Abstract
Objective:
The current study sought to explore the impact of a novel noninvasive treatment called transcranial photobiomodulation (PBM) on resting-state functional connectivity (rsFC) of the cerebellum in individuals with a history of repetitive head acceleration events (RHAEs).
Background:
RHAEs are associated with cumulative neurological compromise, including chronic alterations in rsFC; however, few treatments have been investigated to mitigate these effects. A recent study by our team demonstrated that PBM treatment led to improvements in measures of balance and motor function in adults with RHAE exposure. Due to the cerebellum’s role in motor functioning and its connectivity with cerebral regions targeted by PBM, we sought to examine changes in the rsFC of the cerebellum following PBM treatment.
Methods:
Thirty individuals with RHAE used an at-home PBM headset with an intranasal cannula for 20 min every other day for a period of 8–10 weeks. A total of 240 J/cm2 of red and near-infrared light was administered to cortical nodes of the default mode network (DMN) and subcortical structures in each session. Using resting-state functional magnetic resonance imaging, rsFC was measured before and after treatment in 11 cerebellar seed regions associated with the DMN, salience network (SN), and frontoparietal network (FPN).
Results:
We observed an overall decrease in between-network connectivity and an increase in within-network connectivity after PBM treatment, particularly in the SN and FPN.
Conclusions:
These results suggest that PBM may improve network efficiency of the cerebellum by regulating overactive connections and increasing network segregation.
Introduction
Repetitive head acceleration events (RHAEs) can result from direct blows to the head or indirect impacts to other parts of the body and do not always result in immediate signs or symptoms of neurological dysfunction. However, there is a growing concern that the cumulative neurological injury associated with RHAEs, despite the absence of immediate symptoms, may generate long-term deficits across multiple domains, including cognitive, psychological, and motor functioning, and potentially contribute to the development of chronic traumatic encephalopathy and other neurodegenerative disorders. 1,2 Recent research suggests that RHAE exposure may be related to changes in regional brain volume, 3,4 task-based functional connectivity, 5 and white matter microstructure. 6 In addition, resting-state functional magnetic resonance imaging (rsfMRI) studies of athletes suggest that alterations in resting-state functional connectivity (rsFC) may occur after RHAE, 7 –9 potentially influencing the development and persistence of symptoms. However, few potential treatments for these effects have been investigated.
Research from our team and others has shown that photobiomodulation (PBM), a noninvasive form of near-infrared (NIR) light therapy that has traditionally been used to promote wound healing, inflammation reduction, and pain management, 10 may improve symptoms of RHAE when applied transcranially. 11,12 RHAEs can lead to diverse physiological changes in the brain that are known to be linked to chronic neurological impairments, including neuroinflammation, blood–brain barrier disruption, and axonal damage. 13 PBM may directly influence these issues by improving mitochondrial function, increasing metabolic activity, bolstering cell survival and proliferation, reducing reactive oxygen species, and modulating inflammatory signaling pathways. 11,14 –16 PBM may therefore have neuroprotective and potentially restorative effects on brain function, particularly after RHAE. 13 In a larger study of participants with RHAE, PBM led to significant improvements in motor reaction time, grip strength, and postural control, as measured by the Mini Balance Evaluation Systems Test. 17
The cerebellum plays a key role in motor functioning due to its intrinsic circuitry and involvement in functional networks. 18 Further, recent literature has established nodes of the default mode network (DMN), salience network (SN), and frontoparietal network (FPN) in the crus I (C1) and crus II (C2) subregions of the cerebellum, 19 suggesting that these networks extend into cerebellar structures. Since the PBM devices used specifically targeted brain regions functionally connected to the cerebellum, cerebellar rsFC may underlie some of the beneficial effects on balance and motor function we previously observed in a subset of this sample.
The current proof-of-concept study sought to investigate whether PBM treatment is associated with changes in rsFC of the cerebellum in individuals with RHAE exposure. We hypothesized that PBM would enhance network segregation between cerebellar nodes of the DMN, SN, and FPN and the rest of the brain by increasing within-network connections and decreasing between-network connections.
Methods
Participants
Forty-nine participants (92% male) were recruited via referral, word-of-mouth, and community flyers. Adults aged 18–69 years with a history of RHAE, based on free-text responses to the Ohio State University Traumatic Brain Injury (TBI) Identification Method, 20,21 were included. The mechanism of RHAE was determined by free response. Participants with a history of neurological disease, psychiatric disorder, or magnetic resonance imaging (MRI) contraindication were excluded. All participants provided written informed consent before enrollment. All study activities were approved by the institutional review board at the University of Utah and conducted in accordance with the standards of ethics outlined in the Declaration of Helsinki.
PBM treatment
The Vielight Neuro RX Gamma (v4) at-home PBM device (Toronto, Ontario, Canada) was used for PBM treatment. Participants were instructed to self-administer treatment every other day for a duration of 20 min for the length of the study (8–10 weeks, as recommended by the device manufacturer) and track their usage of the device. The reported compliance rate for PBM usage was 92%. The PBM device emits a total of 240 J/cm2 of NIR through four light-emitting diodes targeting the cortical nodes of the DMN. Detailed PBM device specifications and settings are provided in Table 1.
Photobiomodulation Device Specifications and Settings
Imaging data acquisition
All MRI data were acquired on a 3 Tesla Siemens MAGNETOM Skyra (Siemens, Best, Germany) whole-body scanner using a 64-channel head coil. A high-resolution, three-dimensional T1-weighted magnetization prepared rapid gradient-echo imaging (MP-RAGE) sequence was used to acquire 208 slices of structural data along the sagittal plane [repetition time/echo time/inversion time (TR/TE/TI) = 2300/4.63/1010 ms, voxel size = 1 × 1 × 1 mm, field of view (FOV) = 240 × 256 mm, matrix size = 256 × 240 mm, flip angle = 9°]. A gradient recalled echo – echo planar imaging (GRE-EPI) sequence was used to acquire 200 volumes of 2D resting-state blood oxygen level dependent (BOLD) data with 48 contiguous axial slices while participants remained awake with eyes open (TR/TE = 3000/30 ms, voxel size = 3.31 × 3.31 × 3.29 mm, gap = 3.96 mm, FOV = 212 × 212 mm, matrix size = 64 × 64 mm, flip angle = 80°). In addition, to correct for susceptibility-induced distortion, EPI images with reversed phase encoding were acquired with 72 axial slices (TR/TE = 8000/66 ms, voxel size = 2 × 2 × 2 mm, FOV = 208 × 208 mm, matrix size = 104 × 104 mm, flip angle = 90°).
