 1. The primary symptoms included skin lesions in the nipple-areola complex (3 cases; 37.5%) and breast masses (5 cases; 62.5%). All 8 patients underwent surgical resection, followed by radiotherapy or chemotherapy and/or endocrine, targeted therapy.
Clinicopathological features of the 8 young women with Paget’s disease in nipple region.
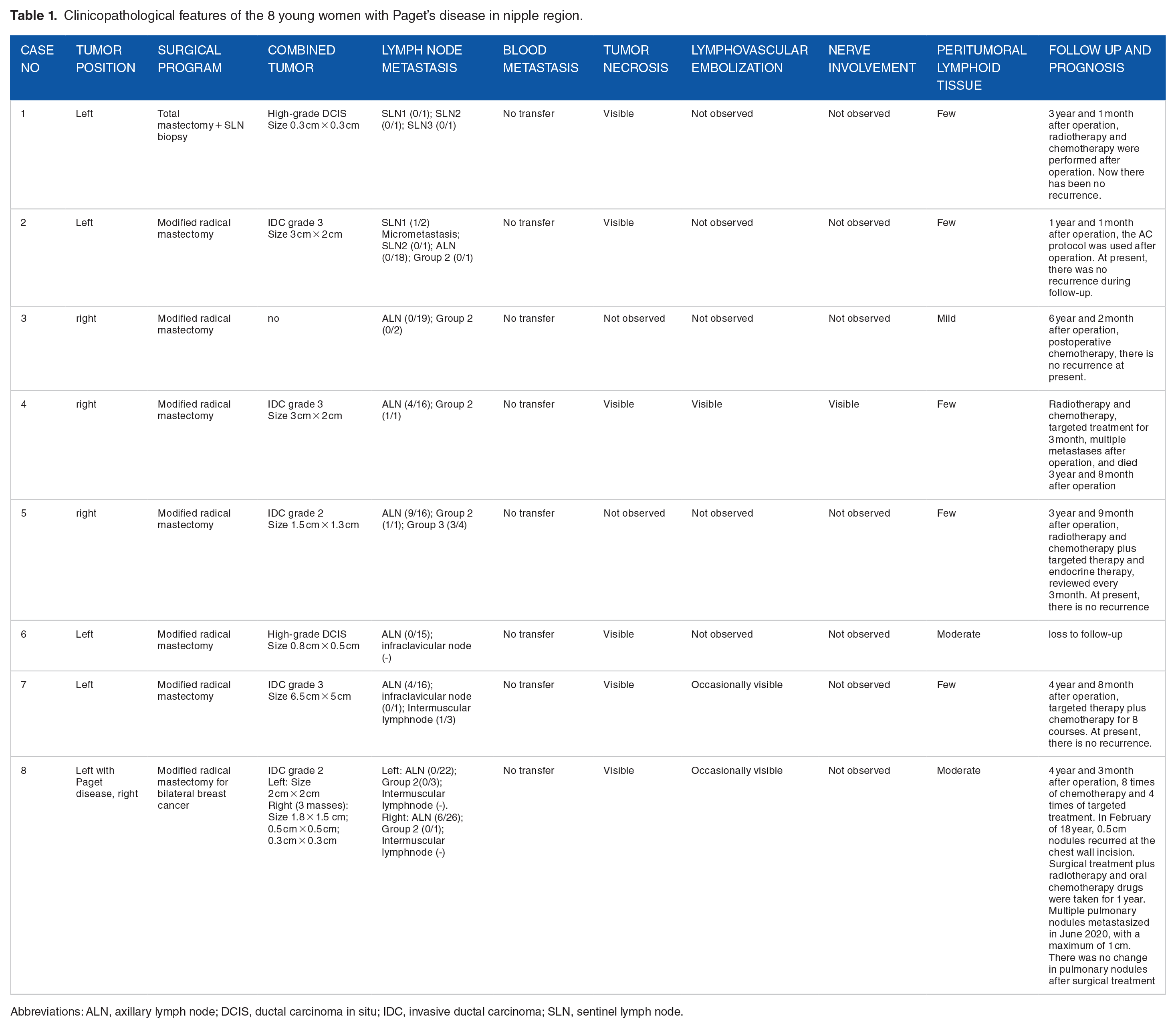
Abbreviations: ALN, axillary lymph node; DCIS, ductal carcinoma in situ; IDC, invasive ductal carcinoma; SLN, sentinel lymph node.
