Abstract
Background and aim:
Endoscopic ultrasound-guided biliary drainage is an alternative to failed endoscopic retrograde cholangiopancreatography. Unfortunately, this procedure remains relatively less explored in Egypt due to its high cost, lack of adequate training, and the perception of increased risk. This study is the first multicenter Egyptian experience of an endoscopic ultrasound-guided biliary drainage in patients with malignant biliary obstruction.
Patients and methods:
We retrospectively reviewed 15 patients (10 men and five women) with malignant biliary obstruction who from October 2013 to May 2019, following a failed or inaccessible endoscopic retrograde cholangiopancreatography, underwent an endoscopic ultrasound-guided choledochoduodenostomy, endoscopic ultrasound-guided hepaticogastrostomy, or endoscopic ultrasound-guided rendezvous. Their mean age was 57.4 years and mean bilirubin was 18.2 mg/dL. The outcome parameters included technical and clinical success. Technical success was defined as the successful placement of a stent in the biliary system, while clinical success was defined as a greater than 50% decrease in the bilirubin levels 2 weeks after the procedure. Patients were monitored for complications during and after the procedure.
Results:
In total, 15 patients underwent endoscopic ultrasound-guided biliary drainage (eight underwent endoscopic ultrasound-guided choledochoduodenostomy, five underwent endoscopic ultrasound-guided hepaticogastrostomy, and two underwent endoscopic ultrasound-guided rendezvous). The technical and clinical success rates were 100% (15/15 patients) and 93.3% (14/15 patients), respectively. The complication rate was 26.6% (4/15 patients). All complications were mild and self-limited, and included fever, mild biliary peritonitis, pneumoperitoneum, and a slight migration of one plastic stent during insertion.
Conclusion:
Although slowly gaining acceptance in Egypt, endoscopic ultrasound-guided biliary drainage is an effective and safe procedure in patients with a malignant biliary obstruction after a failed or inaccessible endoscopic retrograde cholangiopancreatography.
Introduction
Endoscopic retrograde cholangiopancreatography (ERCP) is the standard procedure for biliary drainage in patients with a malignant biliary obstruction. 1 Despite having a high success rate of more than 90%, even skilled endoscopists have difficulty in successfully achieving biliary access using ERCP. This could be due to many factors, such as a surgically altered anatomy (i.e. Billroth II surgery, Whipple procedure), malignant obstruction of the lumen, periampullary diverticula, and gastric outlet obstruction.2,3
Surgical biliary bypass and percutaneous transhepatic biliary drainage (PTBD) are the alternative options for draining the biliary system after an ERCP failure. 4 Although PTBD is more commonly preferred to surgery, it is associated with high complication rates of up to 23%; mostly due to cholangitis, obstruction or dislocation of the tube, higher probability of multiple sessions, and patient’s dissatisfaction with the external tube drain.5,6
In recent years, endoscopic ultrasound-guided biliary drainage (EUS-BD) has been reported as an alternative to PTBD or surgical biliary bypass in patients with failed ERCP. 7 The technique is still evolving with different success and complication rates across the literature, which may be attributed to the type of procedure chosen and the accessories used. Unfortunately, this procedure is rarely performed in Egypt due to its high cost, lack of adequate training, and the perception of increased risk. This study is the first multicenter Egyptian experience with EUS-BD in patients with malignant biliary obstruction after a failed or inaccessible ERCP.
Patients and methods
Patients
This study is a multicenter, retrospective study, which was conducted at three tertiary hospitals, namely, the Mansoura Specialized Medical Hospital, Mansoura University; the Alexandria Fever Hospital; and the Kasr Al-Ainy Hospital, Cairo University. Data of all patients who underwent an EUS-BD from October 2013 to May 2019 were collected.
