Abstract
Introduction
This retrospective study investigates the clinical features, survival outcomes, and treatment-related results of patients with soft tissue sarcoma (STS) at a tertiary referral center in India. STSs are rare malignant tumors comprising over 50 subtypes, representing approximately 1% of adult and 15% of pediatric neoplasms.
Materials and Methods
We reviewed 91 patients with STS diagnosed in the Rajiv Gandhi Cancer Institute and Research Center, India, from 2020 to 2023. Data were collected on demographics, disease characteristics, and treatment outcomes. Statistical analyses were performed with survival rates estimated via the Kaplan–Meier method. Prognostic factors were evaluated through univariate and multivariate analyses using log-rank and Cox regression models.
Results
The study cohort included slightly more males (64.8%) than females, with 51.6% of patients under 50 years. Most tumors were over 5 cm (70.3%) and classified as Fédération Nationale des Centres de Lutte Contre le Cancer (FNCLCC) grade 3 (62.6%). Local recurrence occurred in 7.7% of cases, and distant recurrence occurred in 24.2%. The 3-year overall survival rate was 60.5%, with a 3-year disease-free survival rate of 58%. Significant prognostic factors for better outcomes included smaller tumor size (≤5 cm), margin status (≥1 cm), lower necrosis, lower intraoperative blood loss (<100 ml), and administration of adjuvant chemotherapy. Multivariate analysis showed only intraoperative blood loss to be significant.
Conclusion
This study highlights the necessity of multidisciplinary care in STS management. Achieving wide surgical margins with minimal blood loss followed by multimodality treatment is crucial. Future prospective trials with larger, diverse cohorts are essential to validate these findings and optimize STS treatment strategies.
Keywords
Introduction
Soft tissue sarcomas (STSs) are infrequent malignant tumors arising from mesenchymal tissues, encompassing over 50 distinct subtypes. 1 These tumors constitute approximately 1% of all adult neoplasms and 15% of pediatric tumors. 2 Multiple authors have reported an escalating incidence rate of STS. 3 The upward trend in incidence rates is likely attributable to recent alterations in diagnostic criteria. Treatment approaches for STS vary significantly based on factors such as tumor location, size, histology, and staging. Generally, the initial intervention for nonmetastatic extremity sarcoma involves complete surgical excision, often supplemented by preoperative or adjuvant radiation therapy to reduce local recurrence risk. Optimal treatment determination is intricate and ideally involves a multidisciplinary team comprising surgeons, medical oncologists, radiation oncologists, pathologists, and radiologists. 4 The necessity for specialized, multidisciplinary care suggests potential benefits from centralized treatment at specialized centers. 5 Local recurrence occurs in approximately 7%–18% of patients, while metastases, predominantly to the lungs, are observed in 14%–28% of cases. The 5-year overall survival (OS) rate for patients with nonmetastatic STSs is approximately 50%. 6 Numerous studies have identified prognostic factors for survival in STS.7, 8 However, findings are frequently inconsistent and often lack homogeneity across patient cohorts. We wanted to assess the influence of various factors on the outcomes of STS within a patient cohort treated over a period of 5 years, during which the treatment methodology remained uniform and standard.
Aim and Purpose
This retrospective analysis aimed to evaluate the clinical features, survival outcomes, and treatment-related results in patients with STS at an Indian tertiary referral center. The primary objectives included assessing diagnostic and staging characteristics, analyzing survival rates and prognostic factors, and evaluating treatment outcomes, including recurrence and metastasis.
Materials and Methods
In this retrospective study, we identified 91 patients diagnosed with STS from 2020 to 2023 using our institution’s database, PARAS. Each patient had confirmed sarcoma through histological examination and comprehensive staging. Data collected included patient demographics, disease characteristics, prior treatments, surgical outcomes, postoperative complications, adjuvant therapy, and recurrence patterns.
