Abstract
Li–Fraumeni syndrome (LFS) is defined by the early emergence of tumors and is associated with mutations in the tumor suppression gene known as tumor protein 53 (TP53). Information is scarce regarding the psychosocial challenges encountered by individuals with LFS and how it affects their treatment decisions. A 21-year-old woman developed multiple breast tumors over a period of nine years. Genetic testing identified a heterozygous missense substitution in exon 6 of the TP53 gene, specific variant chr17:7578190T>C c.659A>G (p.Tyr220Cys) listed in the dbSNP database. The report also highlights the psychosocial implications of the diagnosis at a young age and the patient’s reluctance to undergo the recommended bilateral mastectomy. The patient exhibited a significantly higher incidence of secondary tumors, underscoring the importance of comprehensive screening and timely diagnosis for managing LFS. This case highlights the challenges faced and the importance of emotional and psychological support after the diagnosis of hereditary syndromes, especially among young women.
Introduction
Li–Fraumeni syndrome (LFS) is a rare, inherited autosomal dominant disorder associated with abnormalities in tumor protein 53 (TP53), a tumor suppression protein gene, also known as sarcoma, breast, leukemia, and adrenal gland syndrome. 1 LFS can also lead to the development of tumors in different sites and in younger age group, unlikely to occur in other hereditary cancer syndromes. A study by Rath et al. among 213 human epidermal growth factor receptor 2 positive (HER2+) breast cancer (BC) patients reported that 1.4% of patients carried a TP53 germline pathogenic variant, among which two patients satisfied the CHOMPRET criteria for LFS. 2 The occurrence of TP53 mutation in BC patients diagnosed before the age of 30 years ranged from 1% to approximately 7%.3, 4 Multiple studies have reported TP53 mutation as a primary factor involved in the development of LFS.5–8 Hisada et al. reported that the overall relative risk of developing a second cancer was 5.3, with a 57% chance of a second cancer occurrence 30 years after diagnosis. 9
According to Peters et al., the majority of patients with LFS experienced notable social and emotional distress. Additionally, they found that religious and spiritual beliefs played a pivotal role in influencing treatment decisions. The study also revealed that over two-fifths of the patients exhibited symptoms of anxiety, depression, and somatization. 10 There is a scarcity of studies addressing the psychosocial dimensions of LFS, 10 and in India, there is limited information available. Therefore, we report the case of a young patient with LFS, who was reluctant to accept recommended prophylactic surgery and lived through multiple primary breast tumors over a period of nine years due to psychosocial issues. It also highlights the lacunae in the awareness and management of common hereditary cancer syndromes in India, such as LFS and hereditary breast and ovarian cancer (HBOC), both among oncologists and lay persons.
Case Presentation
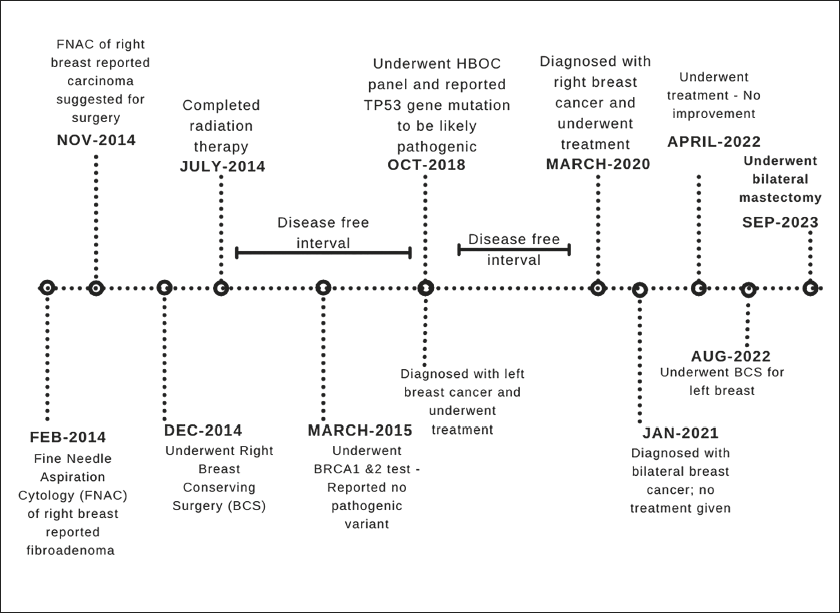
In February 2014, a 21-year-old premenopausal woman, who was a nonsmoker with a history of alcohol consumption, discovered two firm, enlarged masses in her right breast. She visited a general practitioner who recommended fine-needle aspiration cytology (FNAC). The FNAC suggested a fibroadenoma, and no further treatment was advised. During this period, she was in shock, her distress score on the distress thermometer was 7, and she experienced anxiety, shock, and distance from her immediate family.
