Abstract
“Responsive regulation” has been proposed to offset pharmaceutical industry illicit behavior in areas such as drug marketing based on self-regulation backed up with threats of government sanctions. We explore the efficacy of responsive regulation by tracing recent investigations by the UK pharmaceutical industry self-regulatory authority into the firm Astellas’s illicit promotion of a top-selling prostate cancer drug. Using documentary data, we reveal a ruthless company culture reflected in the illicit, so-called off-label promotion across Europe and the deceptive “impression management” by company managers during the course of investigations in the UK. We argue for a more probing, adversarial and government-led regulatory approach instead of the self-regulatory approach that shields breaching companies from enforcement actions and associated public scrutiny.
Keywords
Introduction
The interrelated issues of pharmaceutical industry regulation, crime control and ethics compliance were key concerns in John Braithwaite’s (1984) corporate criminology classic “Corporate Crime in the Pharmaceutical Industry.” When Braithwaite and colleagues revisited these issues 30 years later, they diagnosed what they saw as “a worsening crisis” since the 1990s, concluding that “corporate crime in the pharmaceutical industry appears to be on the rise” (Dukes et al., 2014, p. 275). In terms of providing a solution to abusive business practices, they recommend—as Braithwaite (2011) has done for many industries—a “responsive” regulatory approach that “decenters criminal enforcement by the state” (Braithwaite, 1993, p. 12) in favor of a web of softer social control and support mechanisms based on cooperation and self-regulation (Ayres & Braithwaite, 1992). Distinctively, responsive regulation also involves the concept of an “enforcement pyramid” whereby regulators seek to motivate compliance in the first instance through use of less punitive, self-regulatory strategies involving education, persuasion and negotiation, and by appeal to the intrinsic ethical and social values of a company. If these “softer” measures fail, regulators respond by progressively moving up the enforcement pyramid—but only as far as necessary. For example, the next step might involve publicly naming and shaming offending companies, with regulators escalating their response to impose severer sanctions for continued non-compliance. Criminal prosecution still has an important role near the peak of the pyramid. However, its main purpose is to act as a threatening element in the background. This regulatory approach is advocated by its proponents in the belief that if regulators resist “the temptation to crowd out corporate ethics with excessive criminalization” they will be “more effective in reducing corporate crime in the pharmaceutical industry” (Dukes et al., 2014, p. 271). Hence the pyramidal design, with increasingly intrusive regulatory interventions corresponding with “ever-decreasing frequency of use” (Ayres & Braithwaite, 1992, p. 6).
There is a substantial literature that endorses responsive regulation across sectors and industries (e.g. Burford & Adams, 2004; Braithwaite, 2008; Schell-Busey et al., 2016; but see Tombs, 2015). Responsive regulation has also proven appealing to regulators and policymakers (Job et al., 2007: Parker, 2013), especially the idea of decentring the state (Tombs & Whyte, 2013) and of using enforcement pyramids (Mascini, 2013). However, to the best of our knowledge, no studies have investigated empirically a responsive approach to pharmaceutical industry crime control and ethics compliance despite longstanding claims of efficacy (Braithwaite, 1993). Indeed, if Braithwaite and colleagues are correct that corporate crime in the pharmaceutical industry has been growing, then this seems to put in question responsive regulation because, over the last decades, the pharmaceutical sector has implemented many of the structures and strategies proscribed by the proponents of responsive regulation (Francer et al., 2014).
Responsive Regulation in the UK Pharmaceutical Sector
A case in point is the system for regulating the UK prescription drug marketing—the topic of this article. This system relies heavily on self-regulation based on codified ethical standards drawn up by the industry that are backed up with threats of punitive government sanctions. Thus, the Medicines and Healthcare products Regulatory Agency (MHRA) is responsible for enforcing regulations but the government medicines regulator has delegated an important part of this responsibility to the Association of the British Pharmaceutical Industry (ABPI), the UK pharmaceutical industry trade group. To discourage illicit marketing, the ABPI has developed a Code of Practice for companies (ABPI, 2014), as well as numerous guidance documents and support mechanisms, administered by the ABPI’s self-regulatory authority—the Prescription Medicines Code of Practice Authority (PMCPA). Significantly, the formal memorandum of understanding between the MHRA, ABPI and PMCPA appears to conform to the model of responsive regulation—specifying that the MHRA “reserves the right to take action if serious public health concerns are raised or if self regulation fails” (ABPI, 2005, p. 1). This arrangement differs from the USA—and certain other European countries, like France—where government agencies regulate marketing with little or no formal delegation to self-regulatory bodies, and where—in the case of the USA—the government has often used litigation to punish companies for illicit marketing (Vilhelmsson et al., 2016). In contrast, the MHRA strongly recommends the use of the self-regulatory system for complaints concerning companies that have accepted the ABPI Code (Zetterqvist et al., 2015).
