Abstract
The legalization of cannabis in several states across the United States has increased the need to better understand its effects on the body, brain, and behavior, particularly in different populations. Previous rodent studies have revealed age and sex differences in response to injected Δ9-tetrahydrocannabinol (THC). However, the pharmacokinetic and pharmacodynamic properties of THC administered through more translationally relevant routes of administration are less well known. Here, we addressed this gap by investigating age and sex differences in pharmacokinetics and the acute behavioral effects of vaporized cannabis e-liquid in mice. Adolescent (postnatal day [P] 35–50) and adult (≥P70) mice of both sexes received noncontingent exposure to vehicle vapor, 150 mg/mL (CAN150), or 300 mg/mL (CAN300) vaporized cannabis extract diluted in either 80% propylene glycol/20% vegetable glycerin (PG/VG) or 100% polyethylene glycol 400 (PEG). Immediately after exposure, body temperature, hot plate withdrawal latency, and locomotion were assessed. Blood was collected at 0, 30, and 60 min after vapor exposure, and plasma THC, 11-hydroxy-THC, and 11-nor-9-carboxy-THC were analyzed. THC concentrations were higher in both the plasma of vapor-exposed mice and the cannabis extract solutions when PEG was the carrier oil compared with PG/VG. Vaporized cannabis (mixed with PEG) at the highest dose tested induced hypothermic, antinociceptive, and locomotor-suppressing effects in all groups of mice. We found a dose-dependent age difference in locomotion, indicating that adolescents were less sensitive to the locomotor-suppressing effects of vaporized cannabis, which may be related to differences in circulating THC levels. Although we found no sex differences in the acute behavioral effects of vaporized cannabis, there were sex differences in plasma THC metabolites, suggesting that female mice may metabolize vaporized cannabis more slowly than male mice. Taken together, these findings add to a growing literature implementing vaporized cannabinoid delivery approaches by revealing PEG as a more effective carrier oil than PG/VG for studies involving cannabis extract.
Introduction
The expansion of recreational cannabis legalization in the United States has been met with decreasing harm perceptions, broader social acceptance, and rising daily cannabis use. 1 Inhalation is one of the most common routes of cannabis use in humans,2,3 and cannabis vaping dramatically increased in adolescents from 2017 to 20192,4 and has remained high since. 5 Despite the decreasing stigma and rise of cannabis vaping, the pharmacological effects of acute cannabis intoxication from vaping, particularly in different populations, remain poorly understood.
Cannabis from the Cannabis sativa plant is composed of over 120 phytocannabinoids, 6 including tetrahydrocannabinol (THC) and cannabidiol (CBD). When consumed via e-cigarettes or vaping, viscous cannabis extract is often mixed with carrier oil (also referred to as thinning agent) to facilitate vaporization. 7 Propylene glycol/vegetable glycerin (PG/VG) and polyethylene glycol (PEG) are organic solvents that are found in commercially available cannabis e-liquids8,9 and are employed in pre-clinical THC or cannabis vapor exposure studies.10–14 Many factors including device type, operating voltage, cannabis e-liquid composition, and puff frequency and duration can influence the composition of e-liquid aerosol. 15 The effects of different carrier oils on THC concentration in cannabis e-liquid and the resulting pharmacokinetic and behavioral effects of the aerosolized product are largely unknown.
Human laboratory studies employing vaporized cannabis have shown sex differences in its acute pharmacological effects, with females exhibiting higher plasma levels of THC metabolites and reporting greater sensitivity to subjective drug effects. 16 Historically, pre-clinical studies of cannabinoids have used either synthetic cannabinoids or isolated constituents (i.e., THC) and predominantly administered them via intraperitoneal injections. Notably, pharmacokinetics11,17 and behavioral effects17,18 greatly differ between injected and inhaled THC. More recently, the vaporized cannabinoid model in rats has gained popularity, with multiple studies recapitulating the sex difference in pharmacokinetics reported in humans who use vaporized cannabis—namely, higher plasma levels of the active THC metabolite 11-hydroxy-Δ9-tetrahydrocannabinol (11-OH-THC) in females.11,12,19–21 In rodents, cannabinoids tend to produce a canonical tetrad of behavioral effects—hypothermia, antinociception, suppressed locomotion, and catalepsy.22,23 In general, female rodents tend to exhibit greater behavioral sensitivity to cannabinoid injections compared with males. 24 However, recent reports using vaporized THC have found no sex differences in vaporized THC-induced hypothermia in adolescent20,25 or adult rats, 11 suggesting that sex differences in some tetrad effects may depend on route of administration and plasma THC levels achieved.
Considerably less is known about age differences in the pharmacology of vaporized cannabis, with no studies to date including a direct comparison of adolescents with adults. However, one human study reported that young adults (aged 21–25 years old) experienced greater craving and deleterious cognitive effects after smoking THC-dominant flower compared with older adults (aged 55–70 years old), despite achieving similar plasma THC levels. 26 These findings are in line with another human laboratory study that employed orally administered THC and found that adolescent humans (aged 18–20 years old) were more sensitive to the acute behavioral and cognitive effects compared with adult humans (aged 30–40 years old). 27 Recent pre-clinical studies have reported higher plasma THC metabolites (11-OH-THC and the inactive metabolite, 11-nor-9-carboxy-THC [THC-COOH]) following intraperitoneal injection of THC in adolescent relative to adult mice of both sexes.28,29 In these studies, the sexes were studied separately, which precluded the analysis of interaction effects of age and sex in mice. Therefore, while existing data suggest that cannabis metabolism and its behavioral effects may differ by age and sex, this has not been systematically assessed using a pre-clinical model that mimics the composition and route of administration most often used by humans. Furthermore, it will be important to examine potential age and sex effects on cannabis pharmacokinetics and behavioral endpoints, as sex differences in cannabis use emerge during the adolescent-to-adult transition in humans.30,31
We sought to fill this gap in knowledge by using a model of vaporized cannabis exposure14,19,21,32,33 in mice to investigate age and sex differences in the pharmacology of acute cannabis exposure. Importantly, our model uses a whole-plant-derived cannabis extract instead of isolated THC, which increases the translational relevance by more closely resembling the commercially available broad-spectrum products used by humans. Establishing this model in mice is an important first step to pave the way for future mechanistic investigations that leverage the extensive genetic toolkit available in mice compared with rats. We acutely exposed mice to vaporized vehicle or cannabis e-liquid that differed by dose and carrier oil and measured the acute behavioral effects and pharmacokinetics via plasma THC and metabolites. We hypothesized that acute exposure to whole-plant cannabis vapor would produce hypothermia, antinociception, and suppress locomotion in a dose-dependent manner. Importantly, we hypothesized that these effects would be influenced by age and sex, with adolescents and females showing greater pharmacological effects of acute vaporized cannabis exposure.
Methods
Experiment
Subjects
Experiments were conducted during the light cycle (7–13 h). 414 C57BL/6J mice (205 females, 209 males) were run in the experiment with the following design: 2 sex × 2 age × 3 dose × 2 carrier oil with blood collected at three timepoints (Fig. 1). Adolescent (postnatal day [P] 35–50) and adult (≥P70) mice of both sexes underwent noncontingent vapor exposure to 0 mg/mL (vehicle), 150 mg/mL (CAN150), or 300 mg/mL (CAN300) cannabis extract diluted in 80% PG/20% VG or 100% PEG 400 with blood collected at 0, 30, or 60 min post-exposure. Since mice were euthanized for blood collection, mice for the 0-min timepoint did not undergo behavioral testing. The 30- and 60-min groups underwent behavioral testing immediately following exposure as described below. All procedures were approved by the Washington State University Institutional Animal Care and Use Committee.
