Abstract
Background and aims:
Endoscopic assessment of the regular arrangement of collecting venules (RAC) is a simple and reliable tool for predicting the absence of Helicobacter pylori (H. pylori) infection in the stomach, particularly in Asian populations. While initial studies in Western countries have yielded similar findings, RAC assessment has not yet been widely adopted in these settings. This study aims to evaluate the diagnostic accuracy of RAC in determining H. pylori status in a non-Asian population.
Methods:
This prospective, multicenter study was conducted in 12 hospitals across non-Asian countries. Patients with no history of H. pylori infection or eradication were included, regardless of proton pump inhibitor (PPI) use. All participants underwent high-definition upper endoscopy without magnification or virtual chromoendoscopy. Endoscopists were trained using a 20-image test to identify the RAC pattern. H. pylori status was determined by histology and/or immunohistochemistry.
Results:
A total of 648 patients were included, with an H. pylori infection prevalence of 34.7%. The RAC+ pattern was observed in 31.5% of patients, with no significant differences between those receiving PPI treatment and those who were not (p = 0.55). Absence of pathological endoscopic findings was significantly associated with a RAC+ pattern (p = 0.01). The sensitivity and negative predictive value (NPV) of RAC+ for ruling out H. pylori infection were 0.97 (95% CI: 0.94–0.99), reaching 1.00 when discordant images were reviewed by a blinded expert endoscopist. No significant differences in sensitivity or NPV were found between PPI users and non-users, or between regions with high and low H. pylori prevalence.
Conclusion:
The presence of the RAC pattern along the minor gastric curvature, as assessed with white-light endoscopy, accurately identifies patients without H. pylori infection in non-Asian countries, regardless of PPI use.
Plain language summary
Helicobacter pylori (H. pylori) is a bacteria that can infect the stomach and is linked to various gastrointestinal problems. Detecting whether a person has this infection can be challenging. A specific pattern observed during a stomach examination called the “Regular Arrangement of Collecting Venules” (RAC) can help predict the absence of H. pylori. Earlier work in Asian hospitals showed that when RAC is visible, H. pylori infection is very unlikely—but Western endoscopists have rarely used this sign. This international study involved 648 patients from 12 hospitals in non-Asian countries. None of the participants had a known history of H. pylori infection or treatment. The patients underwent a stomach examination using high-definition endoscopy to look for the RAC pattern. Endoscopists were trained to recognize the RAC pattern before the study. Results showed that the RAC pattern was present in about 31.5% of patients. Notably, there was no difference between patients who were using acid-reducing medication (PPIs) and those who were not. Additionally, patients with the RAC pattern were less likely to show signs of gastric lesions. The ability of the RAC pattern to correctly indicate the absence of H. pylori was very high (97%), and in some cases, it reached 100% when reviewed by an expert. These findings suggest that using white-light endoscopy to look for the RAC pattern is a reliable way to identify patients who do not have H. pylori, even in Western clinical practice and regardless of medication use.
Background
Helicobacter pylori (H. pylori) is the main risk factor for gastric cancer worldwide.1–3 Its eradication and the early detection of preneoplastic conditions are the essential strategies in the primary and secondary prevention of gastric cancer. 4
Recent advances in endoscopic technology have significantly improved the ability to observe the gastric mucosa, even in the absence of magnifying endoscopy. A variety of endoscopic findings have been correlated with the presence of H. pylori infection.5–7 Among these findings, the regular arrangement of collecting venules (RAC) is particularly noteworthy, demonstrating high reliability with sensitivity and negative predictive value (NPV) exceeding 90% for identifying H. pylori-negative mucosa.8–11
Although many of these studies were conducted in Asia using magnifying endoscopy, 12 recent research performed in Europe utilizing high-definition (HD) endoscopes without magnification reported a sensitivity and NPV of around 100%.6,7,13 These preliminary findings are unsurprising because the gastric mucosal micro-anatomy observed endoscopically is comparable in Asian and Western populations. What remains largely unexplored, however, is the reliability of RAC assessment as a routine diagnostic surrogate for H. pylori infection in everyday Western practice—its reproducibility and diagnostic accuracy have yet to be rigorously quantified. Moreover, RAC evaluation is rarely performed outside Asia and is still absent from Western guidelines for high-quality endoscopy.