Image processing and statistical analysis
Eleven spherical seed regions of interest (ROIs; radius = 4 mm; Fig. 1) were identified in C1 and C2 lobes of the cerebellum using a set of functionally constrained regions shown to be associated with the DMN, FPN, and SN. 19 A breakdown of ROI coordinates and corresponding functional networks is provided in Table 2. Details of imaging preprocessing are provided in the Supplementary Data.
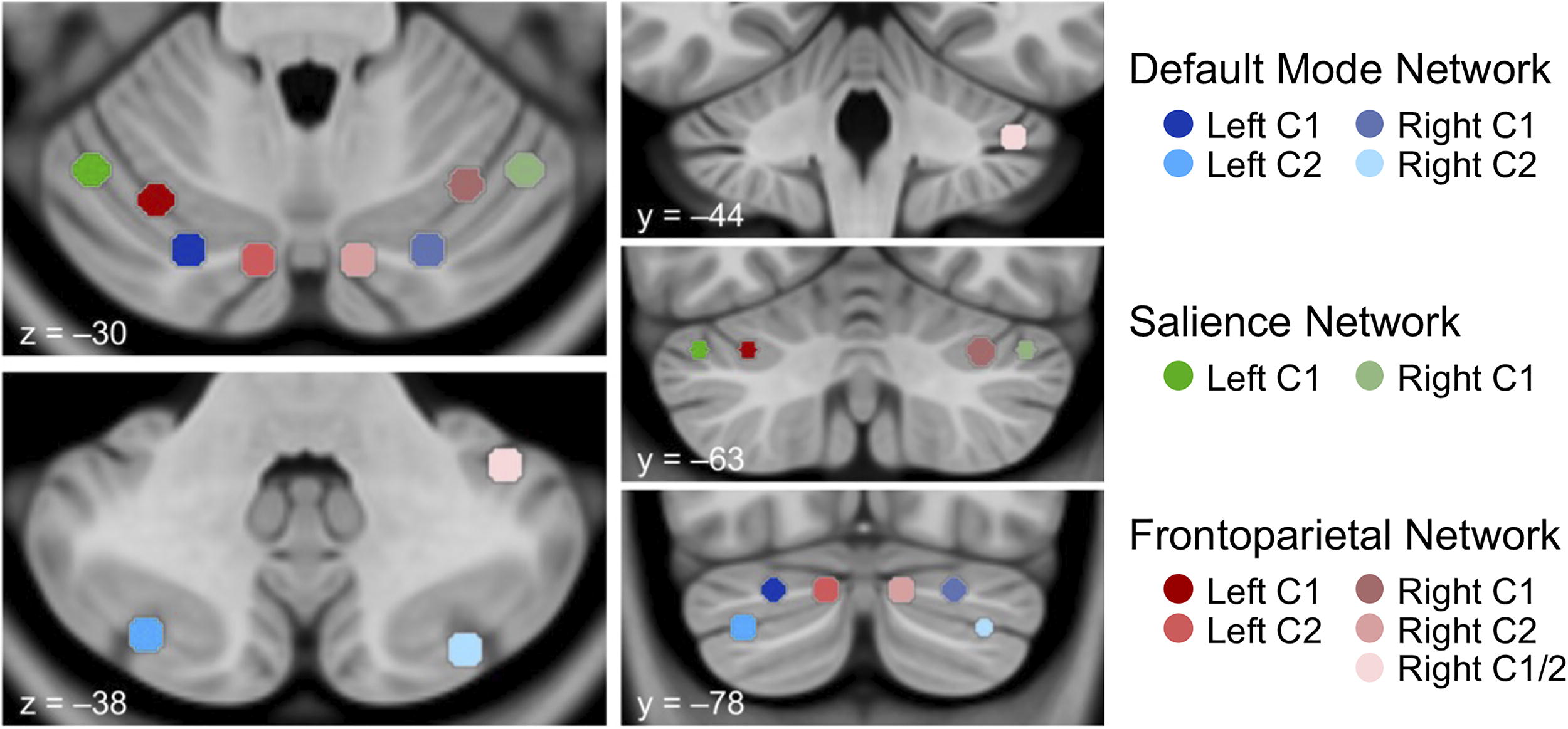
Locations of seed regions of interest. Seed locations are overlaid on axial (z = –30, z = –38) and coronal (y = –44, y = –63, y = –78) slices of the cerebellum in MNI template space (LPI orientation; left = left) and color-coded according to associated network (default mode network = blue, salience network = green, frontoparietal network = red). C1, crus 1; C2, crus II; LPI, lateral–posterior–inferior.
Seed Regions of Interest
All seeds have a radius of 4 mm. MNI coordinates are reported in lateral–posterior–inferior (LPI) orientation.
C1, crus I; C2, crus II; DMN, default mode network; FPN, frontoparietal network; ROI, region of interest; SN, salience network.