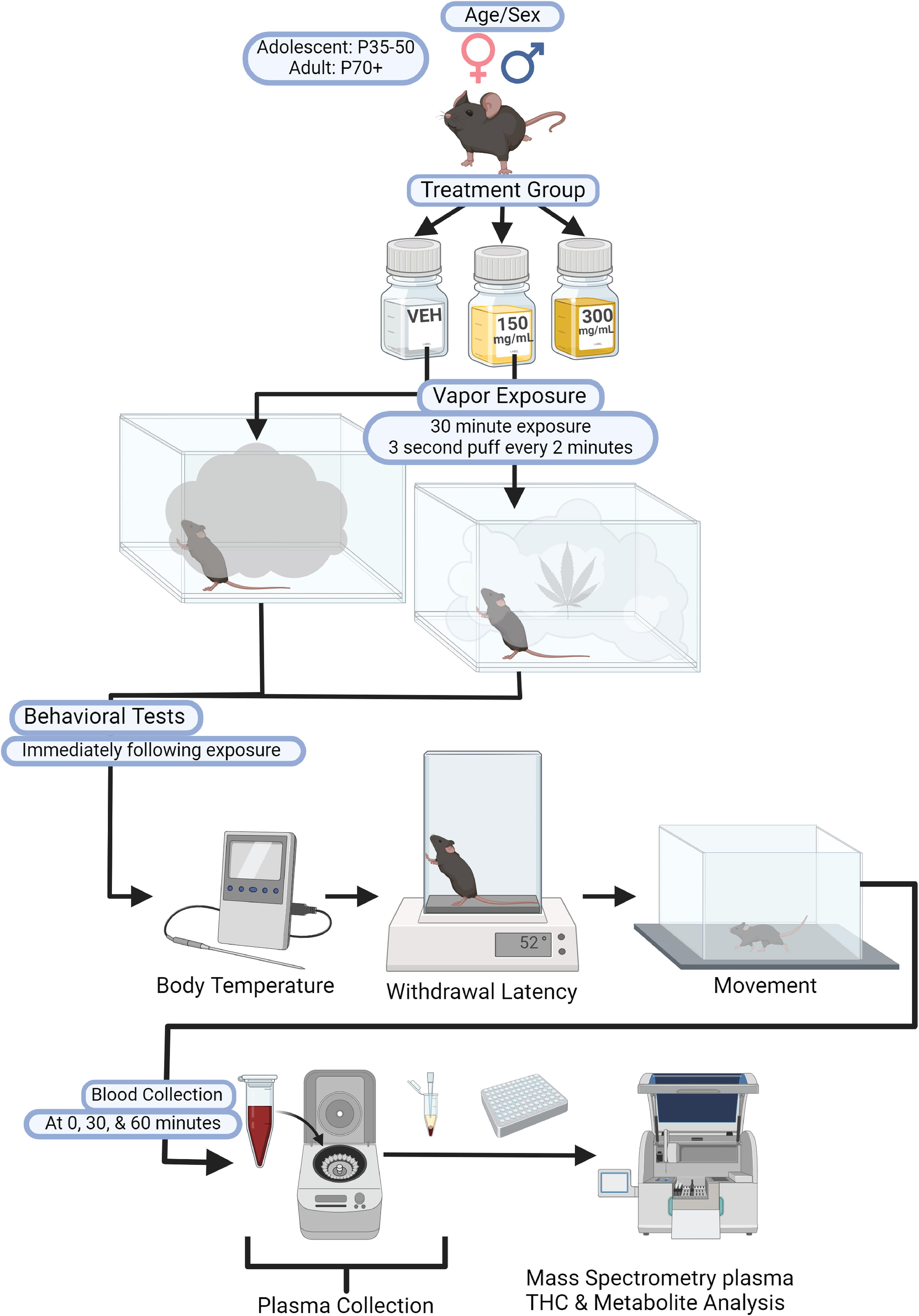
Schematic of the experimental paradigm. Adolescent and adult mice of both sexes were exposed to either vehicle, 150 mg/mL, or 300 mg/mL cannabis vapor for one 30-min session. Blood was collected either immediately after the session (0-min timepoint) or after completion of behavioral testing (body temperature, hot plate, and open field) at 30 or 60 min after the vapor session. Blood samples were centrifuged, and plasma was collected for THC and metabolite measurements via mass spectrometry. THC, Δ9-tetrahydrocannabinol. Created in BioRender. Buursma, J. (2024). https://BioRender.com/c01m565.
Drugs
Whole-plant cannabis extract was obtained from the National Institute on Drug Abuse (NIDA) Drug Supply Program. Raw extract contained 60.31% THC, 0.15% CBD, 2.38% cannabigerol, 2.03% cannabinol, 1.18% cannabichromene, 0.78% tetrahydrocannabivarin, and 0.25% Δ8-THC. Cannabis e-liquid was prepared at 150 mg/mL (CAN150) and 300 mg/mL (CAN300) concentrations by dissolving raw cannabis extract into a vehicle of 80% PG/20% VG or 100% PEG 400 under continuous stirring at 60°C, as described previously. 24
Experimental design
Pre-exposure baselines
On the first experimental day, mice were habituated to the testing room for 1 h prior to hot plate exposure (Hot Plate Analgesia Meter—Columbus Instruments). Mice were placed within an acrylic cylinder (12.7 cm diameter × 25.4 cm height) on the hot plate, which was heated to 52°C. Hot plate withdrawal latency was determined by the number of seconds it took for the mouse to flick or lick a hind paw or jump. Mice were removed from the hot plate if no response was exhibited within 30 sec. Pre-exposure hot plate measurements included three trials with an intertrial interval of 5 min. The first pre-exposure trial was used to habituate the mouse to the hot plate, whereas trials two and three were averaged to generate the pre-exposure withdrawal latency.
On exposure day (24–48 h later), mice were weighed and then habituated to the room for 30 min. Body temperatures were taken immediately before and after vapor exposure using a rectal thermometer (Physiotemp digital thermometer) lubricated with Vaseline and inserted 2 cm.
Vaporized cannabis exposure
Eight 22 × 20 × 14 cm (L × W × H) vapor chambers from La Jolla Alcohol Research Inc. (LJARI; La Jolla, CA, USA) were controlled by a tablet with custom-designed software from LJARI, Inc. to deliver noncontingent vapor puffs as previously described. 25 One to two mice (cagemates only) were placed in a vapor chamber with an iso-PAD (Braintree Scientific). A 3-sec vapor puff was administered every 2 min with simultaneous illumination of a cue light for a total of 15 puffs during the 30-min session.
Post-exposure behavior
At the end of exposure, 30- and 60-min blood collection groups had temperatures taken, one trial of post-exposure hot plate conducted, and were put into the locomotion chamber, a clear acrylic box (28 × 28 × 20.5 cm) (W × L × H). Locomotion was video-recorded using a webcam (Logitech C920e HD 1080p) for 15 min. Catalepsy is another component of the canonical cannabinoid tetrad that assesses an animal’s ability to correct an experimenter-imposed posture. 23 In preliminary experiments, we found no evidence of catalepsy in the bar hang test following vapor exposure at the doses tested, consistent with published reports that catalepsy occurs at THC doses (i.p.) three- to sixfold higher than those that cause locomotor suppression 34 and at blood THC concentrations greater than what is reported here. 35 Therefore, we did not include the bar hang test in our behavioral measures.
Blood collection and metabolites
Mice with 0-min timepoints were euthanized for blood collection immediately after vapor exposure. Mice from the 30- and 60-min timepoint groups underwent behavioral testing immediately post-exposure before being euthanized for blood collection at their respective times post-exposure. Mice were euthanized using CO2, after which intracardiac blood collection was performed. Blood was collected within 15 min of target collection time and put in an Eppendorf tube with 30 µL of EDTA. Samples were centrifuged at 2,000 rcf for 15 min at 4°C, and plasma was extracted into 100 µL duplicates and stored at −80°C. Cannabinoids in plasma (THC, 11-OH-THC, and THC-COOH) were analyzed by the Washington State University Tissue Imaging, Metabolomics, and Proteomics Laboratory, Pullman, WA, as previously described 36 using a Synapt G2-S (Waters) mass spectrometer. Samples with a value of 0 were included, and samples with a value below the limit of detection (<1 ng/mL) were excluded from statistical analyses.
Data analysis
All behavioral videos were analyzed for total locomotion (cm) using offline behavioral tracking software, DeepLabCut 37 and SimBA. 38 Statistical analyses were performed in SAS OnDemand for Academics: Studio statistical software using PROC MIXED for analyses of variance (ANOVAs) and PROC FREQ for chi-square tests of independence. Partial correlations were performed in the Statistical Package for the Social Sciences (SPSS) program version 29 (IBM, Armonk, NY, USA). GraphPad Prism software (version 10, San Diego, CA, USA) was used to generate all graphs.
Behavioral dependent measures were hot plate withdrawal latency difference score (calculated as post-exposure−pre-exposure average), body temperature difference score (calculated as post-exposure−pre-exposure), and total locomotion (cm) in the open arena. Plasma data were analyzed with plasma THC, plasma 11-OH-THC, and plasma THC-COOH as dependent variables. Final sample sizes for behavior and plasma data are provided in Supplementary Tables S1 and S2, respectively, while information for mice that met exclusion criteria is provided in Supplementary Table S3.
All factors (age, sex, dose, carrier oil, timepoint) were between subjects. Chi-square tests of independence were conducted on plasma data to determine whether there was an association between carrier oil and the proportion of plasma samples with THC, 11-OH-THC, and THC-COOH values below the level of detection (<1 ng/mL). Five-way ANOVAs were conducted on plasma levels of THC, 11-OH-THC, and THC-COOH with age, sex, dose (CAN150, CAN300), carrier oil, and timepoint (0, 30, and 60 min) as factors. Four-way ANOVAs were used to assess the interaction of age, sex, dose (vehicle, CAN150, CAN300), and carrier oil (PG/VG, PEG) on body temperature difference score, hot plate difference score, and total locomotion. When significant effects of carrier oil were detected, separate analyses were conducted for each carrier oil.
p-Values below 0.05 were considered statistically significant. Significant interaction effects were followed up with separate sequentially smaller ANOVAs and simple main effects analyses. To account for multiple testing, we used the false discovery rate adjustment for p-values (less than 5%). 39 For significant effects of factors with more than two levels, Tukey’s post hoc comparisons were used.