A significant challenge in the diagnosis of H. pylori is the use of proton pump inhibitors (PPI), which can lead to the bacteria migrating to more proximal stomach regions and a reduction in colonization density.14,15 PPIs are known to cause false-negative results when biopsies are taken from the antrum and may also alter the appearance of the gastric mucosa and the RAC pattern. Therefore, discontinuing PPIs at least 2 weeks before testing for H. pylori is standard practice,14,16 and all studies examining the RAC pattern have excluded patients actively using PPIs. However, up to 36% of elderly patients are on PPIs at the time of endoscopy in routine clinical settings,17,18 underscoring the importance of developing a reliable diagnostic method for H. pylori infection in these circumstances.
The objectives of this study were to evaluate the value of RAC as a real-time diagnostic method to rule out H. pylori infection in routine clinical practice and the impact of PPI usage on the performance of RAC in non-Asian countries with different prevalence of H. pylori infection.
Methods
Study design and population
A multicenter prospective study was carried out across 12 hospitals (11 in Europe and one in South Africa). The study included consecutive patients undergoing non-urgent diagnostic esophagogastroduodenoscopy (EGD) for any clinical indication using HD white-light endoscopes without magnification from December 2021 to March 2024. Exclusion criteria were defined as follows: patients under 18 years of age; recent antibiotic use (within 14 days before the procedure); history of H. pylori eradication; intake of nonsteroidal anti-inflammatory drugs (NSAIDs), aspirin, antithrombotic, or oral anticoagulants (OAC); history of portal hypertension, lymphoma involving mucosa-associated lymphoid tissue (MALT) or previous gastric surgery; refusal to participate in the study. Patients on PPIs were not excluded from the study.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 19 The study protocol was approved by the Ethics Committee at the Hospital Clinic of Barcelona (HCB/2021/0898) and all the participating centers. Patients gave informed consent that included the use of pictures.
Endoscopic procedures and endoscopists
All EGDs were performed using HD endoscopes (Olympus, Tokyo, Japan; Fujifilm, Tokyo, Japan; and SonoScape Medical, Shenzhen, China) without the use of magnification were conducted by 17 different endoscopists. Following the routine examination of the esophagus, stomach, and duodenum, a detailed inspection of the distal part of the lesser curvature near the incisura angularis was performed using white light and appropriate gastric insufflation. 20 The presence of a regular or irregular arrangement of collecting venules was evaluated in real time. The RAC sign originally described in 2002 was defined under conventional white-light endoscopy. Since then, virtually all studies—whether in pediatric or adult populations and with or without magnification—have likewise relied on white light, with only a single report having explored its assessment using narrow-band imaging (NBI).6–13,21 We therefore elected to retain white-light endoscopy without magnification as the primary detection method, both to maintain continuity with the existing body of research and to facilitate implementation in everyday clinical practice. Electronic chromoendoscopy was, of course, available during the procedures for the detection of additional signs related to chronic gastritis or preneoplastic lesions and could be used at the discretion of the endoscopist whenever additional mucosal evaluation was deemed necessary. If minute red points were visible consistently and uniformly, the finding was considered RAC positive (RAC+) (Figure 1(a)). If the finding was absent or displayed a patchy distribution, it was categorized as RAC negative (RAC−) (Figure 1(b) and (c)). 21 For each patient, the most representative image that was used for the assessment of RAC was anonymized and stored. Any discordant cases classified as RAC+ but confirmed to have H. pylori infection were blindly reviewed, along with other RAC+ and RAC− cases, by an experienced endoscopist familiar with the RAC pattern. To standardize RAC interpretation, a training session was conducted using cases not included in the study. A set of 20 images illustrating both positive and negative RAC cases was presented to the participant endoscopists remotely.