Statistical analyses were conducted using IBM SPSS (version 29.0). Categorical variables are presented as counts and percentages. The Kaplan–Meier method estimated OS, disease-free survival (DFS), local recurrence-free survival (LRFS), and distant metastasis-free survival (DMFS). Survival times were measured from treatment initiation to the event or last follow-up. Events were defined as follows: OS as death from any cause, DFS as any failure (local or distant), LRFS as local failure, and DMFS as metastasis. Calculations of OS, DFS, LRFS, and DMFS included all incidents until censoring. Univariate and multivariate analyses of prognostic factors were performed using log-rank and Cox regression models, respectively. Statistical significance was set at P < .05.
Results
Our analysis included 91 patients diagnosed with STS between 2020 and 2023 (Tables 1 and 2). Males slightly outnumbered females, constituting 64.8% of the cohort. The age distribution was nearly equal, with 51.6% of patients under 50 years and 48.4% over 50 years. Most cases (71.4%) were primary diagnoses, while the remaining were recurrent. Tumor size exceeded 5 cm in 70.3% of cases. Preoperative grading revealed that 62.6% were Fédération Nationale des Centres de Lutte Contre le Cancer (FNCLCC) grade 3, followed by grade 2 (21.9%) and grade 1 (15.5%). The majority (83.5%) had tumors located deep in the superficial fascia. Extremity tumors were more common (74.6%) than those at non-extremity sites, with upper limb tumors being more prevalent than lower limb tumors (54.9% vs. 19.7%).
Distribution of Patients According to Histology
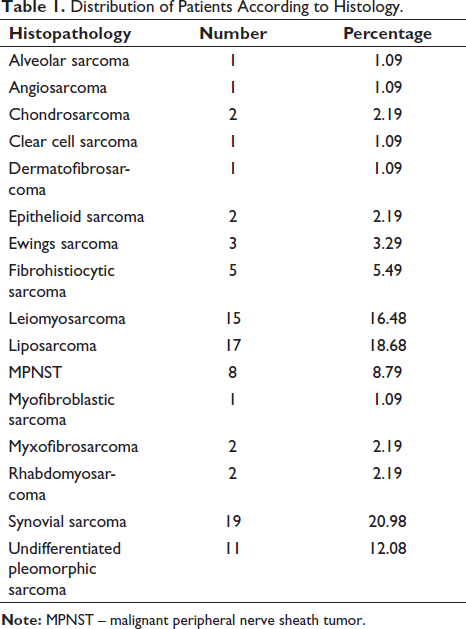
Distribution of Patients According to Patient Characteristics.
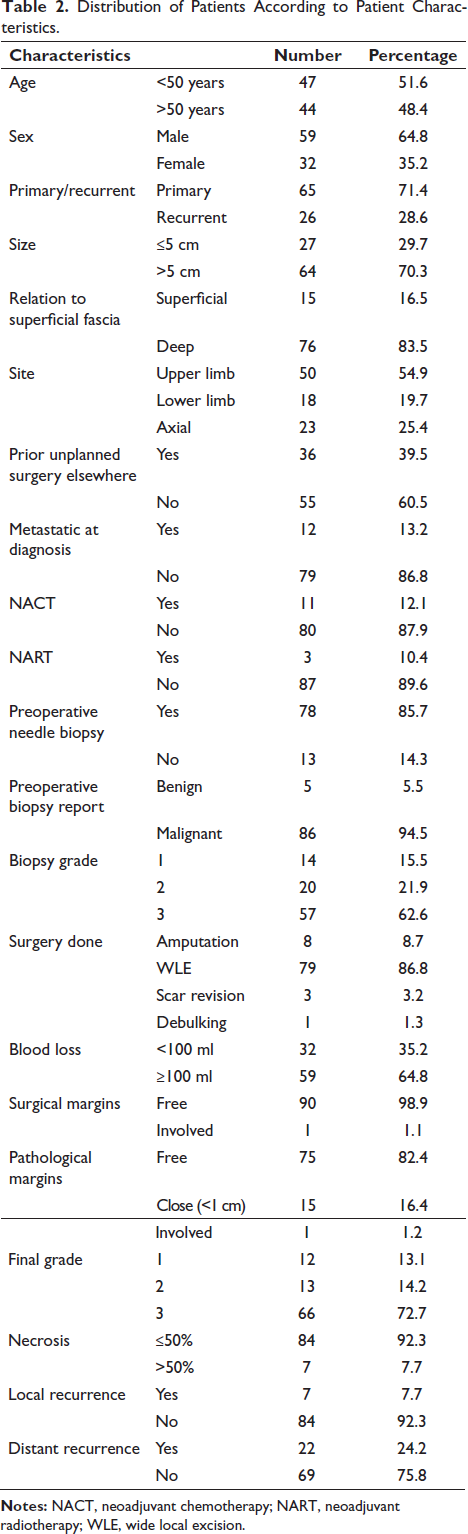
Among the cohort, 36 patients (39.5%) had previously undergone unplanned surgery at another facility, where histopathology reports identified malignant sarcoma. These diagnoses were reviewed and confirmed at the Rajiv Gandhi Cancer Institute and Research Center.
A preoperative needle biopsy was performed on 78 patients (85.7%) at either our institute or another hospital or clinic. The remaining patients underwent either incisional or excisional biopsies at other hospitals or clinics. Notably, five patients (5.5%) received an initial preoperative diagnosis of benign tumor, which was later revised to malignant upon final histopathological examination. The preoperative grading and postoperative final specimen grading demonstrated a strong correlation, with nearly equal percentages present in each grade group.
