Abstract

As the leading malignancy in women worldwide, breast cancer (BC) imposes a considerable health burden due to its high incidence and fatality rates. 1 Germline BRCA1/2 gene variants are well-known high-risk factors for the causation of BC. The cumulative risk for a BRCA1/2 mutation carrier is significantly elevated, reaching up to 70% by the time they are 70 years old. 2 With an emphasis on a deeper understanding of the genetic drivers of the disease and extension beyond the well-known, highly penetrant BRCA1 and BRCA2 mutations, the American Society of Breast Surgeons, in 2019, achieved a major milestone in the field of oncology by advocating universal genetic testing for all BC patients. 3 Emerging evidence has highlighted the critical role of pathogenic variants in a range of other genes, such as TP53, CDH1, PALB2, STK11, PTEN, CHEK2, Ataxia telangiectasia mutated (ATM), BARD1, BRIP1 and RAD51D, in elevating BC risk. 4 Indeed, studies show that over one-third (35%) of individuals with an inherited cancer-related mutation have variants in non-BRCA genes. 5 Such compelling data has led the latest National Comprehensive Cancer Network (NCCN) to expand its screening guidelines to include a more comprehensive list of genes, including ATM, BARD1, BRIP1, CDH1, CHEK2, MSH2, MSH6, MLH1, PMS2, EPCAM, NF1, PALB2, RAD51C, RAD51D and STK11. 6 The management of patients with these variants is now based on careful consideration of their individual lifetime propensity for BC. The ATM gene is particularly noteworthy, as it represents one of the most frequently identified non-BRCA pathogenic variants in BC. 7 New research has highlighted the pathogenicity of ATM gene variants in the early development of BC, notably before 50 years of age.8, 9 A meta-analysis by Marabelli et al. concluded that inheritance of deleterious ATM gene variants steadily augments the cumulative incidence of BC. The researchers reported that 6 out of every 100 individuals with pathogenic ATM variants will have developed primary BC by the time they reach age 50, and 33% by age 80. 10
We report a case of invasive breast carcinoma in a patient who, despite a notable family history of breast and prostate cancers, tested negative for germline variants in the BRCA1 and BRCA2 genes. However, a broader panel for germline screening of hereditary cancer revealed an ATM gene variant. The case underscores the critical need for expanded screening in relevant clinical contexts.
Case Presentation
The patient, a 58-year-old postmenopausal female, sought medical attention for a palpable lump in her left breast. A magnetic resonance (MR) mammogram revealed a spiculated mass in the left breast at the 2 o’clock position, measuring 2.5 × 2.1 × 2.1 cm, with left axillary lymphadenopathy. A tru-cut biopsy revealed an invasive breast carcinoma of no special type. The tumour cells were strongly positive for oestrogen receptor (ER) and progesterone receptor (PR) but did not show any immunoexpression with Human Epidermal growth factor Receptor 2 (HER2) (Score 0). The Ki-67 mitotic labelling index was 40%. The patient underwent lumpectomy with axillary lymph node dissection and was counselled for germline BRCA1/BRCA2 testing as a part of universal screening guidelines. 6 During genetic counselling, a detailed family history was undertaken (Figure 1: Pedigree).
The Pedigree Chart Shows Family History of Breast Cancer on the Maternal Side and Prostate Cancer on the Paternal Side.
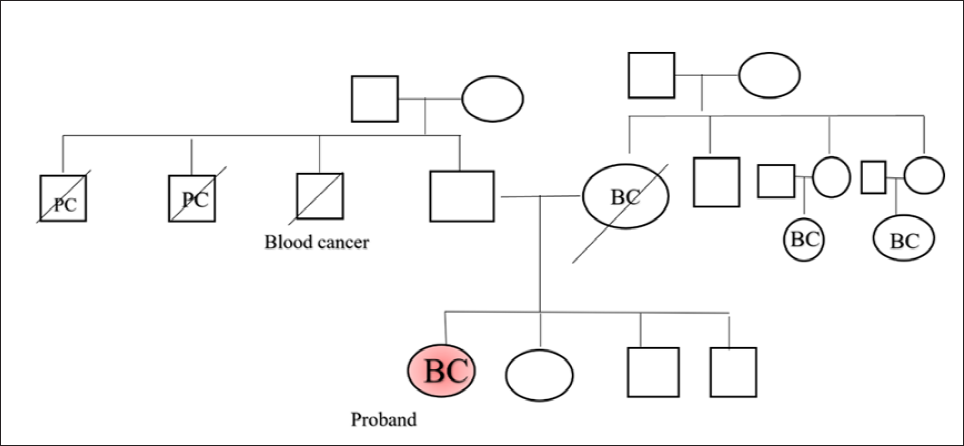
DNA was extracted from a sample of the patient’s peripheral blood using the Qiagen DNA extraction kit (Qiagen DNeasy Blood and Tissue kit; Qiagen NV, Hilden, Germany). An amplicon-based Next-Generation Sequencing (NGS) library was then prepared with the Devyser BRCA kit (Devyser AB, Hägersten, Sweden) to analyse the BRCA1 and BRCA2 genes. Genetic testing of the BRCA1/2 genes revealed no evidence of pathogenic or likely pathogenic variants. However, because the patient had a significant family history that included multiple first-degree relatives with breast, prostate and haematological cancers (Figure 1), an expanded genetic test beyond BRCA was performed. The panel included 37 cancer predisposing genes, interrogating single-nucleotide variants and copy number alterations. A germline deleterious variant was detected in the ATM gene [(NM_000051.4):c.6154G>A ;p.(Glu2052Lys)] at a variant allele frequency of 45%. The patient received post-test counselling and was advised to extend testing to all first-degree relatives. The patient underwent neoadjuvant chemotherapy consisting of four cycles of anthracyclines and twelve cycles of paclitaxel over a period of four months. She has been maintained on a combination of Ribociclib and Anastrozole for the past 1.5 years, with no evidence of disease recurrence, and continues on regular follow-up without any reported complications.
Deleterious variants in these genes are linked to Hereditary Breast and Ovarian Cancer (HBOC) syndrome, which significantly elevates the risk for breast and ovarian cancers in both men and women. Inherited pathogenic variants also confer a moderately increased predisposition for other malignancies, including prostate, pancreatic and melanoma, particularly in individuals with a BRCA2 variant. Research has consistently documented a high lifetime risk of primary BC for carriers of a pathogenic or likely pathogenic variant, ranging from 60% to 72% for BRCA1 carriers and 55% to 69% for BRCA2 carriers. 11
The current NCCN guidelines outline specific testing criteria for BRCA1/2 susceptibility genes in BC patients 6 (Figure 2).
Testing Criteria for BRCA1/2 in Breast Cancer (NCCN Guidelines 2024).