The inclusion criteria were as follows: patients with an inoperable or metastatic malignant biliary obstruction, so identified on the basis of computed tomography or EUS findings; an inaccessible biliary system (either due to difficult cannulations, failure to pass the stricture by ERCP, or a tumor-altered or surgically altered anatomy); and an inadequate biliary drainage after an ERCP. The exclusion criteria were as follows: major organ failure, unfit for sedation, coagulation disorders, and refusal to sign the consent. The study protocol was approved by our ethical committee (IRB of Mansoura University, R.19.04.478) and written consents were taken from all patients after explaining the technique of the procedure and possible complications.
Thus, 15 patients (10 men and five women) were selected and retrospectively reviewed. Their mean age was 57.4 years (range: 42–80) and mean bilirubin level was 18.2 mg/dL.
Methods
All the procedures were performed in the prone position and under sedation with midazolam and propofol. EUS was performed using either the Pentax linear echoendoscope EG-3870UTK (PENTAX Medical, Tokyo, Japan) or the Olympus linear echoendoscope GF-UCT180 (OLYMPUS Medical, Tokyo, Japan). Three experienced endosonographers performed all the EUS-BD procedures, following which, the patients were admitted for at least 2 days and monitored for potential complications such as fever, abdominal pain, pneumoperitoneum, bleeding, and peritonitis. If there were no complications after 24 h, the patients were allowed to consume clear fluids.
EUS-BD techniques
EUS-guided choledocoduodenostomy
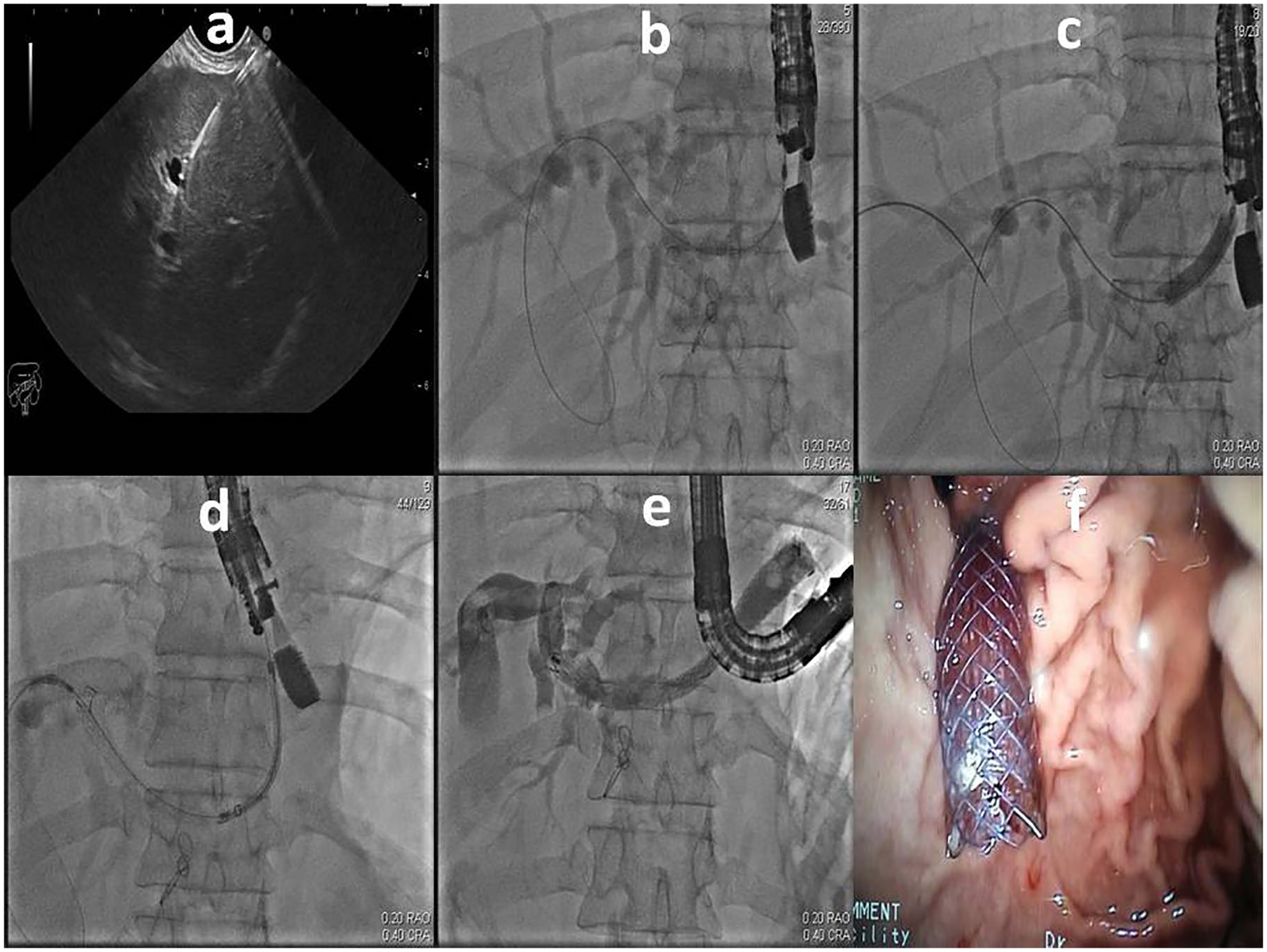
Eight patients underwent EUS-guided choledocoduodenostomy (EUS-CD). The echoendoscope was positioned in the duodenal bulb in a long-scope position. After the dilated common bile duct (CBD) was located and the absence of an interposing vessel was confirmed by a color doppler, a transduodenal CBD puncture was performed using a 19-G needle (Cook Medical, Bloomington, IN, USA). The stylet was then removed and the position was confirmed by aspirating the bile. This was followed by a contrast injection into the biliary tree. Subsequently, a 0.035-in. wire (Boston Scientific, Natick, MA, USA) was inserted through