The PMCPA consists of the Code of Practice Panel, which deals with complaints under the ABPI Code from whatever source (e.g., rival companies, healthcare professionals, company employees), and the Code of Practice Appeal Board, which handles appeals to the Panel’s decisions, as well as situations where a company’s conduct is of special concern. Consistent with the concept of “decentered regulation,” i.e. involving many non-state actors (Black, 2001), and consistent with Ayres and Braithwaite’s (1992) model of democratic involvement to prevent capture, the Appeal Board is made up of eight members drawn from the industry, and eight members representing other interests—three medical practitioners, one pharmacist, and one nurse prescriber (who are appointed following consultation with the British Medical Association, Royal Pharmaceutical Society, and Royal College of Nursing, respectively); and one patient advocate, one lay member, and one representative from an independent body providing medicines information, appointed following consultation with the MHRA (Zetterqvist et al., 2015).
Finally—as recommended by Braithwaite and colleagues—the UK prescription drug marketing oversight system seeks to improve conduct by foregrounding education, persuasion and negotiation and non-damaging sanctions, rather than routinely resorting to adversarial and punitive strategies (Zetterqvist et al., 2015). For example, a key procedural feature is that companies should first attempt to settle disagreements through inter-company dialogue; the complainant company should only lodge a complaint for arbitration to the PMCPA if such efforts should be unsuccessful. Another key feature is that the charges incurred on violating companies are not designed to damage corporations financially; rather, they reflect the cost of administering the self-regulatory system. However, the PMCPA does have the possibility to escalate sanctions in cases of repeat or serious misbehavior via audits, public reprimands, corrective ads or letters; and in very severe cases the ABPI may suspend or expel a company and report it to the MHRA.
In sum, the UK system seems,
Through detailed analysis of the Astellas case, this article seeks to address two gaps in the corporate criminology literature on the pharmaceutical industry. The first is that most of the examples of illicit marketing and criminogenic cultures drawn on by Braithwaite and colleagues (Dukes et al., 2014) to support their contention of a “worsening crisis” pertain to the USA where companies have been subject to criminal prosecution rather than self-regulation (Mulinari, 2016). This is important because it is possible, but not proven, that the adversarial and punitive US system has failed to deter corporate crime in contrast to European self-regulatory systems, which would lend support to a responsive regulatory approach. However, the Astellas case provides a counter-point to this argument because it offers a glimpse into a criminogenic culture also in a European self-regulatory context, reflected both in the co-ordination of illicit marketing
The second gap pertains to the paucity of empirical analyses of how an ostensibly responsive approach operates
This article proceeds as follows. First, because of the complexities of the case, we provide a synopsis of the PMCPA investigations. After this we describe the methods used. We then proceed with a more forensic analysis of the PMCPA investigations and of the minimal national and international regulatory ramifications of the case. In the final sections we discuss the implications of our findings for pharmaceutical industry regulation, crime control and ethics compliance.
Synopsis of the Astellas Investigations
The chronology of events resulting in Astellas’s ABPI suspension is summarized in Table 1. The case against Astellas was based on two successive complaints that related to off-label promotion of the prostate cancer drug enzalutamide during a
Chronology of Events Resulting in Astellas’s Suspension from the ABPI.
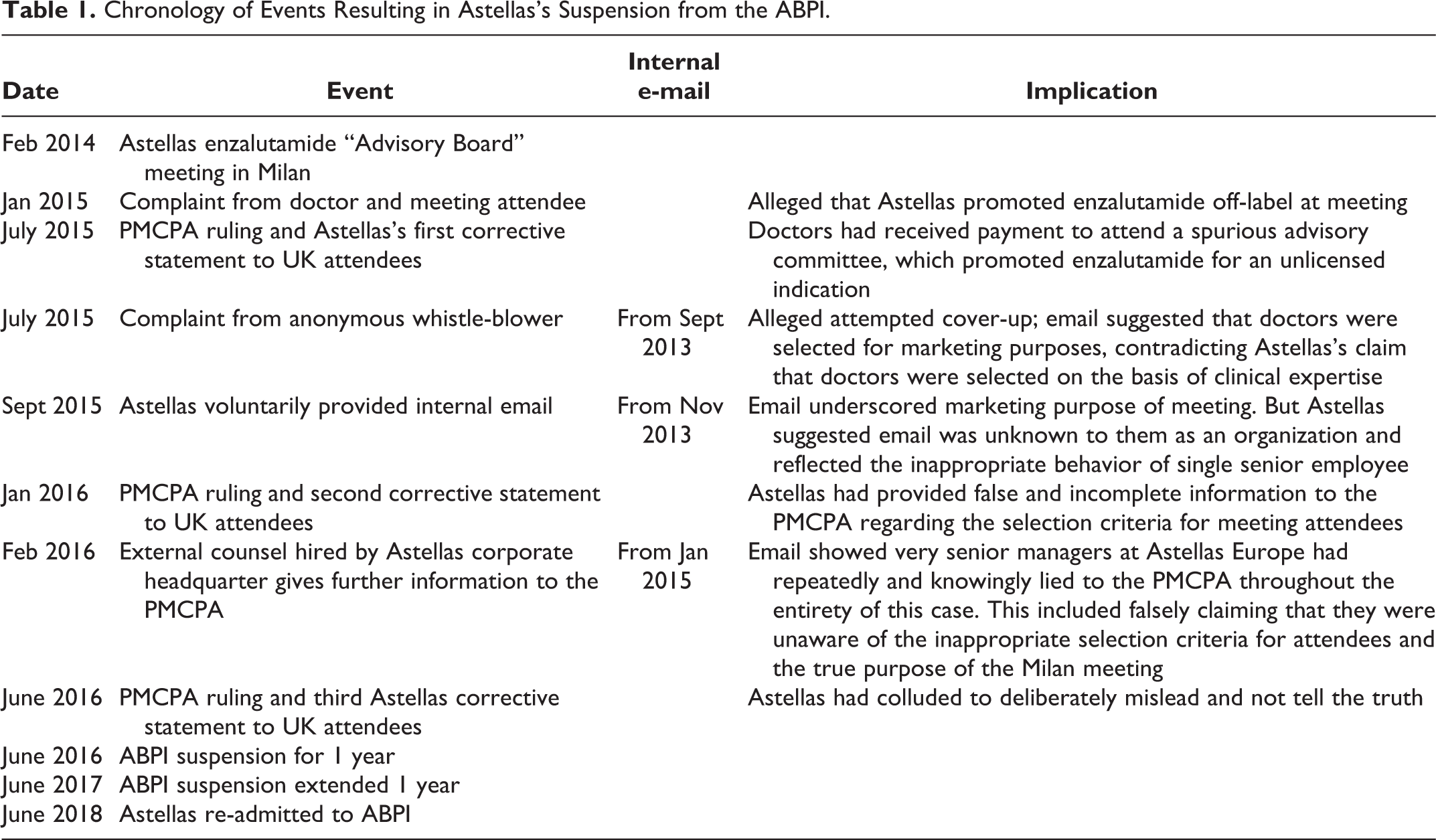
Shortly after the completion of this first investigation, in July 2015, a second complaint was filed with the PMCPA (2015b), this time by an anonymous Astellas Europe employee. This whistle-blower provided an internal company email from September 2013, i.e. from several months