Breast Cancer Susceptibility Genes.

Consequently, multigene panel testing is frequently recommended for individuals in the following circumstances:
When, even in the presence of a known genetic variant, family history indicates a different syndrome. Who meet the testing criteria but have no identifiable disease-causing variant within the pedigree. Who fulfils the testing criteria even with a negative prior single-gene analysis.
Eligibility for multigene panel analysis also extends to close familial relationships of individuals who qualify for testing criteria, the exception being second-degree relatives of pancreatic or prostate cancer patients.
ATM is a moderate-risk BC susceptibility gene, other than the much prevalent BRCA1/2, with a two- to thirteen-fold elevated likelihood of developing BC in heterozygous carriers. A documented cumulative risk of BC in ATM mutation carriers has been reported as 16%-43% by age 80 years. 14 The ATM V2424G variant has been identified as a major risk factor for BC, conferring the highest risk among all known variants.
Beyond BC, pathogenic ATM variants also confer an enhanced predisposition for a broad spectrum of other malignancies, which include pulmonary, gastro-oesophageal tract, colorectal, renal and urinary tract, prostate, hepatic, endometrial, ovarian, salivary gland and thyroid. 15
Conclusion
The present case emphasises the multigene hereditary panel institution in an appropriate clinical setting of a significant family history and an early onset of cancer. The extended testing in view of a strong family history of cancers identified a moderate penetrant pathogenic ATM variant in the present case, which initiated the cascade testing and thus identification of at-risk members of the family.
Footnotes
Data Availability
The data shall be made available as per request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
No ethical approval was taken as it was not required for the study.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Informed consent has been taken from the patient for the publication of this article.
