Abstract
Introduction
Data on mortality trends for colorectal cancer (CRC) in young adults is sparse. So, this study aims to evaluate trends in CRC-related death rates in young adults.
Methods
Data on mortality from the Centers for Disease Control and Prevention (CDC) Wide-Ranging Online Data for Epidemiologic Research database were examined in this retrospective cohort analysis. We focused on young adults aged 25 to 45 who died with colorectal cancer (CRC) listed as an underlying cause from January 1999 to December 2020. The study was conducted in August 2023. Individuals aged 25 to 45 years with colorectal cancer (CRC) are noted as a factor in or the root cause of death. The study measured age-adjusted mortality rates (AAMR) for colorectal cancer (CRC) per 100,000 people in the United States, categorized by sex, ethnicity, race, and geographical location.
Results
From 1999 to 2020, there were 37,601 deaths related to colorectal cancer (CRC) among young adults. Of these, 20,621 (54.8%) were men, and 16,980 (45.2%) were women. The deaths included 7,039 (18.7%) Black individuals, 4,667 (12.4%) Hispanic individuals, and 23,684 (63.0%) White individuals. The overall age-adjusted mortality rates (AAMR) for colorectal cancer (CRC) among young adults rose from 2.0 in 1999 to 2.4 in 2020. Both adult women and men had an increase in mortality, with males consistently exhibiting higher age-adjusted mortality rates (AAMRs). All ethnic and racial groups saw an increase in age-adjusted mortality rates (AAMR), with Black individuals having the highest rates (3.2 in 1999 to 3.3 in 2020). During the same period, the age-adjusted mortality rates (AAMRs) of White and Hispanic people increased from 1.8 to 2.4 and 1.5 to 2, respectively. age-adjusted mortality rates (AAMRs) were either steady or dropping for most demographic and regional categories between 1999 and 2012; however, between 2012 and 2020, there was an uptick. States like Mississippi, Arkansas, Alabama, Oklahoma, and West Virginia were in the upper 90th percentile rank for colorectal cancer (CRC)-caused mortality, reflecting a higher burden in comparison to states in the lowest 10th percentile rank.
Conclusion
Young adults in the United States had higher rates of colorectal cancer (CRC) death between 1999 and 2020; these rates first stabilized before rising in the latter years. Black individuals faced a 1.38 to 1.65-fold higher age-adjusted mortality rates (AAMR) compared to Hispanic and White individuals, respectively. Targeted health policies are essential to address the rising burden of colorectal cancer (CRC) in young adults, emphasizing prevention, early detection, treatment, and reducing disparities.
Introduction
In the United States, colorectal cancer (CRC) ranks third in terms of cancer-related death among both males and females; furthermore, globally, it ranked second as the cause of mortality in men less than 50 years of age. 1 Although the age-standardized rates have been declining since the 1990s, it is paradoxically rising in young people <50 years, most likely due to low rates of screening among them2–4 and the increasing risk factors like Western diet, less fiber, red meat, obesity, smoking, and alcohol use. 5 Research has indicated differences in the occurrence of CRC among young adults, particularly in gender and tumor location. Rectal tumors are found more frequently in males, while females exhibit a higher likelihood of right-sided tumors. When examining age-specific incidence rates in conjunction with anatomical subtypes, it is observed that North Americans have a higher prevalence of rectal cancers. In contrast, in Europe, colon cancer occurs more often. Investigating the rise in CRC cases among young adults and understanding the disease’s stage at presentation is a crucial aspect that has yet to be thoroughly explored. It would be interesting to see if the younger demographic has any changes in the presentation stage or migration across stages of colorectal malignancies. 6 With the modern era and growing resources, incidence rates of CRC are declining in the United States, but disparities in screening and treatment still exist. This descriptive study sought to assess CRC death rates in the United States, spanning 1999 to 2020. The primary focus was on examining trends based on sex, race/ethnicity, region, gender, and location of death. The objective was to analyze how mortality trends for CRC varied among different population groups, providing insights into the occurrence of CRC-related deaths.
Methods
Study Setting and Population
The CDC WONDER (Centers for Disease Control and Prevention Wide-Ranging Online Data for Epidemiologic Research) database provided the death certificate data used in this descriptive analysis.