Examples of RAC. RAC+ pattern: minute, starfish-like red points are visibly distributed regularly and homogeneously in the lower part of the lesser curvature (a). Examples of RAC-patterns include inflammatory changes in the lesser curvature without visible collecting venules (b, c).
Recording data
Personal data (sex, age, and PPI use) and endoscopic findings (such as erosive gastritis, gastric ulcers, gastric adenoma, gastric cancer, and signs of atrophic gastritis or intestinal metaplasia) were recorded. We did not consider recording the endoscopic severity of gastritis or intestinal metaplasia, and we only stored the pictures taken for the evaluation of RAC. In the training session, we only standardized RAC interpretation, and each endoscopist evaluated the presence of atrophy or intestinal metaplasia based on their own experience.
H. pylori infection status was determined by gastric biopsies following the updated Sydney protocol. An immunohistochemical (IHC) study was systematically performed in patients with PPI intake and a negative histologic study for H. pylori. Study data and images were collected and managed using Research Electronic Data Capture (REDCap) tools hosted at the Spanish Gastroenterology Association (AEG).
Statistical methods for data analysis
Sample size calculation
The estimated prevalence of H. pylori in Europe varies significantly (it is less than 20% in Northern Europe and exceeds 30% in Mediterranean countries). 22 Assuming a prevalence of 20%, a minimum sample size of 488 patients was calculated to retain 415 analyzable subjects (including 98 true H. pylori-positive cases) and maintain 80% power to show that the sensitivity is at least 90% with the lower bound of the one-sided 95% confidence interval not dropping below 80%. The trial was initially launched at 10 centers, each assigned a recruitment target of 50 patients. After two additional centers were added, the per-center quota was deliberately maintained to preserve uniform enrollment goals, which proportionally increased the overall sample size.
Quantitative data are presented as medians and interquartile ranges (IQR) or as means and standard deviation (SD), depending on the distribution, while qualitative data are shown as proportions. Categorical variables were compared using the chi-squared test, and continuous variables were analyzed using the t-test. A p-value of less than 0.05 was considered statistically significant. The sensitivity, specificity, positive predictive value (PPV), NPV, and accuracy of RAC in diagnosing H. pylori-negative patients, with a 95% confidence interval (CI), were calculated using standard formulas. Multiple logistic regression analysis was performed to identify predictor factors of RAC+ pattern and H. pylori infection. Candidate variables for inclusion in the model were those achieving a p-value ⩽0.1 in the univariate analysis. Odds ratios (ORs) and associated 95% CIs were used to quantify the level of association. All statistical analyses were conducted using SPSS version 26 (IBM SPSS Inc., Chicago, IL, USA).
Results
Study participants and baseline characteristics
A total of 648 patients were included in 12 centers, with an average age of 52 years (IQR: 40–63) and 363 (56%) were female. One hundred and seventy-three (26.7%) patients were on active treatment with PPIs. Significant endoscopic abnormalities (erosive gastritis, gastric ulcer, gastric adenoma, gastric cancer, and signs of atrophic gastritis or intestinal metaplasia) were found in 180 (27.7%) patients, including six cases of gastric cancer. The baseline characteristics of the patients are detailed in Table 1. H. pylori infection was confirmed in 225 (34.7%) patients with a high variability among countries (median 28%). Only two out of 131 patients taking PPIs and testing negative for H. pylori in histopathology were found positive using the IHC test.
Baseline characteristics of the included individuals (N = 648).
