Abstract
Introduction:
Invasive Breast carcinoma—No special type (NST) is the most common breast malignancy accounting for 95% of breast cancers. Study of predictive and prognostic immunohistochemical markers estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (Her2neu) expression are crucial for treatment planning.
Materials and Methods:
In the present study we studied the hormonal profile in 303 sporadic breast cancers and BRCA1 protein expression in these patients along with its clinico-pathological correlation.
Results:
In our patient population, Triple negative Breast carcinoma (TNBC) (104/303; 34.3%) was the most common luminal subtype followed by Luminal A 74/303; 24.4%), Her2 enriched (65/303; 21.5%), and Luminal B (60/303; 19.8%) respectively. This contrasts with many western studies which commonly report Luminal A being the largest subgroup. BRCA1 protein loss was more prominently seen in TNBC (64/104;61.5%) highlighting the possibility that high grade tumors are more susceptible to some epigenetic modifications leading to higher likelihood of loss of BRCA1 protein.
Conclusion:
Hence, we conclude that like hereditary cases of breast carcinoma with BRCA1 mutation; BRCA1 loss is also more likely in sporadic TNBC cases.
Introduction
According to American cancer society, breast carcinoma accounts for 24.2% of female carcinoma worldwide. 1 In Asian countries including India, breast carcinoma has become leading cause of mortality in females surpassing cervical carcinoma with annual rise in incidence of 0.5% to 2% per annum. 2 There is wide diversity in breast carcinoma morphology, immunohistochemistry (IHC), histopathology, molecular subtypes, clinical features, prognosis and outcome. Few well described and studied prognostic and predictive factors for breast carcinoma are tumor size, histological type, grade and nodal status. Important immunohistochemical predictive and prognostic markers include estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2(Her2neu). 3 The Cancer Genome Atlas (TCGA) Network categorized breast carcinoma into 4 main subtypes, based on ER/PR/Her2neu/Ki67 expression as Luminal A and B subtypes, Her2neu enriched and Triple negative breast carcinoma (TNBC). 4 Patients with TNBC do not benefit from either hormonal or trastuzumab based targeted therapy because there is loss of specific ER/PR or Her2neu receptor expression respectively.
Breast Cancer Susceptibility Gene 1 (BRCA 1) was first described in 1990 by Mary-Claire King’s laboratory at UC Berkeley. 5 BRCA 1 located on chromosome 17 is classic tumor suppressor gene, found in hereditary breast and ovarian malignancies. It is involved in DNA repair, homologous recombination, and transcription. BRCA1 is most associated with familial breast carcinomas but few studies have demonstrated the role of BRCA1 in causation of sporadic breast tumors too.6-9
In sporadic breast carcinomas, there is decreased expression of BRCA1 gene and protein compared to normal mammary epithelial cells. In majority of these cases, there is loss of BRCA1 nuclear protein, while around 19% of sporadic breast carcinomas show both nuclear and cytoplasmic BRCA1 protein loss. 10 Loss of heterozygosity of BRCA1 in sporadic breast carcinoma share genotypic and phenotypic features with familial breast carcinoma leading to the concept of “BRCAness.” 11
BRCA1 loss has been associated with increased sensitivity to drugs that induce cross links (platinum chemotherapy) and single and double stranded breaks (etoposide) in DNA. These single stranded breaks in DNA are repaired by base excision repair pathway in which Poly ADP- Ribose Polymerase1 (PARP1) is one of the major components, thus paving way for use of PARP inhibitors as targeted therapy in BRCA1 loss cases. 12
Due to young age of presentation and high prevalence of TNBC in Indian females with breast cancer, limited therapy options are available for them. BRCA1 loss in sporadic TNBC has been suggested in some studies. Hence, females with sporadic breast carcinoma with BRCA1 loss may benefit from cisplatin based chemotherapy along with PARP inhibitors.
With the above outline the objective of this study was to estimate BRCA1 status and co-relate it with hormonal profile in sporadic breast cancer patients in our patient population to identify females who may be benefited from PARP inhibitor therapy. BRCA1 genetic testing is expensive and a time consuming. In developing countries like India, where molecular diagnostics are beyond reach and paying capacity of many, we studied BRCA1 by immunohistochemistry (protein expression) in tissue sections.