Pathological characteristics
All tumors conformed to the WHO histological criteria (2019) for PD. Paget cells were characterized by their round to oval nuclei, large pleomorphic nucleoli and abundant amphophilic or clear cytoplasm (Figure 2A and B), scattered with single cells and small clusters in the spinous and/or basal layer (Figure 3A), and showed diverse proliferation patterns in the squamous epithelium (Table 2). The most common pattern was the prototypical pattern (PTP; 62.5%), followed by the acantholysis-like pattern (ALP; 50%; Figure 3B), glandular pattern (GP; 50%; Figure 3C), tall nest pattern (TNP; 37.5%), and budding pattern (BP; 12.5%; Figure 3D). No upper nest pattern (UNP) was observed in the young group. The Paget cells extended into the skin appendages in 3 cases and invaded locally in only 1 case. The prototypical pattern was observed in all middle-aged and elderly patients (100%), followed by ALP (80%), sheet-like pattern (SLP; 30%), TNP (20%), UNP (20%), and GP (20%), and the patterns of tumor cell proliferation in the epidermis were inconsistent with those of younger patients (P < 0.01).
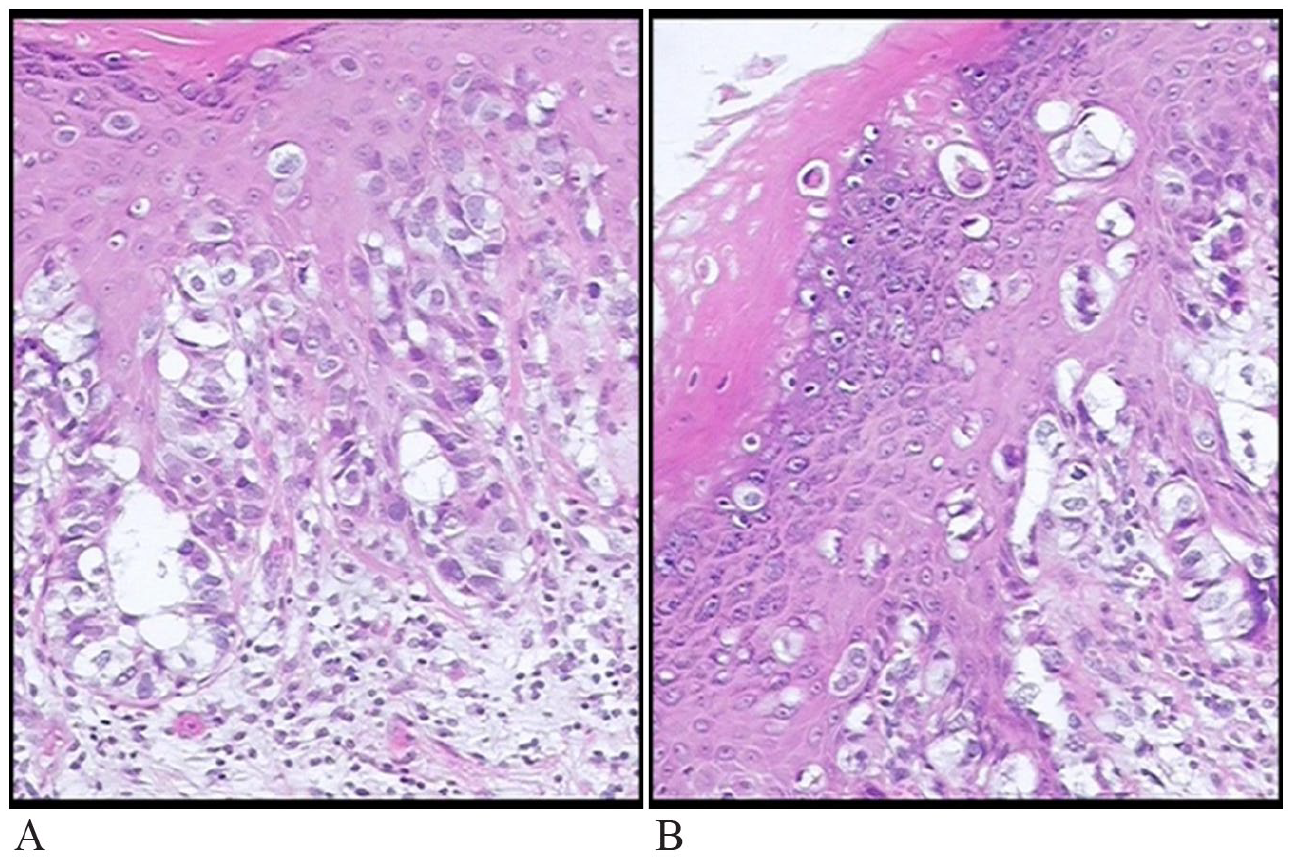
The morphology of Paget cell: (A) the characteristics of Paget cell (HE staining, 10×) and (B) the cytoplasm around some tumor cells is empty and bright (HE staining, 10×).