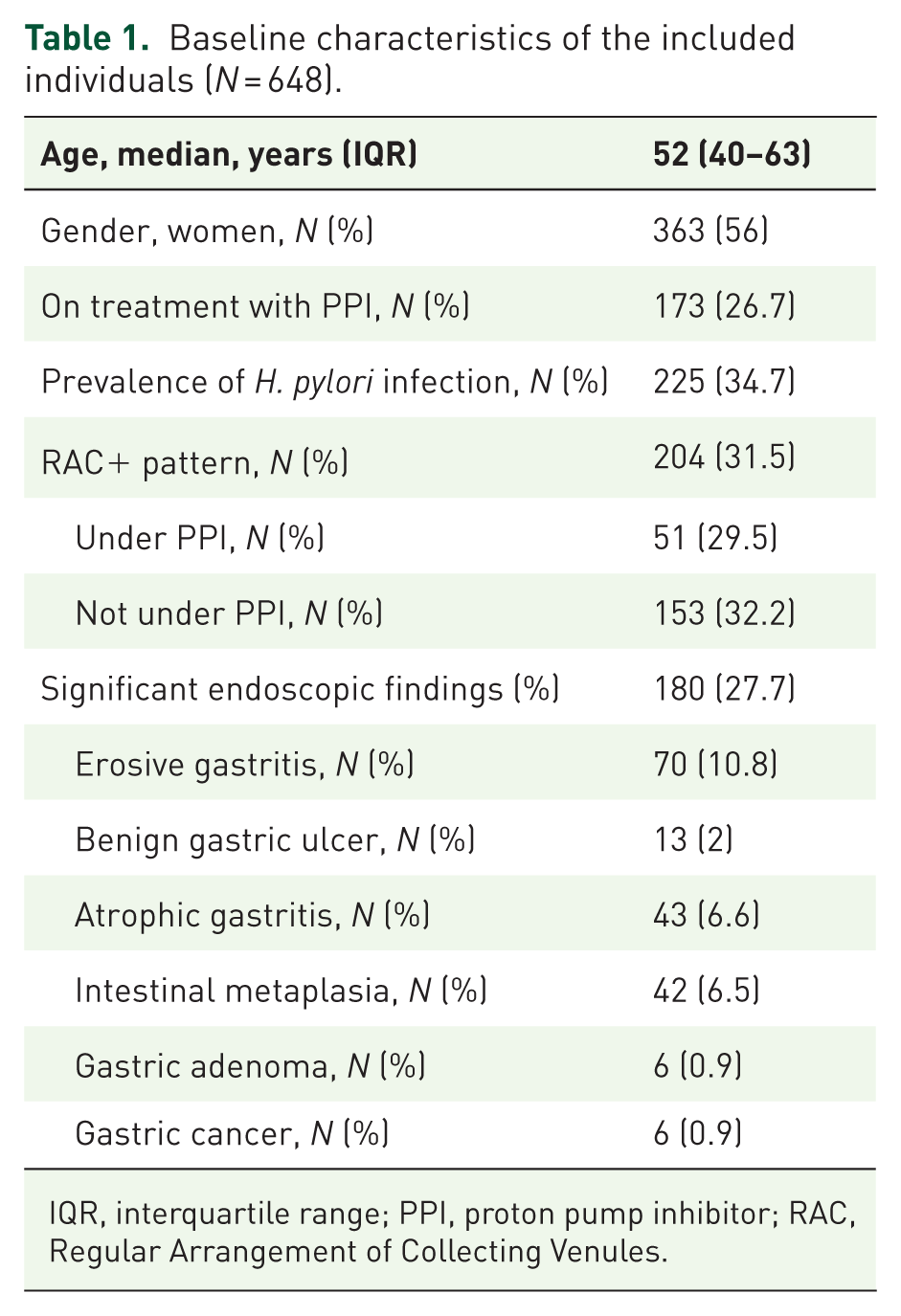
IQR, interquartile range; PPI, proton pump inhibitor; RAC, Regular Arrangement of Collecting Venules.