Materials and Methods
We conducted the study for 1.5 years and enrolled 303 breast carcinoma cases. Various factors like age, laterality, size, necrosis, lymph node status, margins, Nottingham grade, DCIS, hormonal profile and BRCA1 immunohistochemistry were considered. Only females with no family history of breast cancer or ovarian cancers in first- or second-degree relatives were included after informed consent.
Formalin fixed paraffin embedded blocks were processed for hematoxylin and eosin staining. A section was selected for immunohistochemistry enclosing unremarkable ducts and tumor. Immunohistochemical staining was done on (3-aminopropyl triethoxysilane) coated slides using ER (Flex polyclonal rabbit -a Hu ER alpha, Clone EP1, RTU (DAKO AS/AS+), PR (Flex Monoclonal Mo a Hu PR, Clone PgR636, RTU (DAKO AS/AS+), Her2neu (polyclonal rabbit a Hu c-erb2 oncoprotein, RTU (DAKO AS/AS+) and BRCA1 (polyclonal rabbit,RTU (AR345-5R) (BioGenex).
Immunostaining
Deparaffinization of sections was done in xylene followed by rehydration through graded alcohol and distilled water. Antigen retrieval was done in pressure cooker in citrate buffer for 15 minutes at 120°C and cooling was done at room temperature. Peroxidase activity blocking was done by using 3% H2O2 for 10 minutes. Sections were treated with protein block for 10 minutes. Slides were incubated overnight with primary antibody at 4°C in moist chamber followed by washing with TRIS buffer (pH 7.6) next morning. Slides were then incubated with polymer for 30 minutes and further with biotinylated secondary antibody for 30 minutes. Diaminobenzidine was used as chromogen.
Immunohistochemistry interpretation
ER and PR nuclear and Her2neu membranous expression was interpreted according to ASCO-CAP guidelines 2018. 13 BRCA1 expression of 25% or less either nuclear or both cytoplasmic and nuclear in tumor cells was considered as loss of expression for BRCA1.
SPSS (Statistical Package for Social Sciences) Version 21.0 statistical Analysis Software was used for statistical analysis. Significance was considered to be P value <.05.
Results
Clinicopathological parameters
Clinical age of presentation varied from 25 to 90 years with median age of 48.12 ± 11.36 years. Most of the females belonged to 41 to 50 years age group (38.9%) followed by 31 to 40 years (23.1%), 51 to 60 years (21.5%), >60 years (10.9%) and ⩽30 years (5.6%) (Table 1). Right sided (53.1%) breast carcinoma was more common than left (46.9%). Size of the tumor ranged from 0.8 to 15 cm with mean size of 3.98 ± 2.49 cm. Most of the tumors were high grade (Nottingham Grade II and III), that is, 74.9% (Table 2). Sixteen cases had positive surgical resection margin that constituted 7.3% of all cases. Necrosis, LVI/PNI, DCIS were present in 59.9%, 20.2%, 26.1% respectively. ⩾3 lymph nodes were positive in 47.4% and 18.6% cases had tumor size >5 cm.
Age wise distribution (n = 303).

Mean age: 48.12 ± 11.36 (range: 25-90) years.
Characteristics of patient population.

Hormonal profile and BRCA1 protein expression
Among the hormonal profile, TNBC was most common Luminal subtype (104/303; 34.3%) (Figure 1) followed by Luminal A (74/303; 24.4%) (Figure 2), Her2 enriched (65/303; 21.5%) (Figure 3), and Luminal B (60/303; 19.8%) (Figure 4) respectively (Table 2).