Twelve patients were diagnosed with upfront metastatic disease, including eight with lung metastases, three with bone metastases, and one with lymph node metastasis.
Preoperative radiotherapy (RT) was administered to three patients (10.4%) at the time of initial diagnosis. The first patient had a recurrent synovial sarcoma (SS) of the knee and underwent extra-articular resection with arthrodesis following neoadjuvant RT (NART) in August 2022. This patient remains alive with no evidence of disease recurrence. The second patient had a recurrent malignant peripheral nerve sheath tumor (MPNST) of the forearm, initially treated with wide local excision (WLE) and ulnar nerve reconstruction in 2017, followed by adjuvant RT. The patient experienced a recurrence in 2022 and underwent NART, followed by WLE and chemotherapy (CT). This patient also remains alive with no disease recurrence. The third patient presented with a recurrent tumor in the gluteal region. Despite undergoing NART and WLE, this patient experienced local recurrence within 6 months and distant recurrence at 1 year, ultimately resulting in an OS of 2 years.
Preoperative CT was administered to 11 patients (12.1%). Among these, three patients had extraosseous Ewing’s sarcoma (EWS). One of these patients presented with bilateral lung nodules and received five cycles of neoadjuvant CT (NACT) with the vincristine, adriamycin, cyclophosphamide followed by ifosfamide and etoposide (VAC/IE) regimen, followed by surgery after a favorable response. This patient subsequently developed lung metastasis and died after 31 months. Another patient with extraosseous EWS in the thigh underwent surgery following eight cycles of IE and has had no recurrence to date. The third patient, with extraskeletal EWS in the abdominal wall, received four cycles of VAC/IE followed by WLE, later developed lung metastasis, and died after 20 months.
Another patient had rhabdomyosarcoma of the gluteal region, received seven cycles of VCR with actinomycin, underwent WLE, developed bilateral lung nodules, and died after 10 months. A patient with recurrent SS of the thigh presented with bilateral lung nodules received three cycles of ifosfamide and Adriamycin (IA), underwent WLE followed by adjuvant CT, developed bilateral lung metastasis, and died after 23 months. Another patient had an MPNST of the gluteal region with bilateral lung nodules at presentation, received six cycles of IA followed by WLE and video-assisted thoracoscopic surgery (VATS), and has had no recurrence to date.
