Abstract
Background
Stereotactic body radiotherapy (SBRT) is an important treatment modality in liver metastasis in which surgery is not feasible or when the patient denies surgery. With the recent advances in treatment planning systems and real-time image-guided treatment delivery, we can now deliver high doses of radiation in a small number of fractions to the target with high precision and minimal dose to the organs at risk (OARs).
Objective
To describe the procedural steps for radiation planning of SBRT of Liver Metastasis.
Methods
The step-by-step procedure for stereotactic planning of liver metastasis has been described using a clinical scenario of liver metastasis.
Results
The stereotactic radiation planning of liver metastasis starts with the basic history and relevant clinical evaluation followed by tissue confirmation of the diagnosis of liver metastasis. The SBRT planning for liver metastasis starts with the positioning of the patient using a body immobilization device. This is followed by the use of any of the motion management techniques during the computed tomography (CT) simulation. Magnetic resonance imaging (MRI) of the abdomen should be done in the prescribed format to achieve uniformity in radiation planning. After CT and MRI image fusion, contouring of the target, OARs, and radiation planning should be done. The plan evaluation includes target and OAR coverage index, conformity, homogeneity gradient index, and beam arrangement. After radiation plan evaluation, quality assurance and dry run are done. Then treatment is delivered using the same motion management technique that was used during the CT simulation.
Conclusion
The article highlights the sequential process of radiation planning for SBRT in liver metastasis―starting from simulation, planning, evaluation of plan, and treatment
Introduction
Liver metastasis from other primary tumors is a common phenomenon that can deleterious effect on the morbidity and mortality of the patients. Adenocarcinoma of the colon and rectum is the most common primary that metastasizes to the liver. 1 The other primaries are carcinoma breast, gastrointestinal tract tumors, lung primaries, melanoma, bladder cancer, and so on. 2 Among all the cases of liver metastasis, only 10%−20% of them are resectable and the rest are usually treated with chemotherapy in palliative intent. 1 Thus, stereotactic body radiotherapy (SBRT) has emerged as an important modality of treatment for the management of liver metastasis given the recent advances in treatment planning and delivery techniques.
Methods
In this paper, the various steps of radiation planning for SBRT have been illustrated in an easy way with the help of a clinical scenario as described below. It will be of utmost help for beginners who are planning for SBRT in a case of liver metastasis.
Results
Clinical Scenario
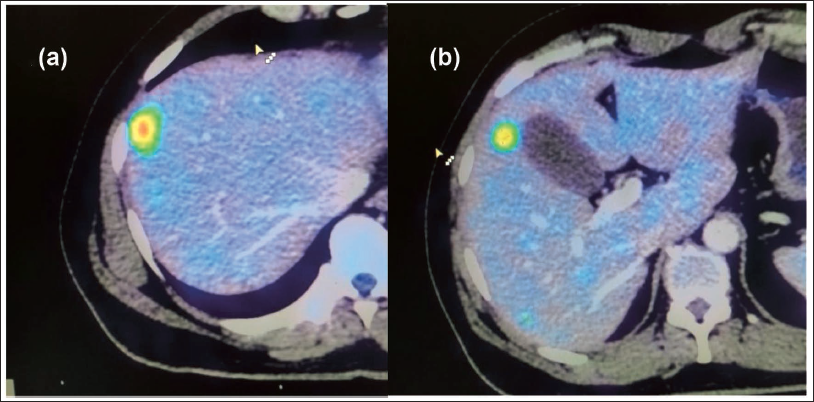
A 50-year male with a clinical diagnosis of carcinoma colon underwent right hemicolectomy. During the surgery, two lesions were found in the liver. Fine Needle Aspiration Cytology (FNAC) proved it to be metastatic adenocarcinoma. On imaging by whole-body positron emission tomography-computed tomography (PET-CT) scan, there was intense fluoro-deoxy glucose (FDG) uptake in 2 lesions – one in segment VIII and another in segment V of the liver. The largest lesion was in segment VIII with a size of 1.2 cm × 1.0 cm and a maximum standard uptake value (SUV) of 10. Out of both the lesions, one was close to the rib (Figure 1a) and the other was touching the gall bladder (Figure 1b).
Final Diagnosis
The final diagnosis of a Post-operative case of carcinoma colon with metastasis to segments V and VIII of the liver was made.