A series of partial correlations were computed to characterize the relationships between plasma THC levels and the behavioral endpoints while controlling for age, sex, dose, and carrier oil. Plasma THC values were converted to z-scores within timepoint groups (30 and 60 min), so that data could be combined for analysis. Moreover, partial correlations were conducted examining the relationships between plasma THC metabolites (11-OH-THC and THC-COOH) levels and the behavioral endpoints while controlling for age, sex, and dose. These partial correlations were only conducted when PEG was the carrier oil (see Results), so the plasma 11-OH-THC and THC-COOH were converted to z-scores within PEG carrier oil and within each timepoint (i.e., 30 and 60 min). Dummy coding of categorical variables for partial correlations was performed as follows: age (adolescent = 0, adult = 1), sex (female = 0, male = 1), dose (CAN150 = 0, CAN300 = 1), and carrier oil (PG/VG = 0, PEG = 1).
Results
Plasma THC and metabolites
Carrier oil as a factor
Blood was collected at three timepoints after acute cannabis vapor (CAN150, CAN300) exposure (0, 30, or 60 min) for analyses of plasma levels of THC and its metabolites, 11-OH-THC and THC-COOH. Chi-square tests of independence revealed that plasma levels below the level of detection (<1 ng/mL) were significantly more common when the carrier oil was PG/VG compared with PEG (Supplementary Tables S4–S6; plasma THC: Χ2 [1, N = 314] = 10.6670, p = 0.0011; plasma 11-OH-THC: Χ2 [1, N = 314] = 55.2650, p < 0.0001; plasma THC-COOH: Χ2 [1, N = 314] = 9.3916, p = 0.0022). This finding led us to test whether carrier oil affected THC levels in the prepared e-liquid and how much e-liquid was vaporized. We compared THC levels immediately after cannabis extract was mixed with PG/VG versus PEG and across days and performed a three-way mixed ANOVA with dose (CAN150, CAN300) and carrier oil (PG/VG, PEG) as between-subjects factors and Day (0, 3, 14) as the within-subjects factor. THC levels were significantly higher when cannabis extract was mixed with PEG compared with PG/VG (Supplementary Fig. S1), as indicated by a main effect of carrier oil (F1,2 = 26.25, p = 0.0360). Significantly more PG/VG was vaporized compared with PEG, as evidenced by a significantly greater decrease in tank weight following a 30-min vapor session (t = 8.727, p < 0.0001; Supplementary Fig. S2). These results suggest that mice exhibited higher plasma THC levels following cannabis vapor exposure in the PEG group due to higher THC levels within the cannabis e-liquid and not due to greater vaporization of PEG versus PG/VG e-liquids.
Plasma THC
A five-way ANOVA (age, sex, dose, timepoint, and carrier oil) on plasma THC levels revealed a significant five-way interaction (F2,251 = 4.83, p = 0.0088). Given these results, we followed up with separate four-way ANOVAs and multiple linear regressions for each carrier oil (see sections below).
PEG as the carrier oil
Plasma THC
Four-way (age × sex × dose × timepoint) ANOVA on plasma THC when PEG was the carrier oil (Fig. 2A, B) indicated sex × dose × timepoint (F2,125 = 3.37, p = 0.0377), age × sex × timepoint (F2,125 = 7.44, p < 0.0001), and age × dose (F1,125 = 4.97, p = 0.0276) interactions. The three-way interactions were followed up with two-way ANOVAs conducted within each factor and further simple main effects analyses where appropriate.
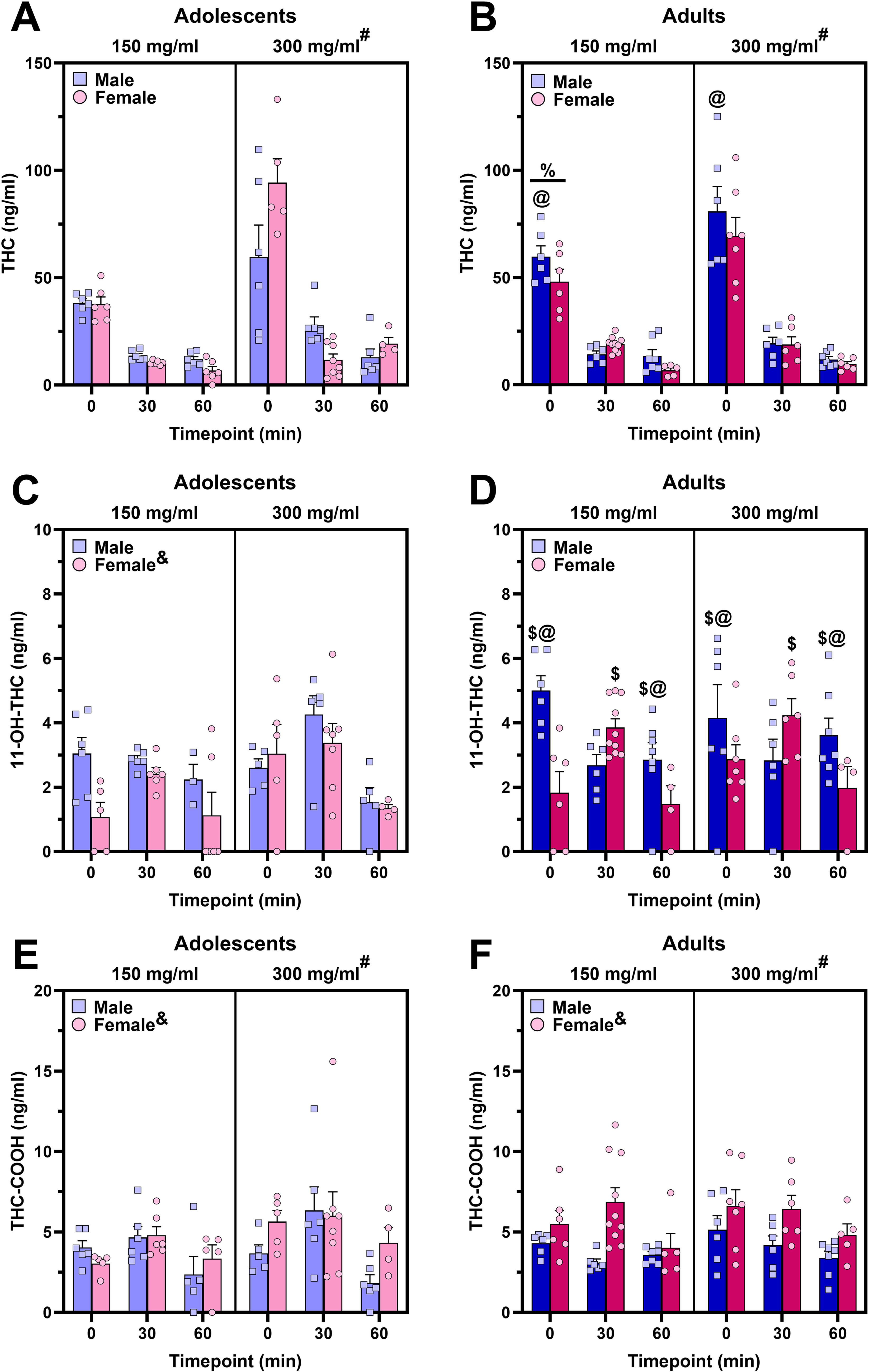
Time course of plasma THC and metabolites following a 30-min cannabis vapor exposure session when the carrier oil was PEG.
Sex × dose × timepoint interaction
Key takeaway: CAN300 produced higher plasma THC levels than CAN150 in males and females, but whether this difference was detected at the 30- or 60-min timepoint differed by sex. To explore how sex affected the relationship between dose and timepoint on plasma THC, two-way (dose × timepoint) ANOVAs were conducted within each sex. There was a significant dose × timepoint interaction in females (F2,69 = 16.17, p < 0.0001) but not males (F2,68 = 3.08, p = 0.0527). CAN300 produced higher plasma THC levels relative to CAN150 in both sexes (females: F1,69 = 51.96, FDR-corrected p ≤ 0.0002; males: F1,68 = 7.95, p = 0.0063), peaking at 0 min (see Supplementary Data S1 for more detail). When examining the interaction of sex × dose within timepoint, at 0 min, plasma THC levels were higher in mice exposed to CAN300 compared with CAN150 (main effect of dose: F1,44 = 19.18, p < 0.0001) with no other significant effects or interactions. At both the 30- and 60-min timepoints, there were significant sex × dose interactions (30 min: F1,50 = 7.90, p = 0.0070; 60 min: F1,43 = 4.45, p = 0.0408). Follow-up simple main effects analyses showed that for males, CAN300 produced higher plasma THC levels than CAN150 at 30 min (F1,50 = 11.75, FDR-corrected p = 0.0021), whereas this dose effect was seen in females at 60 min (F1,43 = 7.08, FDR-corrected p = 0.0144). This led to a sex difference, whereby males reached higher plasma THC levels compared with females at 30 min within the CAN300 group (F1,50 = 10.66, FDR-corrected p = 0.0033) and at 60 min within the CAN150 group (F1,43 = 6.36, FDR-corrected p = 0.0198). Two-way (sex × timepoint) ANOVAs conducted within each dose are reported in Supplementary Data S2.
