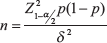Abstract
Background
The aim of this article is to evaluate radiological and pathological response rates and correlate them after neoadjuvant short-course radiotherapy (SCRT) followed by chemotherapy in resectable locally advanced adenocarcinoma of the rectum (LARC).
Materials and Methods
It is a single institutional, prospective observational study from January 2021 to March 2022 including 45 patients with resectable adenocarcinoma of the rectum. Radiotherapy was delivered to a dose of 25 Gy in 5 Gy per fraction using image-guided radiotherapy followed by mFOLFOX-based chemotherapy. After completion of 4–8 cycles of chemotherapy, tumor regression grade (TRG) was evaluated using magnetic resonance imaging and whole-body positron emission tomography. In case of a satisfactory response on imaging (mrTRG), patients underwent appropriate locoregional surgery. Histopathological examination of the surgical specimen was carried out and response to neoadjuvant treatment was assessed (pTRG).
Results
A significant downstaging of T and N stages (60% and 72%, respectively, p < .0001) was observed after neoadjuvant therapy with a pathological complete response rate of 16%. Conversion of positive circumferential resection margin (CRM) to negative CRM was observed in 71.4% patients. A poor agreement between mrTRG and pTRG (k 0.133, p .16) was noted.
Conclusion
Preoperative SCRT in LARC is feasible and well-tolerated with encouraging radiological and pathological response rates. A significant number of patients achieve negative CRM, thereby improving the chances of R0 resection. Significant relief in pain and bleeding can be achieved rapidly leading to an improved quality of life.
Introduction
Carcinoma rectum is the third most common gastrointestinal malignancy worldwide. 1 Histologically, majority of rectal cancers are adenocarcinomas. Surgery remains the mainstay of curative treatment. However, an unacceptable high local recurrence rate (20–50%) was observed after surgery alone. 2 Adjuvant radiotherapy or chemoradiation significantly reduced local recurrence with prolonged disease-free survival, 3 however at the cost of higher grade and incidence of radiation-related gastrointestinal toxicity due to inclusion of large postoperative radiation volume.
On the other hand, neoadjuvant therapies seem to offer several potential advantages including downstaging of the tumor, increased possibility of resection, low incidence of treatment-related toxicity, and potential for sphincter preservation in lower rectal cancers. Various studies4, 5 from across the world concluded that neoadjuvant treatment approach had better pathological complete response (pCR) as well as lower local recurrence rate compared with surgery alone or surgery followed by adjuvant treatment.
Pertaining to neoadjuvant therapy, several randomized control trials5, 6 have demonstrated the benefits of short-course radiotherapy (SCRT) followed by surgery compared to surgery alone, with better pCR rates after delayed surgery (6–8 weeks after completion of radiotherapy). The incorporation of chemotherapy between SCRT and surgery showed even better tumor-related outcomes with fewer toxicities compared to postoperative adjuvant chemoradiotherapy.
Despite ample published literature in favor of preoperative SCRT followed by chemotherapy, the adoption of this approach has not been widespread in our country. This study analyses radiological and pathological response rates in patients who underwent SCRT followed by chemotherapy and surgery for locally advanced rectal cancer (LARC) in our Indian population. We often encounter bulky tumors in our patients. The results of this study will further help us design studies to analyse the outcomes based on the volume of disease and the requirement for dose escalation in selected cases in the same cohort of patients.
Materials and Methods
Study Design: Single institutional, prospective, and observational.
Sample Size: Forty-five.
Sample size calculation: Calculated based on the following assumptions and formula:
5% level of significance (α), 9% margin of error (δ), and Of a total of 217 patients with carcinoma rectum treated at our institute from November 1st, 2019 to October 31st, 2020, 22 patients were treated with all three modalities (chemotherapy, radiotherapy, and surgery), the prevalence of patients treated with triple modality being 10.1% (p). Zα = Value of standard normal variate corresponding to α level of significance = 1.96
Inclusion criteria: Histologically proven newly diagnosed primary adenocarcinoma of the rectum, clinically any T and N0–N2 disease with an Eastern Cooperative Oncology Group (ECOG) performance status of 1 or 2.