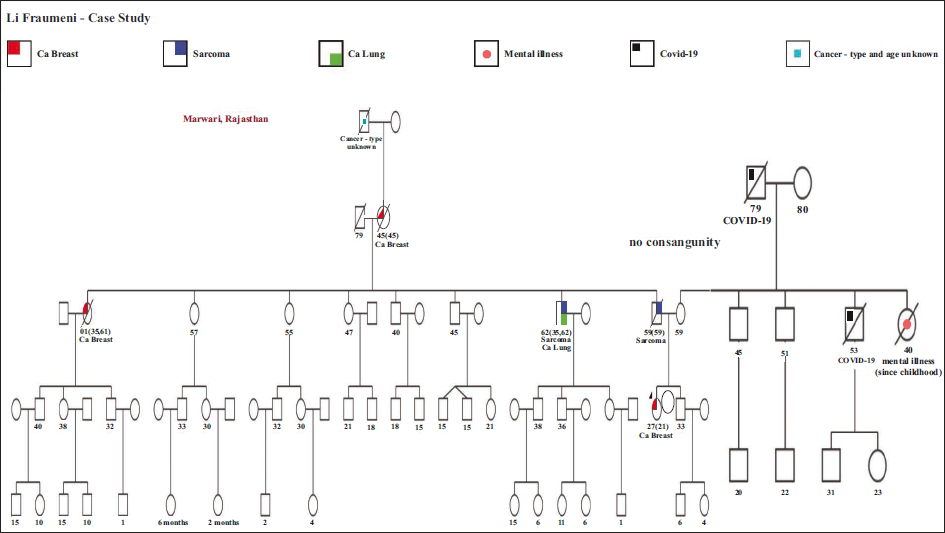
By November 2014, the breast lump had increased in size. Another FNAC was performed, revealing carcinoma, and surgery was recommended. In December 2014, she underwent a right breast-conserving surgery (BCS) with wide local excision and axillary clearance. The histopathology reported invasive ductal carcinoma (IDC), grade III, with a single focus of 2-mm residual disease. The surgical margins were tumor-free, but 2/11 lymph nodes showed metastasis with perinodal spread. Immunohistochemistry results showed estrogen receptor-positive (ER+) (70%), progesterone receptor-positive (PR+) (95%), HER2 (3+), and Ki67 at 60%. She received six cycles of adjuvant docetaxel/carboplatin/trastuzumab (TCH) and 17 doses of maintenance trastuzumab every three weeks. Adjuvant radiotherapy was delivered to the right breast and right supraclavicular fossa until July 2015. Post-radiation, she began hormonal therapy with tamoxifen and remained relapse-free for three years. Adjuvant radiotherapy was delivered to the right breast and a similar dose to right supraclavicular fossa. The treatment continued till July 2015. Post-radiation, she started hormonal therapy with tamoxifen and was relapse-free for three years. However, given her strong family history of various cancers as shown in pedigree chart (see Figure 1), she underwent germline BRCA1 and 2 gene sequencing analysis in March 2016, which reported no pathogenic variant.
Pedigree Chart of Li–Fraumeni Case.
Disease timeline of Li–Fraumeni Case.
As the treatment started, she was quite optimistic, however, she also expressed a sense of shame and isolation. Her coping mechanism was the familial support, however, by this time she had isolated herself from her friends. However, her strong spiritual beliefs and practices helped her cope with her emotional crisis.
In October 2018, a follow-up examination revealed a lump in her left breast. A core biopsy identified IDC—HER2 (3+), ER, and PR negative. Germline HBOC panel testing revealed a heterozygous “likely pathogenic” variant in the TP53 gene (chr17:7578190T>C c.659A>G p.Tyr220Cys). Despite being advised to undergo a bilateral prophylactic mastectomy, she and her family refused. She was treated with 24 cycles of Kadcyla (T-DM1), a targeted therapy regimen, and was on regular follow-up for one and a half years.
In March 2020, another lump was detected in her right breast. Sonography revealed a hypoechoic scar with microcalcification in the superior quadrant of the right breast, but no solid or cystic mass. A core biopsy from the microcalcification confirmed IDC–ER-negative, PR+ (70%), HER2 negative (score 1+). As her treatment was undergoing, she started integrating into regular society, her distress score was 4. However, the problem she started facing issues associated to her academics. She refused bilateral prophylactic mastectomy as it meant a loss of her femininity and womanhood. Marriage was important to her and her family and a bilateral mastectomy would make this impossible. Hence, she opted for medical management, receiving nine months of capecitabine and remained disease-free for eight months.
By January 2021, she was diagnosed with bilateral breast carcinoma. No treatment was taken. In August 2021, breast sonography showed an irregular heterogeneous mass in the left breast. An ultrasound-guided (USG) core biopsy-reported IDC Grade III HER2+ (score 3). Next-generation sequencing (NGS) panel testing on tissue confirmed the same pathogenic TP53 variant. She continued without treatment.
In April 2022, she was treated with four cycles of lapatinib and five cycles of capecitabine, which showed a response in the left breast. However, the right breast lump increased in size, prompting an additional three cycles of lapatinib with a combination of capecitabine and cyclophosphamide. Despite treatment, a new lump developed in the left breast. These frequent diagnoses affected her greatly and her distress score was 7, which was accompanied by fear of loss of control, and uncertainty of her future. Also, added to her distress was the loss of her parent who was a great support.