The data, spanning from 1999 to 2019, was examined to assess CRC-related mortality in young adults. The following International Classification of Diseases, 10th Revision (ICD-10) codes were used to identify CRC-related deaths: C-18 for colon cancer, C-19 for rectosigmoid junction cancer, and C-20 for rectum cancer. 7
This dataset, which includes causes of death from death certificates, has been utilized in numerous researches to investigate cancer mortality trends. The study focused on death certificates that listed CRC as either an underlying or contributing cause of death. Individuals between the ages of 25 and 45 at the time of death were considered young adults.
As the study made use of a government-provided de-identified public use dataset, it was excused from local institutional review board clearance and adhered to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines for reporting.
To calculate the age-adjusted mortality rate (AAMR), each age group’s age-specific death rate was multiplied by the equivalent weight from a reference population. All age group results were added up and then multiplied by 100,000.
The exact formula used was:
Age-adjusted mortality rate=(∑(Age-Specific death rate×standard population weight))×100,000
The age-specific death rate was determined by dividing the number of deaths in an age group by the population of that age group:
Age-specific death rate=Number of deaths in an age group/population of age group
The standard population weight for an age group was calculated by:
Standard population weight=Population of age group/Sum of population of all age groups
To evaluate yearly national trends in CRC-related mortality, the study used the National Cancer Institute’s Joinpoint Regression Program (version 4.9.0.0).
Using log-linear regression models to account for time variations, this program calculated the annual percent change (APC) in AAMR, along with 95% confidence intervals (CIs). This approach allowed for the identification of significant changes in AAMR over different periods. With the use of this method, notable variations in AAMR throughout various periods might be found.
Data Abstraction
Data on population size, year, location of death, demographics, urban-rural classification, region, and state were extracted. Demographic data included sex, age, race/ethnicity, and location of death, which encompassed medical facilities (outpatient, emergency room, inpatient, death on arrival, or unknown status), home, hospice, and nursing home/long-term care facilities. Race/ethnicity was categorized as non-Hispanic (NH) White, NH Black or African American, Hispanic or Latino, NH American Indian or Alaska Native, and NH Asian or Pacific Islander. This information was derived from death certificate reports and has been used in previous analyses of the WONDER database. Based on the 2013 U.S. Census classification, 8 the National Center for Health Statistics Urban-Rural Classification Scheme was used to classify populations by urban (large metropolitan area [population ≥ 1 million], medium/small metropolitan area [population 50,000-999,999]), and rural (population <50,000) counties. The U.S. Census Bureau’s designations of the Northeast, Midwest, South, and West were used to categorize the regions. 9
Statistical Analysis
We computed both crude and AAMRs per 100,000 population yearly, segmented by year, sex, race/ethnicity, state, and urban–rural status, with 95% CIs, to evaluate national trends in CRC-related mortality from 1999 to 2019.
The number of deaths from CRC each year divided by the corresponding U.S. population yielded the crude mortality rates. By normalizing CRC-related fatalities in the U.S. population in 2000, AAMRs were calculated. 9 .
To evaluate national trends in CRC-related mortality annually, we used the Joinpoint Regression Program (Joinpoint V 4.9.0.0, National Cancer Institute), which calculates the APC in AAMRs with 95% CIs. 10 This program identifies significant shifts in AAMR over time by fitting log-linear regression models to periods where temporal changes were evident. An APC was deemed increasing or decreasing if the slope of the mortality trend was significantly different from zero, assessed using a two-tailed t-test. A P value of less than .05 was considered statistically significant.