A seed-based analysis was conducted because we had hypothesized that specific regions of the cerebellum (seeds of the cerebellar DMN) would have altered functional connectivity with the cerebellar and cortical DMN and other networks. Because this was an exploratory study, we chose to correlate the seeds with the whole brain. To determine changes in rsFC of the cerebellum after PBM, correlation coefficients were computed between the BOLD time course fluctuations of each seed region individually and all other voxels in the brain before and after treatment. A Fisher’s r-to-z transformation was applied to ensure that the coefficients were normally distributed. As this was an exploratory analysis and due to the small sample size, the cluster-defining height threshold was set at an uncorrected p < 0.005, with a cluster size of 84, determined using Monte Carlo simulation, and correction for multiple seeds was not performed. Significance at the cluster level of inference (over time) was defined by an uncorrected cluster threshold of p < 0.001. For individual associations at each time point, Cohen’s d was calculated to provide effect sizes. 22 In addition, standard errors (SE) and 95% confidence intervals were computed for each effect size using 10,000 iterations of bootstrapping. The first-level analysis utilized a seed-based analysis with a one-sample t-test to examine functional connectivity of the 11 cerebellar seed regions by calculating the correlation between their time series and every other voxel in the brain. The second-level analysis utilized a paired t-test to identify voxels where the connectivity strength with each seed and the rest of the brain was significantly different between at pre>post (1–1) and at pre<post (−1 1) across all subjects.
The brain regions included in each cluster were labeled automatically by the two default atlases built into the CONN Functional Connectivity Toolbox, 23 the Harvard-Oxford Atlas for cortical regions and the Automated Anatomical Labeling atlas for cerebellar regions. Any proportion of voxels that fell outside of the atlas boundaries for a given cluster were manually traced in DSI Studio, and each voxel was labeled according to the Montreal Neurological Institute (MNI) nearest gray matter atlas, HCP842 tractography, 24 or the FLIRT/FNIRT cerebellum 25,26 atlases and confirmed through visual inspection by an expert in neuroanatomy (HML).
Results
Of the initial 49 participants, 30 participants (28 males), 22–69 years of age (M = 46.9 ± 15.07), were included (Table 3). Six were lost to follow-up, and 11 had missing or low-quality rsfMRI data. Imaging data from six participants were excluded due to wrap artifact from an FOV that was too small.
Sample Demographic and Injury Characteristics
PBM, photobiomodulation; RHAE, repetitive head acceleration event; SD, standard deviation; TBI, traumatic brain injury.
Since we were comparing the same participants at two time points, a one-sample t-test was used to generate seed-based connectivity maps for the whole sample averaged over time for each seed region (Fig. 2). In the following sections, we report the locations of peak voxel activity for each cluster associated with the seed regions for each network at pre-treatment > post-treatment (pre > post) and at post-treatment > pre-treatment (post > pre). All other regions covered by each cluster are listed in Table 4 (pre > post) and Table 5 (post > pre).
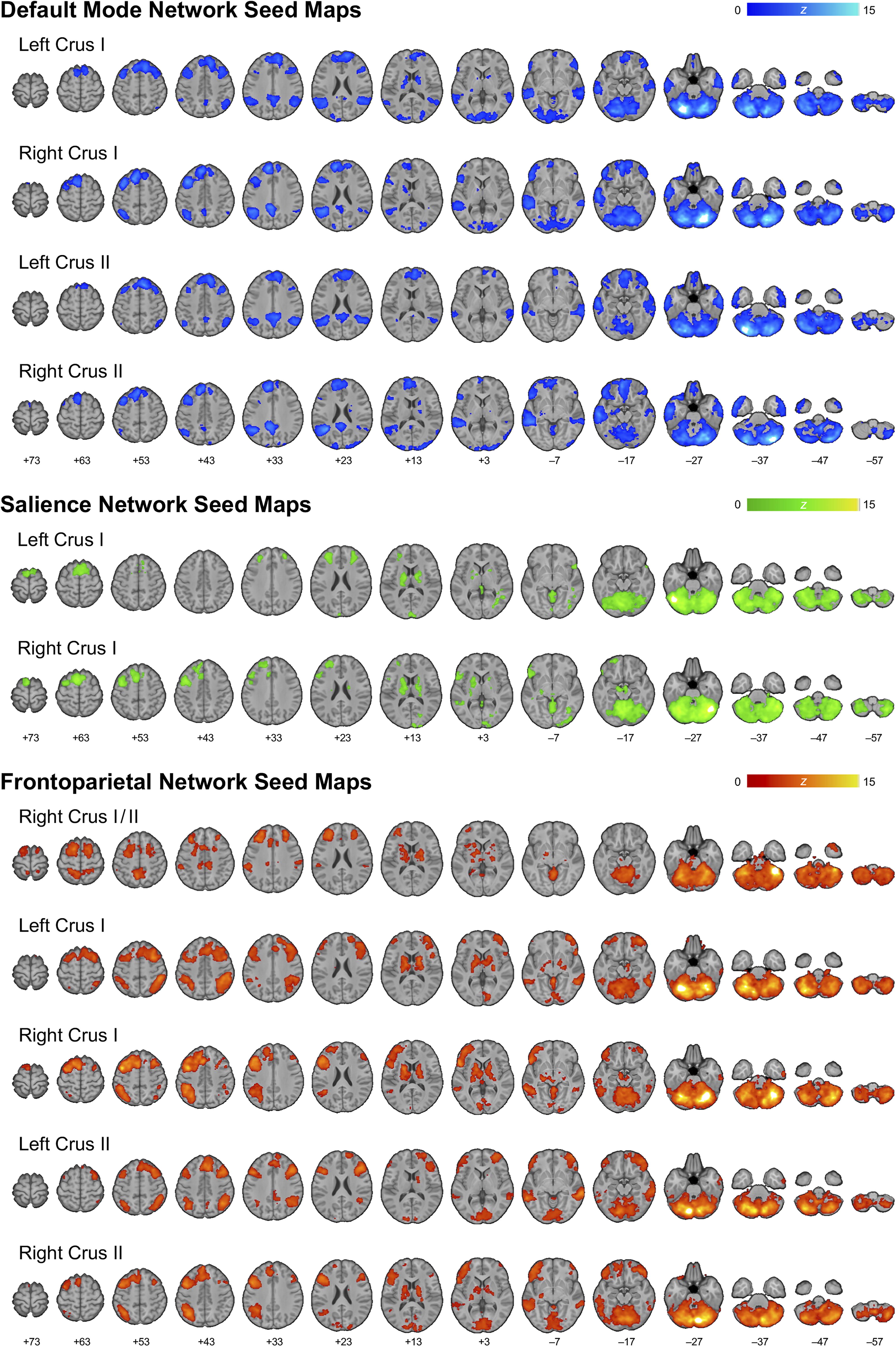
Seed connectivity maps. Sample-specific seed-based connectivity maps for each region of interest are overlaid on an array of axial images in MNI template space, lateral–posterior–inferior (LPI) oriented, and represented in neurological view (left = left).
