Abstract
Trypanosomes are single-celled protozoa that cause severe diseases in both humans and livestock in sub-Saharan African countries. The disease in the West African Dwarf (WAD) sheep is often neglected due to the issue of trypanotolerance. The current study is aimed to evaluate some biochemical changes in this breed that may modify the understanding of trypanotolerance. Fifteen WAD sheep were assigned into 3 groups (A, B, and C). Baseline (day 0) values of the parameters assayed were obtained before groups A and B were infected with Trypanosoma brucei brucei and Trypanosoma congolense, respectively, by intraperitoneal inoculation with 106 trypanosomes per animal. Standard procedures using Quimica Clinica Applicada (Spain) and Randox (UK) test kits were used to evaluate serum levels of AST, ALT, ALP, total protein, albumin, total cholesterol, urea, and creatinine on days 0, 14, 28, 42, 56, and 70 post infection. The infections caused sustained pyrexia, hypoproteinaemia, hypocholesterolaemia, weight loss, hepatitis, and mortalities although parasitaemia was greatly controlled especially in the T congolense infected rams. The findings suggest that the WAD rams are not just passive reservoirs of trypanosomes for human and animal infections, but experience active host-parasite interactions with huge price for resilience, biochemically.
Introduction
Trypanosomes are unicellular parasitic protozoa belonging to the Trypanosoma Genus and Family Trypanosomatidae. 1 The parasites live and thrive in the blood and other body fluids of vertebrate hosts, and some of them cause disease known as trypanosomiasis, leading to significant morbidity and mortality in both man and animals with enormous economic losses. 2 Trypanosoma brucei rhodesiense and Trypanosoma brucei gambianse cause acute (haemolymphatic) and chronic (meningoencephalitic) forms of human African trypanosomiasis (HAT), respectively, whereas Trypanosoma brucei brucei, the third member of the Trypanosoma brucei group together with Trypanosoma congolense and Trypanosoma vivax are the causative agents of ‘Nagana’ or African animal trypanosomiasis (AAT) in a wide range of livestock. 3
The Djallonke or West African Dwarf (WAD) sheep, as they are often referred to, inhabit the area south of latitude 14° N including the coastal areas of west and central Africa. These include Nigeria, Dahomey, Ghana, Ivory Coast, Guinea, Senegal, Cameroon, Gabon, Congo, and Southern Mali. They are also found in Angola and Botswana. The WAD sheep are known for their adaptation to tropical hot and humid environment of West Africa, and they are also considered tolerant to trypanosomiasis.4,5 The WAD sheep (Djallonke) and goats, as well as the Taurine cattle (Bos taurus or N’Dama), which entered Africa from the near east around 5000 BCE and 7000 BCE, respectively, developed innate tolerance to African animal trypanosomosis (AAT), probably as a result of natural selection pressures.6-8 The innate ability of these livestock breeds to survive and remain productive under AAT challenge, with very low mortality and without the use of trypanocidal drugs, is referred to as trypanotolerance. It was estimated that approximately 32% of sheep and 47% of goats in West and Central Africa are trypanotolerant, and this is so because the WAD breeds are predominant in these regions. 9 In N’Dama cattle specifically, trypanotolerance was shown as a better ability to control both parasitaemia and, more importantly, anaemia, and this permits the host to remain productive under disease challenge. 7
Trypanosomiasis is known to cause anaemia, hypoproteinemia, leukocytosis, immunosuppression, hypoglycemia, and changes in serum enzyme and cholesterol levels. 10 However, these changes have not been well evaluated in the WAD sheep and goats and N’Dama cattle breeds partly because of the presumptions associated with trypanotolerance.
The present study evaluated some biochemical changes in WAD rams infected separately with T brucei brucei and T congolense to elucidate what trypanotolerance may preclude and widen its understanding or limit overemphasis as well as highlight targets for therapeutic intervention in the so-called trypanotolerant breeds.