Age × sex × timepoint interaction
Key takeaway: adult males had higher plasma THC levels than adolescent males at 0 min, while adolescent females had lower THC levels than both adolescent males and adult females at 30 min. To examine how sex affected the relationship between age and timepoint on plasma THC, we conducted two-way (dose × timepoint) ANOVAs within each sex. In males, there was a significant age × timepoint interaction (F2,68 = 4.55, p = 0.0139), whereas there was only a main effect of timepoint in females (F2,69 = 70.98, p < 0.0001). Simple main effects analyses showed that adult males reached higher plasma THC levels at 0 min compared with adolescent males (F1,68 = 11.18, FDR-corrected p = 0.0023) with no age differences at 30 or 60 min. As expected, there were significant main effects of timepoint within each age (adolescent: F2,68 = 17.88, FDR-corrected p ≤ 0.0002; adult: F2,68 = 52.21, FDR-corrected p ≤ 0.0002) (see Supplementary Data S2 for post hoc tests). When examining two-way ANOVAs of age × sex within timepoint, there were no significant main effects or interactions at 0 or 60 min. However, at 30 min, there was a significant age × sex interaction (F1,50 = 9.56, p = 0.0033), with follow-up simple main effects analyses indicating that adolescent females had lower plasma THC levels compared with adolescent males (F1,50 = 12.50, FDR-corrected p = 0.0017) and adult females (F1,50 = 9.22, FDR-corrected p = 0.0061). There were no significant sex differences within adults nor age differences within males. Finally, two-way (sex × timepoint) ANOVAs conducted within each age are reported in Supplementary Data S2.
Age × dose interaction
Simple main effect analyses conducted to further examine the significant age × dose interaction indicated that CAN300 produced higher plasma THC levels than CAN150 in adolescents (F1,125 = 32.19, FDR-corrected p ≤ 0.0002) and adults (F1,125 = 7.35, FDR-corrected p = 0.0106). Moreover, adults reached higher plasma THC levels than adolescent mice when exposed to CAN150 (F1,125 = 5.41, FDR-corrected p = 0.0242).
Plasma THC metabolites
Separate four-way ANOVAs were conducted on levels of plasma THC metabolites—namely, 11-OH-THC and THC-COOH—with age, sex, dose, and timepoint as between-subjects factors.
Plasma 11-OH-THC
Key takeaway: age and sex influenced the timing and magnitude of plasma 11-OH-THC levels, with adult males reaching higher 11-OH-THC levels at 0 and 60 min compared with both adolescent males and adult females. Relative to adult males, 11-OH-THC levels peaked later in adult females (30 min), consistent with slower THC metabolism. Meanwhile, adolescent males had generally higher 11-OH-THC levels compared with age-matched females. Analysis of plasma levels of 11-OH-THC (Fig. 2C, D) revealed a significant age × sex × timepoint interaction (F2,115 = 5.71, p = 0.0043). Subsequent two-way (age × timepoint) ANOVA in females indicated a significant main effect of timepoint (F2,64 = 13.07, p < 0.0001) with no significant effect of age or interaction, with plasma 11-OH-THC peaking at 30 min (Tukey’s: 0 vs. 30 min, p = 0.0045; 30 vs. 60 min, p < 0.0001). Two-way (age × timepoint) ANOVA in males indicated a significant age × timepoint interaction (F2,63 = 6.13, p = 0.0037), with follow-up simple main effects analyses indicating a significant effect of timepoint in adolescent and adult males (F2,63 = 4.07, FDR-corrected p = 0.0242 and F2,63 = 5.90, FDR-corrected p = 0.0071, respectively), with plasma 11-OH-THC significantly declining at 60 min in adolescent males (Tukey’s: 30 vs. 60 min, p = 0.0034), but 30 min in adult males (Tukey’s: 0 vs. 30 min, p = 0.0171). Furthermore, adult males had higher plasma 11-OH-THC levels compared with adolescent males at 0 min (F1,63 = 9.41, FDR-corrected p = 0.0052) and 60 min (F1,63 = 5.74, FDR-corrected p = 0.0227). To examine the effect of sex and timepoint on plasma 11-OH-THC within age, we conducted two-way (sex × timepoint) ANOVAs within adolescent and adult groups. Among adolescents, there were significant main effects of timepoint (F2,58 = 9.33, p = 0.0003), indicating that plasma 11-OH-THC levels drop off at 60 min (Tukey’s: 30 vs. 60 min, p = 0.0002), and sex (F1,58 = 4.12, p = 0.0469), indicating that adolescent males reached higher 11-OH-THC levels compared with adolescent females. There was no significant interaction of sex and timepoint among adolescents. In adults, two-way (sex × timepoint) ANOVA revealed a significant sex × timepoint interaction (F2,69 = 11.19, p < 0.0001), with simple main effects analyses showing that adult males had higher plasma 11-OH-THC levels than adult females at 0 min (F1,69 = 15.59, FDR-corrected p = 0.0004) and 60 min (F1,69 = 6.00, FDR-corrected p = 0.0206), but females had higher plasma 11-OH-THC levels compared with males at 30 min (F1,69 = 5.50, FDR-corrected p = 0.0242). There were significant timepoint effects within adults of both sexes (females: F2,69 = 8.71, FDR-corrected p = 0.0008; males: F2,69 = 5.62, FDR-corrected p = 0.0082). Plasma 11-OH-THC levels peaked at 30 min in females (Tukey’s: 0 vs. 30 min, p = 0.0031; 30 vs. 60 min, p = 0.0004) compared with 0 min in males (Tukey’s: 0 vs. 30 min, p = 0.0171), suggesting that female mice metabolize vaporized cannabis more slowly than male mice in adulthood. Subsequent two-way (age × sex) ANOVAs within each timepoint are reported in Supplementary Data S1.
Plasma THC-COOH
Plasma levels of THC-COOH (Fig. 2E, F) showed dose-dependent increases (main effect of dose: F1,120 = 4.41, p = 0.0377). Female mice had higher THC-COOH levels compared with male mice (main effect of sex: F1,120 = 12.55, p = 0.0006). A main effect of timepoint (F2,120 = 9.56, p = 0.0001) indicated that plasma levels of THC-COOH significantly declined by 60 min, as they were significantly lower compared with 0 (Tukey’s: p = 0.0118) and 30 min (Tukey’s: p < 0.0001), with no significant differences between 0 and 30 min timepoints. There were no other significant main effects or interactions.
PG/VG as the carrier oil
Plasma THC
As a follow-up to the significant five-way ANOVA (above), the four-way (age × sex × dose × timepoint) ANOVA conducted on plasma THC when PG/VG was the carrier oil (Fig. 3A, B) indicated a significant four-way interaction of age × sex × dose × timepoint (F2,126 = 7.53, p = 0.0008), which was followed up with three-way ANOVAs conducted within each factor, with further simple main effects analyses where appropriate. Where four- or three-way interactions were explored, a key takeaway is provided at the beginning of the subsection. Key findings are presented below, and separate follow-up three-way ANOVAs within each of the other factors are reported in Supplementary Data S2.