Resting-State Functional Connectivity with the Cerebellum Before (Pre > Post) Photobiomodulation Intervention
x, y, and z refer to peak voxel coordinates in MNI152 space, after the lateral–posterior–inferior (LPI) convention. All findings are thresholded at a cluster-defining (height) threshold of an uncorrected p < 0.005 after a Monte Carlo simulation (extent 84 contiguous voxels), and all between-group differences are significant at p < 0.001. Standard errors and confidence intervals for Cohen’s d effect size estimates were determined using 1000 bootstrap iterations. Regions covered by each cluster were identified using CONN’s default Harvard-Oxford cortical parcellation atlas for cortical regions and the Automated Anatomical Labeling (AAL) atlas for cerebellum regions. Regions where peak activity occurred are indicated by an asterisk (*). Any regions that were not labeled according to these built-in atlases were identified using the MNI nearest gray matter atlas (†) in Mango, the HCP842 tractography atlas (‡), or expert identification (+). Within- and between-network cluster regions are indicated by bold and italicized text, respectively. C1 = crus I. C2 = crus II. Hemi = hemisphere. k = cluster size/volume. L = left. R = right. Effect size is reported as Cohen’s d, where |d| ≥ 0.2, 0.5, and 0.8 are considered small, moderate, and large effects, respectively.
SE, standard error; 95% CI, 95% confidence interval.
Resting-State Functional Connectivity with the Cerebellum After (Post > Pre) Photobiomodulation Intervention
x, y, and z refer to peak voxel coordinates in MNI152 space, after the lateral–posterior–inferior (LPI) convention. All findings are thresholded at a cluster-defining (height) threshold of an uncorrected p < 0.005 after a Monte Carlo simulation (extent 84 contiguous voxels), and all between-group differences are significant at p < 0.001. Standard errors and confidence intervals for Cohen’s d effect size estimates were determined using 1000 bootstrap iterations. Regions covered by each cluster were identified using CONN’s default Harvard-Oxford cortical parcellation atlas for cortical regions and the Automated Anatomical Labeling (AAL) atlas for cerebellum regions. Regions where peak activity occurred are indicated by an asterisk (*). Any regions that were not labeled according to these built-in atlases were identified using the MNI nearest gray matter atlas (†) in Mango, the HCP842 tractography atlas (‡), or expert identification (+). Within- and between-network cluster regions are indicated by bold and italicized text, respectively. C1 = crus I. C2 = crus II. Hemi = hemisphere. k = cluster size/volume. L = left. R = right. Effect size is reported as Cohen’s d, where |d| ≥ 0.2, 0.5, and 0.8 are considered small, moderate, and large effects, respectively.
Pre-treatment > post-treatment rsFC
Widespread and primarily between-network rsFC was observed pre > post across the three networks of interest (Table 4; Fig. 3). Specifically, pre > post rsFC of the DMN was observed with one region within the DMN (right C2—left frontal pole) and six task-positive network regions: the cerebellar network (left C1—left cerebellum lobule 7 b), FPN (left C1—right superior frontal gyrus), auditory network (right C1—right superior temporal gyrus), visual network (VN; left C2—right posterior sagittal striatum/occipital lobe), SN (right C2—left and midline anterior cingulate cortices), and sensorimotor network (SMN; right C2—right postcentral gyrus).
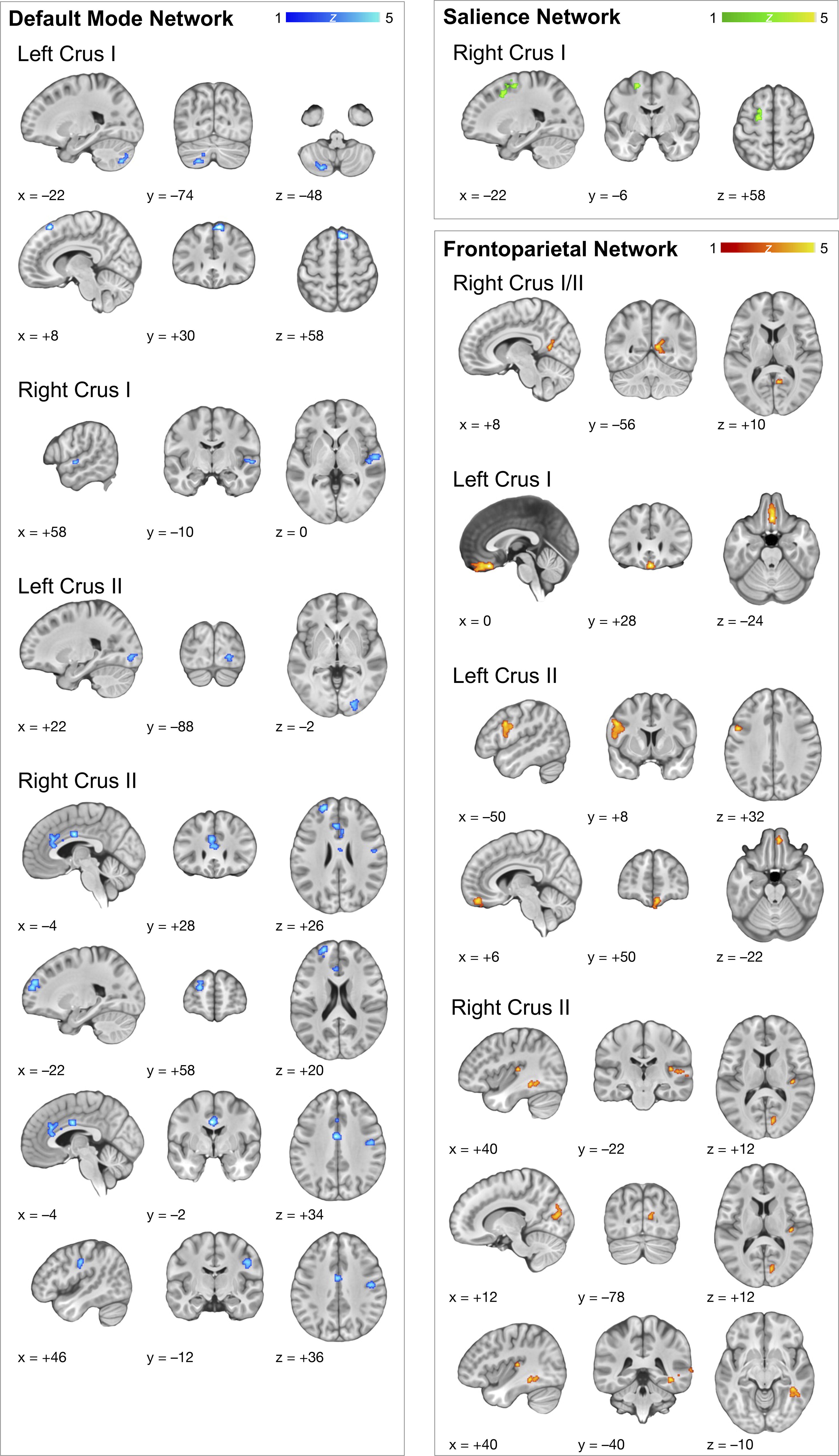
Resting-state functional connectivity (rsFC) of the cerebellum before PBM intervention. Pre > post-treatment rsFC of the default mode, salience, and frontoparietal network seed regions of interest. Peak voxel activity of each associated cluster is overlaid on a template brain standardized in MNI space, lateral–posterior–inferior (LPI) oriented, and represented in neurological view (left = left).