the needle toward the intrahepatic biliary tree. The needle was then removed and a track was created by guiding either a standard cannula or a 6- to 10-Fr cystotome over the wire (6-Fr cystotome from Taewoong Medical, South Korea and 10 Fr from Cook Medical). When necessary, a 6- to 8-mm dilation balloon was used to dilate the track. Finally, a 10-Fr double pigtail plastic (Cook Medical) or a 6- to 8-cm partially covered self-expandable metal stent (PCSEMS; Boston Scientific and HANAROSTENT, M.I. Tech, Korea) was deployed under fluoroscopic and endoscopic guidance into the track created between the CBD and the duodenal bulb (Figure 1).

(a) EUS guided puncture of the bile duct, (b) contrast injection and wire passed towards the intrahepatic ducts, (c) track creation with cystotome, (d) deploying the stent under fluoroscopic guidance, (e) final result after deployment and contrast injection, (f) endoscopic view of the stent.
EUS-guided hepaticogastrostomy
Five patients underwent EUS-guided hepaticogastrostomy (EUS-HG). The echoendoscope was positioned in the stomach in the short-scope position. After the dilated intrahepatic biliary tree was located and the absence of an interposing vessel was confirmed by a color doppler, a transgastric puncture of the dilated left hepatic duct was performed using a 19-G needle (Cook Medical). The stylet was then removed and the bile was aspirated for confirmation, followed by a contrast injection into the biliary tree. Subsequently, a 0.035-in. wire (Boston Scientific) was inserted through the needle toward the liver hilum or the CBD. The needle was then removed and a track was created by guiding a 6-Fr cystotome (Taewoong Medical, Goyang-Si, Gyeonggi-do, South Korea) over the wire. When necessary, a 6-mm dilation balloon was used to dilate the track. Finally, an 8-cm PCSEMS was deployed under fluoroscopic and endoscopic guidance into the track created between the left hepatic duct and the stomach (Figure 2).
(a) EUS guided puncture of intrahepatic duct, (b) contrast injection and wire passed towards the common bile ducts, (c) track dilation with balloon, (d) deploying the stent under fluoroscopic guidance, (e) final result after deployment and contrast injection, (f) endoscopic view of the stent.