A patient with MPNST of the groin received six cycles of IA, followed by WLE and adjuvant RT, developed lung metastasis, and is currently alive and undergoing CT. Another patient with SS of the thigh and a femoral vein thrombus received four cycles of IA, followed by surgery, and is alive with no recurrence. A patient with high-grade STS and bilateral lung nodules received three cycles of IA, followed by surgery and adjuvant CT, developed local and distant recurrence, and died after 17 months.
Finally, a patient with recurrent SS of the knee, who underwent surgery elsewhere and experienced local recurrence, received three cycles of gemcitabine and docetaxel, followed by above-knee amputation and VATS for lung metastasis, and is alive to date.
Upfront surgery was performed on 78 patients, representing 85.7% of the total cohort. Among these, 8.7% underwent amputation, 3.2% underwent scar revision, and one patient underwent debulking; the remainder had WLE. Regarding surgical blood loss, 32 patients experienced less than 100 ml of blood loss, while the rest had blood loss of 100 ml or more.
Among the 91 patients analyzed, all were retrospectively assessed for resection status. The results indicated that 90 patients achieved R0 excisions, denoting complete tumor removal with negative margins. However, one case resulted in an R1 resection. This particular patient, a 57-year-old woman with a grade 3 leiomyosarcoma measuring 5 × 5 cm on the lower back, which had invaded the vertebrae and presented with indeterminate bilateral lung nodules, underwent palliative debulking due to the impossibility of achieving clear margins. She received postoperative adjuvant RT but subsequently developed multiple lung metastases. She began palliative CT with ifosfamide and Adriamycin-based regimen; however, imaging showed progressive disease in the lungs. She then commenced second-line CT. After three cycles, a positron emission tomography CT scan revealed further progression of the lung disease. The patient was subsequently treated with pazopanib. Her surgical site developed gaping, crusting, and foul-smelling discharge. Ultimately, the patient passed away with an OS of 19 months.
Adjuvant RT was employed in 49 cases (53.8%) as part of postoperative management strategies.
Postoperative systemic treatment was prescribed for 25 patients (27.4%) following primary tumor resection. Notably, a subset of 62 patients did not receive any systemic treatment.
Local recurrence was observed in seven patients, accounting for approximately 7.7% of cases, while distant recurrence was documented in 22 patients, representing approximately 24.2% of cases.
The median time from primary tumor diagnosis to onset of local relapse was 7.5 months, with a range of 4–23 months. The 3-year OS rate for patients experiencing local recurrence was 53%. For patients with both local and distant recurrence, the OS was 15 months. Furthermore, five patients who experienced local relapse subsequently developed metachronous metastasis.
In the cohort, 12 patients (13.2%) presented with upfront metastatic disease, with four cases being recurrent tumors. Among these patients, 11 exhibited lung metastasis, while only one had lymph nodal metastasis. The predominant histological type was SS (n = 5), followed by MPNST (n = 2). Eight patients underwent WLE with VATS, and one patient underwent WLE with pelvic lymph nodal dissection. The remaining patients underwent amputation with VATS. The average duration of metastasis-free survival was 9.5 months. Four patients received NACT, while the others underwent upfront surgery. The 2-year and 3-year OS rates for patients were 84% and 42%, respectively.
In our comprehensive analysis, which included patients diagnosed with SS across all stages and clinical presentations at initial diagnosis, the 2-year OS and 3-year OS rates were 64% and 60.5%, respectively.
Among the cohort, 29 patients experienced some form of relapse following primary resection. The 2-year and 3-year DFS rates were 67.6% and 58%, respectively. The 3-year LRFS rate was 88.6%. Additionally, the 2-year and 3-year DMFS rates were 72.1% and 69.1%, respectively.