Pre > post rsFC of the SN demonstrated a single association with a major FPN hub (right C1—left superior frontal gyrus); no associations were observed with the left C1, left C2, or right C2 seeds.
Pre > post rsFC of the FPN included seven associations with regions of the DMN (right C1/2—right precuneus, left C1—midline subcallosal cortex, left C2—right medial frontal gyrus), regions of the motor (left C2—left precentral gyrus), auditory (right C2—right Heschl’s gyrus), and visual (right C2—right intracalcarine cortex and right fusiform gyrus) networks.
Pre-treatment < post-treatment rsFC
Fewer clusters were associated with the networks of interest after the PBM intervention compared with pre-treatment rsFC (post > pre). The observed increase in post-treatment rsFC occurred both within and between-network regions (Table 5; Fig. 4).
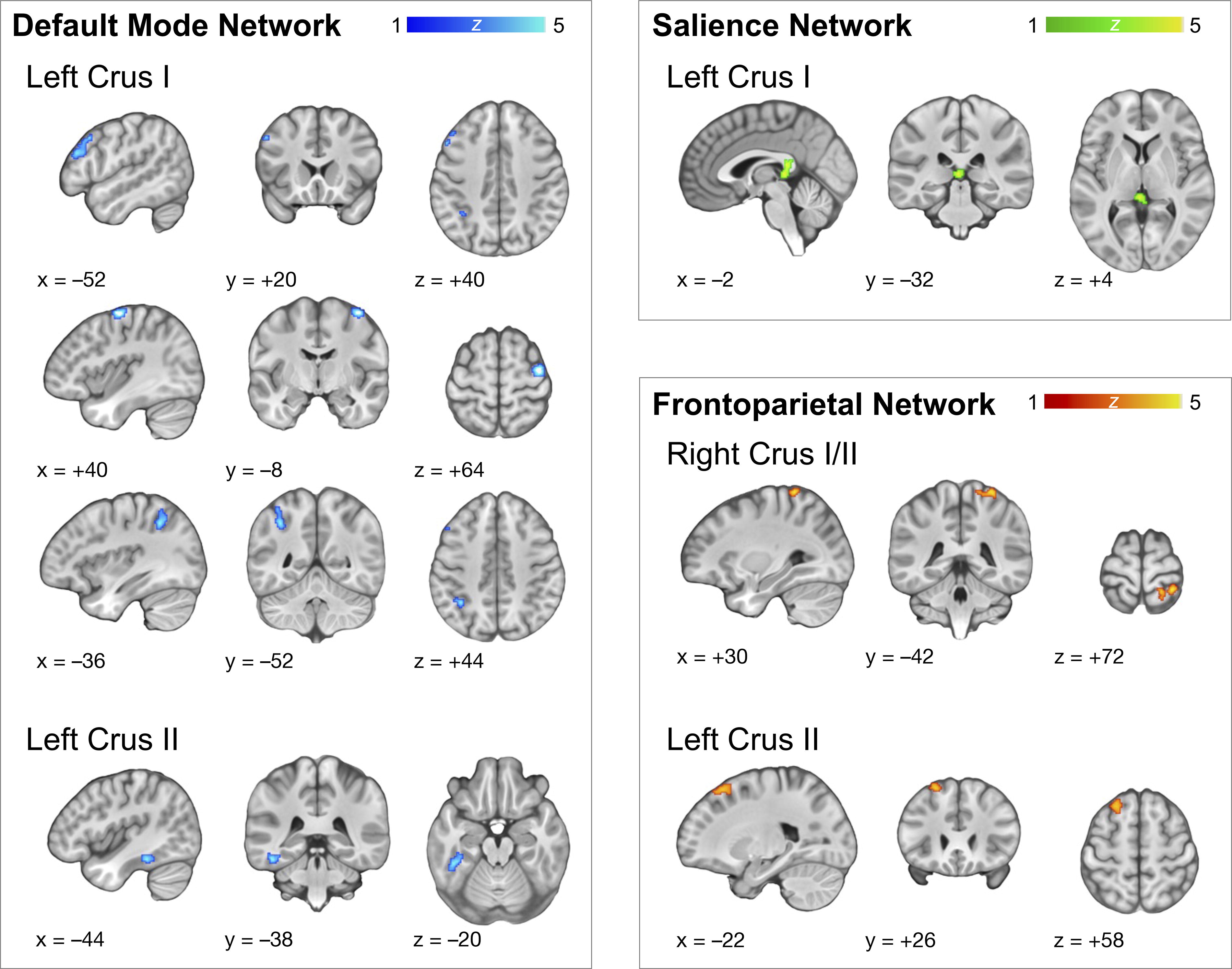
Resting-state functional connectivity (rsFC) of the cerebellum after PBM intervention. Post > pre-treatment rsFC of the default mode, salience, and frontoparietal network seed regions of interest. Peak voxel activity of each associated cluster is overlaid on a template brain standardized in MNI space, lateral–posterior–inferior (LPI) oriented, and represented in neurological view (left = left).