Exclusion criteria: Unresectable primary disease, metastatic or recurrent rectal cancer, presence of any concomitant malignancies, any contraindication to MRI, medical or psychiatric conditions that compromise the patient’s ability to give informed consent, concurrent uncontrolled comorbidities, pregnancy or breastfeeding, presence of malabsorption syndrome, and history of peripheral neuropathy.
Study population: Forty-five patients with resectable adenocarcinoma rectum planned for neoadjuvant SCRT followed by chemotherapy, who meet the inclusion criteria and who do not have any of the exclusion criteria.
Time period: From January 7th, 2021 to March 30th, 2022.
Data collection and processing: Demographic profile (age, gender, origin, performance status, presence of comorbidities, smoking, consumption of smokeless tobacco or alcohol, type of diet, and body mass index), patient and disease-related characteristics and histopathological features reported on a resected TME specimen were recorded in the patient data sheet. Finally, all the data were entered into the master sheet and Statistical Package for Social Sciences (SPSS) software for statistical analysis.
Pretreatment evaluation: Complete history and physical examination including digital rectal examination, complete blood count, kidney and liver function test, CEA level, colonoscopy, MRI pelvis with contrast (Contrast-Enhanced Magnetic Resonance Imaging [CEMRI]), whole body PET-CT, and cardiac evaluation with an ECG and 2D echocardiogram.
Primary objectives: Analysis of radiological response using MRI (mrTRG), pathological response (pTRG) by histopathological examination of the postoperative specimen, and correlation of mrTRG and pTRG.
Secondary objectives: Analysis of correlation between various demographic and tumor-related MRI parameters with mrTRG and pTRG, evaluation of acute bowel and skin toxicities during and after SCRT and rate of conversion of an involved circumferential resection margin (CRM) to uninvolved CRM after neoadjuvant therapy.
Treatment Detail
SCRT was planned and delivered using image-guided radiotherapy (IGRT) to a dose of 25 Gy in daily fractions of 5 Gy per fraction over eight days. mFOLFOX (Oxaliplatin 100 mg/m2 on day 1, folinic acid 400 mg/m2 on day 1, 5-FU 400 mg/m2 bolus on day 1, 5-FU 2,400 mg/m2 continuous infusion over 24 h on day 1 followed by 22 h continuous infusion on day 2) chemotherapy was started within two weeks after completion of SCRT.
Radiotherapy Detail
The patients were simulated and treated in the supine position, hands placed above the head and with a full bladder protocol. Immobilization was achieved using a thermoplastic cast for pelvis, whole abdomen, and upper part of the thighs.
Contrast-enhanced CT simulation scans taken from L2 vertebral body to the proximal one-third of the femur with a slice thickness of 3 mm and an FOV of 60 cm was fused with CEMRI and PET-CT. The fusion between the various datasets (CT/MRI and PET/CT) was done on ECLIPSE version 10 (Varian Medical Systems, Palo Alto, CA) using an automatic image registration algorithm. The gross tumor volume, defined as the primary tumor and the suspicious metastatic lymph nodes visualized on the CT scan, MRI, and/or PETCT, was contoured on the simulation scan. Information from colonoscopy and physical examination was also taken into account.
Target volumes, namely clinical target volume (CTV) and planning target volume (PTV), and organs at risk were contoured according to the international consensus guidelines on clinical target volume delineation in rectal cancer by Valentini et al. 8 CTV included the presacral space, mesorectum, posterior group of lateral lymph nodes (LLN), and external iliac nodes. Anterior LLN anatomically corresponding to the obturator nodes was included in the CTV in case of cT4 tumors/multiple positive lymph nodes in the mesorectum and/or positive nodes in the posterior LLN. The entire ischiorectal fossa (IRF) was included if the primary tumor directly invaded the IRF or when the external anal sphincter was infiltrated. Inguinal nodes were included in a case with involved inguinal nodes, massive tumor extension into the internal or external anal sphincter, or when the lower third of the vagina was infiltrated. PTV was generated by giving a 7-mm isotropic margin to the CTV.
A dose of 25Gy in 5 fractions was prescribed to the PTV.