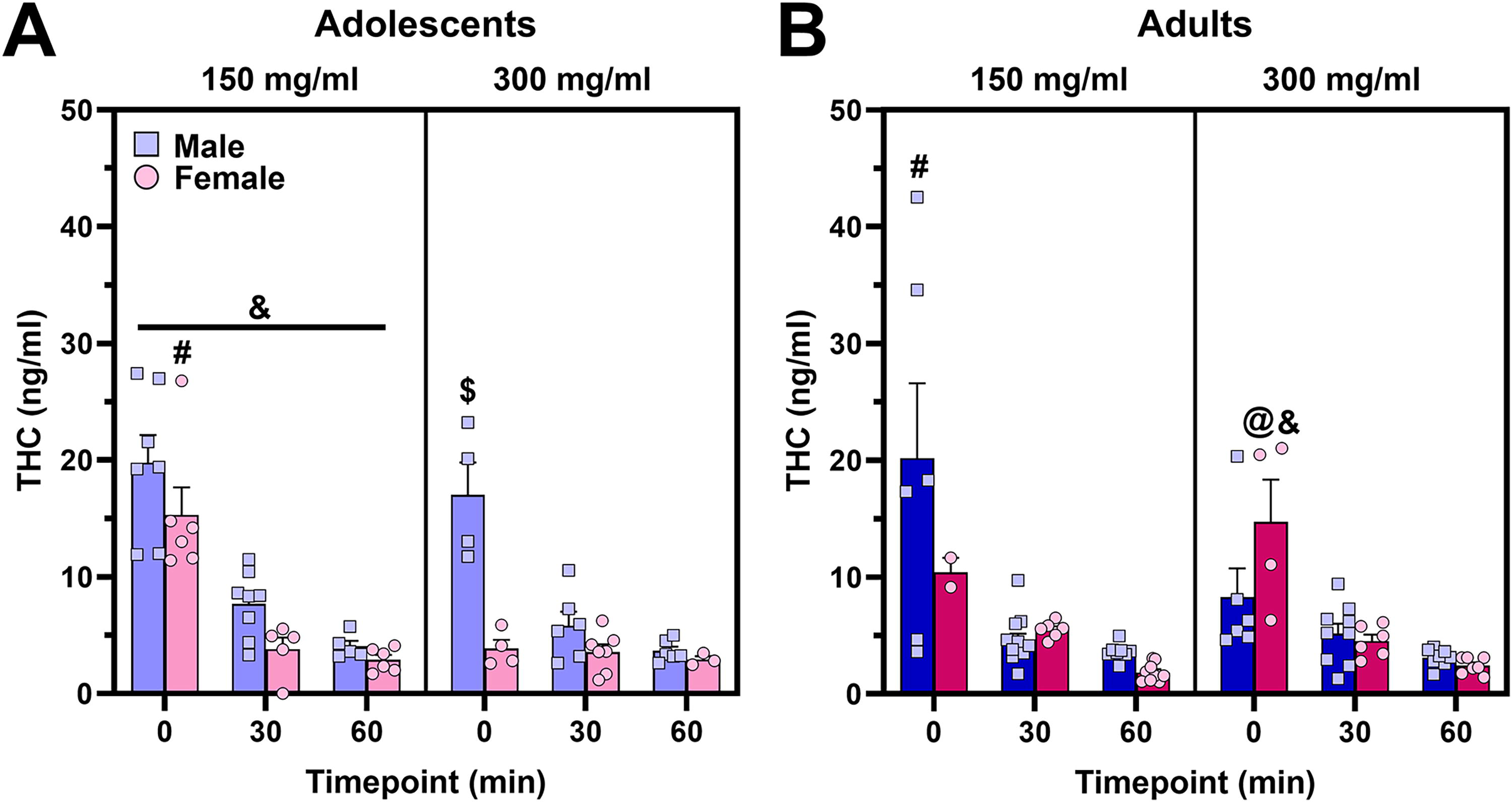
Time course of plasma THC following a 30-min cannabis vapor exposure session when the carrier oil was PG/VG.
Age × sex × vapor × timepoint interaction
Key takeaway: CAN300 did not consistently produce higher plasma THC levels compared with CAN150 at 0 min, and timepoint effects were not consistently detected across groups. Therefore, subsequently reported sex or age effects on plasma THC should be interpreted with caution. Three-way (sex × dose × timepoint) ANOVAs were conducted within each age. In adolescents, there was a significant sex × dose × timepoint interaction (F2,55 = 3.50, p = 0.0372), which was followed up with separate two-way ANOVAs split by each factor and simple main effects analyses to explore how sex affected the relationship between dose and timepoint on plasma THC. In adolescent females, there was a significant dose × timepoint interaction (F2,25 = 11.87, p = 0.0002), and simple main effects analyses revealed that, paradoxically, CAN150 led to higher plasma THC levels at 0 min compared with CAN300 (F1,25 = 36.18, FDR-corrected p ≤ 0.0002), with no effect of dose at 30 or 60 min. Among adolescents, two-way (sex × timepoint) ANOVAs performed within dose indicated that males reached higher plasma THC levels compared with females following CAN150 exposure (main effect of sex: F1,31 = 5.72, p = 0.0230). For CAN300, there was a significant sex × timepoint interaction (F2,24 = 14.32, p < 0.0001), with simple main effects analyses showing that, at the 0 min timepoint only, male mice reached higher plasma THC levels compared with females (F1,24 = 51.19, FDR-corrected p ≤ 0.0002). Among adolescent mice, only the CAN300 female data did not show a main effect of timepoint (see Supplementary Data S2 for details). Both the observed sex difference at 0 min and the lack of timepoint effect are most likely explained by the paradoxically lower plasma THC in the CAN300 versus CAN150 group in females.
In adults, there was a significant sex × dose × timepoint interaction (F2,71 = 4.35, p = 0.0166), which was followed up with separate two-way ANOVAs split by each factor and simple main effects analyses to explore how sex affected the relationship between dose and timepoint on plasma THC. In adult males, there was a significant dose × timepoint interaction (F2,43 = 4.64, p = 0.0150), and simple main effects analyses revealed that CAN150 led to paradoxically higher plasma THC levels at 0 min compared with CAN300 (F1,43 = 12.00, FDR-corrected p = 0.0021), with no effect of dose at 30 or 60 min. Among adults, two-way (sex × timepoint) ANOVAs performed within dose indicated only a main effect of timepoint for the CAN150 dose (F2,37 = 10.13, p = 0.0003) but a significant sex × timepoint interaction for CAN300 (F2,34 = 3.82, p = 0.0319). Follow-up simple main effects analyses indicated that females exposed to CAN300 reached higher plasma THC levels than males at the 0 min timepoint only (F1,34 = 8.36, FDR-corrected p = 0.0094), the opposite direction of the sex difference observed in adolescents. Among adult mice, only the CAN300 male data did not show a main effect of timepoint (see Supplementary Data S2 for details). Subsequent two-way (sex × dose) ANOVAs conducted in adults within each timepoint are reported in Supplementary Data S2.
Plasma THC metabolites
The large proportion of samples with plasma THC metabolite—11-OH-THC and THC-COOH—values below the limit of detection when the carrier oil was PG/VG precluded our analyses of plasma THC metabolites.
Behavior
Carrier oil as a factor
Behavioral measures (body temperature difference score, withdrawal latency difference score, and total locomotion) were assessed with four-way (age, sex, dose, carrier oil) ANOVAs. Across behaviors, we observed significant dose × carrier oil interactions, with follow-up analyses consistent with cannabis e-liquids containing PEG producing greater, more reliable cannabimimetic effects compared with e-liquids containing PG/VG. A full description of analyses can be found in Supplementary Data S1. Below, we report partial correlations assessing the relationship between plasma measures and behavioral outcomes, as well as separate ANOVAs of behavioral endpoints for PEG- and PG/VG-containing e-liquids.
Partial correlations
To assess the relationship between plasma THC and behavioral endpoints, partial correlations were conducted controlling for age, sex, dose, and carrier oil. For partial correlations with plasma THC metabolites (11-OH-THC and THC-COOH), only data when PEG was the carrier oil were used, due to the large proportion of samples below detection levels when PG/VG was the carrier oil. Thus, partial correlations with metabolites were conducted controlling for age, sex, and dose. As shown in Table 1, the results revealed significant negative correlations between plasma THC concentrations as well as metabolite concentrations (11-OH-THC and THC-COOH) and body temperature and locomotion, indicating that higher plasma THC and metabolites were associated with lower body temperature and lower locomotion. In contrast, plasma THC and plasma 11-OH-THC showed significant positive correlations with withdrawal latency difference scores during hot plate, suggesting higher levels of plasma THC and 11-OH-THC are associated with longer withdrawal latencies after exposure (i.e., greater antinociception). While the correlation between plasma THC-COOH and withdrawal latency difference scores during hot plate fell shy of statistical significance (p = 0.06), the relationship was similar to that of plasma THC and 11-OH-THC.