EUS-guided rendezvous
Two patients underwent EUS-guided rendezvous (EUS-RV). The echoendoscope was positioned in the stomach in a short-scope position. After the dilated intrahepatic biliary tree was located and a color Doppler confirmed the absence of an interposing vessel, a transgastric puncture of the dilated left hepatic duct was performed using a 19-G needle (Cook Medical). The stylet was then removed and the bile was aspirated for confirmation, followed by a contrast injection into the biliary tree. Subsequently, a 0.035-in. wire (Boston Scientific) was inserted through the needle toward the CBD. The needle was then exchanged for a standard cannula (Cook Medical) to manipulate the wire into the duodenum through the papilla. The echoendoscope was then removed, leaving the guidewire in place, and replaced with a standard duodenoscope, which was inserted into the second part of the duodenum. To perform the conventional ERCP procedure, a pair of forceps was used to grasp and withdraw the guidewire through the accessory channel of the duodenoscope. Finally, a 6- to 8-cm PCSEMS was deployed under fluoroscopic and endoscopic guidance through the papilla using the conventional technique (Figure 3).

(a) EUS guided puncture of intrahepatic duct, (b) contrast injection showed dilated biliary system, (c) guidewire passed to the duodenum, (d) guidewire grasped to the accessory channel of the duodenoscope, (e) deploying the stent under fluoroscopic guidance, (f) final result after deployment.
To summarize, in all patients, a 19-G needle and a 0.035-in. guidewire were used to access the biliary system. A cystotome was used for track creation in 12 cases (6 Fr in six cases and 10 Fr in six cases) and a standard cannula in three cases. Both PCSEMS and plastic stents were used for draining the biliary system (PCSEMS in nine cases, double pigtail plastic stent in five cases, and standard a plastic stent in one case).
Outcome parameters
The outcome parameters included technical and clinical success. Technical success was defined as the successful deployment of the stent into the biliary system. Clinical success was defined as greater than 50% reduction in the bilirubin value after 2 weeks from the procedure, when compared with the preprocedural value. All complications during the procedures and follow-up were recorded. Patients were monitored during and after the procedure for complications such as fever, bleeding, biliary peritonitis, and pneumoperitoneum. The severity of adverse events was graded on the basis of the need for hospitalization according to the American Society for Gastrointestinal Endoscopy (ASGE) lexicon. 8 Four grades of severity were used, mainly: mild (events requiring hospitalization for 1–3 days), moderate (4–9 days in the hospital), severe (more than 10 days in the hospital or needing surgery or intensive care), and fatal (death attributable to the procedure). After the discharge, patients were followed clinically and with laboratory studies for signs of recurrent biliary obstruction. Imaging was done during follow-up when necessary.
Results
A total of 15 patients (10 men and five women) with malignant biliary obstruction, who underwent EUS-BD, were selected on the basis of the inclusion and exclusion criteria and were retrospectively reviewed. The causes of biliary obstruction were: (1) an inoperable pancreatic head mass in seven patients, (2) a cholangiocarcinoma in four patients, (3) a metastatic pancreatic head mass in two patients, and (4) an ampullary mass in two patients. An inaccessible biliary system was observed in nine patients due to a difficult cannulation, in two due to a tumor-altered anatomy, in one due to a surgically altered anatomy, in one due to a failure to pass the stricture by ERCP, and in two due to an inadequate biliary drainage (draining only from the right anterior or posterior duct) after ERCP (Table 1).
Baseline characteristics of included patients.

BD: biliary drainage; ERCP: endoscopic retrograde cholangiopancreatography; EUS: endoscopic ultrasound.
The technical and the clinical success rates were 100% (15/15 patients) and 93.3% (14/15 patients), respectively. The complication rate was 26.6% (4/15 patients). All complications were mild and self-limiting, including fever, mild biliary peritonitis, pneumoperitoneum, and a slight migration of one plastic stent during insertion (proximal migration with an impacted distal tip in the duodenal wall). The summary of all cases is shown in Table 2.
Summary of cases that underwent EUS-BD.

BD: biliary drainage; CD: choledocoduodenostomy; EUS: endoscopic ultrasound; G: gauge; HG: hepaticogastrostomy; PCSEMS: partially covered self-expandable metallic stent; RV: rendezvous.