Response and Toxicity Assessment
Symptom relief (rectal bleed and pain) and acute bowel (diarrhea) and skin toxicity was recorded weekly as per the Radiation Therapy Oncology Group toxicity grading during the treatment. The week during which SCRT was delivered was considered week 1. After completion of four cycles, patients were evaluated for surgery by CEMRI and PET-CT. In case of a good partial response on imaging, patients were taken up for appropriate locoregional surgery. In others, further chemotherapy was continued for another four cycles before reassessment for surgery. Histopathological examination of postoperative specimen was carried out and the response of the neoadjuvant treatment was assessed as per the guidelines by the College of American Pathologists (CAP) using the modified Ryan TRG system, based on the volume of residual primary tumor cells. 7
Statistical Analysis
Association of the quantitative variables not normally distributed in nature was analysed using the Mann–Whitney test (for two groups) and the Kruskal–Wallis test (for more than two groups). Spearman rank correlation coefficient was used to correlate TRG with various parameters and with each other. Interrater kappa agreement was used to find a strength of agreement between mrTRG with pTRG. McNemar test was used for comparison of the CRM at baseline and CRM after neoadjuvant treatment and Bhapkar test was used for comparison of cN and ypN and between cT and ypT. For statistical significance, p value of less than .05 was considered statistically significant. All analysis was done with the use of SPSS software (IBM, Chicago, USA, ver 21.0).
Results
Demographic Characteristics
Nearly 50% patients were more than 50 years of age with a predilection toward male gender. Notably 80% patients were nonvegetarian.
Other demographic characteristics are shown in Table 1.
Demographic Characteristics.
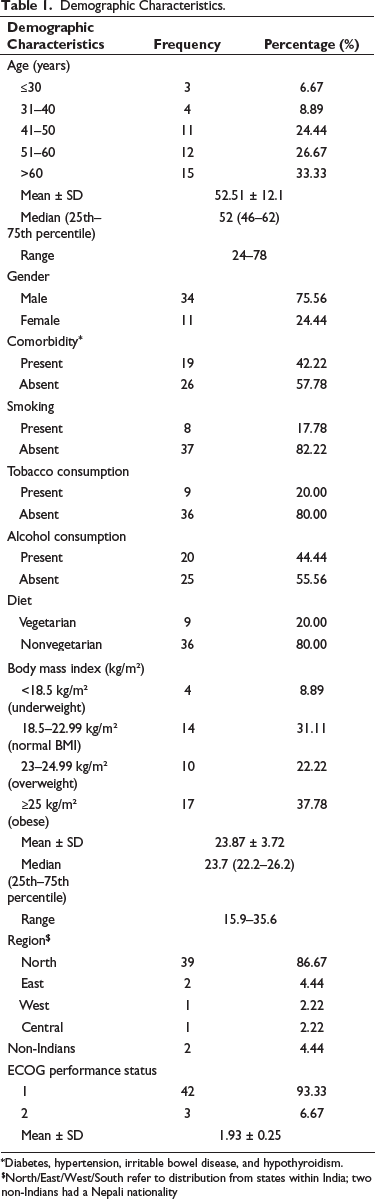
*Diabetes, hypertension, irritable bowel disease, and hypothyroidism.
$North/East/West/South refer to distribution from states within India; two non-Indians had a Nepali nationality
Nearly 36% patients had tumor in the lower third of rectum (Table 2).
Distribution of MRI Parameters.
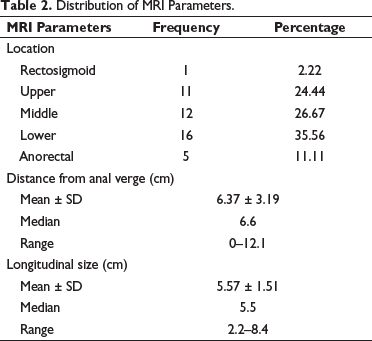
Mean value of pre-neoadjuvant treatment CEA and post-neoadjuvant treatment (presurgery) CEA was 25.32 and 4.26 ng/ml, respectively. The mean value of pre-neoadjuvant treatment SUVmax and presurgery SUVmax on PET was 13.56 ± 5.89 and 5.36 ± 3.26, respectively.
Response Assessment
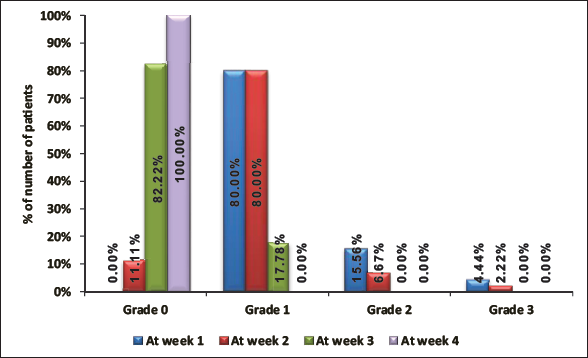
Relief from bleeding per rectum was observed week 2 onward when 11% achieved complete relief and 100% achieved complete relief during week 4 (Figure 1).