While initially denying wrongdoings, Astellas has fully accepted the findings and rulings, and global company executives have referred to the situation as “a corporate crisis” (PMCPA, 2015b). The company’s official explanation for wrongdoings is one of “significant cultural and compliance failings created and caused by the actions and behaviours of some of its very senior managers” in Europe (PMCPA, 2015b, p. 37). In its report, the PMCPA (2015b, p. 38) attributed Astellas’s gross misconduct and dishonesty to “multiple organisational and cultural failings” within the company, and to a corporate culture that prioritized “the bottom line” over compliance obligations and ethical norms.
Methods
The PMCPA assembles complaints into cases that pertain to alleged breaches of the ABPI Code. Our primary data consists of publicly available case reports of two sequential PMCPA investigations (PMCPA, 2015a, 2015b). In line with previous US research into pharmaceutical industry promotion drawing on documents released via leaks or litigations (Steinman et al., 2006), we analyzed the PMCPA case reports to construct a detailed narrative covering all key events constituting our case as well as the likely causal relationships between them. We also analyzed the data thematically, focusing on (1) the nature of the breach of the ABPI Code; (2) the methods of investigating the case by the PMCPA, including possible strengths and weaknesses; and (3) the arguments and tactics used by Astellas during the course of the investigations.
In addition to documentary research, we contacted, through email, the PMCPA, MHRA, European Federation of Pharmaceutical Industries and Associations (EFPIA, the European pharmaceutical industry trade group) and Astellas Europe with specific questions relating to this case. We received responses from all except for EFPIA, which did not reply to three email requests. This research did not require a full ethics approval by the Social Sciences Research Ethics Committee at the University of Bath; instead it was approved through peer-ethics review.
First Complaint: Anonymous Health Professional vs. Astellas Pharma Europe
International Co-Ordination of Off-Label Marketing Via an “Advisory Board” Meeting
An anonymous UK doctor made the initial complaint to the PMCPA and recounted having participated, together with more than 100 other doctors from various countries in Astellas’s “Pan-European Uro-oncology Advisory Board Meeting” in Milan, Italy on 27–28 February 2014 (PMCPA, 2015a). According to the complainant, the meeting had been a large congregation intended to promote enzalutamide. In June 2013, enzalutamide was approved in the EU for the treatment of adult men with metastatic castration-resistant prostate cancer (mCRPC) whose disease had progressed on, or after chemotherapy. At the time of the Milan meeting the supranational European Medicines Agency (EMA) was reviewing an application by Astellas for an additional “chemo-naïve” indication (approved in December 2014). According to the anonymous complaint, Astellas had used the purported advisory committee meeting to illicitly promote enzalutamide for this “chemo-naïve” use before the EMA’s approval.
In Astellas Europe’s deposition to the PMCPA, the company confirmed that it had gathered doctors from EU countries as well as Turkey, Russia and South Africa for the Milan meeting. It claimed that doctors had been selected solely for their experience of treating patients with mCRPC and not for marketing purpose (PMCPA, 2015a, pp. 6–7). Participants were selected by the company’s local affiliates. For example, Astellas UK had nominated the UK attendees. All attendees were paid €1,000 with the exception of those from South East Europe, who were paid €500, and two speakers who were each paid €1,500.
Astellas Europe also explained that the itinerary of the meeting was such that after an initial joint session with two speakers who presented the benefits of enzalutamide in mCRPC, attendees were split into their respective country/regional advisory board meetings, which were also attended by no more than three Astellas employees from the European organization and the local country affiliate (e.g. Astellas UK). There were 16 such meetings that were attended by 108 “advisors” from 23 countries, including five from the UK. Astellas Europe denied any wrongdoing, and submitted that meetings had been “non-promotional, scientific/medical-led…with an agenda focused on legitimate scientific exchange about the treatment of mCRPC” (PMCPA, 2015a, p. 8).