The proliferation patterns of tumor cell in the epidermis: (A) the tumor cells are mottled (CK7 staining, ×10), (B) acantholysis-like pattern, ALP (HE staining, 10×), (C) glandular pattern, GP (HE staining, 10×), and (D) budding pattern, BP (HE staining, 10×).
Histopathological observation of Paget’s disease in nipple region of 8 young women.
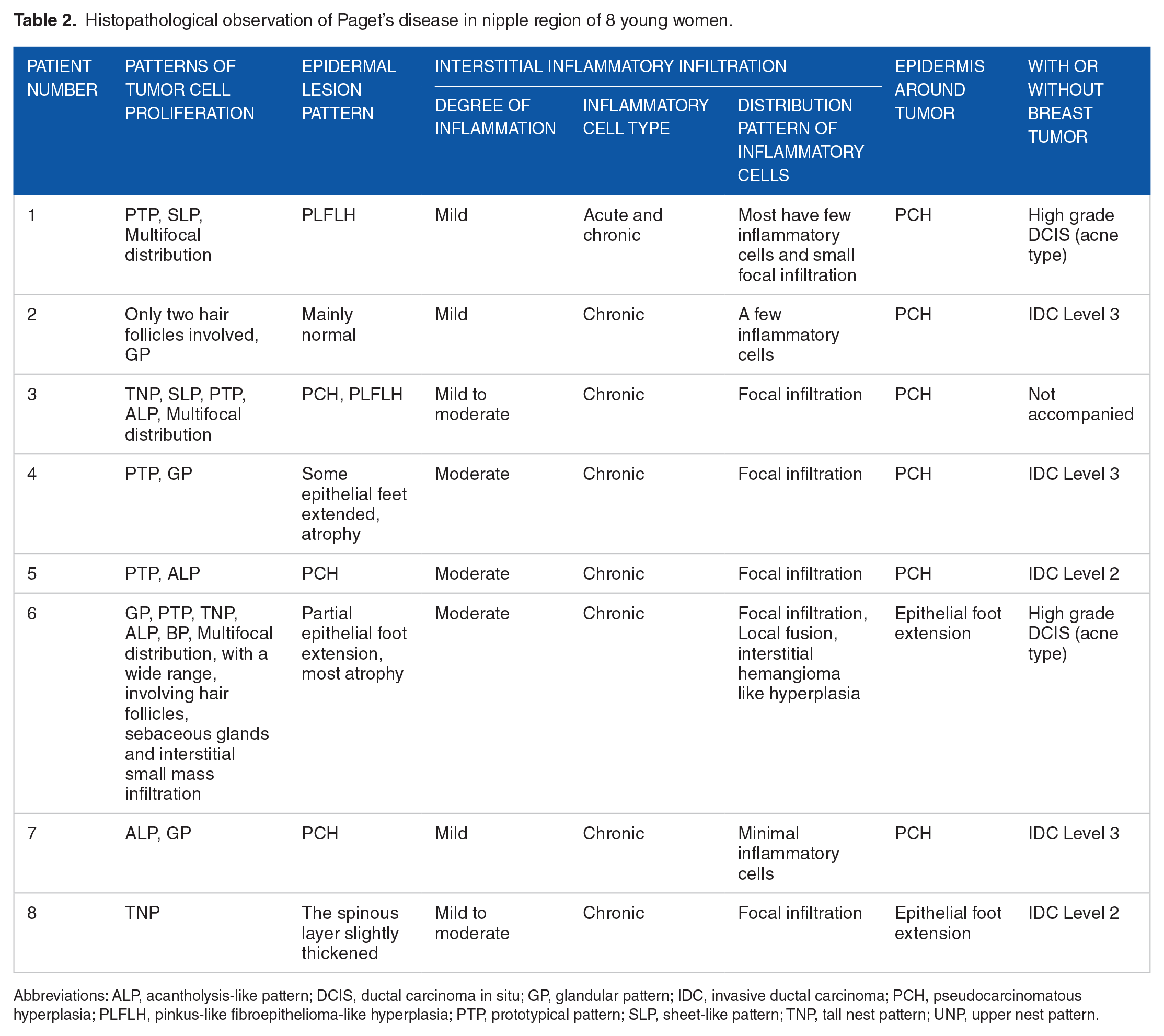
Abbreviations: ALP, acantholysis-like pattern; DCIS, ductal carcinoma in situ; GP, glandular pattern; IDC, invasive ductal carcinoma; PCH, pseudocarcinomatous hyperplasia; PLFLH, pinkus-like fibroepithelioma-like hyperplasia; PTP, prototypical pattern; SLP, sheet-like pattern; TNP, tall nest pattern; UNP, upper nest pattern.