Discussion with the Patient
The patient was explained about the bouquet of treatment options such as re-surgery, radiotherapy, and chemotherapy. The patient was not interested in a second surgery.
Treatment Decision by the Tumor Board
The patient details were put in the tumor board for a decision regarding the line to treatment. After a group discussion with the surgeon, medical oncologist, and radiation oncologist, the board decided to plan for SBRT followed by palliative chemotherapy.
Decision of Radiation Tumor Board
SBRT was planned with multiple fractions on alternate days.
Dose Selection
According to the study by Eriguchi et al., while planning for SBRT in liver tumors close to central biliary structures such as gall bladder, a total dose of 40 Gy in 5 fractions can be used safely with minimal biliary toxicity. 3 Therefore, in the current case, the SBRT was planned with a dose of 40 Gy in 5 fractions keeping in mind that one of the lesions was adjacent to the gall bladder.
Radiation Planning
Here we describe the steps of treatment of liver metastasis from simulation to plan execution.
Step 1: CT Simulation
Motion Management Technique
As the liver is a mobile organ that moves with respiration, we need to use any of the motion management techniques such as active breath control (ABC) technique, abdominal compression, tumor tracking, or gating technique to account for liver movement during respiration. 4 In the current situation, we used ABC as the motion management technique for the simulation.
Patient Set-Up
During the simulation, the patient was set up in the supine position with a neutral neck position and arms above the head. The immobilization was done using an individualized vacuum mold.
Synchronizing Breath Hold with Triple Phase CT Scan
It is the most important step in SBRT planning for liver metastasis. Before triple-phase computed tomography (CT) scan-based simulation, we need to note the phase (arterial phase, portal venous phase, or delayed phase) during which the tumor was well defined from the prior images done during diagnostic CT scan or magnetic resonance imaging (MRI) of the abdomen. The breath hold was then synchronized with this particular phase of the triple-phase CT scan. For example, suppose the lesion was better seen in the portal venous phase, then the patient was asked to hold his breath for 35 sec, starting to hold his breath 10 sec after the contrast was given in order to synchronize the breath hold with the portal venous phase of the scan.
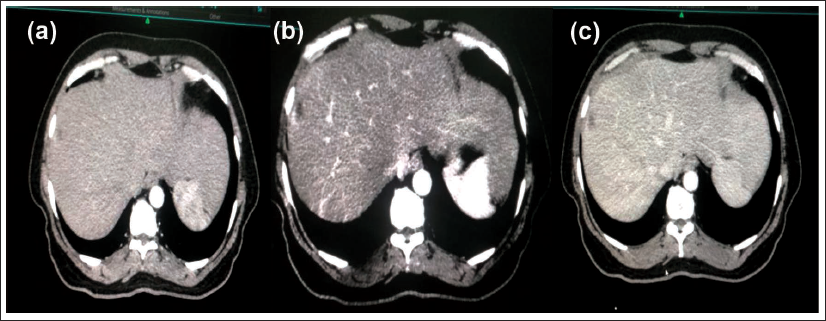
Then, a CT scan was taken from the nipple to the pelvis with a CT slice thickness of 1mm. After simulation, the digital imaging and communications in medicine (DICOM) CT images (Figure 2a−c) were sent to our treatment planning system which was then imported for delineation of target and organ at risk.
Step 2: Image Fusion
It is better not to fuse the diagnostic MRI images with the planning CT images as the MRI was usually done with free breathing while the CT simulation was done using the ABC technique. But we can take the help of complimentary image data of the MRI while targeting volume delineation.
Step 3: Target Delineation
The lesions seen on the portal venous phase of the CT scan were delineated as gross tumor volume (GTV). As there were 2 lesions, they were delineated as GTV1 and GTV2 respectively (Figure 3a,b). There was no necessity for delineation of clinical target volume (CTV) in SBRT. Riou et al., in their paper, have defined 5 mm radial and 10 mm cranio-caudal margin around the GTV as the planning target volume (PTV). 4 In the current case, we have taken a 7 mm margin both radially and craniocaudal due to the availability of daily image-guided radiotherapy and the use of the ABC technique (Figure 3c,d). Multi-planar evaluation of GTV and PTV contours was done in all three planes that are, axial plane, coronal plane, and sagittal plane.