Out of the 91 patients, 67, constituting 73.6% of the total, were alive at the time of data compilation.
Baseline Prognostic Factors
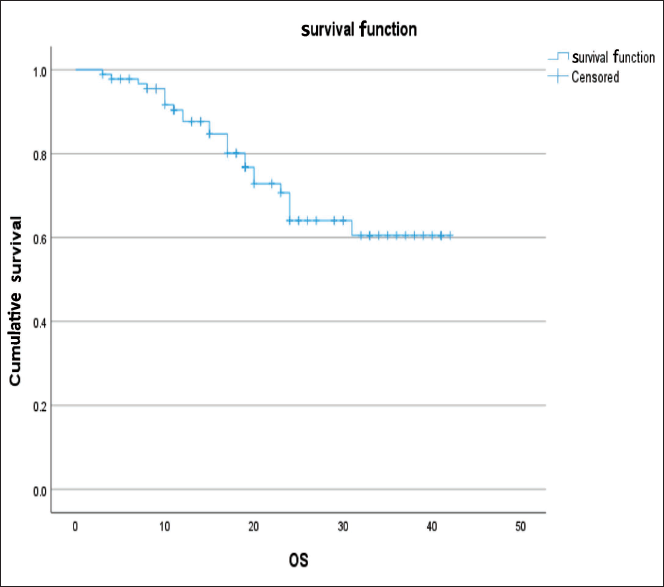
The analysis of OS revealed significant relationships with several factors (Figure 1). Notably, tumors of size ≤5 cm (P = .013), patients not receiving NACT (P = .001), and intraoperative blood loss <100 ml (P = .008) all had a significant impact on OS. The 3-year OS for patients who experienced local recurrence was 53%. NACT may be considered as a confounding factor, as all patients who presented with upfront metastatic disease were given NACT, thus naturally affecting OS.
Furthermore, a trend toward better OS was observed in patients who received adjuvant CT and exhibited a lower degree of necrosis in the final specimen.
Graph Showing the OS Curve of the Entire Cohort.
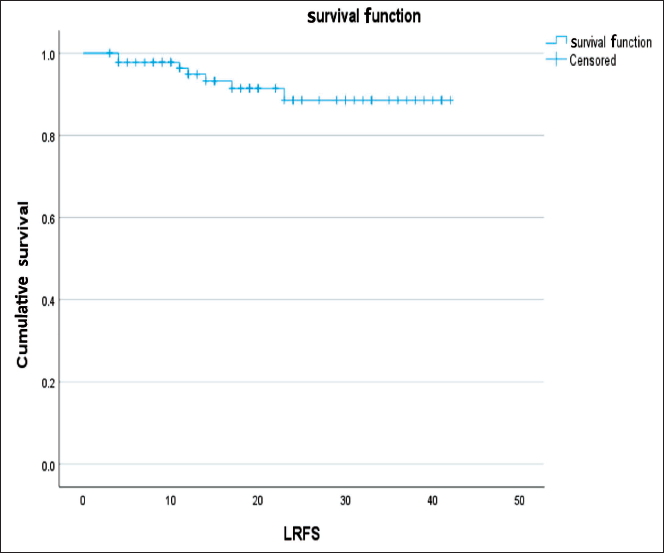
The assessment of LRFS revealed significant relationships with several factors (Figure 2). Specifically, tumors exhibiting lower or no necrosis (P < .005) and margins greater than 1 cm in the surgical specimen (P < .005) were associated with significantly longer LRFS. Additionally, there was a trend toward better LRFS performance in tumors smaller than 5 cm.
Graph Showing the LRFS Curve of the Entire Cohort.