Materials and Methods
Animals
Fifteen sexually mature WAD rams, weighing between 7 and 13 kg, and aged 1 to 1½ years were used for the study. They were procured from local breeders. They were allowed to acclimatize for 3 weeks, during which they were dewormed with albendazole at 10 mg/kg body weight (b.w.) per os and ivermectin at 1 mL/50 kg b.w. By buffy coat examination, those already infected with trypanosomes or any other haemoparasite were screened out and excluded from the study. The rams were thereafter assigned into 3 groups (A, B, & C) of 5 rams each. Group A rams were infected with T brucei brucei, group B rams were infected with T congolense and group C served as the uninfected control. The trypanosomes were sourced from the Nigerian Institute for Trypanosome Research (NITR, Nigeria). Infection of groups A and B was by intraperitoneal injection of 106 trypanosomes per head. The rams were placed on fresh forage and drinking water ad libitum. Baseline values of the mean body weight, rectal temperature, and serum biochemical assays were determined before infecting groups A and B. The infected animals and the control were monitored for 70 days.
Ethical approval
The study was approved by the Institutional Animal Care and Use Committee (IACUC) of the Faculty of Veterinary Medicine, University of Nigeria.
Parasitaemia
Blood samples of the infected groups were examined for parasitaemia daily until parasitaemia was established, then the level of parasitaemia were measured on days 7, 14, 21, 28, 35, 42, 56, and 70 post infection (PI) by buffy coat scoring method. 11
Biochemical analyses
On days 0, 14, 28, 42, 56, and 70 PI, 3 mL of blood was collected from the external jugular vein of the rams into clean tubes and allowed to clot followed by centrifugation at 3000 rpm for 10 minutes to obtain clear serum samples for biochemical analyses.
The biochemical parameters assayed in serum were aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP) activity, total protein, albumin, total cholesterol, urea, and creatinine. All the biochemical assays were done using Diatek Biochemistry Analyzer (Wuxi Hiwell Diatek Instruments Co Ltd, China). The test kits used for the assay of serum AST, ALT, ALP, total cholesterol, and urea were sourced from Quimica Clinica Applicada (QCA), Spain, while the ones used for the assay of serum levels of total protein, albumin, and creatinine were sourced from Randox Laboratories Ltd, UK.
The serum AST and ALT activity were assayed based on the Reitman-Frankel colorimetric method, 12 while assay of the ALP activity was based on the phenolphthalein monophosphate method.13-15 The serum total protein levels were determined by the direct biuret method, 16 while the serum albumin levels were determined based on the bromocresol green method.17,18 The serum globulin level for each sample was calculated by subtracting the serum albumin level from total protein level. The serum total cholesterol was determined based on the enzymatic colorimetric method, 19 while the serum level urea was determined based on the Berthelot-Searcy method. 20
Histopathology
Rams that died due to the infection were thoroughly necropsied and tissue samples were generously collected and fixed in 10% neutral buffered formalin. The tissues were routinely processed, sectioned at 5 µm thickness and stained with haematoxylin and eosin (H&E). However, focus was mainly on the liver tissue because it is the site for the synthesis of most the biochemical elements evaluated in the study.
Statistics
Data generated from the study were subjected to 1-way analysis of variance using SPSS version 21. The level of parasitaemia was analysed by Student t test. The variant means were separated post hoc using the least significant difference (LSD) method. The level of significance was accepted at probability P < .05.
Results
Physiological parameters and parasitaemia
There was no significant (P > .05) changes in the mean body weights of the rams except on day 70 PI on which T brucei brucei infected rams had significantly (P < .05) lower mean body weight compared with the control (Figure 1).