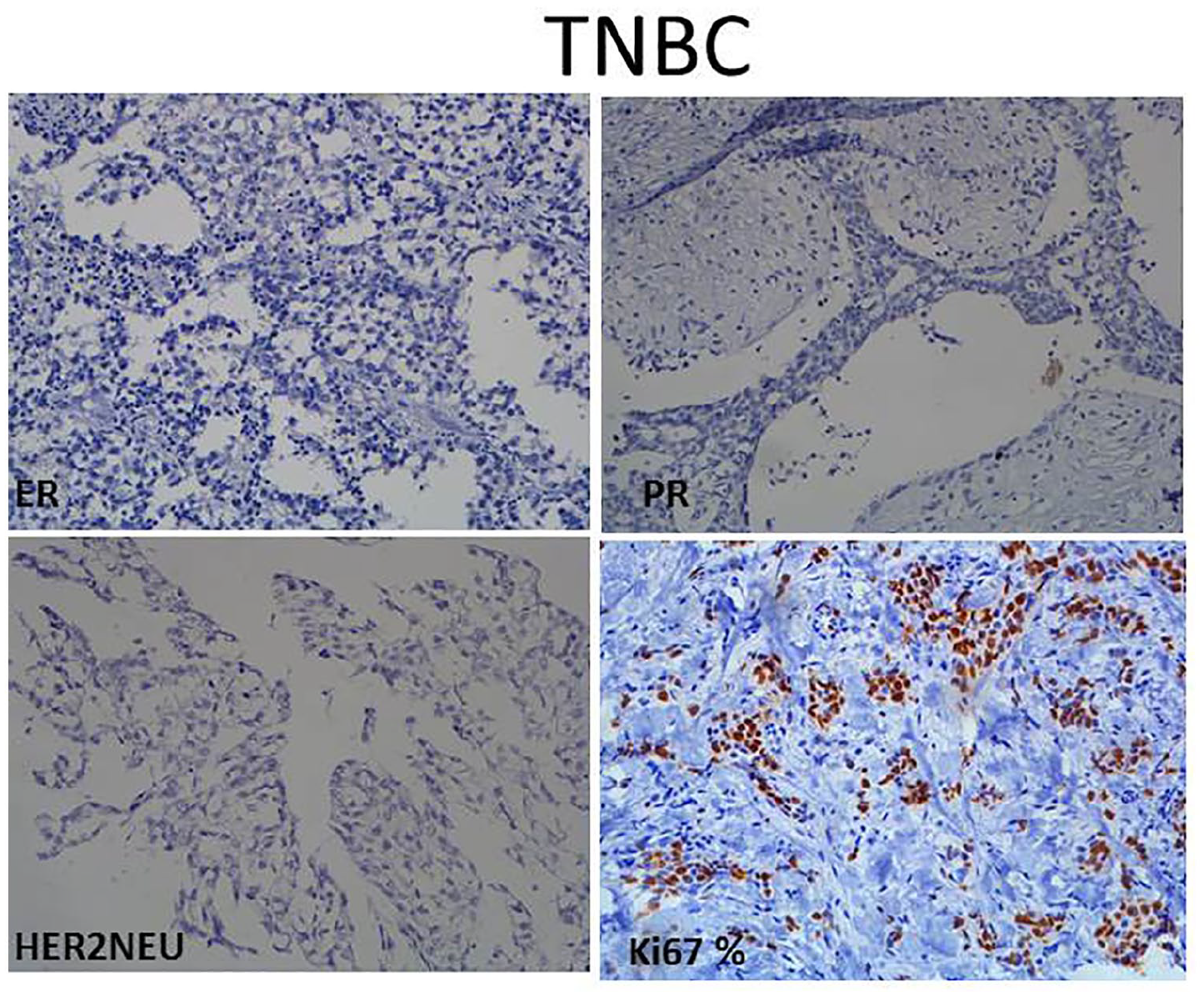
(200 X) Triple negative breast carcinoma (TNBC) subtype showing ER, PR, Her2neu negative expression with Ki67 60%.