Assessment of Bleeding Per Rectum.
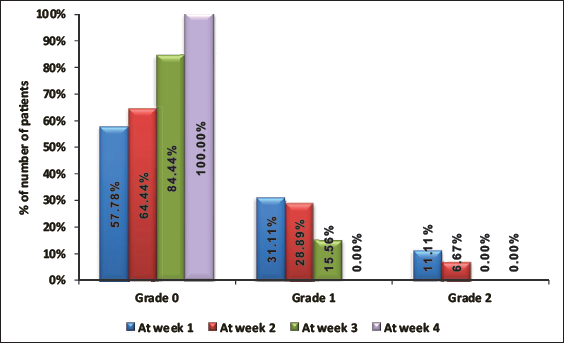
Perianal/Sacral pain was reported by 42% patients at presentation. Improvement in pain score was noted by end of week 2 and all patients achieving complete pain relief during week 4 (Figure 2).
Assessment of Pain.
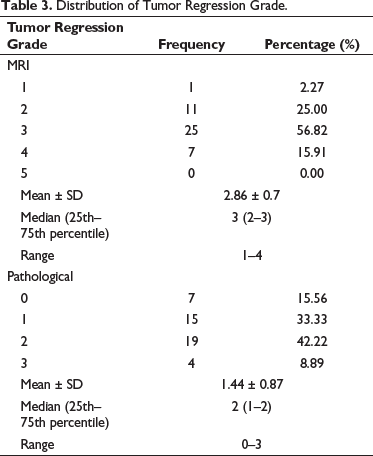
An mrTRG of 3 and a pTRG of 2 were observed in the majority (56.82% and 42.22%, respectively). mrTRG and pTRG observed after neoadjuvant therapy are shown in Table 3.
Distribution of Tumor Regression Grade.
No significant association was seen between mrTRG or pTRG with age, gender, presence of comorbidities, smoking, consumption of smokeless tobacco or alcohol, type of diet, body mass index, location of the tumor, distance of tumor from anal verge, longitudinal tumor size, baseline CEA level or the SUVmax of tumor at baseline (supplementary material).
Correlation Between mrTRG and pTRG
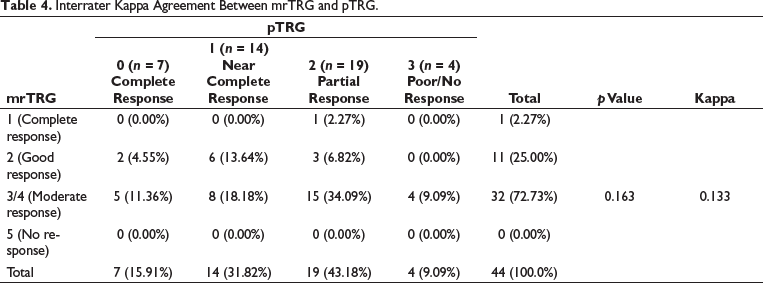
Among seven patients with a pTRG of 0 (complete response), none of the patients had an mrTRG of 1 (complete response on MRI). Among 14 patients with a pTRG of 1 (near-complete response), six patients had a good response on MRI (mrTRG 2). Among 19 patients with pTRG of grade 2 (partial response), 15 had a moderate response on MRI (mrTRG 3/4). Of the four patients with pTRG of 3 (poor/no response), all showed an mrTRG of 3/4 (moderate/slight response). The overall concordance rate between pTRG and mrTRG, taking into account the TRG of all 45 patients, was 47.73% (Table 4). Poor agreement exists between pTRG and mrTRG with a kappa of 0.133 and a p value of 0.163.
Interrater Kappa Agreement Between mrTRG and pTRG.
Of the 21 patients with involved CRM at the time of diagnosis, only six continued to have pathologically involved CRM after neoadjuvant therapy, signifying a 71.4% conversion rate (p value = .0001) (Table 5).