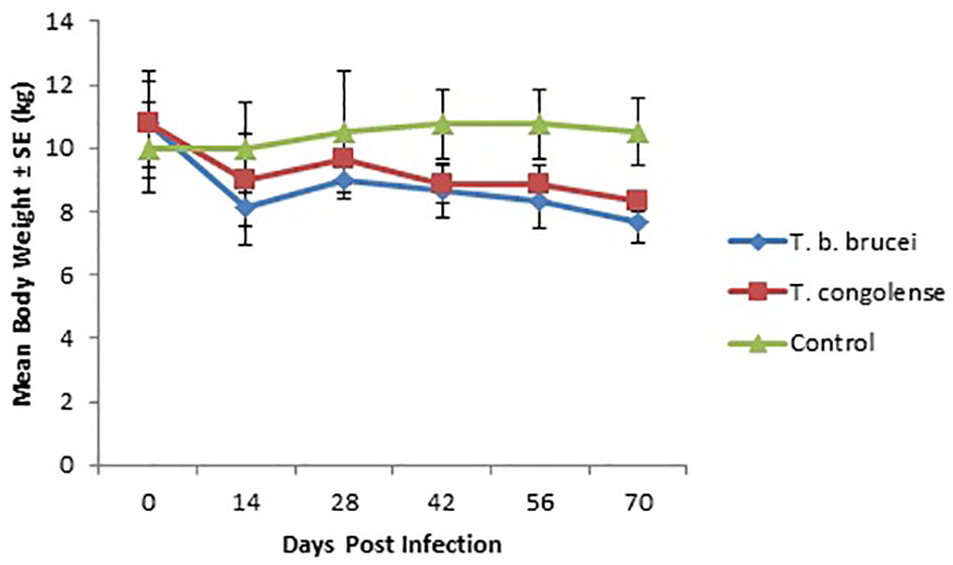
Graph showing the mean body weight (kg) of WAD rams infected with T brucei brucei and T congolense.
The rectal temperature of the infected groups were significantly (P < .05) higher than the control from day 14 to the end of the experiment (day 70), with T congolense producing relatively higher temperature elevations than T brucei brucei (Figure 2).

Graph showing the mean rectal temperature (°C) of WAD rams infected with T brucei brucei and T congolense.
Parasitaemia was first detected on day 5 PI in both T brucei brucei and T congolense infected rams and by day 6 PI all the infected rams had become parasitaemic. Three peaks of parasitaemia were observed in T brucei brucei on days 14, 35, and 56 PI. T congolense peaked on day 14 PI after which no sharp fluctuations were observed. Also, T brucei brucei produced significantly (P < .05) higher levels of parasitaemia compared with T. congolense especially on the peak days (Figure 3).
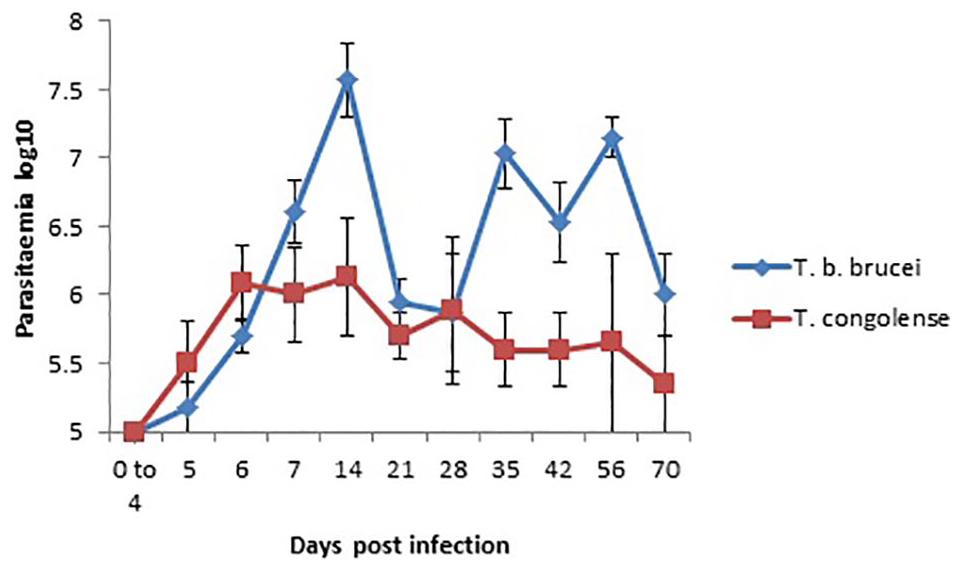
Graph showing the mean log10 parasitaemia of WAD rams infected with T brucei brucei and T congolense.