Specifically, post > pre rsFC of the DMN occurred with three regions associated with the FPN (left C1—left middle frontal gyrus and left inferior parietal lobe), SMN (left C1—right precentral gyrus), and VN (left C2—left fusiform gyrus).
Greater rsFC was observed between the SN and one cluster that covered other SN regions (i.e., thalamus), but peak voxel activity occurred within the white matter (left C1—splenium of corpus callosum).
Finally, post > pre rsFC of the FPN occurred with one other FPN region (left C2—left superior frontal gyrus) and one region associated with the SMN (right C1/2—right postcentral gyrus).
Discussion
Previous research from our laboratory found that PBM treatment was associated with improvements in balance and motor functioning in individuals with RHAE. 17 Given the cerebellum’s role in motor functioning and its connectivity with the DMN, changes in cerebellar rsFC may underlie the beneficial effects of PBM previously observed in a subset of this sample. We sought to determine if PBM would be associated with changes in rsFC between cerebellar seed regions of the DMN, SN, and FPN and the rest of the brain. We found significant alterations in cerebellar rsFC after PBM treatment (Fig. 5). Further, we observed both a decrease in between-network connections and a subtle increase in within-network connections after PBM treatment, suggesting that PBM may increase network communication and efficiency.
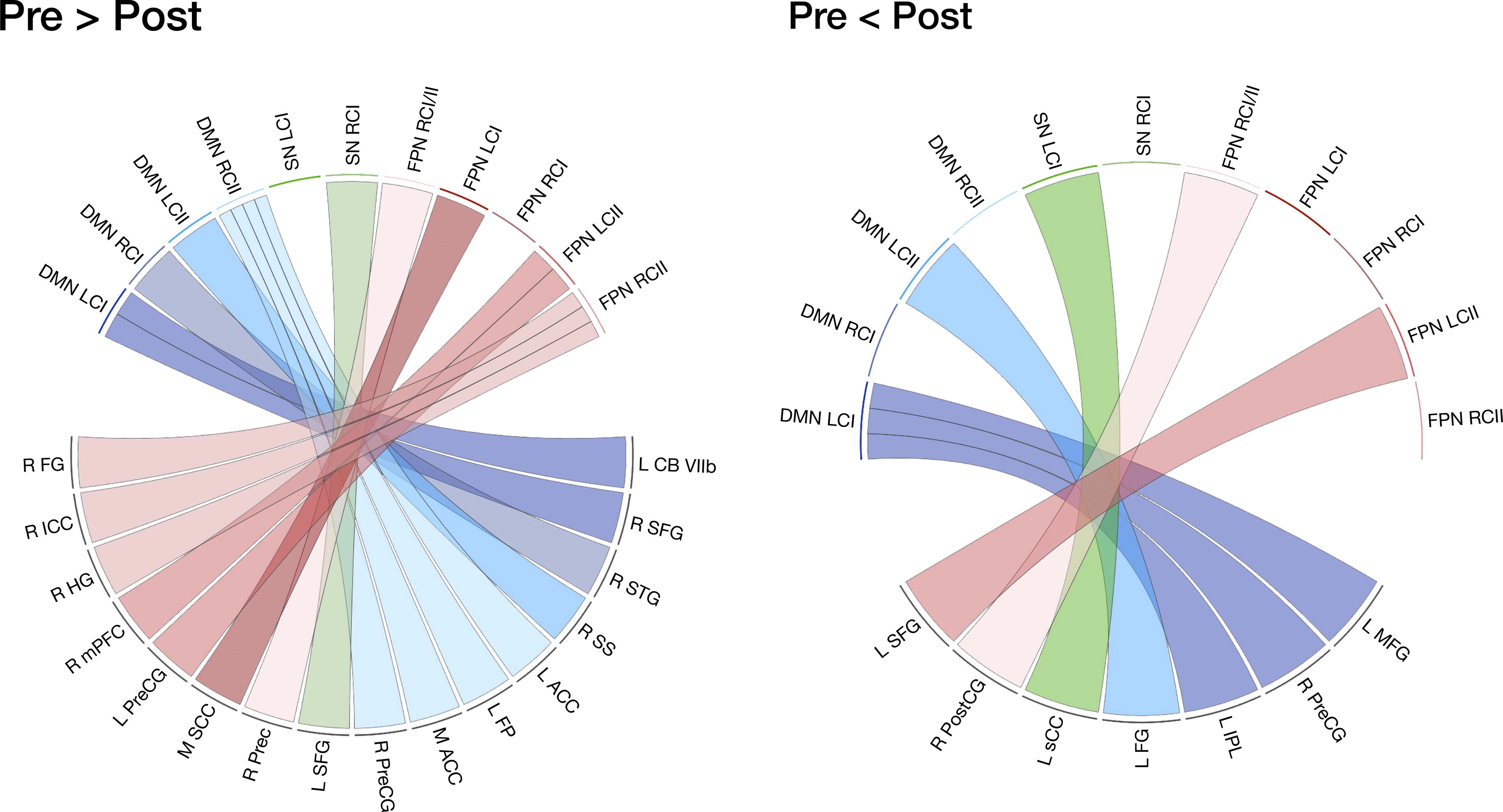
Chord diagram representing cerebellar rsFC pre > post and pre < post-treatment. Significant associations in pre > post-treatment rsFC (left) and pre < post-treatment (right) of the default mode network (DMN), salience network (SN), and frontoparietal network (FPN) seed regions of interest. ACC, anterior cingulate cortex; CB VIIb, cerebellar lobule VIIb; CI, crus I; CII, crus II; FG, fusiform gyrus; FP, frontal pole; HG, Heschl’s gyrus; ICC, intracalcarine cortex; IPL, inferior parietal lobule; L, left; M, midline; MFG, middle frontal gyrus; mPFC, medial prefrontal cortex; PreCG, precentral gyrus; SCC, subcallosal cortex; Prec, precuneus; PostCG, postcentral gyrus; R, right; sCC, splenium of the corpus callosum; SFG, superior frontal gyrus; SS, sagittal stratum; STG, superior temporal gyrus.