Results
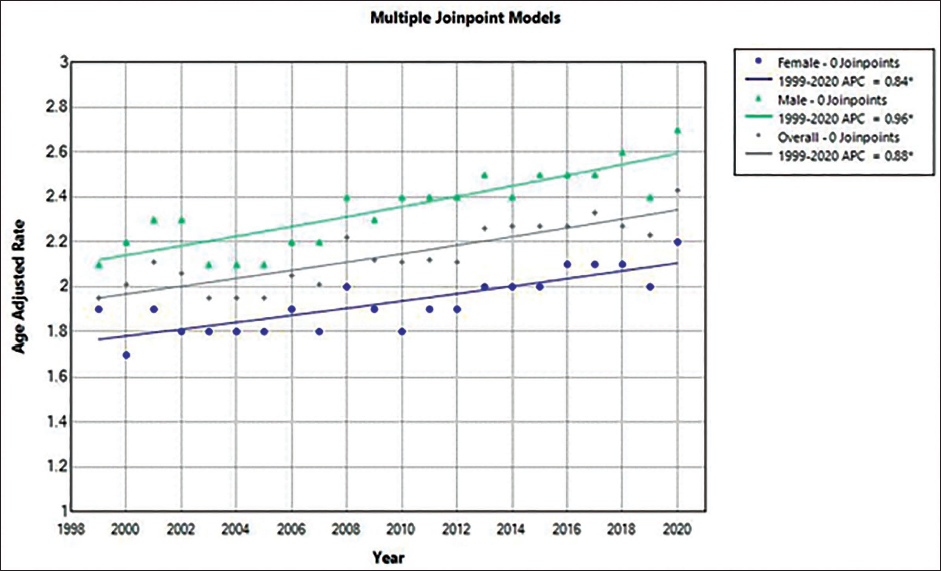
A total of 37,601 fatalities attributable to CRC occurred in young adults between 1999 and 2019. Of these, 20,621 (54.8%) were men and 16,980 (45.2%) were women, and 7,039 (18.7%) were Black, 4,667 (12.4%) were Hispanic, 23,684 (63.0%) were White, 328(0.9%) were Alaskan Indian or American Native, and 1,799(4.5%) were Asian or Pacific Islanders. Total 13,768 (36.8%) of the 37,458 deaths for which the location of death was known happened in medical facilities, 1,670 (4.5%) in nursing homes or long-term care facilities, 3311 (8.8%) in hospice care, and 16,459 (44.0%) happened at home (eTable 6 in Supplemental). Throughout the study, the AAMR was 2.2. Men’s AAMR was greater than women’s (2.3 vs. 1.9), and Black adults’ AAMR was higher than that of Hispanic and White adults (3.1 vs. 1.5 vs. 2.2, respectively).
Overall, from 2.0 in 1999 to 2.4 in 2019, the AAMR for CRC in young adults rose in 2019 (Figure 1; eTable 2 in Supplemental). AAMR increased from 1999 to 2019 (APC, +0.9% [95% CI, 0.6-1.2]). The death rate from CRC rose in young men and women. APC, 1.0% [95% CI, 0.7-1.3]) shows that the AAMR for men went from 2.2 in 1999 to 2.7 in 2019, whereas the AAMR for women climbed from 1.7 in 1999 to 2.2 in 2019 (APC, 0.8% [95% CI, 0.5-1.2]). The highest AAMR was seen in Black adults, who saw an increase from 3.2 in 1999 to 3.3 in 2019 (APC, –0.17% [95% CI, –0.0.8 to 0.5]). Adult White and Hispanic individuals saw an increase from 1.5 to 2. For Hispanics, it remained the same from 1999 to 2012 (APC, 0.3 [95% CI, –5.5 to 1.5]) and increased from 2012 to 2019 (APC 4.03% [95% CI, 2.0-11.9]). For White, it increased from 1999 to 2014 (APC 1.8% [95% CI, 1.3-9.4] and remained same from 2014 to 2019 (APC –0.07 [95% CI –5.5 to 1.5]) (Figure 2; eTables 4 and 5 in the Supplemental). Nonmetropolitan areas had the highest AAMR, which increased from 2.6 in 1999 to 3.1 in 2019 (APC 1.0% [95% CI: 0.4-1.6]). Mortality rates similarly increased in the large and medium/small metropolitan areas during the study period (Figure 3; eTable 8 in the Supplemental). The burden of CRC-related mortality varied greatly by region, with states in the upper 90th percentile (Mississippi, Arkansas, Alabama, Oklahoma, and West Virginia) having a mortality load that was noticeably higher than those who fall within the lowest 10 percentile (Wisconsin, Montana, Wyoming, Alaska, Connecticut) (eTable 7 in the Supplemental).
Trends in Age-adjusted Mortality Rates of Colorectal Cancer Among Young Adults in the United States, 1999-2020, Sorted by Sex.
Trends in Age-adjusted Mortality Rates for Colorectal Cancer Among Young Adults in the United States From 1999 to 2020, Both Overall and Grouped by Race and Ethnicity.
Trends in the United States Among Young Adults for Colorectal Cancer in Terms of Age-Adjusted Mortality Rates Segregated by State and Region.