Biochemical perturbations in T brucei brucei and T congolense infections in the rams
There were significantly (P < .05) lower levels of AST activity in the infected groups on days 14, 28, 42, and 70 PI compared with the control (Figure 4). Serum activity of ALT was significantly (P < .05) higher in T brucei brucei infected rams than the control on day 42 PI while T congolense was significantly (P < .05) lower than the control on day 56 PI (Figure 5). No significant (P > .05) variations were observed in the serum activity of ALP in both the infected rams and uninfected control (Figure 6).

Graph showing the mean serum AST activity (IU/L) of WAD rams infected with T brucei brucei and T congolense.

Graph showing the mean serum ALT activity (IU/L) of WAD rams infected with T brucei brucei and T congolense.
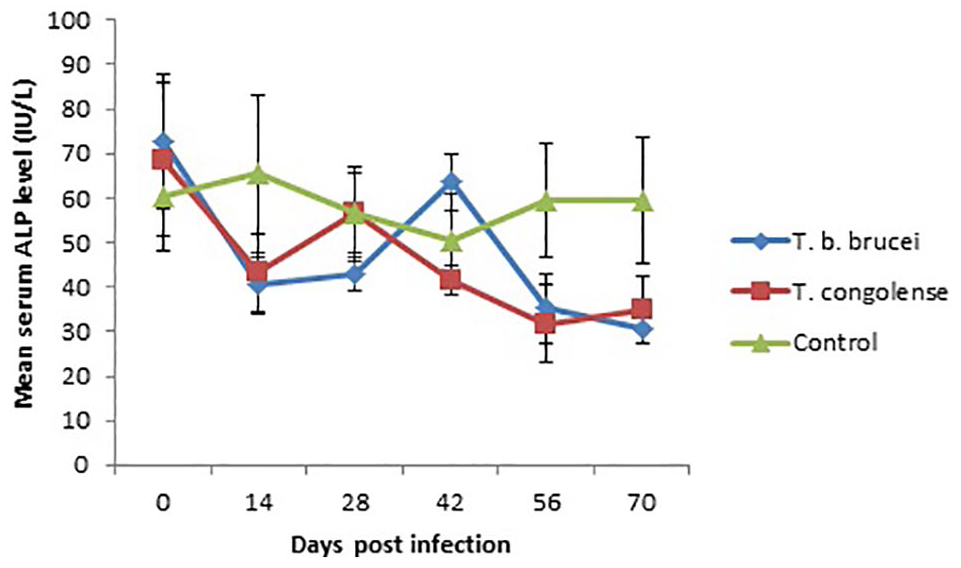
Graph showing the mean serum ALP activity (IU/L) of WAD rams infected with T brucei brucei and T congolense.
Serum total protein level was significantly (P < .05) higher in T congolense infected rams than in T brucei brucei infected rams on day 42 PI and was also significantly (P < .05) higher than both T brucei brucei infected rams and the uninfected control on day 70 PI, whereas T brucei brucei infected rams had significantly (P < .05) lower total protein level than the uninfected control on day 70 PI (Figure 7). Serum albumin levels of T brucei brucei infected rams were significantly (P < .05) lower than the uninfected control on days 14, 28, 42, 56, and 70 PI, while that of T congolense was significantly (P < .05) lower than the uninfected control on days 14, 28, 56, and 70 PI (Figure 8). Serum globulin level of T congolense infected rams was significantly (P < .05) higher than those of T brucei brucei infected rams as well as the uninfected control on days 42 and 70 PI (Figure 9).

Graph showing the mean serum total protein level (g/dL) of WAD rams infected with T brucei brucei and T congolense.

Graph showing the mean serum albumin level (g/dL) of WAD rams infected with T brucei brucei and T congolense.