In August 2022, oncologists once again strongly recommended bilateral mastectomy due to recurrent bilateral tumor development, but she refused and opted for BCS of the left breast again. She underwent wide local excision in multiple locations along with left axillary dissection up to level III. Metastatic carcinoma was found in 1/5 level I and 1/4 level III axillary lymph nodes. A right breast lumpectomy was also performed. Immunohistochemical assessment showed the left breast was ER−, PR−, and HER2 (2+) with Ki67 at 70%, while the right breast was triple-negative breast cancer (TNBC) with Ki67 at 90%. Fluorescence in-situ hybridization (FISH) testing was not carried out. Psychological education helped her adjust to the various changes brought about by not only her diagnosis of cancer but also the presence of the genetic mutation in her body. Her family still considers marriage and their belief in astrology furthers it. However, she has since then accepted the diagnosis and has made many lifestyle choices to help her feel better about herself. Frequent counseling made her undergo bilateral mastectomy on September 2023 (Figure 2).
Discussion
In LFS, 15–35% of cancer cases will have multiple primary tumors throughout their lives.9, 11 Over two-thirds of individuals who meet the LFS diagnostic criteria have detectable TP53 gene pathogenic variants. Individuals with TP53 variant have 70–90% chance of lifetime risk of developing cancer by 70 years, with females having a higher risk of BC. 12 The identified TP53 gene pathogenic variant, c.659A>G (p.Tyr220Cys) in our patient causes a single amino acid and has a dominant negative effect, potentially increasing cancer risk.
Gonzalez et al. conducted a study among 341 families to perform TP53 analysis which includes 195 families that fulfilled CHOMPRET criteria. Among the 195 families, the mutation detection rate was 35%. Hence, according to National Cancer Institute (NCI), CHOMPRET criteria aid in diagnosing LFS.13, 14
Cancer screening for TP53 mutation carriers has reported to reduce morbidity and mortality. Also, early cancer screening in LFS will identify precancerous lesions, before they turn malignant. 15 Hence, The National Comprehensive Cancer Network (NCCN) guidelines suggest early cancer screening of breast for TP53 mutation carriers. 16
Our patient was diagnosed with HER2+ BC at a young age (<30 years) and exhibited a classic pattern indicative of LFS. At diagnosis, it should have been evident that there was a 67% likelihood of her carrying a pathological LFS variant. Ideally, she should have been initially referred to Cancer Genetic Service for a pregenetic testing consultation with a genetic counselor, where genetic testing could have been recommended.
However, in India, addressing the needs of young BC patients presents distinct challenges compared to Western countries. There is a lack of trained genetic counselors in the country. Genetic testing also has become available since 2017, but it is not widely accessible and affordable today as well. While the core principles of BC treatment remain consistent, it is imperative to account for practical, cultural, and psychosocial factors that can influence the care and outcomes of young BC patients in India, which was observed in our case report. Unfortunately, genetic testing was not available during the patient’s diagnosis and treatment time period, which further compounded the challenges. The patient only had access to a psychological counselor, which highlighted the critical need for genetic counseling in such cases. At that time, awareness among oncologists regarding hereditary syndrome diagnosis and treatment in India was low.
Furthermore, limited evidence exists on LFS in India. Indian studies such as Mishra et al. found TP53 more prevalent in intestinal subtypes, while Singh et al. reported a 7.2% TP53 mutation rate in BC patients.17, 18 Emphasizing NGS panels for CHOMPRET criteria-fulfilling patients or those with TP53 germline mutations is crucial for diagnosis and management. Additionally, alongside genetic testing, providing counseling to patients and their caregivers or families is crucial to reduce psychosocial distress.
Conclusion
This case highlights the challenges faced and the importance of emotional and psychological support after the diagnosis of hereditary syndromes, especially among young women. The management of LFS patients should include comprehensive cancer screening, surveillance, and psychosocial support. By addressing these social taboos and stigmas and promoting understanding and acceptance, individuals, and families affected by genetic predisposition to cancer can better navigate the challenges and make informed decisions about their healthcare and well-being. In addition, further research is warranted to enhance understanding and management strategies for LFS in India.
Authors’ Contribution
Dr Shona Nag (SN), Dr Shriniwas Subhas Kulkarni (SK), Dr Atul Dongare (AD), Rebecca deSouza (RD), Dr Aparna Dhar (ApD), Abhilash Patra (AP): conceptualization, investigation, resources, visualization, writing—original draft.
Dr Varun Agiwal (VA), Hira B. Pant (HBP), Dr Nirupama A. Y. (NAY), Dr Sirshendu Chaudhuri (SC), Dr Yashaswini Kumar (YK): project administration, writing—review and editing; Dr G. V. S. Murthy and SN: supervision, writing—review and editing, and all authors approved the manuscript.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient Consent Statement
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