Comparison of CRM Before and After Neoadjuvant Therapy.

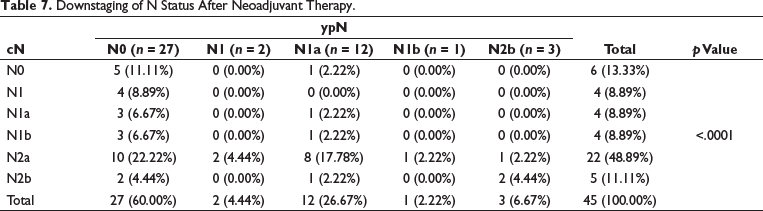
A significant downstaging of cT and cN was observed (p value < .0001 for each parameter) (Tables 6 and 7). Out of 39 patients with cT3 disease, eight had ypT0 stage after neoadjuvant therapy. Out of four patients with cT4a stage, all were downstaged to ypT3. Of the 39 patients with node-positive disease, 22 (56.6%) were downstaged to node negative.
Downstaging of T Status After Neoadjuvant Therapy.

Downstaging of N Status After Neoadjuvant Therapy.
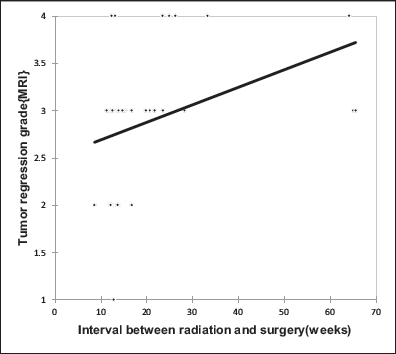
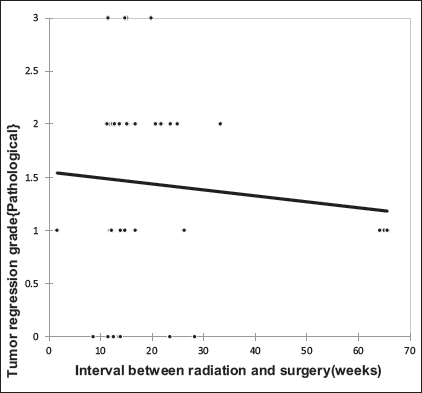
The median time from completion of SCRT to surgery was five months (20 weeks), where 44.44% and 55.55% of patients underwent surgery within <20 weeks and ≥20 weeks, respectively. A significant positive correlation was seen between the interval from completion of radiation to surgery with mrTRG with a correlation coefficient of 0.452 (Figure 3); however, no such correlation was observed with the pTRG (Figure 4).
Correlation of Interval Between Radiation and Surgery with mrTRG.
Correlation of Interval Between Radiation and Surgery with pTRG.
Toxicity Assessment
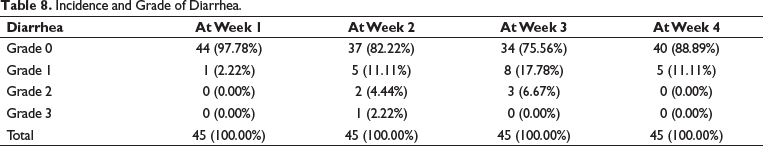
Acute toxicity seen in the form of diarrhea peaked during week 2 (the week immediately after completion of SCRT) with one patient experiencing grade 3 and other seven experiencing grade 1–2 diarrhea. However, during week 3, no patient had grade 3 but 11 continued to have grade 1–2 diarrhea. This toxicity abated by the completion of fourth week (Table 8).
Incidence and Grade of Diarrhea.
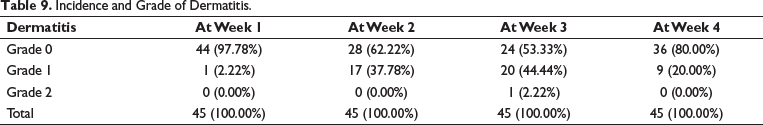
Maximum grade of acute dermatitis (grade 2) was seen in week 3 in only one patient (Table 9).
Incidence and Grade of Dermatitis.