The company also submitted that this had been the third of a sequence of non-promotional pan-European advisory meetings with a common format. The previous two meetings had taken place before and immediately after the first marketing authorization of enzalutamide in June 2013. Astellas Europe described the meetings as follows (PMCPA, 2015a, pp. 10–11): November 2012 in Barcelona. The stated objective was “to seek advice on best practice in clinical management of advanced renal cell carcinoma (RCC) and castrate resistant prostate cancer relating to tivozanib and enzalutamide respectively” A total of 53 “advisors” attended from: France, Germany, Italy, Spain, UK. July 2013 in Frankfurt. The stated objective was “to seek advice on enzalutamide in metastatic castrate resistant prostate cancer following the first approval of enzalutamide in the post-chemotherapy indication and how enzalutamide could be introduced into clinical practice in the light of the changing treatment landscape.” A total of 61 “advisors” attended from: Austria, Czech Republic/Slovakia, Germany, Greece/Cyprus, Ireland, Netherlands, the Nordics, Poland, Switzerland, UK.
To aid the PMCPA investigation into the allegations regarding the 2014 Milan meeting, the PMCPA asked Astellas Europe to provide additional information, in accordance with established self-regulatory procedures. In particular, the PMCPA requested information on the criteria and process for selecting “advisors” as well as the meeting objectives and schedule. On the basis of the obtained information, the PMCPA found that several manifest aspects of the Milan meeting did not meet the established criteria for advisory meetings, e.g. the time spent obtaining advice was low and no preparation was needed from participants. Instead, the PMCPA considered that the meeting was used to present current data and to encourage participants to assess “the impact of potential promotional claims” related to the “chemo-naïve” off-label indication (PMCPA, 2015a, p. 13) The implication of this was that UK doctors had been paid to attend a meeting that “clearly promoted Xtandi for an unlicensed indication” (PMCPA, 2015a, pp. 14–15).
Because of the severity of the breach, the PMCPA ruled a breach of §2 of the ABPI Code (promotion that “brings discredit to, and reduction of confidence in, the industry”) as a sign of particular censure. According to the PMCPA, “Publicity is the main sanction when breaches of the Code are ruled” (PMCPA, 2018a), and as a matter of policy, when a breach of §2 is ruled, a public reprimand is issued in the form of adverts in the professional press (e.g., The British Medical Journal). In addition, Astellas Europe was asked to issue a corrective letter to all UK attendees. The letter, issued on July 1, 2015, stated “that the arrangements did not meet the criteria for an advisory board and that UK health professionals had received payment to attend the meeting, which promoted Xtandi for an unlicensed indication” (PMCPA, 2015a).
Two additional aspects of this initial investigation need highlighting as they illustrate a possible weakness of the UK self-regulatory system and one key “impression management” tactic used by Astellas vis-à-vis the PMCPA. First, despite unambiguous statements in the ruling, advert and corrective letter that Astellas Europe had promoted enzalutamide off-label, the PMCPA did not actually rule a breach of the clause of the ABPI Code that bans off-label promotion (i.e. §3.2). The reason for this was a procedural rule that constrains the PMCPA to only consider potential breaches of clauses that are cited by the individual member of the PMCPA that prepares the case, the so-called case preparation manager. In the present case, the complainant doctor had alleged that Astellas had promoted enzalutamide
Second, and of relevance to the issue of impression management, at the consideration of the PMCPA’s verdict, Astellas Europe representatives asserted that they fully appreciated and accepted the verdict, and evidenced the company’s commitment “to continual improvement of compliance activities and standards” by citing a long list of measures that it had voluntarily undertaken (PMCPA, 2015a, p. 14). These included: The move of healthcare compliance to the legal department to become the Legal and Compliance Department; growth of the compliance team; updated/new regional policies and procedures including advisory boards; rollout of a global policy for review of materials used to promote to health professionals; Legal and Compliance day; quarterly compliance updates; final signatory training [for the health professional certifying promotional material on behalf of the company]; in-house PMCPA seminar; [ABPI] 2015 Code update; revised ZINC [compliance management solution] process and system training; regional Healthcare Compliance and reporting workshop; face-to-face/on-line training on new regional policies and SOPs; internal monitoring of compliance review and approval process; communication cascade of the Panel’s ruling including the affiliate teams; further case review at quarterly compliance updates; planned training on advisory boards including details of this case including the UK affiliate [i.e., Astellas UK] and the agencies [hired by Astellas Europe] involved in the meeting at issue.
Second Complaint: Anonymous, Non-Contactable Employee Vs. Astellas
Self-Assessed Beneficial or Inaccurate Information by Astellas
The June 2015 corrective statement, the advert, and Astellas’s pledge of commitment to the ABPI Code and extensive list of measures could have been the end of the investigation had additional damaging information not been provided to the PMCPA in July 2015 by an anonymous Astellas Europe whistle-blower (see also Cohen et al., 2019). This employee provided evidence, detailed below, and alleged that the account given by Astellas Europe to the PMCPA in relation to the previous investigation was “knowingly false and intentionally misleading” (PMCPA, 2015b, p. 12). Specifically, in communications with the PMCPA, Astellas Europe had certified that all meeting invitees were selected based on “clinical expertise” and “experience of treating patients with mCRPC,” consistent with Astellas’s argument that the meeting’s purpose was to seek experts’ advice, and not to promote the drug off-label (see above). Yet, according to this whistle-blower “nothing was further from the truth and Astellas knew that but deliberately chose to conceal it from the PMCPA” (PMCPA, 2015b, p. 12).