Graph showing the mean serum globulin level (g/dL) of WAD rams infected with T brucei brucei and T congolense.
Serum total cholesterol level of T brucei brucei infected rams was significantly (P < .05) lower than the uninfected control on days 14, 28, 56, and 70 PI. While that of T congolense infected rams was significantly (P < .05) lower than the uninfected control on days 14, 56, and 70 PI (Figure 10).

Graph showing the mean serum total cholesterol level (mg/dL) of WAD rams infected with T brucei brucei and T congolense.
Serum urea level of T congolense infected rams were significantly (P < .05) higher than T brucei brucei infected rams on day 28 PI and significantly (P < .05) higher than the uninfected control on days 28 and 70 PI (Figure 11). There were no significant (P > .05) variations in the serum creatinine levels of the infected and uninfected rams excepts on day 28 PI on which the serum creatinine level of T congolense infected rams was significantly higher than that of T brucei brucei infected rams (Figure 12).
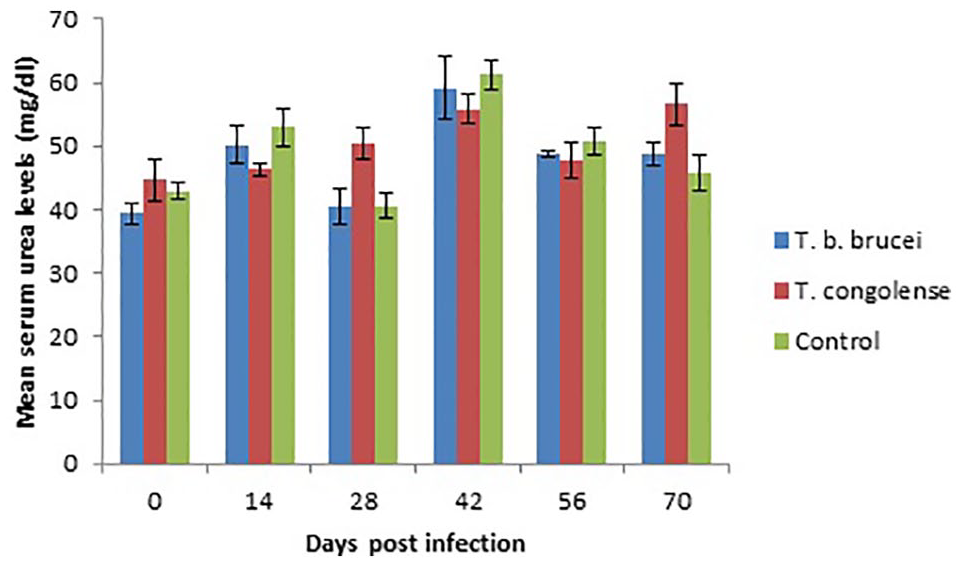
Bar graph showing the mean serum urea level (mg/dL) of WAD rams infected with T brucei brucei and T congolense.

Bar graph showing the mean serum creatinine level (mg/dL) of WAD rams infected with T brucei brucei and T congolense.
Histopathology
Mortality occurred in the T congolense infected rams on days 14 and 66 PI and in T brucei brucei infected rams on days 14 and 50 PI. The liver tissue of T brucei brucei infected rams showed severe multifocal random hepatocellular necrosis in the form of uncapsulated microgranulomas with massive infiltration of mononuclear inflammatory cells (Figure 13). The liver of the T congolense infected rams also showed mild coagulative hepatocellular necrosis and perivascular inflammation in the portal area (Figure 14). There were other organs with significant lesions but they were not relevant to the focus of the research.

Liver section of WAD ram infected with T brucei brucei showing large focal uncapsulated microgranuloma comprising of necrotised hepatocytes and infiltrated mononuclear inflammatory cells. H&E stain ×100.