Discussion
Management of carcinoma rectum has undergone dramatic changes over the past two decades. Surgery, established as the curative treatment modality for early and LARC, as a single modality was associated with an unacceptably high recurrence rate. Local recurrence rates decreased with the introduction of total mesorectal excision (TME) as compared to APR or LAR alone but remained unacceptably high, 9 prompting the use of preoperative modalities such as radiation. Potential advantages of neoadjuvant radiotherapy include a reduction in tumor size leading to a higher chance of sphincter preservation, smaller treatment field with less small bowel irradiation leading to less acute toxicities, and irradiation to well-oxygenated tissue. SCRT with 25 Gy over five days followed by surgery within one week in the Stockholm I trial significantly reduced the rate of pelvic recurrence and prolonged the recurrence-free interval. 10 However, no overall survival benefit was observed. Further, the Swedish rectal cancer group demonstrated a significantly higher overall survival in the radiotherapy-plus-surgery group than in the group treated with surgery alone (p .02). 11
Despite significant benefits seen in preoperative SCRT followed by TME, short interval between radiotherapy and surgery led to inadequate downstaging. A need to increase the gap between SCRT and surgical resection was realized in order to improve the chances of sphincter sparing and achieving a negative CRM.
Stockholm III trial designed to optimize timing of surgery after preoperative radiation, reported a higher rate of pCR (11.8% vs. 1.7%; p .001) and Dworak grade 4 tumor regression (10.1% vs. 1.7%; p < .001) in patients undergoing delayed surgery than patients undergoing immediate surgery, thereby concluding that SCRT induces tumor downstaging if surgery is performed after an interval of 4–8 weeks. 5
Further, Polish II trial incorporated chemotherapy in the interval between SCRT and surgery. 12 RAPIDO study recruited patients with cT4a or cT4b, extramural vascular invasion, cN2, involved mesorectal fascia or enlarged mesorectal lymph nodes. At three years after randomization, the cumulative probability of disease-related treatment failure was 23.7% (95% CI 19.8–27.6) in the SCRT arm and 30.4% (26.1–34.6) in the standard of care long-course radiotherapy (LCRT) arm (hazard ratio 0.75, 95% CI 0.60–0.95; p .019). 13
With the publication of Stockholm III and RAPIDO, SCRT followed by chemotherapy followed by surgery has been widely and readily adopted across the radiation oncology community for its ease of implementation, better patient compliance, and improved downstaging in LARC.
Excellent symptomatic palliation of bleeding per rectum and perianal/sacral pain was achieved with SCRT in a short span of four weeks resulting in improved quality of life and better compliance to further treatment.
CEA, an established biomarker for predicting prognosis and monitoring the progression of colorectal cancer, 14 provides a good tool for response assessment. In our study, 66% had elevated CEA levels at presentation of whom 66% (20/30) attained normal levels after neoadjuvant treatment.
Rectal MRI plays an important role in the pre- and posttreatment evaluation of rectal cancer. A concordance rate of 94% and 85% in the T stage and N stage, respectively, has been observed between MRI and pathology.15, 16 One of the most commonly used grading systems (grade 1–4) is based on MERCURY study based on different signal intensity by residual tumor, amount of fibrosis, or mucin. 17 MRI-based mrTRG has recently emerged as a dynamic, noninvasive, surrogate method for the assessment of tumor regression following neoadjuvant treatment and is being increasingly used as a decision-making tool for patient management following the completion of standard neoadjuvant chemoradiotherapy. Koëter et al. showed a moderate or good response (mrTRG ≤ 3) in 73% patients which was 84% in our study. 18
CRM, defined as the closest distance between tumor, area of neurovascular invasion, or mesorectal lymph node to the radial resection margin, is considered involved if the nearest distance from a tumor or involved lymph node is ≤1 mm from mesorectal fascia. Involvement increases the risk of both local recurrence and distant metastases. 19 Baseline CRM-positive rate of 62% was noted in the RAPIDO 13 study, which remained positive in only 9% in the pathological specimen. In the current study, of the 21 patients (53%) with an involved CRM at presentation, six (28.6%) were reported to be involved on MRI after completion of neoadjuvant treatment (p value = .02), while only two (9.52%) were pathologically involved (p value = .1).
PET-CT is increasingly being adopted for staging and response assessment after neoadjuvant therapy. Altered stage as a result of PET-CT changes treatment intent in 8% of cases, mainly due to revised nodal staging. 20 Capirci et al. evaluating the role of SUV values of 18F-FDG PET to assess response after neoadjuvant chemoradiation in rectal cancer observed a 35% drop in the SUVmax after neoadjuvant treatment, 21 similar to 39.5% fall in SUV values observed in the current study.