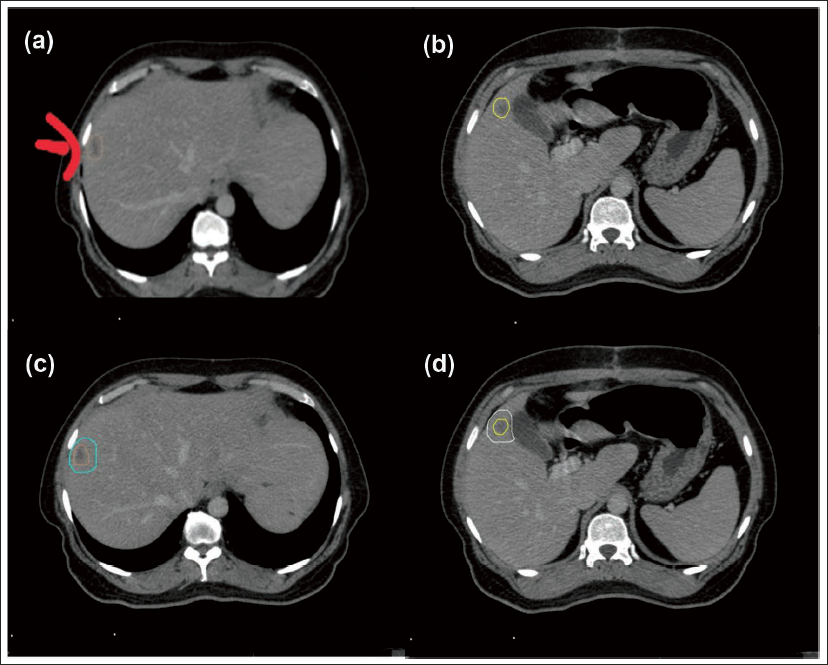
In the present case, the GTV1 volume was 2.96 cc while the GTV2 volume was 1.4 cc adding up the Total GTV was 3.63 cc. The PTV1 volume was 17.8 cc and the PTV2 volume was 10.015 cc making the Total PTV volume 27.815 cc.
Step 4: Organs at Risk Delineation
The organs at risk (OARs) for delineation included the liver, common bile duct, gall bladder, duodenum, stomach, bowel, lung, spinal cord, rib, chest wall, and both kidneys. All the OARs were contoured using the Radiation Therapy Oncology Group (RTOG) consensus guidelines. 5 Also (liver minus GTV) and (liver minus PTV) were considered as OAR.
Step 5: Radiation Technique
Radiation planning can be done using any of the radiation techniques such as Intensity Modulated Radiotherapy (IMRT), Volumetric Modulated Arc Therapy (VMAT), Dynamic Conformal Arc Therapy (DCARC), or 3-Dimensional Conformal Radiotherapy (3DCRT) according to the convenience of the radiation physicist and the physician.
In the present case, planning was done using the VMAT technique.
Step 6: Plan Evaluation
After the completion of planning by the physicist, the evaluation for the treatment plan was done using the following indices as noted below.