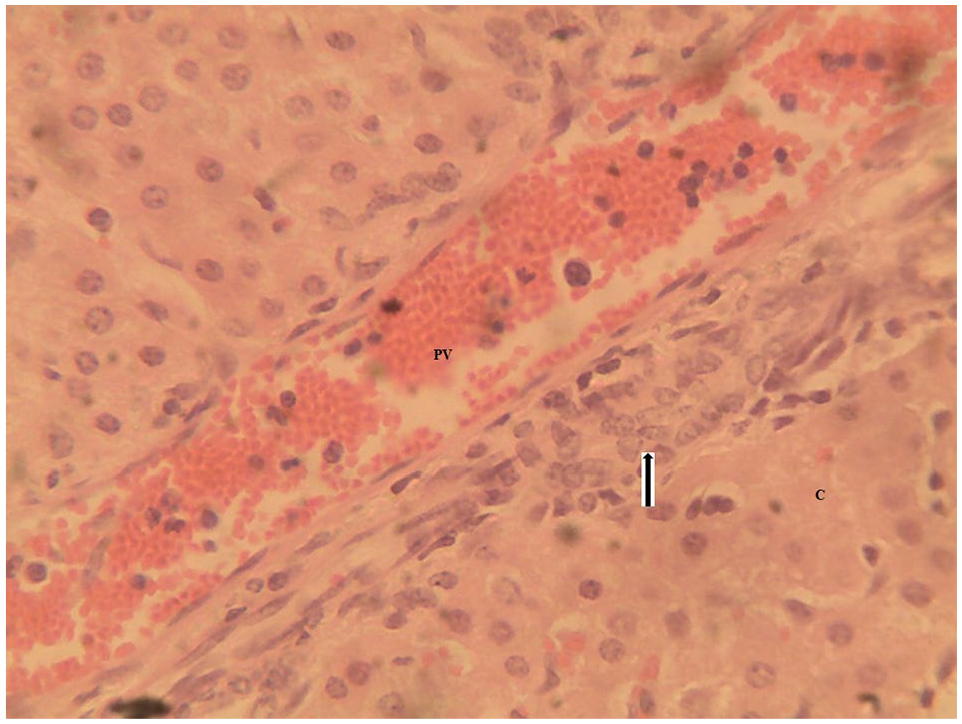
Liver section of WAD ram infected with T congolense showing mild infiltration of mononuclear inflammatory cells (arrow) around the portal vein (PV) with mild coagulative hepatocellular necrosis (C). H&E stain ×400.
Discussion
From the results of our study, the body weight of the infected rams did not drop significantly and this agrees with the reported observations in trypanotolerant breeds.6-8 However, the significant drop in the body weight on day 70 PI in the T brucei brucei infected group may suggest that weight loss may eventually occur in tolerant animals chronically infected with highly virulent strains of T brucei brucei.
The significant rise in rectal temperature in the infected animals is consistent with trypanosome infection and T congolense appears to produce higher level of pyrexia than T brucei brucei probably due to its affinity for the microvasculatures of the brain. 21
The prepatent period of 5 to 6 days for intraperitoneal inoculation of T brucei brucei and T congolense in our study compares well with experimental infections in mice in which the mean prepatent period was 3.8 days for T brucei brucei and 6.5 days for T congolense. 22 Also, in our study, T brucei brucei produced higher level (4.0 × 107 trypanosomes/mL of blood) and more fluctuating peaks of parasitaemia than T congolense (1.3 × 106 trypanosomes/mL of blood). The understanding of trypanotolerance includes the ability of the tolerant breeds to control parasitaemia. This is very evident in our study because very much higher levels of parasitaemia, up to 1 × 109 trypanosomes/mL of blood, are seen in rodent models and other very susceptible animals. 23 It appears that parasitaemia was better controlled in the T congolense infected WAD rams than in T brucei brucei infected ones. Variations in parasitaemia within same species or breed of animals may depend on the strain and pathogenicity of the trypanosome isolate.