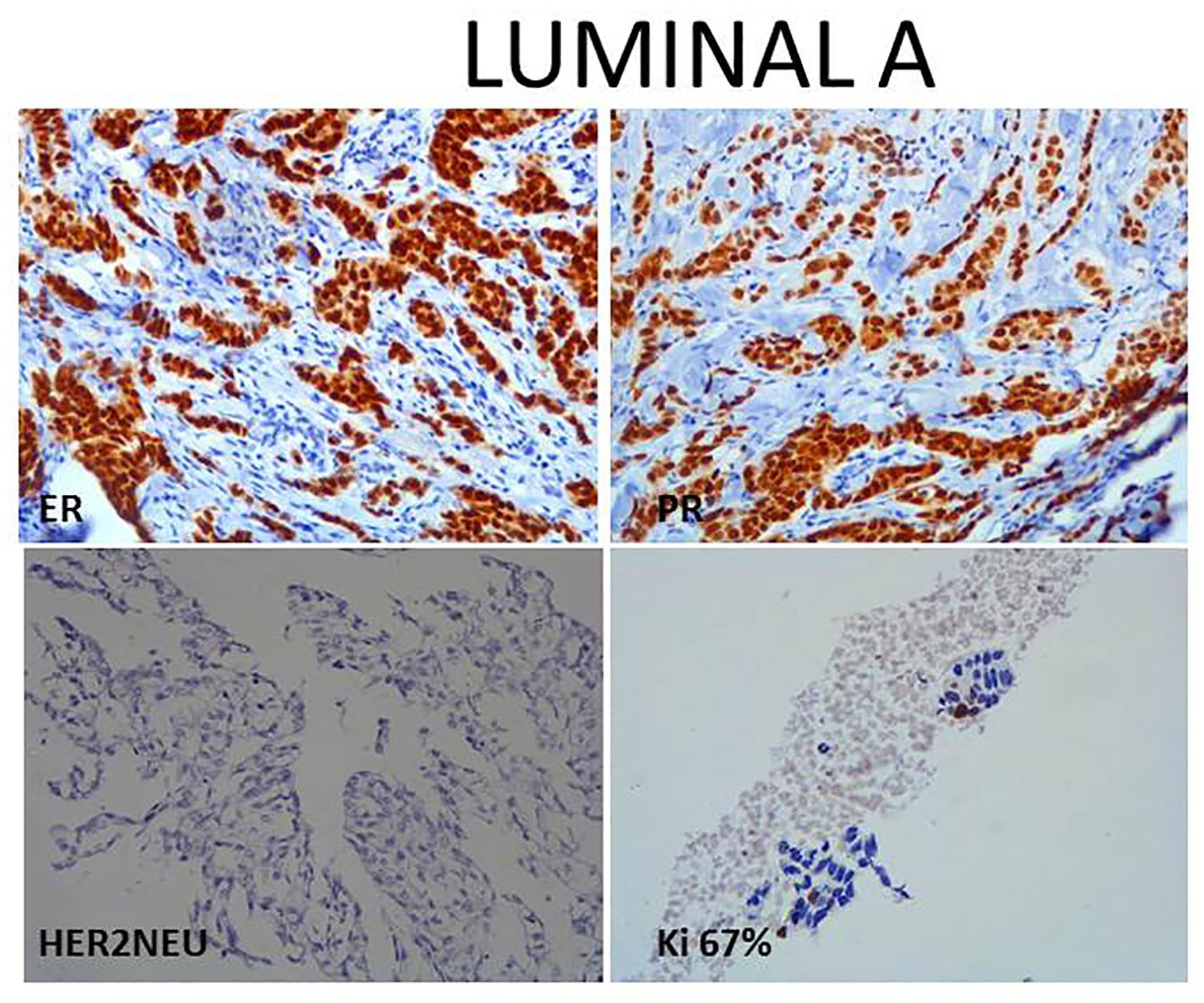
(200 X) Luminal A subtype showing ER, PR strong nuclear positivity, Her2neu negative expression and Ki67 <5%.