It is thought that networks in the brain are segregated to maximize efficiency while minimizing metabolic cost. 27 Consequently, task-positive networks, such as the SN and FPN, often maintain a reciprocal relationship with the task-negative DMN in healthy brains. 28,29 Network desegregation is related to poorer cognitive and motor function and occurs naturally as part of the aging process. 30,31 Desegregation also occurs after TBI, such that task-positive networks and the DMN display synchronized activity. 32,33 Given the positive effects of PBM previously observed, 17 we anticipated that PBM would enhance network segregation by increasing within-network rsFC and decreasing between-network rsFC.
We found that connections stronger before PBM treatment were primarily between-network, aligning with prior studies suggesting that network desegregation often follows TBI 32 –34 and with our hypothesis that PBM contributes to a reduction in between-network rsFC with the cerebellum. However, stronger within-network DMN connectivity was observed with the frontal pole and anterior cingulate gyrus, and one within-network FPN connection was observed with pars opercularis. Although these results do not support our hypothesis, they may reflect a reduction of the within-network hyperconnectivity that may occur in response to brain injury. Hyperconnectivity is an adaptive response that allows the brain to maintain communication within damaged networks by optimizing network efficiency and minimizing metabolic cost. 35 However, chronic hyperconnectivity is linked to an increased risk of neurodegeneration and impaired motor and cognitive functions. 35 Hyperconnectivity disproportionately occurs in the brain’s most interconnected network hubs, such as the dorsal and ventral DMN, right executive control network, and SN. 36 In a study of chronic moderate-to-severe TBI, hyperconnectivity in these network hubs remained within-network and did not contribute to postinjury desegregation. 27 Thus, the stronger within-network connections observed before PBM may be the result of chronic hyperconnectivity. This is further supported by the overall decrease in rsFC observed after treatment, suggesting that PBM may regulate excessive within-network connections.
After PBM treatment, we also observed an increase in within-network connectivity with the cerebellar seeds. Within-network connectivity was increased for the FPN, particularly with one cluster on the left superior and middle frontal gyri, which work together to support a variety of cognitive functions, such as attention, working memory, and episodic memory. 37,38 Another cluster was located predominantly in the superior parietal lobule, with peak voxel activity in the postcentral gyrus. As part of the SMN, the postcentral gyrus has been shown to coactivate with the FPN during tasks involving cognitive control, particularly those that require spatial attention and complex sensorimotor integration. 38,39
Increased rsFC was observed post-treatment between the SN and a cluster on the splenium of the corpus callosum and medial pulvinar nucleus of the left thalamus. The SN modulates the balance between task-negative and task-positive network activity based on the salience of incoming stimuli, with the thalamus aiding efficient cognitive resource allocation by segregating internally and externally oriented subsystems. 40 The pulvinar is involved in visual attention 41 ; however, the role of the medial pulvinar nucleus is less understood. Given its widespread cortical and subcortical connections, it is considered to be a multi-modal integration nucleus implicated in the modulation and selectivity of directed attention, 42 potentially through the binding of emotionally relevant features during working memory updating. 43 The corpus callosum facilitates information transfer between hemispheres and maintains homotopic FC. 44 The splenium handles the interhemispheric transfer of visual information, and thalamocortical commissures 45 contribute to this function. 46 Our results suggest that PBM may enhance SN rsFC in regions tied to interhemispheric transfer of visually and emotionally relevant information. Notably, post-treatment changes occurred predominantly in cortical areas ipsilateral to the seed region. Given the cerebellum’s contralateral organization, this pattern suggests overall improvement in interhemispheric rsFC after PBM.
In contrast to the observed increase in within-network connectivity in the FPN and SN, increased between-network connectivity was observed between the DMN and regions of the FPN, SMN, and VN after PBM. While the DMN is traditionally associated with resting-state activity, 47 it is also linked to internally focused cognition and overlaps with various executive, sensorimotor, and perceptual functions. 48,49 Notably, DMN-FPN coactivity has been associated with goal-directed cognitive tasks 50 and the interaction of emotional and executive control, 51 and increased connectivity between the DMN and FPN has been shown in relation to improved cognitive functioning after cognitive rehabilitation in adults with chronic TBI. 34 Our findings of increased rsFC between the DMN and key FPN regions, including the middle frontal gyrus and posterior parietal cortex, suggest that PBM may enhance connectivity in areas critical for executive control, emotional regulation, and goal-oriented attention. 52,53 As hypoconnectivity can occur in these regions after TBI and may contribute to functional deficits, the enhanced connectivity observed may contribute to improvements in executive function. However, further studies are required to confirm this.