The epidermis around the nest of tumor cells proliferated or shrank in PD. Four of 8 (50%) were normal or less normal; 3 of 8 (37.5%) showed pseudocarcinomatous hyperplasia (PCH); 2 (25%) revealed pinkus-like fibroepithelioma-like hyperplasia (PLFLH), and psoriasis-like hyperplasia (PLH) was not detected in young patients. All middle-aged and elderly patients displayed epidermal hyperplasia around the tumor nest, and the modes of epidermal hyperplasia were PCH (100%), PLH (40%) and PLFLH (20%). Epithelial hyperplasia seemed to be more pronounced in middle-aged and elderly patients (P < 0.01).
Eight young patients presented a minimal degree of inflammation, while 62.5% showed mild (or moderate) inflammation. The distribution of infiltrating cells was as follows: focal infiltration (3 cases; 37.5%), regional blending infiltration (1 case; 12.5%), and only a few inflammatory cells (4 cases; 50%). The degree of inflammation in middle-aged and elderly patients was mostly moderate (80%), and a few were mild to moderate (20%); the distribution of infiltrating cells was focal infiltration (8 cases; 40%), regional blending infiltration (8 cases; 40%), and zonal infiltration (4 cases; 20%). The degree and distribution of inflammation were lower in young patients (P < 0.005). The superficial dermal vessels were observed to have proliferated and dilated to varying degrees. Only in 1 young patient was abundant interstitial vessels and hemangioma-like hyperplasia observed (Figure 4).

Hemangioma-like hyperplasia was observed in 1 case.
Paget cells extended into the skin appendages (Figure 5), concomitant breast ductal carcinoma (2 cases; 25%) and invasive ductal carcinoma (5 cases; 62.5%). Paget cells expressed CK-Pan, CK7, CK8/18, CAM5.2, EMA, and HER-2 (Figure 6A and B), partially expressed CEA and GCDFP-15, and did not express HMB45, S-100, ER, or PR. CK5/6 and p63 were positive in the surrounding normal or residual epithelium, which stood in sharp contrast with the tumor cells (Figure 6C and D). The positive rate of cell proliferation-related antigen Ki67 was 10% to 80% (average 38% ± 30%). AB-PAS staining was positive in some cases.

Paget cells involved hair follicles and/or large ducts (CK8/18 immunohistochemical staining, 4×).

Immunohistochemical staining: (A) CK8/18, (B) Her-2, (C) CK5/6, and (D) P63.
Follow-up
Follow-up was until March 2022 or until death if earlier. Follow-up was completed for 7 young patients, while 1 patient was lost to follow-up (12.5%). Six of 7 patients were alive. One patient with an obvious vascular tumor thrombus died. The young patients with bilateral breast cancer were still alive, with local recurrence and metastasis to the lung. The follow-up and prognosis of PD patients are shown in Table 1.
The comparative results of clinicopathological characteristics of patients under 40 years old and over 40 years old are exhibited in Table 3. The tumor distribution pattern, interstitial inflammatory infiltration (Degree and Distribution pattern), concomitant or non concomitant breast tumor and lymph node metastasis between patients younger and older than 40 years old were various (P < 0.05). The 2 groups are basically the same in the lymphovascular embolization and the prognosis (P > 0.05).
Comparing clinicopathological features between patients younger and older than 40 years old.