In support of the allegation of cover-up, the whistle-blower provided a copy of an email (see Table 1) dated 5 September 2013 sent on Astellas Europe behalf by the agency coordinating the February 2014 Milan meeting to the general manager of Astellas Turkey, and copied to a senior employee at Astellas Europe and six others. The email stated that each national company affiliate should provide a list with 30 potential attendees to the Milan meeting who should be “mid to top level opinion leaders
This second complaint initiated a sequence of events that eventually led to Astellas’s ABPI suspension. In defending itself against the cover-up allegation, Astellas Europe first issued a clarification to the PMCPA regarding the selection of meeting attendees, in an effort to justify why it had kept the September 2013 email provided by the whistle-blower from the PMCPA, in violation of self-regulatory procedures. Prompted by the second complaint, Astellas Europe said it had traced the origin of the admittedly inappropriate selection criteria contained in the email to a presentation held at a February 2013 meeting by its Oncology Steering Committee, which consisted of representatives from Astellas’s country affiliates in the Nordic countries, France, Germany, UK, Spain, Czech Republic/Slovakia, Italy and Poland. (PMCPA, 2015b, p. 13) This presentation referred to the aims and suggested meeting attendees (i.e. the customers/local product champions) for the Milan meeting as (PMCPA, 2015b, pp. 2–3):
Objectives for meeting Increase Astellas’s profile in the field of oncology Communicate Astellas’s strategy and oncology pipeline to key target customers Communicate Xtandi [enzalutamide] and tivozanib [an unlicensed drug] data and common set of messages to EU affiliates’ key target customers Gain an increased understanding of the current landscape in RCC [renal cell carcinoma] and prostate cancer & the challenges Astellas will face when launching Xtandi and tivozanib in the EU
Target audience for meeting Mid—top level product OLs [opinion leaders]—those with the potential to be local product champions within the relevant EU markets Data-naïve customers, ie those who have not been involved in any APEL [Astellas Pharma Europe Ltd] or national/local advisory board meetings prior to the pan EU ad [advisory] board meeting 10 per affiliate [e.g., Astellas UK]: 5 prostate/Xtandi and 5 RCC [renal cell carcinoma]/tivozanib
Astellas Europe also reported that it had now uncovered additional emails, very similar to the one provided by the whistle-blower, that were sent to senior Astellas’s employees in many other countries (i.e., not only Turkey) in September and October 2013 (PMCPA, 2015b, pp. 13–14). Furthermore, and of relevance to the PMCPA’s jurisdiction, Astellas Europe acknowledged that on 21 October 2013 a senior UK employee had forwarded the email in its entirety to a management team that consisted of UK medical and commercial members noting that, “we need to get back with nominations of “[city] is one of…main key accounts,” “…one of our high users [prescribers] and would respond well to such a meeting…,” “…is influential at a [city] level and more and more nationally with time” and “This is a business move…barely sees industry, not using prechemo abi [competitor’s product abiraterone] and once using he rarely changes.” [Description of] the reserve nominations included “…I believe he has the reputation with us for being an abi [competitor’s drug] man, however, this would give us the opportunity to convert him to the new way” and “…is on our list and is influential, and would be good to engage at this level.” (PMCPA, 2015b, p. 23)
Astellas Europe defended itself by claiming that while its responses during the previous PMCPA investigation might suggest that there was a conscious decision not to provide the emails with the inappropriate selection criteria and the Oncology Steering Committee presentation to the PMCPA, this had not been the company’s intention: “At no point during the preparation of its [Astellas Europe’s] response…was there a discussion as to whether the emails…or the presentation of February 2013 [of the Oncology Steering Committee] should or should not be submitted; these were not considered at all” (PMCPA, 2015b, p. 20). According to Astellas Europe, this omission might have been due to “the significant time that has elapsed since the email was sent meaning that those preparing the letter of response [to the PMCPA] had no awareness of or had simply forgotten about the existence of the letter” (PMCPA, 2015b, p. 21).
The PMCPA, however, largely rejected Astellas Europe’s claim of involuntary omission and, once again, ruled against Astellas. This time the ruling applied to both Astellas Europe and UK, and stated that the case “raised fundamental concerns regarding Astellas Europe and Astellas UK’s approach to compliance and self-regulation” (PMCPA, 2015b, p. 5). In particular, Astellas had misinformed the PMCPA regarding several important details, for example, by incorrectly declaring that the UK sales force had not been involved in the nomination process and omitted key information related to the selection of meeting attendees; therefore “Astellas had either not paid sufficient attention to ensuring that all relevant information had been supplied in its response…or had made a conscious decision to omit relevant details from that response” (PMCPA, 2015b, p. 4).