The left C1 seed of the DMN also demonstrated increased rsFC post-treatment with a cluster in the right precentral gyrus, a key SMN region responsible for the execution of motor movements known to coactivate with the DMN during certain goal-oriented movement tasks. 54 The brain regions involved in goal-oriented movement are subserved by the dentatorubrothalamic tract, which projects directly from the frontal cortex, including the precentral gyrus, to the middle cerebellar peduncle, which includes contralateral C1 terminations. 55,56 Thus, our findings of increased rsFC between the DMN and precentral gyrus likely reflect enhanced structural integration of pathways involved in the coordination and execution of purposeful movement, further supporting our previously reported findings of improved motor functioning after PBM treatment in a subset of this sample. 17
Finally, in support of our previously described findings that PBM improves rsFC of cerebellar SN seed regions with areas involved in visual and emotional processes, we also observed increased rsFC between the DMN and the fusiform gyrus, a core VN region that plays a critical role in high-level visual processing 57,58 and is implicated in emotion perception and social cognition. 59 Strengthened connectivity between the DMN and the fusiform gyrus suggests enhanced integration of internally directed cognitive processes with visual–perceptual mechanisms, which may facilitate more efficient processing of emotionally salient stimuli. Emotions manifest as distributed representations of interactions across multiple networks, including the SN, DMN, and VN. 60,61 Recent work suggests that the DMN and VN are especially strong contributors to the emotional experience by integrating visual cues with internally generated thoughts and memories. 62 Given that the fusiform gyrus is a central node in the VN, its increased connectivity with the DMN may reflect an enhanced capacity for processing emotional stimuli in a way that incorporates both external perceptual and internal cognitive-affective elements.
Overall, our findings indicate that PBM treatment leads to significant changes in cerebellar rsFC in individuals with a history of RHAE. The observed decrease in between-network connectivity across networks suggests that PBM may improve network efficiency by regulating overactive connections. The increased within-network connectivity of the FPN and SN reflects a strengthening of network segregation, potentially promoting more specialized and efficient within-network communication. These changes in rsFC may underlie the previously reported improvements in motor function and balance after PBM, 17 particularly by enhancing connectivity in motor control regions and sensorimotor integration pathways. Moreover, the observed increases in DMN connectivity with regions involved in executive control and emotional regulation, as well as VN and SMN regions, suggest that PBM may also support broader cognitive and emotional processes. While the mechanisms of these changes in rsFC are unclear, they may relate to PBM’s physiological effects, 13 which may directly target these outcomes by enhancing mitochondrial function, reducing oxidative stress, and modulating inflammatory signaling pathways, thereby restoring more balanced neural signaling and improving neural efficiency. These findings provide evidence that PBM could be an effective intervention for regulating overactive brain networks and restoring functional network organization, offering a potential therapeutic avenue for individuals affected by RHAE and other forms of TBI.
Limitations and future directions
While this study offers promising preliminary evidence of the potential of PBM to improve rsFC in individuals with RHAE, several limitations need to be addressed. The small, predominantly male sample limits generalizability, and future studies should include larger, more diverse groups for improved statistical power. Participant variability in RHAE severity, frequency, and symptom duration also impacts outcomes, highlighting the need for more homogeneous samples or subgroup analyses. In addition, while we considered adjusting for age, the small sample size led us to prioritize maintaining statistical power over controlling for this variable. Future research should aim to correct for age and other confounding variables, such as body mass index, to better understand their influence on the effects of PBM. The absence of a control group prevents definitive conclusions about the effects of PBM, so future research should include matched controls. In addition, we did not measure the frequency with which treatments were completed, so it is possible that the total duration of exposure was inconsistent across the sample. Finally, the long-term effects of PBM are unknown, and extended follow-up studies are needed to determine whether benefits persist or require supplemental sessions.
Conclusions
Our findings suggest that PBM treatment leads to significant changes in the rsFC of the cerebellum, specifically by regulating overactive network connections and improving network segregation. The observed decrease in between-network connectivity, coupled with increases in within-network connectivity, indicates that PBM may reduce chronic hyperconnectivity associated with exposure to neurotrauma and promote more efficient communication within and across brain networks. These results support the use of PBM as a potential therapeutic intervention for the rehabilitation of functional brain networks. These findings provide a compelling foundation for future research on the mechanisms of PBM, particularly its role in regulating resting-state network activity and improving functional outcomes in individuals with RHAE and other brain injuries.
Footnotes
Acknowledgments
The authors gratefully acknowledge the participants of this study and their family members. The authors also acknowledge the thoughtful contribution of Margaret Naeser and H. Christian Davidson and the technical support provided by Jessica Guinn, Stephani Debenham, Maria Gonzalez, Catharine Taylor, Brandon Johnson, Brittney Sosa, and Kasey Kawakami. The authors also acknowledge Vielight, Inc. for providing the equipment used in this study.
Authors’ Contributions
F.K.: Conceptualization, formal analysis, investigation, visualization, writing—original draft, and writing—review and editing. C.E.: Conceptualization, data curation, formal analysis, writing—original draft, and writing—review and editing. H.M.L.: Conceptualization, data curation, formal analysis, visualization, writing—original draft, and writing—review and editing. M.R.N.: Conceptualization, data curation, formal analysis, writing—original draft, and writing—review and editing. P.K.J.: Conceptualization, methodology, and writing—review and editing. D.J.: Conceptualization, data curation, formal analysis, writing—original draft, and writing—review and editing. E.S.H.: Investigation and writing—review and editing. D.T.: Investigation and writing—review and editing. C.M.: Investigation and writing—review and editing. H.A.R.: Investigation and writing—review and editing. R.C.W.: Software, validation, and writing—review and editing. C.M.M.: Writing—review and editing. C.V.: Investigation and writing—review and editing. E.N.R.: Investigation and writing—review and editing. M.J.L.: Validation and writing—review and editing. L.E.D.: Validation and writing—review and editing. S.W.L.: Writing—review and editing. D.F.T.: Validation and writing—review and editing. L.S.C.: Resources and writing—review and editing. E.A.W.: Conceptualization, methodology, project administration, resources, supervision, and writing—review and editing.
Disclaimer
The views, opinions, interpretations, conclusions, and recommendations expressed in this article are those of the authors and do not reflect the views or official policy of the U.S. Department of Veterans Affairs.
Author Disclosure Statement
L.S.C. has a consulting role with Vielight, Inc., but had no role in collecting or analyzing data for this study.
Funding Information
This research was supported in part by the Traumatic Brain Injury and Concussion Center PBM Research Fund and the Keith Rivera Fund. Vielight, Inc. donated the PBM equipment used in the present study and covered costs associated with open-access publication of this article. Vielight, Inc. had no role in the study design, data collection, analysis, interpretation of results, or manuscript preparation.
Supplementary Material
Supplementary Data
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