Partial Correlations Between THC and Metabolites and Behavioral Outcomes
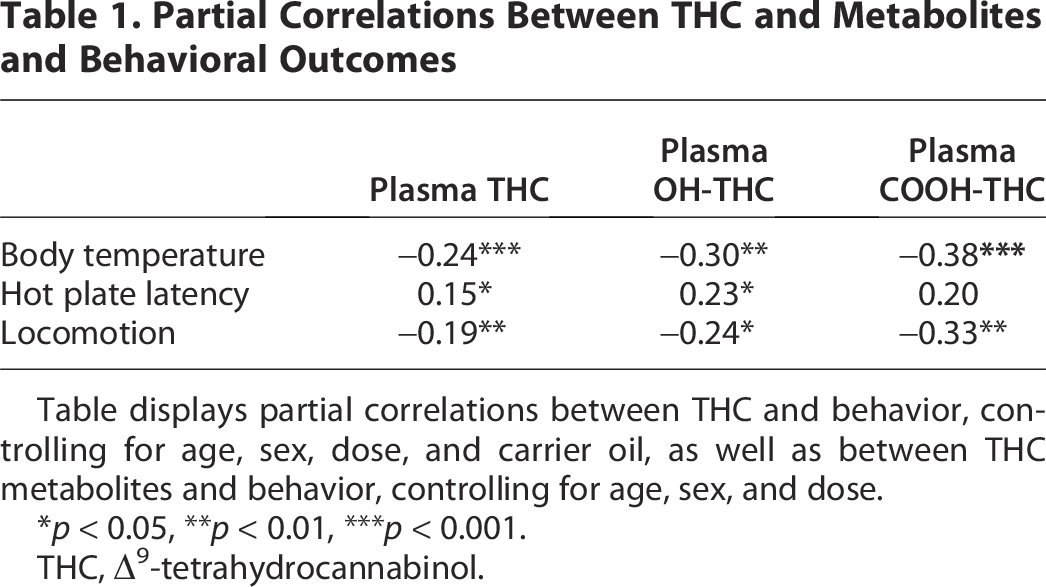
Table displays partial correlations between THC and behavior, controlling for age, sex, dose, and carrier oil, as well as between THC metabolites and behavior, controlling for age, sex, and dose.
*p < 0.05, **p < 0.01, ***p < 0.001.
THC, Δ9-tetrahydrocannabinol.
PEG as the carrier oil
Key takeaway: mice exhibited dose-dependent cannabimimetic effects across all three behavioral measures, but adolescents were less sensitive to the locomotor-suppressing effects of cannabis vapor compared with adults. Group differences in behavioral measures when PEG was the carrier oil were analyzed with three-way (age × sex × dose) ANOVAs. Mice exposed to the higher dose of cannabis vapor (CAN300) had more negative body temperature difference scores (main effect of dose: F2,134 = 28.02, p < 0.0001; Fig. 4A), indicating a hypothermic effect relative to CAN150 and vehicle vapor (Tukey’s: p’s < 0.0001). Exposure to CAN300 vapor also produced a more positive withdrawal latency difference score (main effect of dose: F2,131 = 16.35, p < 0.0001; Fig. 4B), indicating greater antinociceptive effects after vapor exposure compared with CAN150 and vehicle (Tukey’s: p’s < 0.0001). There was a significant age × dose interaction (F2,132 = 4.13, p = 0.0182) on total locomotion (Fig. 4C), with simple main effects analyses indicating that adolescent mice moved significantly more than adults when exposed to CAN150 (F1,132 = 5.49, FDR-corrected p = 0.0236) but not vehicle vapor or CAN300. In both adolescents (F2,132 = 11.84, FDR-corrected p ≤ 0.0002) and adults (F2,132 = 21.47, FDR-corrected p ≤ 0.0002), there was a significant dose effect, reflecting that significant cannabis-induced hypolocomotion was present at both doses for adults (Tukey’s: vehicle vs. CAN150 and vehicle vs. CAN300, p’s < 0.0001), but only at the higher dose for adolescents (Tukey’s: vehicle vs. CAN300, p < 0.0001; CAN150 vs. CAN300, p = 0.0021). Female mice generally moved more than male mice (main effect of sex: F1,132 = 4.27, p = 0.0407).
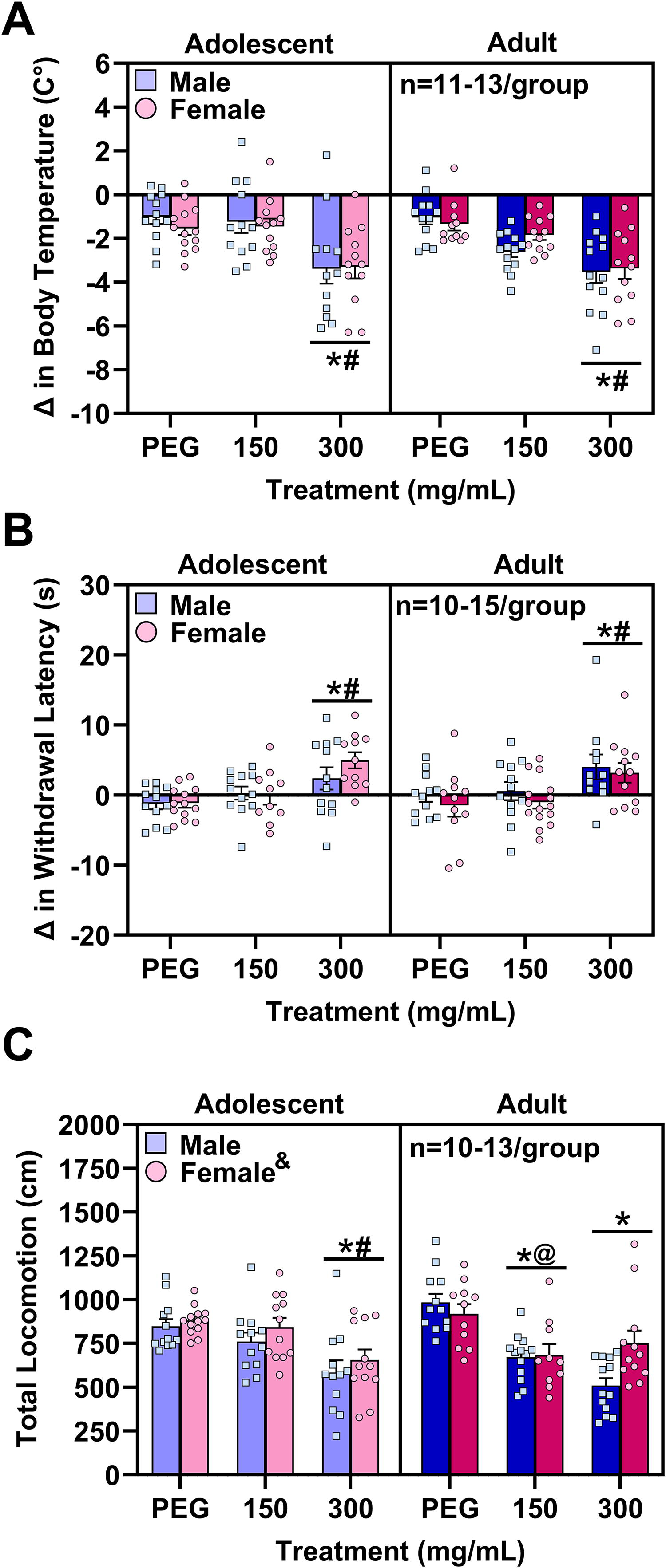
Vaporized cannabis extract when PEG was the carrier oil produced dose-dependent hypothermia, antinociception, and hypolocomotion in adolescent and adult mice of both sexes.
PG/VG as the carrier oil
Key takeaway: CAN300 induced greater hypothermia in males than females and stronger antinociceptive effects in adolescents versus adults, especially in males. Adolescent females moved more than adult females, but cannabis vapor did not reduce locomotion in any group. Group differences in behavioral measures when PG/VG was the carrier oil were analyzed with three-way (age, sex, dose) ANOVAs. There was a significant sex × dose interaction (F2,159 = 3.29, p = 0.0398) for temperature difference scores (Fig. 5A). Simple main effects analyses revealed that male mice exposed to CAN300 had more negative body temperature difference scores (i.e., greater hypothermic response) compared with female mice exposed to the same dose (F1,159 = 5.80, FDR-corrected p = 0.0207). There were no significant sex differences in mice exposed to vehicle vapor or CAN150. In males, there was a significant effect of dose (F2,159 = 8.70, FDR-corrected p = 0.0006), such that male mice exposed to either cannabis dose experienced greater hypothermic responses compared with those exposed to vehicle vapor (Tukey’s: vehicle vs. CAN150, p = 0.0081; vehicle vs. CAN300, p = 0.0008). There was no significant effect of dose in females, suggesting that cannabis vapor at the doses tested did not induce significant hypothermia compared with vehicle vapor exposure when the carrier oil was PG/VG.