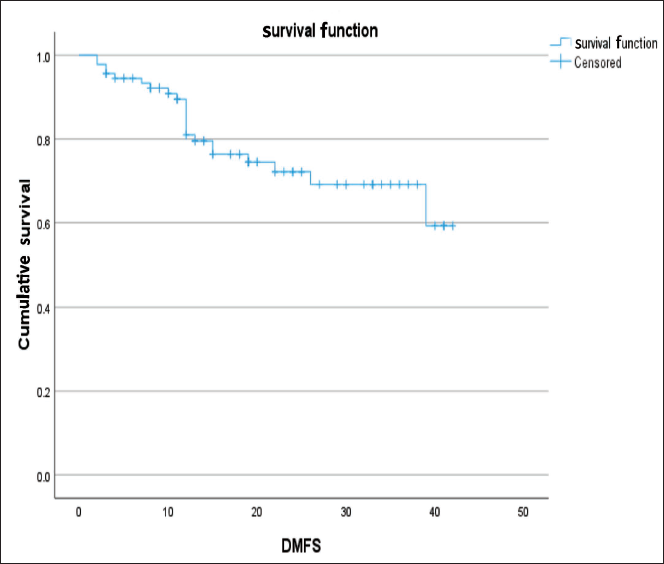
The analysis of DMFS unveiled several factors that exhibited a significant relationship with DMFS (Figure 3). Specifically, patients receiving NACT (P = .016), tumor grade (P = .043), intraoperative blood loss <100 ml (P < .005), and adjuvant CT (P < .005) were identified as significant factors.
While the size of the tumor and the type of surgery performed showed a potential impact on DMFS, these associations were not statistically significant.
Graph Showing the DMFS Curve of the Entire Cohort.
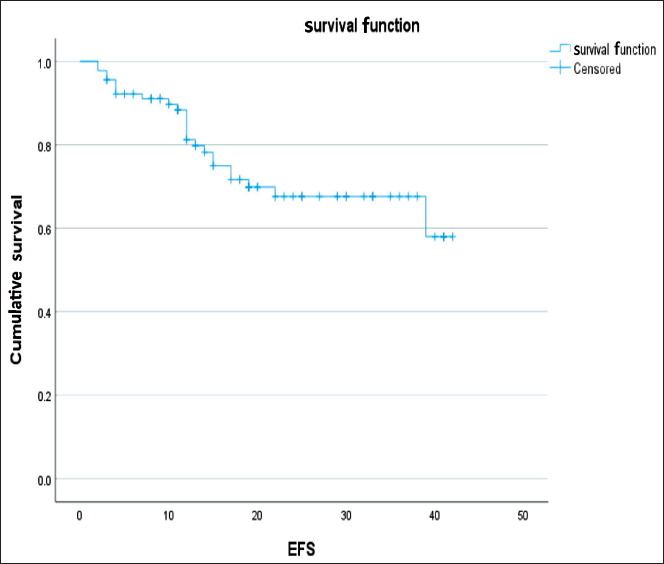
On multivariate analysis, the only significant factor that remained was intraoperative blood loss (P = .010, 95% confidence interval [CI]: 0.074–0.701) (Figure 4). This indicates that patients with intraoperative blood loss <100 ml performed significantly better in all aspects compared to patients who experienced greater blood loss intraoperatively.
Graph Showing the Event Free Survival (EFS) Curve of the Entire Cohort.
Discussion
The primary treatment for STSs is wide surgical resection. 9 Following surgery, adjuvant RT and/or CT may be administered as deemed necessary; however, the utilization of adjuvant CT remains a topic of debate and may not confer benefits to all patients with localized resectable sarcoma. 10 In the current series comprising 91 patients diagnosed with grade 1/2/3 STSs of the extremities and trunk, the 2-year and 3-year OS rates are 64% and 60.5%, respectively, and the 2-year and 3-year DFS rates are 67.6% and 58%, respectively. A study of 79 patients by Garg et al. 4 reported a 3-year OS of 77.6%. Zhao et al., 11 in a study involving 166 patients with extremity/trunk STS, reported a 5-year OS of 77.5%, with a 5-year local failure-free survival rate of 74.3%. One recent study reported a median OS of 24 months (95% CI: 21–33) for patients with metastatic STS, with 1-, 2-, and 5-year OS rates of 70.0% (95% CI: 64–77), 49.9% (95% CI: 43–58), and 24.8% (95% CI: 19–33), respectively. 12 . It is noteworthy that the majority of local recurrences and instances of systemic disease, particularly in malignant tumors, manifest within the first 2 years following initial surgery.13–15
The investigation yielded no discernible correlation between the localization of STS (whether appendicular or axial), age (<50 years vs. ≥50 years), or gender, and their impact on OS, LRFS, or DMFS.