Discussion
The nipple is the cylindrical part protruding from the center of the breast, and it is also the most important part of the whole breast tissue. The most important role is to breastfeed and raise the next generation, which will also increase the physical beauty. The stimulation of sexual life to the breast will cause increased sexual tension. Nipple disease will inevitably affect children’s feeding, physical beauty, and harmonious sexual life. This effect is particularly evident for young women. In this article, we discuss Paget’s disease of the nipple, especially the pathological manifestations of young women, to provide a basis for the prevention and treatment of nipple disease in young women.
The age of common onset of Paget’s disease (PD) of the nipple region of patients is middle-aged and elderly, and patients with PD younger than 40 years old are rare. In this article, we report 8 patients with PD younger than 40 years old for the first time. The first symptoms, physical examination and treatment methods are mainly consistent with the literature, and young patients and middle-aged and elderly patients are basically the same.12 -16 There is no special attention and treatment for nipple retention in young women, nor can it meet the needs of young women’s beauty and China’s current 3-child policy.
Some researchers believe that PD cases have a tall nested tumor cell distribution pattern, which may be related to the infiltration and metastasis of patients. 11 In this paper, we pay attention to the tumor distribution pattern of young patients. The results show that 8 patients had no tall nest pattern, while 20% of the middle-aged and elderly patients in the control group had a tall nest pattern. It seems that the tumor pattern of young patients belongs to the type that is not easy to metastasize. This study is limited by the small number of samples. The author believes that the discussion in this regard needs to be strengthened.
The proliferation of the tumor perinest epidermis in middle-aged and elderly PD patients is significantly higher than that in young female patients. Is this conducive to the absorption of local drugs? Further research is needed to clarify this. This study found that the number and degree of inflammatory cell infiltration around tumors in young patients are lower than those in middle-aged and elderly patients. Does this suggest that young patients benefit less from the immunomodulator imiquimod? 17 The proliferation of blood vessels in the dermis under Paget cells is obvious, consistent with literature reports. 18 Does it mean that the effect of local application of antitumor angiogenesis drugs Bevacizumab will be better?
Paget cells have a large volume, rich cytoplasm, large nucleus, obvious nucleolus, occasional mitotic images, and varying degrees of pigmentation, which are easily confused with malignant melanoma. 19 Tumor cells express CK7 and CK8/18 but do not express CK5/6, p63, and HMB45, which can be differentiated from Paget-like melanoma in situ, squamous cell carcinoma in situ and Paget-like reticulohistiocytosis.20 -22 The characteristic tumor cells of PD patients are the same in young patients and middle-aged and elderly patients. Therefore, there is no difference between them in pathological diagnosis and differential diagnosis.
Most patients have malignant lesions of the deep breast tissue, namely, DCIS, with or without invasion. This makes it more difficult to save the nipples of young patients. Fortunately, Amin et al 23 found by whole-exome sequencing that frequent mutations in chromatin remodeling genes in mammary and extramammary PD and mammary PD and underlying breast ductal carcinomas represent independent oncogenic events. Does this mean that Paget’s disease of the breast and the underlying cancer can be treated separately? And can breast PD also be treated with local drugs such as vulva PD? 24 25% of patients with intraductal cancer can appropriately narrow the scope of surgery, and 12.5% of women with no deep breast cancer can even make efforts to preserve the nipple. The prognosis of young PD patients is basically the same as that of middle-aged and elderly PD patients. The in-depth study of young PD patients and the formation of personalized nipple PD treatment are bound to be more helpful in improving the quality of life of young patients.25,26
Conclusion
PD is a malignant skin lesion in the nipple areola area that is prone to occur in older women, and young patients can also be involved. The first symptom, physical examination, Paget cell morphology, and immunohistochemical marks were the same in different age groups. Although the tumor distribution, infiltration of interstitial inflammatory cells, and hyperplasia of the peripheral epithelium between younger and middle-aged and elderly patients were diverse in this study, more cases are needed to provide a basis for better/early diagnosis and potential for histology directed therapeutic interventions in future studies.
Footnotes
Acknowledgements
The authors would like to thank Su Qianyao of Chengdu Third People’s Hospital for providing technical support.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Chunyan Lv, Xiankui Cheng, and Yan Tang conceived the study, participated in its design and coordination, and drafted the manuscript. Li Liu and Jian Cai collected clinical samples and background. Tao Lei and Zhong-Yue Guo performed the immunohistochemical evaluation.