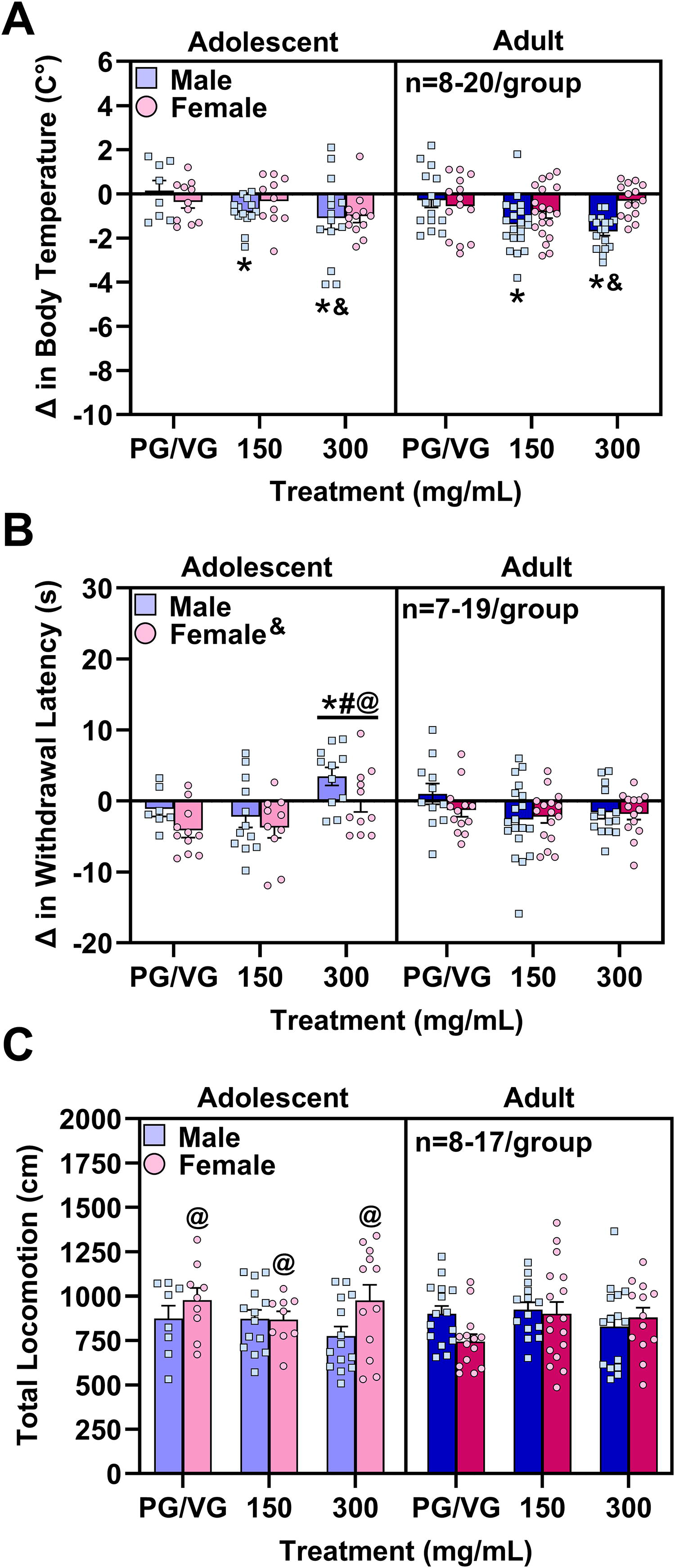
Vaporized cannabis extract when PG/VG was the carrier oil did not consistently produce canonical cannabinoid effects of hypothermia, antinociception, and hypolocomotion in adolescent and adult mice of both sexes.
In the hot plate test (Fig. 5B), there was a significant age × dose interaction (F2,137 = 5.73, p = 0.0041). Simple main effects analyses indicated that CAN300 induced greater antinociceptive effects in adolescent compared with adult mice (F1,137 = 7.88, FDR-corrected p = 0.0082), but there were no age differences in mice exposed to vehicle vapor or CAN150. In adolescents, but not in adults, there was a significant effect of vapor (F2,137 = 8.01, FDR-corrected p = 0.0010), reflecting that CAN300 induced greater antinociceptive effects in adolescent mice relative to vehicle vapor and CAN150 (Tukey’s: p = 0.0054 and p = 0.0035, respectively). It is important to note that only adolescents exposed to CAN300 had positive withdrawal latency difference scores indicative of antinociceptive effects. All other groups had negative values, suggesting a post-exposure sensitization of nociception. Additionally, males had higher withdrawal latency difference scores compared with females (main effect of sex: F1,137 = 5.73, p = 0.0179), reflecting that withdrawal latency shortened in females after exposure, suggesting pain sensitization, while withdrawal latency in males remained similar to the pre-exposure baseline levels.
For locomotor activity (Fig. 5C), there was a significant age × sex interaction (F1,142 = 4.25, p = 0.0410). Simple main effects analyses revealed that adolescent females moved significantly more than adult females (F1,142 = 3.98, FDR-corrected p = 0.0480). This age effect was not present in males, and there were no significant sex effects within each age.
Discussion
The current study sought to use the whole-plant-derived cannabis vapor exposure model in mice to determine whether there are age and sex differences in the acute behavioral effects and pharmacokinetics of vaporized cannabis. We first discovered that PEG was a better carrier oil for the cannabis extract than PG/VG. Vaporized cannabis e-liquids containing PEG induced significant hypothermia, antinociception, and hypolocomotion at the highest dose tested (CAN300) with no significant sex differences. Adults displayed vaporized cannabis-induced hypolocomotion regardless of dose, but only the highest dose suppressed locomotion in adolescents. Immediately after vapor exposure, adult males reached higher plasma THC levels compared with adolescent males, which could contribute to the behavioral difference. The current study found sex differences in plasma THC metabolites that suggest females (and particularly adult females) may metabolize vaporized cannabis more slowly than males.
Carrier oil influences e-liquid properties and resulting plasma THC levels
Both PG/VG, and to a lesser extent PEG, are used as carrier oils in vaping products for humans. 40 We found that more whole-plant-derived cannabis extract went into solution when mixed with PEG compared with PG/VG, and plasma THC and metabolite levels were higher and less variable in mice exposed to vaporized PEG e-liquids compared with PG/VG. When PEG was the carrier oil, our vaporized cannabis protocol produced average plasma THC levels (CAN150: 46 ng/mL; CAN300: 75 ng/mL) comparable with other passive THC vapor studies in rodents (28–73 ng/mL),11,13,20,41 and there was a clear relationship between vapor dose and plasma THC. When PG/VG was the carrier oil, paradoxically higher plasma THC levels were measured in the CAN150 compared with CAN300 groups, and dose-dependent cannabinergic behavioral effects were not observed for most groups. Cannabis e-liquids face inhomogeneity issues, with a particulate phase made up of cannabinoid-rich plant tissue debris typically removed from the extract and vehicle emulsion during e-liquid preparation. 7 Comparison of the Hansen Solubility Parameters for THC 42 versus PEG 400 43 and PG 44 suggests that THC is more soluble in PEG compared with PG. Therefore, we speculate that the chemical properties of PG/VG versus PEG contribute to the differing degree of cannabinoid loss due to separation during e-liquid preparation, resulting in higher bioavailability of THC from vaporized e-liquid containing PEG. We therefore caution interpretation of dose by age or sex interactions on pharmacokinetic and behavioral measures for the PG/VG data and do not discuss them further. Finally, from a translational standpoint, it should be noted that factors beyond solubility including taste and carcinogen production 8 must be considered when assessing the merits of a particular e-liquid carrier.
Sex influences plasma THC and metabolite levels
We found no significant sex differences in initial plasma THC levels when PEG was the carrier oil, consistent with vaped THC studies in adult rats.41,45 However, sex differences emerged at later timepoints, when plasma THC was generally higher in male compared with female mice. This time-dependent sex difference in plasma THC may reflect underlying differences in THC metabolism. In adults, males had higher plasma 11-OH-THC levels immediately after exposure compared with females but lower levels at 30 min, consistent with females metabolizing THC more slowly than males. Adolescent mice replicated this sex effect, but males reached higher 11-OH-THC levels compared with females across timepoints. On the one hand, these sex differences in plasma 11-OH-THC suggest that males may metabolize vaporized cannabis more readily than females. On the other hand, females reached higher plasma THC-COOH levels compared with males across timepoints. The latter finding is in line with females metabolizing THC more readily than males, which is consistent with previous studies in rats that have observed higher plasma THC metabolite levels (both 11-OH-THC and THC-COOH) in females compared with males at adolescent20,46 and adult12,21,46 timepoints following vaped cannabis12,21 or injected THC.20,46 Since vaporized cannabis bypasses hepatic first-pass metabolism, the sex difference in plasma 11-OH-THC immediately after exposure could be attributed to central mechanisms (e.g., sex differences in distribution or metabolism in the brain) or reflect differences in hepatic clearance that begin during the 30 min vapor exposure session as inhaled THC rapidly enters the bloodstream. Future studies assessing THC and metabolites in the brain after vaporized cannabis exposure in mice are needed to examine these possibilities. It is also possible that interactions among phytocannabinoids found in the whole-plant-derived cannabis extract used in our study—as opposed to isolated THC—may impact the pharmacokinetic properties of THC47–50 differently between the sexes.