Discussion
The 20-year CRC mortality data obtained from the CDC WONDER database was analyzed, revealing several notable results. A steady rise in the mortality rate was noted. From 1999 to 2019, in the younger adults, the APC was 0.9%. Furthermore, throughout the two-decade span, it was consistently observed that men had a higher AAMR in comparison to women.
Distinct racial and ethnic groups displayed varying CRC outcomes over the 20 years. The patients with the highest AAMR were always African Americans, after White patients, Asian or Pacific Islanders, American Indian or Alaska Native patients, and Hispanic or Latino. 11 These disparities were partly caused by inequalities in the prevalence of risk factors and access to healthcare. The higher CRC incidence among Black men in comparison to White individuals likely results from a complex interaction between socioeconomic, lifestyle, genetic, and health inequality-related factors.
Current data suggests a historical gap between CRC screening rates among Black and White individuals, 12 although recent trends indicate a gradual narrowing of this disparity. The decrease in CRC cases among African Americans might be largely attributed to their increased use of colonoscopic screening. African Americans continue to have a higher incidence of CRC, which is linked to variables such as a lack of knowledge about ancestors’ cancer history and a reluctance to disclose colonic polyps. 13 Furthermore, the likelihood of a CRC diagnosis occurring before the age of 50 is higher in African Americans, highlighting the shortcomings of earlier screening recommendations.
African Americans are more susceptible to CRC due to several additional factors, such as genetic susceptibility and presentational variations. For example, Mendelsohn et al. 14 found a higher prevalence of advanced adenomas among African Americans, though these findings were not always replicated. African Americans have historically presented with more advanced metastatic colon cancer at diagnosis, possibly due to a greater likelihood of proximal CRC, which is harder to detect through colonoscopy. According to Lieberman et al., 15 African Americans have a higher prevalence of advanced-stage polyps in the proximal site, which supports this finding.
The US Multi-Society Task Force on Colorectal Cancer initially suggested screening for CRC at 50 years of age for average-risk individuals 16 ; other organizations like the American College of Gastroenterology and the American College of Physicians suggested earlier screening for African Americans. The United States Preventive Services Task Force (USPSTF) updated guidelines in 2021, recommending screening at 45 for average-risk individuals. 17 Collaborative efforts and advancements in treatment modalities have significantly improved AAMR, particularly in African American patients, within the last two decades. Declining AAMR has also been attributed to innovations in screening methods such as fecal occult blood testing, sigmoidoscopy, colonoscopy, and computed tomographic colonography.
The lower AAMR consistently observed in females during the study period compared to males may be attributed to multifaceted factors. This could be due to various reasons. Hormone replacement therapy (HRT) has been associated with a decreased risk of tumor instability. 18 Postmenopausal women using HRT experienced a 40% decrease in the risk of CRC. 19 However, among women with CRC already diagnosed, those undergoing HRT tended to have more advanced cancer stages. Overall, prior and ongoing HRT use may lower the risk of developing CRC, whereas prolonged exposure to natural estrogen after menopause might heighten this risk in women.20, 21
CIMP-high (CpG island methylator phenotype-high) incidence increased from the rectum to the cecum, where tumors were more common in females. 22 The p161NK4a tumor suppressor gene’s 5’ region contains methylated CpG island, which has been related to the gender of women. 23 The location of the tumor affects the correlation between dietary variables and CRC. In women, an increased intake of carbohydrates is linked to a greater risk of right-sided colon cancer, whereas in men, it has been tied to an increased risk of rectal cancer. 24 Moreover, elevated fat and protein consumption heightens the risks of developing CRC on the right and left sides, respectively.25, 26 Increased consumption of polyunsaturated oil, cholesterol, trans-fat, sugar, and lactose has been linked to an elevated risk of colon cancer on the right side. 27
Higher levels of total iron and supplemental iron are inversely linked to cancer of the distal colon. Moreover, there is an inverse relationship between distal colon cancer and greater serum levels of 25-hydroxyvitamin D and increased calcium intake. 28 Estrogen’s potential role in lowering CRC risk in women is recognized, although the exact mechanism remains uncertain. 29
This study finds that the AAMR in the young population has increased with the passing years till 2019, which will create a burden on the health system of the United States. Understanding the causes and mechanisms behind early-onset colorectal cancer (EOCRC) remains a challenge, particularly since many young individuals with the disease lack known risk factors. Between 16 and 35% of EOCRC cases are observed in individuals with hereditary cancer syndromes, predominantly Lynch syndrome. 30
The percentage of young adults receiving a diagnosis of CRC at advanced stages is higher, possibly due to the absence of regular screening, limited understanding among patients and healthcare providers, and insufficient focus on the warning signs of early commencement of CRC. Compared to those born in 1950, 40-year-olds born in 1970 had a greater prevalence of CRC in the United States (24.4 per 100,000), where 18.3 per 100,000 people is the rate. The birth cohort effect suggests that certain factors during early life may increase the risk of EOCRC. The development of CRC has been linked to Western diets, which are high in processed and fatty foods, sugary drinks, and desserts and low in fiber and micronutrients like calcium, vitamin D, beta-carotene, and vitamin E. 31 It also creates a need for health-oriented guidelines improvement over the increasing risk factors that can be modified and restricted to lower the incidences of CRC. Moreover, initiation of screening should be done in people less than 50 years old who present with risks of developing CRC. This data will lead to awareness among the people and contribute to strategically targeting the resource for better outcomes.