RAC pattern and H. pylori infection
Two hundred and four patients (31.5%) had a RAC+ pattern; 198 (97.1%) of these patients were free from H. pylori infection according to mucosal biopsy and IHC results (Figure 2). Conversely, the absence of RAC was associated with H. pylori infection in 49.1% (218 out of 444 cases). Six out of the 225 (2.7%) patients with confirmed H. pylori infection exhibited a RAC+ pattern. This finding resulted in an NPV and sensitivity of 97.1% (95% CI: 95–100) and 97.3% (95% CI: 94–100), respectively. When analyzing the diagnostic performance of the RAC pattern in patients treated with PPIs, no statistically significant differences were found regarding sensitivity, specificity and NPV, whereas PPV was lower in patients under PPI (Table 2).
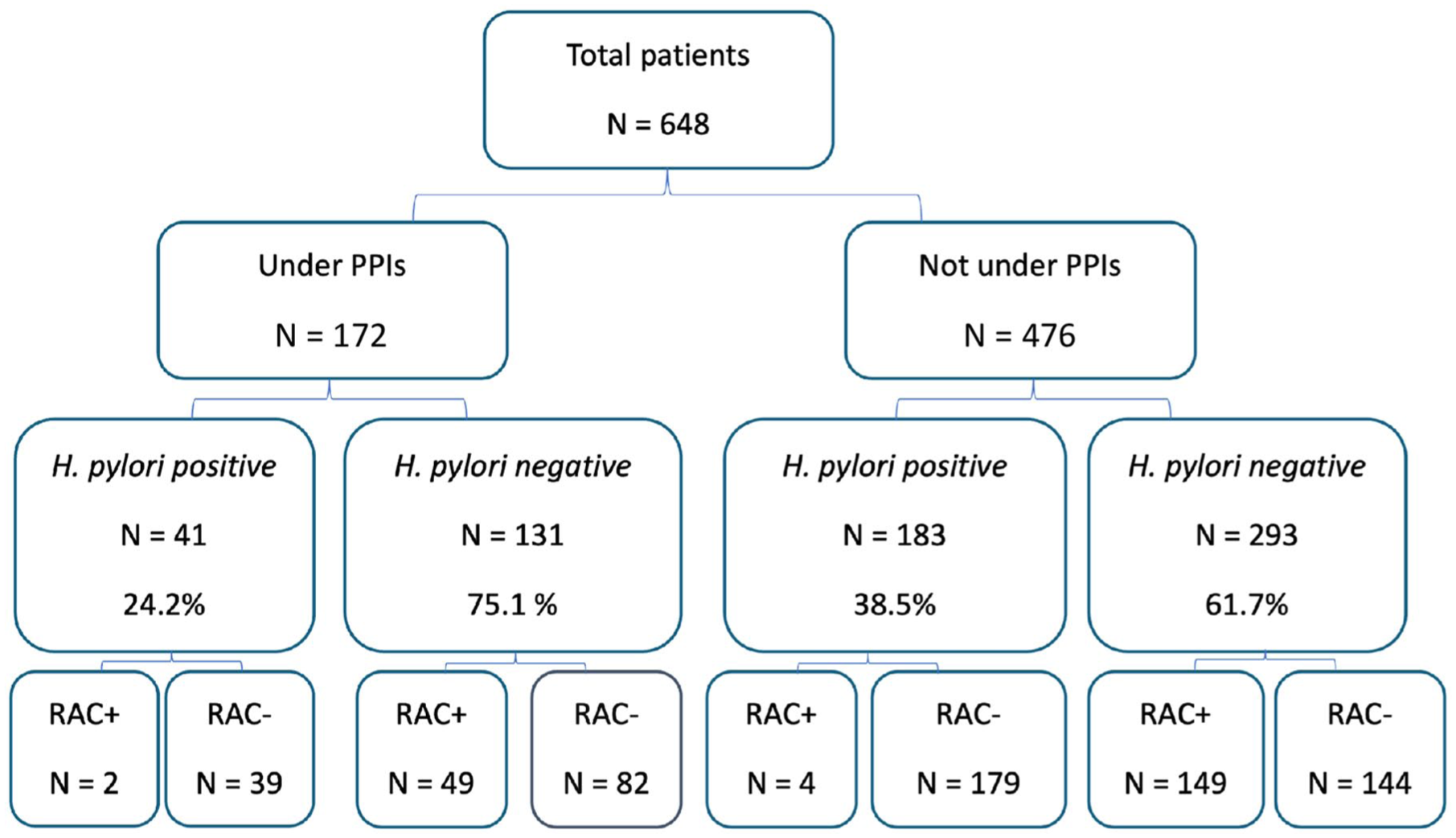
Flow-chart of the study.