Lack of sex differences in vaporized cannabis behavioral effects
The current study did not find evidence for significant sex differences in cannabis vapor-induced hypothermia, antinociception, or hypolocomotion when PEG was the carrier oil. The lack of sex differences contrasts with the previous literature using injected THC in rats, which has predominantly reported that female rats are more sensitive to these effects than male rats.36,41,51,52 One potential explanation for these discrepant findings is the route of administration. Intraperitoneal injection versus inhalation have been shown to greatly influence plasma and brain levels as well as the time course of THC and its metabolites in rats. 11 Specifically, inhaled THC produced a more rapid peak in plasma and brain THC levels compared with injected THC. Importantly, injected THC compared with inhaled THC leads to higher and more sustained levels of the active metabolite, 11-OH-THC, in plasma and the brain, 11 which amplifies the magnitude of the sex difference and could impact behavior, as 11-OH-THC is more potent in behavioral assays than THC in mice. 53 In line with this, sex differences in acute behavioral effects of vaporized THC seem to be less consistent than with injected THC. Here, we found no evidence of sex differences in cannabis vapor-induced hypothermia in mice. Meanwhile, rat studies have reported that females are more 10 or less sensitive 41 to the hypothermic effects of vaporized THC, with others finding no sex differences.20,45 Previous studies using vaporized THC in rats have reported no sex differences in antinociception,41,45 consistent with our current findings. Although we did not find evidence for sex-dependent effects of cannabis vapor on locomotion in mice, two recent studies in rats found that vaporized THC suppressed locomotion in adolescent males and females at different doses 20 or had a locomotor-enhancing effect in adult females that was absent in adult males. 13 Differences across these studies may point to variation in species, strain,54,55 and/or the aforementioned drug composition.
Age influences plasma THC and metabolite levels
Here, we found significant age differences in plasma THC and metabolite levels. Specifically, adult mice had higher plasma THC and 11-OH-THC levels compared with adolescent mice, with males showing this age difference at 0 min and females showing this age difference at 30 min post-cannabis vapor exposure. While this age difference is in line with rat data,46,51 recent studies in mice reported that adolescent males 28 and females 29 reached higher plasma THC and metabolite levels compared with adult mice. Again, these discrepant findings may be explained by differences in route of administration and/or drug composition. Even substrain differences (C57BL/6J in the current study vs. C57BL/6NCrl in the other studies28,29) could be involved, as these strains have been shown to differ in response to other drugs, for example, alcohol. 56
Age influences locomotor-suppressing effect of vaporized cannabis but not hypothermia or antinociception
We found significant age differences in the locomotor-suppressing effects of vaporized cannabis, with adult mice showing hypolocomotion in response to both doses (CAN150, 300) relative to vehicle, whereas adolescents only showed hypolocomotion in response to CAN300. This dose-dependent age difference is consistent with THC injection studies in mice28,57 and could be explained by age differences in pharmacokinetics and/or pharmacodynamics. Studies in both male 28 and female mice 29 have shown lower levels of THC in the brain in adolescents compared with adults following THC injection, and in male mice, adults displayed locomotor suppression, but adolescents did not. 28 Thus, an age difference in THC distribution may explain our observed age difference in vaporized cannabis-induced hypolocomotion. Another potential explanation is that adolescents may be less sensitive to the locomotor-suppressing effects of vaporized cannabis due to differences in pharmacodynamics. While we did not assess pharmacodynamics in the current study, based on known changes in cannabinoid receptor 1 (CB1R) expression58,59 and dopamine receptor signaling 60 across adolescence, we speculate that age differences in the downstream effects of CB1R activation in motor-related regions including the striatum 61 could account for the observed locomotor responses in adolescents versus adults.
Although mice displayed significant dose-dependent hypothermic and antinociceptive effects of vaporized cannabis when PEG was the carrier oil, age did not affect these outcomes. Studies in rats reported no age differences in hypothermia with lower THC doses (0.5 and 5 mg/kg) administered intraperitoneally 46 and greater sensitivity of adolescent male rats to hypothermia compared with adult male rats with higher THC doses (10–300 mg/kg i.p.). 52 Studies have found that adult rats are more sensitive to the antinociceptive effects of injected THC across several doses (1–18 mg/kg) 51 with adult female rats displaying greater sensitivity than adolescent female rats particularly at the highest dose tested in another study (176 mg/kg). 52 Together, these findings raise the question of whether age differences in hypothermia and/or antinociception may have emerged if mice were exposed to higher cannabis vapor doses than in the current study.
Study limitations
Our current study has several limitations. First, we collected behavioral data from mice immediately following vapor exposure and performed terminal blood collection in these same mice at either 30 or 60 min post-exposure. To relate plasma THC and metabolites collected at 30 or 60 min back to behavioral outcomes, we compared z-scored plasma values, which may have weakened the relationship to behavioral outcomes. Next, behaviors were always assessed in the same order—body temperature, hot plate, and locomotion. We found a dramatic decrease in plasma THC levels between 0 and 30 min post-exposure, and all behavioral measures were completed ∼20–25 min post-exposure. Thus, it is possible that the effects of vaporized cannabis were more robust on behavioral measures that occurred closer to the end of exposure (i.e., body temperature and hot plate vs. locomotion). In line with this, we observed that hypothermia was the only behavioral outcome that significantly predicted plasma THC. In addition, blood collection at only three timepoints between subjects did not permit detailed pharmacokinetic analysis. Furthermore, we only measured THC and metabolites in the plasma. Plasma THC and metabolite levels have been shown to differ from brain THC and metabolite levels in adolescent and adult mice of both sexes following intraperitoneal injections of THC,28,29 so future work is necessary to determine whether there are age and sex differences in brain levels of THC and metabolites after vaporized cannabis exposure. Due to the number of samples under the limit of detection in the PG/VG sample groups, we were unable to compare the pharmacokinetics of THC metabolites in the PEG versus PG/VG groups. There are several questions unaddressed by our study that remain to be explored. First, the current study focused exclusively on C57Bl/6J mice, but it is known that mouse strain can influence the pharmacokinetics and behavioral effects of cannabinoids, sometimes in sex-dependent ways.54,55 Future studies could investigate how genetic factors including strain contribute to cannabis vapor metabolism and behavioral effects.
Conclusion
Overall, the current study adds to a growing number of studies implementing vaporized cannabinoid delivery approaches by revealing that PEG is a more efficient carrier oil compared with PG/VG for studies involving cannabis e-liquids and demonstrating the typical acute cannabinoid-induced behavioral effects in adolescent and adult mice of both sexes. Future work is needed to fully understand the mechanisms contributing to age- and sex-dependent differences in the pharmacokinetics of vaporized cannabis in mice. Further establishing this model in mice will allow future studies to take advantage of the extensive genetic toolkit available in this species for mechanistic investigations.
Authors’ Contributions
S.R.W.: Writing—original draft (lead), writing—review and editing (equal), conceptualization (supporting), formal analysis (lead), visualization (lead), supervision (supporting). A.L.J.: Writing—original draft (supporting), writing—review and editing (supporting), visualization (supporting), investigation (equal), data curation (equal), supervision (supporting). V.C.-S.: Investigation (equal), data curation (equal). J.B.: Investigation (equal), methodology (equal). G.F.: Investigation (supporting), methodology (equal). T.M.: Investigation (supporting). T.E.: Investigation (supporting). C.C.: Formal analysis (supporting). K.H.: Conceptualization (equal), funding acquisition (equal). R.M.: Writing—review and editing (equal), conceptualization (equal), funding acquisition (lead), project administration (supporting), supervision (supporting). K.M.D.: Writing—review and editing (equal), conceptualization (equal), funding acquisition (equal), project administration (lead), supervision (lead).
Footnotes
Acknowledgments
The authors would like to thank Maury Cole and LJARI, Inc. for their continued support with vapor chamber troubleshooting and optimization. The authors wish to thank Dr. Mark Lange for helpful discussion. The authors would also like to thank Qing Wang for her assistance with colony maintenance and Courtney Klappenbach for her assistance with DeepLabCut and SimBA methodologies. Finally, the authors would like to thank Dr. Anna Berim and the Washington State University Tissue Imaging, Metabolomics, and Proteomics Laboratory for their excellent technical assistance with analyzing plasma samples.
Author Disclosure Statement
The authors have no competing personal or financial interests or any other conflicts of interest to disclose.
Funding Information
This study was supported by an NIH R21 grant from the National Institute on Drug Abuse (R21DA057245-01) awarded to K.M.D., R.J.M., and K.H., as well as funds provided for medical and biological research by the State of Washington Initiative Measure No. 502 (K.M.D., R.J.M., K.H.). S.R.W. was supported by an NIH F32 fellowship from the National Institute on Drug Abuse (F32DA060685) and the State of Washington Initiative Measure No. 502 (S.R.W., K.M.D., R.J.M.).