Considerable ethnic and racial differences have also been observed as AAMR among Black adults increased almost 1.6 and 0.9 times higher than Hispanic and White adults, respectively, and the rate is higher among Hispanic and White adults from 1999 to 2019. Bitterly, race is also one of the factors that care providers do not recommend screening for Black people. 32 Socioeconomic status and health insurance status have been identified as a cause of healthcare differences for Black people, and some studies show that Black people are less treated with radiation and chemotherapy than White people. 33 Other than that discrepancies among screening and the stage-specific relative CRC survival are thought to cause differences in healthcare. 34
The burden of CRC-related death varied greatly by region, with states in the upper 90th percentile of CRC-related death (Mississippi, Arkansas, Alabama, Oklahoma, and West Virginia) having a substantially greater death load than individuals in the bottom 10th percentile (Connecticut, Montana, Wyoming, Alaska, Wisconsin). The large geographical variation that is found in this study can be due to the access to the outpatient CRC department, the effects of the state law in the expansion of Medicare and Medicaid, and the presence of comorbidities that largely impact the outcome of the disease.
Health policy plays a key component in approaching better health by screening, diagnosing, and treating the disease. Between 1987 and 2010, screening is thought to have prevented over 500,000 new instances of CRC. 35 Affordable care act (ACA) was aimed to decrease the cost-effective coverage for the diagnostic screening of CRC, but still, it contains legal restrictions that cause disparities in CRC outcomes for Medicare beneficiaries. So, improved health policies are required because they will allow low-income individuals to have a more accessible approach to being screened and treated. 36 Fecal blood tests (FBTs) using a guaiac fecal occult blood test (gFOBT), colonoscopy, flexible sigmoidoscopy, computed tomographic colonography, and/or fecal immunochemical (FIT) test, with or without multitarget stool DNA, 37 are among the various US-guided medical screening modalities for diagnosing CRC that should be aggressively implemented. Patient navigation after the confirmed diagnosis is the barrier to providing care, so a trained individual can fully guide the patient in overcoming the social, complex healthcare, and educational barriers to minimize the loss of follow-up. 38
Data Availability Statement
The CDC WONDER database, developed by the CDC in the United States, is an open-access online platform that offers a wide range of public health data. The database is freely accessible, enabling users to easily obtain information through its website.
Limitations
This dataset relies heavily on information from healthcare providers, which can introduce potential errors or inaccuracies, particularly with key variables like race and ethnicity. Additionally, the rise in digital health records documenting CRC diagnoses might increase CRC reporting on death certificates, potentially inflating mortality trends and distorting actual trends in CRC mortality among young adults by conflating statistics or comorbidities.
Moreover, the absence of detailed data on individual risk factors or potential confounders influencing CRC development or progression restricts the comprehensive analysis of complex relationships between variables. Our data indicates an increase in AAMRs among White individuals under the age of 55. Although EOCRC typically refers to cases diagnosed before age 50, our findings showed a significant rise in this age group, which we included in the study despite not strictly meeting the EOCRC criteria. We acknowledge the complexities of grouping diverse populations, such as categorizing Pacific Islanders and Asians together, as different subgroups within this category may exhibit varied mortality rates. However, it’s important to note that this analysis might not fully capture these differences due to the grouping method used.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Ethical Approval
This study was exempt from local institutional review board approval because it used a de-identified government-issued public use data set and followed the STROBE guidelines for reporting.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