In our study, we compared 3-year OS and DFS between patients who underwent WLE followed by adjuvant RT and those who had an amputation. We found no significant difference in OS, DMFS, or LRFS. These results emphasize the impact of treatment choice on long-term survival and provide insights into managing STSs. Similarly, there was no significant difference observed in OS, DMFS, and LRFS at the time of data compilation (P = .848 for OS, P = .909 for DMFS, P = .455 for LRFS, respectively) between patients who received adjuvant radiation therapy and those who did not. Adjuvant radiation therapy was administered in 53.8% of patients in our study. According to information from the National Cancer Institute, the impact of adjuvant CT on survival in STSs remains controversial, with conflicting results from various trials. While some studies reported benefits in terms of relapse-free interval and distant relapse-free interval with adjuvant CT, the OS benefit was not statistically significant. 16
Our study identified a significant relationship between local recurrence and OS (P < .005), with patients not having local recurrence performing significantly better. This echoes the findings of Lewis et al., 17 who also observed a strong correlation between local control, metastasis, and tumor mortality.
Peiper et al. 18 elucidated that tumor grade emerges as an independent prognostic factor for survival through univariate analysis. Similarly, surveillance, epidemiology and end results (SEER) data highlighted the significance of grade as a predictor of OS in both univariate and multivariate analyses. 19 Contrary to these findings, our investigation unveiled that the ultimate specimen grade holds significance solely for DMFS (P = .042), with no notable impact observed for OS (P = .150) or LRFS (P = .438). Hong et al. 20 remarked that the grading system reflects the degree of resemblance between tumor cells and normal cells, thereby correlating with the tumor’s propensity for metastasis. Notably, tumors of higher grade often necessitate adjunctive therapies to mitigate their heightened metastatic potential.
Most studies have reached a consensus regarding the significance of surgical margin as a strong negative prognostic factor for local control.21, 22 However, the anatomical constraints surrounding the tumor often make achieving a true wide resection with a 2-cm safety margin impractical without sacrificing critical anatomical structures such as major nerves, blood vessels, bone, and joints. In such cases, a close surgical margin is deemed acceptable.23, 24 Additionally, several series have reported that postoperative RT for patients with close margins can improve local control.25, 26
In patients undergoing RT and limb-sparing surgery for STSs, obtaining a negative margin is crucial for optimizing both local control and survival. Nevertheless, the absolute quantitative width of the negative margin does not significantly influence the outcome, suggesting that attempts at achieving wide resection margins may be unnecessary. 27
Within our study, no statistically significant variance was detected in either OS, DMFS, or LRFS across cohorts categorized by free or close pathological margins (P = .494 for OS, P = .981 for DMFS, and P = .495 for LRFS). Among the instances of local recurrence, comprising seven cases, five exhibited free margins, while two presented with close margins subsequent to resection.
A significant correlation was observed between the volume of blood loss and outcomes, evident in both univariate and multivariate analyses. A lower blood loss had significantly better outcomes. Luo P et al. 28 conducted a study focusing on retroperitoneal sarcoma, revealing that the quantity of blood loss serves as an independent prognostic indicator. Importantly, their findings indicated markedly improved outcomes associated with reduced blood loss.