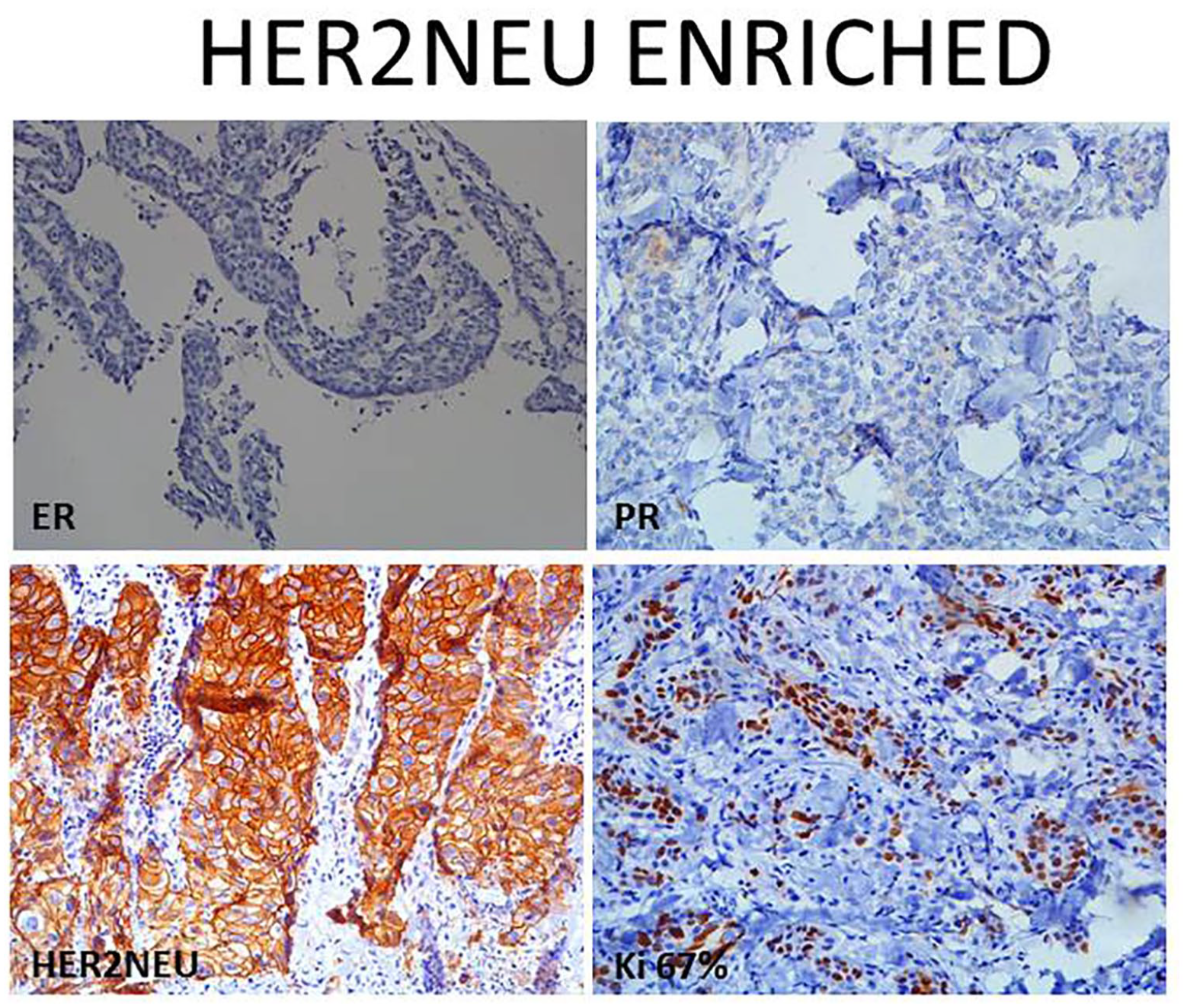
(200 X) Her2neu enriched subtype showing ER, PR negative expression, Her2neu intense complete membranous expression and Ki67 55%.
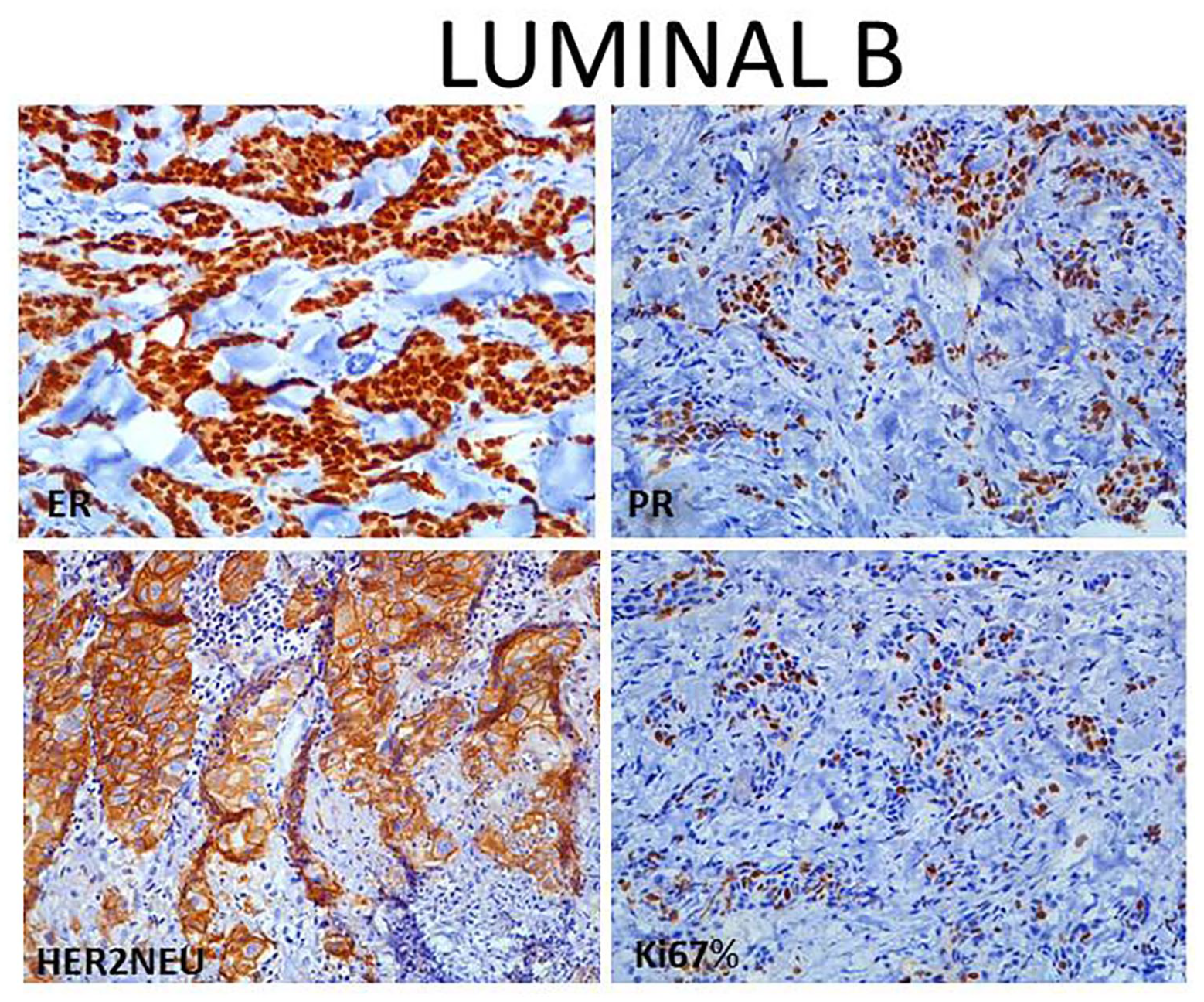
(200 X) Luminal B subtype showing ER, PR strong nuclear positive expression, Her2neu intense complete membranous expression and Ki67 30%.
In the adjacent terminal ducts BRCA1 was strongly expressed in the lining ductal cells (internal control) (Figure 5A). Loss of BRCA1 protein expression was seen in 146 cases out of 303 evaluated (Figure 5B), out of which maximum loss was seen in TNBC 43.8% (64/146) of the total cases displaying BRCA1 protein loss and the association was statistically significant (P = .003) (Table 3).
Association of BRCA (IHC) expression and hormonal profile.

χ² = 13.926(df = 3); P = .003.

BRCA1 expression seen in (A) (200 X) Normal TDLU, (B) (200X) Faint cytoplasmic expression in <25% of cells, and (C) (400X) Strong nuclear and cytoplasmic expression in >75% of tumor cells.
Discussion and Conclusion
Globally breast carcinoma is one of the leading causes of cancer related mortality among women. Hormonal profile of breast carcinoma plays an important role, not only in determining prognosis of the patients but also in treatment planning. BRCA1 is a tumor suppressor gene usually associated with inherited breast cancers. 14 We intend to identify BRCA1 loss in sporadic breast carcinoma and co-relate it with hormonal profile to detect high grade tumors and manage them accordingly.