Significant reductions were observed in the serum AST activity of the infected rams. Also, significant reduction was observed in the serum ALT activity of T congolense infected rams on day 56 PI, but that of T brucei brucei was significantly higher than the control on day 42 PI. Furthermore, no variations were seen in the serum ALP levels of the infected rams. These finding differ moderately from serum enzymes activity reported in other susceptible animals. Increases in the serum activity of AST, ALT, and ALP have been reported in T brucei brucei infection, and were attributed to liver injury caused by the parasite. However, in T congolense infection, the serum enzymes activity remained unchanged.10,24 Reductions in serum AST and ALT observed in our study may be attributed to inhibition of their cofactor (vitamin B6) following losses of total protein and reductions in hepatic synthesis due to hypoproteinemia. 25 Although the significant increase in ALT in T brucei brucei infected rams on day 42 PI may be due to liver damage induced by the tissue invasive nature of the parasite, this did not affect serum AST probably because AST is a less specific marker for liver damage and has a shorter half-life. 26
Total proteins and gamma globulins increase while serum albumin decreases in several trypanosomes infections. 27 In the present study, serum total protein and globulin levels significantly increased only in the T congolense infected rams, but serum total protein level of T brucei brucei infected rams was significantly lower than those of both T congolense infected rams and the uninfected control. Decrease in serum total protein in T brucei brucei infection has also been reported in boars. 28 However, significant decrease in serum albumin level in the infected groups in our study agrees with the findings in other susceptible species. 27 The degree of hypoalbuminaemia was related to the level of parasitaemia and/or severity of the disease and occurrence of haemodilution. 29
Significant reductions in serum total cholesterol levels were observed in the infected rams. This has also been reported in susceptible goat breeds. 30 It has been reported that the blood-stream forms of trypanosomes are unable to synthesize cholesterol although they require it for growth and synthesis of their membranes and this could contribute to lowering of the serum levels of lipids and cholesterol in infected hosts. 31
Serum urea level was significantly higher in T congolense infected rams on days 28 and 70 PI. Also, serum creatinine level of the infected groups did not vary significantly with the uninfected control. High urea levels were reported in cattle infected with T congolense and it occurred early when parasitaemia was high and returned to normal level in chronic trypanosomiasis. 32 It appears that excessive protein catabolism caused by fever contributes to increased blood urea nitrogen. In the present study, both serum total protein and pyrexia were more pronounced in the T congolense infected rams. Moreover, trypanosome infection in the WAD sheep may not be associated with significant muscle or kidney damage that may increase serum creatinine level as reported in the more susceptible species. 24
The histopathological changes in the liver in our study, especially in the T brucei brucei infected group may be responsible for the significantly higher level of serum ALT activity observed in this group. The liver lesions may have been triggered by massive invasion of trypanosomes into the liver parenchyma with attendant inflammatory response and hepatocellular necrosis.
Conclusions
The results of the present study revealed significant clinical and biochemical perturbations in the so-called trypanotolerant WAD rams ranging from pyrexia, hypoproteinaemia, and hypocholesterolaemia to weight loss, liver damage, and death. These findings suggest that the WAD rams are not passive reservoirs of trypanosomes for human and animal infections,33,34 but also suffer significantly from the infection where the disease surveillance, control of the insect vector, as well as therapeutic interventions are neglected while trypanotolerance is overemphasized.
Footnotes
Acknowledgements
The authors are grateful to the management of the Nigerian Institute for Trypanosome Research (NITR), Kaduna, Nigeria, for generously donating the trypanosome isolates used.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Tertiary Education Trust Fund (TETFund).
Ref. No.: TETFUND/DESS/UNI/NSUKKA/2018/RP/VOL. I
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
D.C.A., V.S.O.S., and J.I.I. conceived and designed the experiments. D.C.A. and J.I.I. performed the laboratory work, analysed the data, and wrote the first draft of the manuscript. D.C.A., V.S.O.S., and J.I.I. contributed to the writing of the manuscript, agreed with manuscript results and conclusions, jointly developed the structure and arguments for the article, made critical revisions and approved final version, and reviewed and approved the final manuscript.