A notable contrast in OS and DMFS was observed, albeit not for LRFS, associated with NACT within the entire cohort (P = .010 for OS, P = .016 for DMFS, and P = .202 for LRFS). This may be associated with the administration of NACT to all patients presenting with upfront metastatic disease, thus exerting an influence on OS and DMFS outcomes.
Consistent findings were observed in the adjuvant setting, wherein adjuvant CT exerted a significant impact on DMFS (P = .013) and displayed a trend toward enhancing OS (P = .061), but without significance in LRFS. Notably, two extensive European Organisation for Research and Treatment of Cancer (EORTC) trials comparing doxorubicin-based adjuvant CT to observation in completely resected STSs failed to unveil any discernible benefit concerning OS. However, adjuvant CT emerged as an independent favorable prognostic factor for relapse-free survival but not for OS. 29
Residual tumor presence is acknowledged as a risk factor for local recurrence, with research indicating its occurrence in up to three-fourths of instances involving unplanned resections.30, 31 Within our study, no statistically significant discrepancies were discerned in OS (P = .632), LRFS (P = .716), or DMFS (P = .837) among patients who underwent prior unplanned surgery elsewhere compared to those who underwent planned surgery at our institution. This may be attributed to the likelihood that tumors of smaller size and lower grade were addressed elsewhere before presentation, whereas more advanced cases were primarily presented to us. This is supported by Fromm et al., 32 who noted that subsequent to the re-resection of STS following unplanned initial resections, OS did not exhibit a significant difference when compared to a cohort of non-re-resected tumors. These findings suggest that re-resection may not be universally necessary in all cases of unplanned resections of STS.
It is notable that despite a considerable portion of patients initially presenting following excisional biopsy or intralesional surgery at other institutions (39.5%), the local recurrence rate within our series stood at 7.7%. This observation indicates that RT and wide re-excision are viable treatment strategies even where tumors were previously subjected to surgery. Moreover, the incidence of local recurrence per se should not dissuade further endeavors aimed at achieving local disease control.
Peabody et al. 33 highlighted the independent significance of both tumor size and depth concerning survival outcomes. Similarly, Ogura et al. 34 highlighted in their series that tumor size and deep-seated location significantly heightened the risk of disease-specific mortality.
Within our investigation, significant outcomes in OS at 3 years were evident among the various tumor size groups (P = .013), with a suggestive trend toward significance observed in both LRFS (P = .056) and DMFS (P = .064). However, the depth of the tumor did not show significance concerning OS, DMFS, or LRFS.
Italiano et al. 35 conducted a comprehensive study involving 1,024 patients diagnosed with metastatic STSs, revealing a 2-year OS rate of 33.2% and a median OS of 14 months. Notably, our investigation unveiled a significant correlation between OS and the presence of metastasis at presentation (P < .005), with corresponding 2- and 3-year OS rates of 84% and 42%, respectively. A sharp decline in survival beyond the 2-year mark among metastatic patients is evident. Although the studies reviewed do not explicitly cite the 2-year OS rate, they indicate that the anticipated median OS in patients with metastatic STS has extended from approximately 12 months to nearly 2 years in more recent clinical trials. 36
While our study relied on retrospective data, it offers significant clinical insights into the management and outcomes of STS. Moving forward, there is a need for real-world data and prospective trials to further validate the effectiveness of current treatment modalities. Through continued research efforts, we aim to refine therapeutic approaches and ultimately improve outcomes for patients suffering from this disease.
Limitations
This retrospective study highlights the importance of conducting additional randomized prospective studies to thoroughly understand STS characteristics. Increasing the sample size would improve statistical power and enhance the reliability of findings. Future investigations should include a wider range of histopathological types of sarcoma and specify CT regimens for a more comprehensive analysis. While some characteristics may not have shown statistical significance in this study, their potential importance in a larger cohort warrants further investigation. Continuous research efforts are crucial for refining our understanding and management of STS.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