Performance characteristics of RAC to detect H. pylori infection globally depending on the use or not of PPI.

PPI, proton pump inhibitor; RAC, Regular Arrangement of Collecting Venules.
All six discordant cases involved adults aged 42–58 years (four women, two men) recruited from different centers. None had received oral anticoagulants, NSAIDs or antibiotics within the preceding 4 weeks, and two had taken proton pump inhibitors during the 2 weeks before endoscopy, and—although collecting venules were visible—their distribution was uneven or patchy. After independent reassessment by an expert endoscopist, all six patients who had been labeled RAC positive yet tested positive for H. pylori were re-classified as RAC negative, resulting in 100% sensitivity and negative predictive value. The six discordant cases (initial RAC+, H. pylori+) are illustrated in Figure 3.

Examples of discordant cases (RAC+, H. pylori+).
We also analyzed the sensitivity and NPV of the RAC+ pattern based on the region and prevalence of H. pylori, and both were better in low-prevalence areas, although there were no statistically significant differences.
Predictor factors of RAC+ and H. pylori infection
The presence of any pathological finding on EGD (erosive gastritis, gastric ulcer, gastric adenoma, gastric cancer, or optical signs of atrophic gastritis or intestinal metaplasia) was associated with the absence of RAC+ pattern (OR 0.23; 0.14–0.38) and the presence of H. pylori infection (OR 1.6; 1.12–2.28). On the other hand, patients under PPI had a lower prevalence of H. pylori infection (OR 0.53; 0.35–0.78). Histology identified 43 patients with atrophic gastritis and 42 with intestinal metaplasia. In this dataset, the “atrophic gastritis” group, by definition, showed no evidence of intestinal metaplasia, whereas the “intestinal-metaplasia” group could exhibit concomitant atrophy. Among patients with atrophic gastritis, the RAC pattern was negative in 93.3% and positive in 6.7%. Likewise, in the intestinal-metaplasia group, 90.5% were RAC negative and 9.5% were initially scored RAC positive. All these findings are summarized in Table 3.
Predictor factors of RAC+ and H. pylori infection.

Erosive gastritis, gastric ulcer, gastric adenoma, gastric cancer, atrophic gastritis or intestinal metaplasia.
IQR, interquartile range; PPI, proton pump inhibitor; RAC, Regular Arrangement of Collecting Venules.
Discussion
This study confirms the reliability and clinical applicability of RAC after brief training using only HD white-light endoscopy without magnification in a non-Asian cohort, irrespective of PPI use and H. pylori prevalence. We employed conventional white-light endoscopy without optical magnification to streamline implementation during routine diagnostic gastroscopies; nonetheless, ongoing technological advances and wider availability of modalities such as virtual chromoendoscopy and high-magnification endoscopy may further enhance RAC detection as well as other normal structures, such as crypt openings or subepithelial capillary network. 23
Training for endoscopic assessment of H. pylori is predominantly conducted in Asian regions where magnifying endoscopy is widely available. In the research conducted by Watanabe et al., 24 novice endoscopists with less than 500 EGDs were trained systematically over 2 years during routine clinical practice. At the conclusion of their training, 11 specific endoscopic features indicative of H. pylori infection were re-evaluated. RAC showed the highest inter-observer agreement (0.58), which improved to 0.81 by the end of the training. Our study demonstrated that, following brief training provided to all endoscopists, the RAC pattern serves as a reliable predictor for excluding H. pylori infection, with a sensitivity and NPV of approximately 97%, rising to 100% when independently reviewed by another blinded experienced endoscopist.