Various grading systems have been proposed to assess tumor response, including TRG systems proposed by Ryan et al., Dworak et al., and Mandard et al.22–24 Among these, the most widely used TRG system is by Ryan et al. in which amount of viable and nonviable tumor cells are assessed on the surgical specimen (0—complete response, 1—near-complete response, 2—partial response, and 4—poor or no response). Accurate determination of pTRG, ypT, and ypN in rectal carcinoma after neoadjuvant therapy reflects therapeutic response and carries prognostic value.25–27 Ninety-one percent of our patients had a pTRG of 2 (signifying residual cancer outgrown by fibrosis) or less. Pathological complete response (pTRG 0) was observed in 15.56% of the patients.
In a prospective study conducted by Markovina et al., 78% of the patients with a clinical T3 stage were downstaged to T2 or less after neoadjuvant SCRT followed by chemotherapy. 28 We observed a significant tumor and nodal downstaging (60% and 72%, respectively) following neoadjuvant treatment (p value < .0001).
One of the questions that remain unanswered is whether and to what extent mrTRG truly corresponds to the degree of pathological regression. An overall discordance rate of 52.27% between pTRG and mrTRG in our study signifies a poor agreement. The probable reasons could be low sensitivity of MRI to differentiate between postradiation changes, fibrosis, and viable tumor and low intra- and interobserver reproducibility. In a study conducted by Sclafani et al., a fair agreement was observed between mrTRG and pTRG with a kappa of 0.24. 29
Only a few studies have assessed the effect of the time interval between surgery and SCRT. An optimum timing between radiotherapy and surgery must be taken into account as it might affect tumor control, and locoregional and distant recurrence after surgery. In our study, the median time from completion of SCRT to surgery was five months (20 weeks), where 44.44% and 55.55% of patients underwent surgery within <20 weeks and ≥20 weeks, respectively. A significant positive correlation was seen between radiation and surgery with mrTRG with a correlation coefficient of 0.452 (p .002). A nonsignificant mild positive correlation was seen with pTRG with a correlation coefficient of 0.077 (p .615).
Increasing the number of chemotherapy cycles after SCRT in the RAPIDO and STELLAR trials did not show any significant effect on pTRG (p value = 0.588).13, 30 Similarly, we observed that the patients who received ≤4 chemotherapy cycles had a median pTRG of 1, and those who received >4 cycles had a median pTRG of 2 (p .2). However, a significant association was seen between mrTRG and the number of chemotherapy cycles (p value <.05).
Polish II trial comparing SCRT followed by chemotherapy and LCRT along with concurrent chemotherapy reported a significant difference (p .006) in acute toxicity rate (45% and 60% had grade ≥2 toxicity in SCRT and LCRT arm, respectively). 31 Incidence of acute toxicity greater than grade 2 remained low in our study.
Limitations
We acknowledge that a study with a longer follow-up and a bigger cohort of patients would have thrown more light on the influence of improved radiological and pathological response rates on overall survival outcomes. Inter- and intra-observer differences in MRI reporting were significant and it resulted in mrTRG and pTRG discordance.
Conclusion
This prospective observational study has highlighted that preoperative SCRT in locally advanced rectal carcinoma is feasible and well-tolerated. Our radiological and pathological response rates are encouraging and comparable to world literature. Significant symptomatic relief in pain and bleeding per rectum was achieved rapidly leading to an improved quality of life and better tolerance to the subsequent treatment. A significant number of patients with CRM positivity at induction became CRM negative both radiologically and pathologically thereby improving the chances of R0 resection. This may result in improvement of disease-free and possibly overall survival on longer follow-up of these patients. We believe that mrTRG and pTRG concordance would have been better if the inter- and intra-observer differences could be reduced.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Clearance for the study was obtained from the Institutional Scientific Research Committee and Institutional Ethics Committee. (IRB letter no. RGCIRC/IRB-BHR/09/2021 issued on January 6, 2021).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient Consent
After counseling the patient regarding the nature of the disease, prognosis, treatment options, need for neoadjuvant and radical treatment, duration and cost of the treatment, and possible side effects, written informed consent was taken.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.