The PMCPA also expressed severe doubts on whether the revised selection criteria for the Milan meeting attendees had been effectively circulated to Astellas’s country affiliates and, therefore, “was not at all surprised…that none of its affiliates had subsequently requested any changes to those nominated as attendees” (PMCPA, 2015b, p. 24). On the basis of this new information the PMCPA also expressed concerns about the two previous meetings held in 2012 and 2013 in Barcelona and Frankfurt. However, because there was no complaint about these two meetings, no further investigation was undertaken, in accordance with PMCPA procedures.
Scapegoating by Astellas
Again, this might have been the end of it were it not for two additional internal emails (see Table 1) that surfaced in the ensuing months, and that provided further evidence of the deliberate co-ordination of off-label marketing and deceptive impression management by top-level managers.
The first email was provided voluntarily by Astellas Europe in September 2015 to the PMCPA with the apparent intention of scapegoating a single senior employee. The email was written by an Astellas Europe senior employee and related to the “internal concerns” that had been raised in November 2013 regarding the selection criteria for nominees, and which Astellas Europe had claimed resulted in the “inappropriate criteria” being revised (see above). Although the email demonstrated that concerns were indeed raised internally, as Astellas had declared, its content confirmed that the revised criteria were a
According to Astellas Europe, “the email indicated that there was a conscious decision internal audit preparation; full gap analysis of healthcare compliance program by external consultant at Astellas Europe; review and revision of existing standard operating procedures (SOPs) and policies, development of new SOPs including: Astellas Europe—Complaint Handling SOP and Deviations SOP, UK Affiliate—Advisory Board SOP, advisory board moratorium within Astellas Europe until 31 December 2015; all UK led advisory boards required medical director approval in addition to routine approvers, communication on publication of case report, communication on advisory board toolkit/templates within Astellas Europe; approval for further increase in healthcare compliance headcount; town hall meetings. (PMCPA, 2015b, p. 32)
Report From External Counsel Provides Strong Evidence of Deceptive Impression Management
During the consideration of the audit reports (which are not public) in February 2016 new information relevant to the case was presented to the PMCPA as a result of an “external” investigation (i.e., independent of the PMCPA) ordered by Astellas’s headquarters in Japan to further probe the activities of Astellas Europe (PMCPA, 2015b, p. 8). This “external” investigation had uncovered a
Evidently, the Astellas Europe manager who received the email from the oncology business unit employee had raised it with the very senior managers who were involved in drafting the PMCPA response. This information contradicted Astellas Europe’s earlier claim that it had only later in 2015 re-discovered the “selection criteria email” and that “At no point during the preparation of its response…was there a discussion as to whether the emails…should not be submitted; these were not considered at all” (PMCPA, 2015b, p. 20). The oncology business unit employee email also demolished Astellas Europe’s attempt to scapegoat a single senior manager. Furthermore, the email demonstrated that some very senior managers were perfectly aware of the inappropriate selection criteria for attendees as well as the true purpose of the Milan meeting, i.e. to promote Astellas’s oncology products, including for an unlicensed indication in violation of the law.
The report from counsel attributed this dishonesty to institutional rather than personal failure. The report is confidential but the PMCPA (2015, p. 36) cites from it as saying, “[Astellas Europe] staff considered business concerns prevailed over compliance concerns and that there were no consequences for compliance breaches.” The report furthermore acknowledged, “that the PMCPA criticisms of the Milan meeting were likely to apply to two other advisory board meetings” in Barcelona and Frankfurt; and revealed that “compliance concerns with regard to the Milan meeting were raised by [Astellas’s affiliate in] the Nordic countries.” These two latter points are important as they show how the PMCPA’s investigation was limited with respect to both the activities included (i.e., only the Milan meeting) and—which is more understandable given the PMCPA’s national jurisdiction—to the corporate entities considered (i.e., only Astellas Europe and UK).
Astellas’s Suspension From the ABPI
In its reaction to this new information, Astellas Europe “sincerely apologised for the significant cultural and compliance failings created and caused by the actions and behaviours of some of its very senior managers” (PMCPA, 2015b, p. 37), but at the same time indicated that these individuals had now left the company. It was acknowledged that these managers “had colluded to deliberately mislead and not tell the truth” (PMCPA, 2015b, p. 38). To ensure that this would not happen again the company, for the third time, referred to its plans to intensify internal ethics and compliance efforts. Specifically: the company’s compliance function would no longer be managed locally but would report to the newly appointed Senior Vice President and Head of Ethics and Compliance who in turn reported to the global company. Senior leadership in medical affairs now reported globally and was no longer managed by Europe. Dedicated compliance professionals would also be employed at every European affiliate. (PMCPA, 2015b, pp. 37–38)
Minimal Reaction of MHRA and Other European Countries’ Authorities
Our analysis shows that hundreds of doctors from most European countries were exposed to off-label marketing at the spurious “advisory” meeting in Milan, and most likely at two other meetings. Therefore, it would be reasonable to expect that other European countries’ self-regulatory or government authorities would have followed suit and investigated Astellas. Such investigations should have been facilitated by the reports from the PMCPA and the Astellas counsel, and by Astellas’s admission of guilt. Similarly, because of the pan-European nature of the meeting, and because Astellas Europe is a member of the European industry’s trade group, EFPIA, it would be reasonable to expect that EFPIA had castigated Astellas Europe, possibly in line with the actions taken by the ABPI. However, in response to our queries, spokespersons from the PMCPA and Astellas Europe confirmed that the Dutch industry self-regulatory authority was the only other European authority or organisation to have followed suit. And although the Dutch self-regulatory authority found Astellas guilty of off-label promotion to Dutch doctors, it decided that no further actions were necessary in light of the sanctions already levied in the UK (CGR, 2016).