Breast carcinoma has been reported a decade earlier in Indian females compared to their western counterparts. Multicenter study carried out by Leong et al 15 reported that peak age of detection of breast carcinoma in Asian females is 45 to 50 years of age, which is a decade younger from western females, that is, 55 to 60 years. Our findings are in coherence of their observation and bulk of our study population belonged to age group of 41 to 50 years (38.9%; 118/303) with mean age of 48.12 ± 11.36 years (Table 1). Involvement of right breast (53.1%) was more as compared to left (46.9%) in the received gross specimens (Table 2). This is in contrary to Tiwari et al 16 where they reported left breast (68.6%) involved more than right.
Majority of our patients (74.9%) presented with high grade tumors (Grade II and grade III), bulk of which (56.1%) belonged to Grade II (Table 2). These findings are similar to the observations of Bal et al 17 where Grade II was maximum (57.1%). Hence, we confer that in our region females present a decade earlier, with higher grade compared to western population.
Multiple morphological and clinico-radiological features have been recognized which either predict or prognosticate in breast carcinoma patients. 18 We studied few of them which were namely surgical resection margin status, necrosis, lymph vascular/perineural invasion (LVI/PNI), ductal carcinoma in situ (DCIS), lymph nodes (LNs) and tumor size.
Sixteen cases have positive surgical resection margin that constituted 7.3% of all cases. Necrosis, LVI/PNI, DCIS were present in 59.9%, 20.2%, 26.1% respectively. ⩾3 lymph nodes were positive in 47.4% and 18.6% cases had tumor size >5 cm.
Contrary to published literature from West as well as from India where Luminal A is more predominant, our study shows that most common molecular subtype of breast carcinoma is TNBC (104/303; 34.3%).19,20 Unlike other luminal subtypes, TNBC is characterized by good initial response to chemotherapy, more aggressive clinical behavior, and lack of targeted therapy options.
Germline mutations BRCA1 has been extensively studied in breast cancer in young females and females with family history of breast and ovarian cancers.21-23 These studies have found that these cancers are usually of higher grade, aggressive and with higher triple negative hormonal profile. BRCA1 loss may also be seen in sporadic breast carcinoma as well and it may be due to silencing because of promoter methylation or downregulation of BRCA1.17,24,25 This subgroup of sporadic breast carcinoma is also associated with reduction in mRNA levels and protein expression. Bal et al 17 found reduced expression of BRCA1 on immunohistochemistry in 38% and absent or markedly reduced expression in 22% of cases of sporadic breast carcinoma respectively in North Indian population showing the role of BRCA1 in sporadic breast carcinomas. In our study we found that loss of protein expression (both nuclear and cytoplasmic) was seen in 48.2% cases, that is, 146/303 (Figure 3) (Table 2) and statistically significant correlation was seen between the BRCA1 protein loss and hormonal profile where BRCA1 loss was more frequent in TNBC 61.5% (64/104) of total TNBC cases (Table 3). This highlights that similar to hereditary cases of breast carcinoma with BRCA1 mutation; sporadic cases too were frequently ER/PR/Her2neu negative (TNBC). The results are in agreement with study of Bal et al 17 , they concluded that BRCA1 negative tumors are more frequently triple negative. These findings designate that high grade tumors usually undergo somatic mutations or epigenetic silencing of BRCA1 protein and hence BRCA1 protein loss on immunohistochemistry. These sporadic breast carcinoma cases can therefore be benefited from PARP inhibitor and/or cisplatin-based therapy as recommended in hereditary breast carcinomas with BRCA mutations.
To conclude, role of BRCA1 gene in sporadic breast carcinoma needs to be further studied in larger breast cancer population. The loss of BRCA1 protein expression shows statistical significance with TNBC (ER/PR/Her2neu negative) cases similar to hereditary breast carcinomas showing BRCA1 mutations. Loss of BRCA1 in sporadic breast carcinoma implies that therapeutics like PARP inhibitors used in targeting BRCA1 in hereditary breast carcinoma might also be applicable to sporadic breast carcinomas. However our observations must be further co-related with molecular expression (under study by the author) or loss of BRCA1 which might be a limitation of the present study.
Footnotes
Acknowledgements
The current pandemic has taken many away from us. Through this article we wish to pay our respects to Late Prof. Raj Mehrotra, Ex Professor and Head, Department of Pathology, KGMU, Lucknow, India who was our mentor. We are thankful to King George’s Medical University for providing the infrastructure to perform our work. We are also thankful to our Immunohistochemistry and histopathology laboratory staff for their support.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