While RAC serves as a valuable marker for real-time identification of H. pylori-negative normal gastric mucosa, its clinical application outside Asia remains limited. Although there are some studies in the European population that demonstrate a high reliability to rule out H. pylori infection,6,7 most western endoscopists are unaware of its existence. With this multicenter study, we aimed to involve as many endoscopists from different countries as possible to disseminate the use of the RAC sign as widely as possible.
Diagnosing H. pylori-related gastritis, atrophy, and/or metaplasia based on endoscopic observations is critically important but can be challenging in non-Asian countries due to inconsistent adherence to guideline recommendations for biopsy sampling.25,26 Real-time endoscopic suspicion of H. pylori infection may improve patient management by suggesting more detailed evaluations, such as magnification or virtual chromoendoscopy, as well as biopsies for antimicrobial susceptibility testing. Conversely, in cases of normal gastric mucosa that test negative for H. pylori, this approach could reduce the necessity for biopsies.25-28
A previous study by our group reported that using RAC with only white-light endoscopy achieved a 100% NPV for excluding H. pylori infection. 6 However, similar to other studies, patients taking PPI were excluded due to the potential negative impact of these drugs on infection diagnosis. 14 In the current study, we observed no significant differences in RAC+ prevalence between patients on PPIs (30% vs 32.2%, p 0.55), with comparable sensitivity and NPV. Furthermore, we demonstrated that the RAC pattern performs exceptionally well in both high and low H. pylori prevalence regions, with comparable sensitivity and NPV.
Although the effectiveness of RAC has been noted to diminish with age due to age-associated inflammatory changes in the gastric mucosa,20,21,29,30 we did not find differences in our study. Contrarily, we observed that the presence of significant endoscopic findings, such as erosive or preneoplastic lesions, was significantly associated with the presence of H. pylori infection and the disappearance of the RAC pattern. The exclusion of individuals taking drugs with a well-known gastroerosive effect, such as aspirin, NSAIDs, or OACs, may have contributed to this association.
This study has several notable strengths. It represents a multicenter cohort including 11 hospitals from European countries and 1 hospital from South Africa, encompassing over 600 patients. To the best of our knowledge, this is the first multicenter international study to assess the performance of the RAC pattern in non-Asian countries, irrespective of PPI use. In addition, photodocumentation of the RAC pattern was conducted in all cases and subsequently reviewed by an independent external endoscopist blinded to the results, further enhancing the consistency of our conclusions.
The limitation of the study is that including patients actively taking PPIs may result in some false negatives for H. pylori due to bacterial migration to the more proximal stomach areas and decreased bacterial load.16,31 To mitigate this, biopsies were taken from both the antrum and corpus, and IHC testing was performed on all PPI-using patients with H. pylori-negative hematoxylin and eosin (H&E) biopsies. IHC staining offers greater sensitivity and specificity than H&E, especially beneficial in cases with PPI use, which can cause atypical cell forms or mucus in H&E samples.32,33 Although additional tests could have been employed to increase diagnostic yield, our approach yielded similar H. pylori prevalence rates regardless of PPI use.
Conclusion
In summary, the use of white-light HD endoscopy to diagnose H. pylori based on the arrangement of gastric collecting venules is a simple and highly reproducible technique, consistent across different examiners and applicable in regions with varying H. pylori prevalence, including patients on PPIs. Our findings support the integration of RAC as a real-time diagnostic tool for confirming H. pylori-negative status in Western clinical settings, potentially eliminating the need for biopsies when RAC+ is observed.