Furthermore, because the case involves off-label promotion, in clear violation of the law, and because the investigations uncovered an appalling corporate culture and strongly suggested that at least two other “advisory” meetings were dubious, one might have expected the MHRA to have expanded and intensified the investigation. However, the MHRA told us in 2016 that they had deliberately chosen not to scrutinize Astellas as its “informal review of the transparent and robust investigation carried out by PMCPA” did not identify any “failure or omission by PMCPA that would provide a reason to intervene.” The MHRA also stressed that it “would only expect to intervene if there was evidence that self-regulatory sanctions had not achieved an appropriate outcome,” showing that it also considered the disciplinary actions to be proportionate and efficient. However, the MHRA did tell us it “have had concerns about the use of advisory boards for some time and have worked with PMCPA on initiatives to promote compliance across the sector including updated guidance.” This latter quote is important as it shows that Astellas was not unique in using bogus advisory meetings, and that the MHRA was perfectly aware of the nature of the problem but still chose not to intervene in the case.
However, further underscoring that regulators should have taken Astellas’s non-compliance seriously, there have been three additional, major PMCPA rulings against Astellas after its suspension from the ABPI in 2016. First, in May 2017, and while still on probation, Astellas UK was—following a voluntary admission by the company—severely criticized by the PMCPA for, among other things, its “wholly unsatisfactory oversight and control” over two patient support programs that had started in 2006 and 2013, and run until 2016, and that displayed “a lamentable lack of concern for patient safety” (PMCPA, 2016). Then, in June 2017, the PMCPA published details of another, even more alarming case revealing how Astellas had produced a large number of promotional materials that had been used for several years and up until 2017, and which did not include the required prescribing information related to some serious or common adverse reactions and warnings (PMCPA, 2017). The case was based on another voluntary admission by the company, admitting that five promotional materials had omitted requisite prescribing information for one drug. However, the subsequent investigation revealed a large number of non-compliant material for a total of eight drugs. At this point, the MHRA did react, but the reaction was minimal. Thus, the annual report of the MHRA Advertising Standards and Outreach Unit (2019, p. 14) notes how it met with Astellas in early 2018: “We heard about the steps being taken to improve compliance and change the company culture and encouraged them to continue these. We were pleased to see that they had made sufficient improvement to be accepted back into ABPI membership in June.”
The MHRA’s optimism was, however, misguided. Coinciding with Astellas readmission into the ABPI in June 2018, the PMCPA reported Astellas once more to the ABPI Board (PMCPA, 2018b). This new ruling was initiated following complaint by a doctor and—as with the enzalutamide case—pertained both to illicit marketing, in this case providing funding (£50,000) to a hospital which was linked to its use of an Astellas product, as well as “failure to provide comprehensive, accurate information” during the investigation, which the PMCPA called “both inexplicable and inexcusable” in light the company’s “recent and numerous commitments…to upholding the highest standards” (PMCPA, 2018b, pp. 7–8). Following this fourth ruling the MHRA Advertising Standards and Outreach Unit (2019) decided it would vet Astellas’s promotional material, which it started in December 2018, almost 3½ years after the first PMCPA ruling. However, vetting cannot identify, for example, bogus advisory meetings. Furthermore, vetting is not a major sanction since the MHRA routinely vets adverting for new medicines, or medicines with major new indications, or those affected by safety concerns. Thus, arguably, the MHRA’s response was too little, too late.
Discussion
We have argued elsewhere that the seemingly more trivial and limited character of detected promotional violations in the UK compared to US cases may be (at least in part) an artifact of the different regulatory systems—including the relative weakness of self-regulatory bodies’ detective and investigative capacities—as opposed to a true reflection of the nature and extent of illegal marketing practices in the UK (Vilhelmsson et al., 2016). Not only does this case study provide further support for that thesis but also suggests that companies have a relatively large scope for (1) creating ambiguity around the true nature of detected violations, and (2) concealing serious offences from the PMCPA in the absence of in-depth investigations, whistle-blower testimony and direct access to company-internal documents.
Corporate Non-Compliance and Impression Management
The initial complaint—as with the majority of cases dealt with by the PMCPA (Zetterqvist et al., 2015)—was made by someone external to the company. The evidence presented to the PMCPA was, therefore, necessarily limited, e.g. pertaining to the inappropriate selection criteria for meeting attendees. In the absence of direct access to internal informants or documents, a true understanding of the facts of the case was dependent, in the first instance, on the willingness of Astellas to investigate itself and to respond fully and honestly to the PCMPA’s information requests. However, this case study suggests that companies cannot be relied on to conduct a thorough and unbiased internal investigation or to disclose the existence and full extent of their marketing activities. On the contrary, Astellas exploited the considerable discretion companies are able to exercise when responding to complaints, by selectively disclosing documents and emails—first to conceal the true nature of the Milan meeting (and possibly the previous meetings in Barcelona and Frankfurt), and then to scapegoat a single employee.