PTV Coverage Index
Following the planning, we need to see the PTV coverage. The prescription isodose level is usually not 100% of the prescribed dose covering 100% of the PTV. Often, 95% of the prescription dose should cover 95% or higher percentage of the PTV otherwise 100% of the prescription dose should cover 95% or higher percentage of the PTV. 6
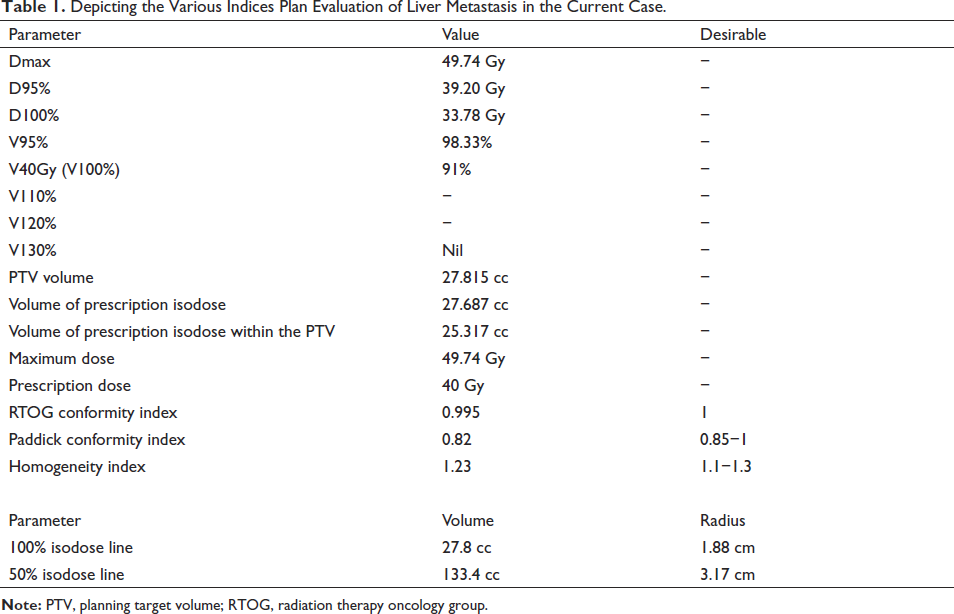
In the present case, 95% of the prescription dose covers 98.33% of the PTV and 100% of the prescription dose covers 91% of the PTV which is acceptable as per the above-mentioned parameter for the PTV coverage and is depicted in Table 1.
Depicting the Various Indices Plan Evaluation of Liver Metastasis in the Current Case.
Stereotaxy Organ Constraints and OAR Coverage
Keeping in mind the desirable dose constraints to the OAR, the dose to individual OARs needs to be checked.7, 8
The dose desirable and dose achieved for all the OARs in the present case are depicted in Table 2.
Showing the Individual OAR with its Desirable Dose and Dose Achieved in the Current Case of Liver Metastasis.

Conformity Index
To note the conformity index of the SRS, here we used two types of conformity indices that is, the RTOG conformity index and the Paddick conformity index.6, 9
RTOG Conformity Index (CIRTOG) was calculated using the following formula.
CIRTOG = Volume of Prescription Isodose/PTV volume.
In this case of liver metastasis, the RTOG conformity index was 0.995 (Table 1).
Paddick Conformity Index (CIPaddick) was calculated using the following formula.
CIPaddick = (Volume of prescription isodose in the area of interest, i.e. PTV) 2 /PTV volume × Volume of prescription isodose.
Here in the current case, the Paddick conformity index was 0.82 (Table 1).
Homogeneity Index
It was calculated using the formula:
Homogeneity Index = Maximum Dose/ Prescription Dose.
In this case, the Homogeneity Index was 1.23 (Table 1).
Dose Fall-off
The dose fall-off observation is very much needed in the plan evaluation under the heading of gradient index. For this, we need to calculate the difference between various isodose lines. In order to calculate the difference between the isodose lines, we need to calculate the equivalent radius.
Equivalent Radius Calculation
To evaluate the dose gradient, we have to find out the difference between the radii of various isodose lines. But none of the isodoses are spherical. So, we use the following formula to calculate the equivalent radius. First: Pick one isodose volume. Second: Calculate the radius of the isodose volume by using the formula:
V = 4/3 ύr3
r = (3V/4 ύ)1/3
The calculation of the volume and radius of various isodose lines in the present case is shown in Table 1.
Gradient Index
The gradient index was calculated as per the 2 formulae given below.
Formula 1: Gradient Index = Equivalent radius of 50% isodose − Equivalent radius of prescription isodose. Ideally, the gradient index should be between 0.3−0.9 mm.
In the current case, the gradient index is 3.17 cm–1.88 cm = 1.2 cm which is near to the ideal gradient index.
Formula 2: Gradient Index = volume enclosed by half of the prescribed isodose divided by prescribed isodose volume. 6
In the current case, Gradient Index = 20 Gy volume/40 Gy volume that is, 133.4 cc/27.8 cc = 4.7. Ideally, it should be less than 3.