Discussion
EUS-guided cholangiography was first described by Wiersema and colleagues 9 in 1996, while EUS-BD was first described by Giovannini and colleagues 10 in 2001. Since then, many reports on EUS-BD have been published. In Egypt, the first EUS-BD was performed by our group in the Mansoura University in 2014. 11 However, this procedure is still slowly gaining acceptance in Egypt and is only performed in a few tertiary centers.
Although EUS-BD is an evolving technique, its success and complication rates are comparable to that of ERCP, which is the standard procedure for biliary drainage in patients with a malignant biliary obstruction. In a large retrospective study by Dhir and colleagues, 12 the success and complication rates for EUS-BD (93.26% and 8.65%, respectively) were similar to that for ERCP. Similarly, a comparison between EUS-BD and PTBD reveals that EUS-BD has a success rate higher than that of PTBD (95% versus 46%) and a lower complication rate than that of PTBD (20% versus 46%). 13 Another advantage of EUS-BD is the ability to switch to biliary drianage within the same procedure, thus avoiding delayed biliary decompression and repeated procedures. Furthermore, EUS-BD improves the patient satisfaction and offers a longer patency of the stents, which naturally reduces the cost of repeated procedures. 14
EUS-guided procedures include rendezvous and drainage procedures with either CDorHG. To date, there is no agreement on the best route to achieve biliary drainage. Thus, it often depends on the judgment and preference of the therapeutic endoscopist. 15 So far, few comparative studies on the efficacy and safety of different biliary drainage techniques have been reported with variable results, and most of the data originates from retrospective studies. In a study by Artifon and colleagues, 16 the technical success rate of EUS-CD was 91% compared with the 96% for EUS-HG. Similarly, a recent meta-analysis by Uemura and colleagues 17 demonstrated an equal efficacy and safety for EUS-CD and EUS-HG.
Regarding the stent type, both PCSEMS and plastic stents were used in this study for draining the biliary system. As of yet, there is no definite EUS-BD study that compares the metal stents with their plastic counterparts; however, metal stents may reduce the risk of a biliary leak due to their radial expansion force. However, ERCP studies have shown that metal stents have longer patency durations than plastic stents. The analysis of a study, which used both stent types in EUS-HG, revealed that the follow-up periods for metal and plastic stents were 325.3 days (range: 120–610 days) and 164.7 days (range: 30–267 days), respectively. It also reported that the patency periods of the metal and plastic stents were 269.3 days (range: 78–610 days) and 136.6 days (range: 30–222 days), respectively. 7
Overall, the EUS-BD procedures have a significantly high success rate, with reviewed data revealing technical and clinical success rates of up to 94% and 90%, respectively. 18 Furthermore, Dhir and colleagues 19 reported a 95.6% success rate, which is comparable to our study, where the technical and clinical success rates were found to be 100% and 93.3%, respectively. The complication rates of EUS-BD, while variable, appear to be declining in the recent years with an increasing experience in the technique in advanced endoscopy centers. 20 In a large meta-analysis of 42 studies that included 1192 patients, the complication rate for EUS-BD was estimated to be 23%. The highest risk was observed for bile leakage and bleeding, followed by a lower risk of cholangitis, abdominal pain, pneumoperitoneum, peritonitis, and stent migration. 18 Similarly, our retrospective study also showed a complication rate of 26.6%; the complications included a self-limited fever, biliary peritonitis, pneumoperitoneum, and slight migration of one plastic stent during insertion.
This study has some limitations, including the relatively small number of cases, different techniques for biliary drainage, and different accessories used in the procedures; making generalizing our results difficult. However, the study also has some strengths: it is a multicenter study that was conducted at three tertiary centers and it describes the first experience of an EUS-BD in Egypt.
In conclusion, EUS-BD is an effective and safe procedure in patients with a malignant biliary obstruction after a failed ERCP or inaccessible papilla. However, it requires a high level of technical skills and should be performed only in tertiary centers by experienced endoscopists.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