While the PMCPA and ABPI repeatedly state that the “integrity of self-regulation” is underpinned by an assumption that companies can be relied on to provide “complete and accurate information” (PMCPA, 2015b, pp. 4, 9, 11), this case suggests that it is debatable whether this degree of trust is warranted. Indeed, companies may attempt to deceive regulators by deploying a well-rehearsed discourse of compliance and ethics as part of their “impression management” strategy. Throughout the course of the investigation, Astellas reiterated its official position “that it took its commitments with regard to the Code very seriously” (PMCPA, 2015a, p. 5) and “supported employees who raised concerns” (PMCPA, 2015b, p. 14). But the facts of the case showed the very opposite to be true. At the very least, it seems clear that companies with the
Astellas corporate leaders in Japan apparently reacted to the revealed state of affairs at the 11th hour. After discussions with the PMCPA, the Astellas leadership contracted a counsel whose investigation provided a damaging account of Astellas Europe corporate culture. Revelations included the deliberate nature of the unethical, and most likely illegal marketing activities (probably dating back to at least 2012); the fact that senior managers had colluded to mislead, and then repeatedly lied to, the PMCPA, and in their corrective statements to UK doctors; and that senior managers had deliberately attempted to scapegoat an employee and ignored concerns expressed by more junior employees. Overall, the totality of the evidence revealed “multiple organisational and cultural failings,” and pointed to an institutional culture that prioritized “the bottom line” over compliance obligations and ethical norms. This non-compliance culture extended beyond Astellas Europe, and shaped the activities of the company’s European country affiliates. Disturbingly, with the possible exception of the Nordic countries, Astellas country affiliates failed to challenge the promotional intent and unethical selection criteria of the Milan meeting, or even to amend their selection of participants in light of the revised criteria. Indeed, the PMCPA found that the UK company had reviewed and approved the arrangements. Significantly, the non-compliance culture also extended beyond the particular case of enzalutamide, as evidenced by the three subsequent PMCPA rulings involving numerous activities and drugs.
Regulatory Failure
The literature on responsive regulation envisages self-regulation as part of a broader, coordinated and
To bring home these points, it is instructive to compare the UK response to Astellas’s manifest illicit promotion and cover ups to that of US authorities which are significantly more probing and punitive. In the USA in 2014, Astellas paid $7.3 million to resolve allegations that, between 2005 and 2010, it off-label promoted the anti-fungal drug Mycamine for pediatric use, which was not an approved indication. The lawsuit was brought by the Department of Justice following a whistle-blower suit filed by a previous sales representative for the company who alleged that Astellas placed particular pressure on sales persons to sell Mycamine to children’s hospitals for use in children without warning of certain health risks, including the risk of liver tumors. According to the Department of Justice (2014) press-release announcing the settlement, the fine was intended to send a “message that should resonate with all drug companies: there are consequences for violating the False Claims Act and putting profit ahead of government safeguards.” Indeed, in 2019 the Department of Justice announced a new settlement ($100 million) regarding alleged Medicare fraud through Astellas’s funding in 2013 of two charities that would illegally subsidize enzalutamide to increase its use, so called kickbacks—which, again, was said to send a clear “adversarial” message across industry, namely that “the government will hold accountable” and “the FBI will aggressively go after drug companies that look to bolster their drug prices by paying illegal kickbacks” (Department of Justice, 2019).
In addition to the lack of any truly significant escalation (i.e. beyond suspension from the ABPI and, eventually, promotional vetting by the MHRA), our analysis reveals a manifest failure of
Finally, the Astellas case provides further evidence for the contention that instances of illegal or unethical marketing are likely to be
Beyond Responsive Regulation
In sum, the Astellas case provides further support for our thesis that limitations of the self-regulatory apparatus in the UK contribute to an impression that illegal pharmaceutical marketing is infrequent and benign. It was only following a second complaint to the PMCPA by an Astellas whistle-blower that any ambiguity concerning the true purpose of the Milan meeting was resolved. Yet, incentives and protections for whistle-blowers in the UK are weak compared to the US (Cohen et al., 2019), a fact that is underscored by the low number of whistle-blower complaints in the UK (Vilhelmsson et al., 2016). Consequently, as the initial case against Astellas indicates, it is possible that PMCPA inquiries may be prematurely closed in the mistaken belief that the true facts have been established and matters have been adequately resolved. This case study, therefore, also provides support for previous arguments—including by Braithwaite and colleagues (Dukes et al., 2014)—for exporting US-style financial incentives and government protections for whistle-blowers, coupled with in-depth government-led investigations and enforcement actions, in order to uncover the true nature and scope of corporate crime (Vilhelmsson et al., 2016). Although a US-style approach would appear to run counter to most key tenets of responsive regulation—including by “re-centering” the state, collapsing the enforcement pyramid, and criminalizing corporate maleficence in both discourse and practice (Tombs, 2015)—the “adversarial” approach, we suggest, is preferable to a self-regulatory approach that evidently shields companies from government enforcement actions and, consequently, the associated public scrutiny and negative publicity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Swedish Research Council for Health, Working Life and Welfare (FORTE) [grant no. 2016-00875 to SM and PO].
Open Research Statement
This research was not pre-registered. The data and materials used in the research are available upon request by emailing