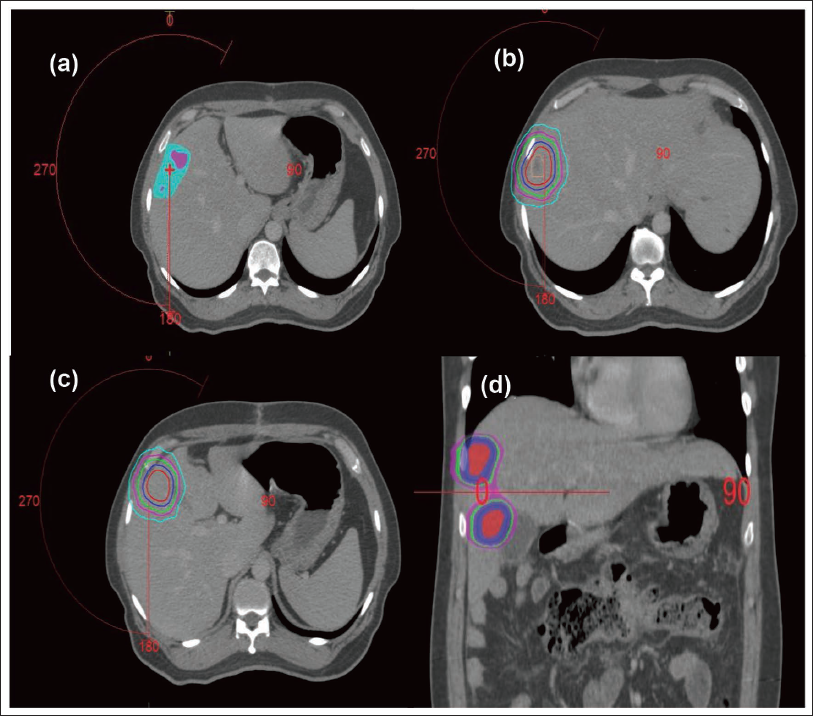
Beam Arrangement
The arrangement of the beams (Figure 4a) was done such that there is adequate coverage of the target while giving less dose to the OARs. The isodose distribution is shown in Figure 4b−d.
Step 7: Quality Assurance (QA)
Mechanical isocenter check was done using the Winston Lutz test and the point dose verification was done keeping the tolerance as 1 mm. 10
Step 8: Dry Run
Treatment verification consists of setup reproduction, isocenter verification and clinically verifying each treatment field―check beam clearance, check any interlock―MLC interlock and potential Monitor Unit (MU) problems. Then clearly mark the immobilization devices after the successful dry run.
Step 9: Pre-medication Protocol
Prior to the start of treatment, pre-medication was delivered in the form of tablets as described below―all starting the day before the start of radiation treatment.
Tablet Dexamethasone 8 mg―one tablet thrice daily.
Tablet Ondansetron 8 mg―one tablet thrice daily.
Tablet Pantoprazole 40 mg―one tablet once daily.
If the patient is diabetic, proper diabetic care needs to be done.
Step 10: Set up Verification and Treatment Delivery
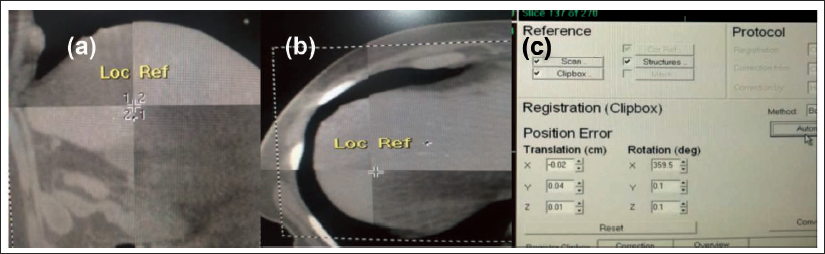
It includes cone beam CT correction (Figure 5a,b) and hexapod corrections (Figure 5c). After all the corrections have been done treatment is delivered.
Step 11: Post-medication
It is an optional protocol that usually includes antiemetics, proton pump inhibitors and tapering the dose of steroids over a week.
Step 12: Advice and Follow-up
After the completion of the treatment the patient was referred to the Department of Medical Oncology for further treatment by chemotherapy as per the multi-disciplinary tumor board decision and was advised to follow up with a PET-CT scan after three months for review.
Conclusion
This paper conceptualizes and acts as an easy guide for beginners for the stereotactic radiation planning for liver metastasis.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Ethical Approval and Informed Consent
Necessary ethical clearances and informed consent were received and obtained respectively before initiating the study from all participants.
