Abstract
Deep eutectic solvents are a new class of biodegradable, inexpensive and environmentally friendly solvents. These solvents are created by combining quaternary ammonium salts, which can be converted into liquids at temperatures lower than their melting points. Recently, researchers in various fields have shown increased interest in deep eutectic solvents as alternatives for dissolving oxides in metal extraction processes. Electrochemical studies have demonstrated successful dissolution of some oxide minerals in these solvents, while results have been less consistent for others. This review focuses on the dissolution of sulphide ores in deep eutectic solvents. Arsenopyrite and pyrite exhibit similar thermodynamic properties, as well as comparable stability and solubility in aqueous solutions. Pyrite has been successfully dissolved using choline chloride-based deep eutectic solvents; however, challenges have been noted in dissolving arsenopyrite with these solvents. The review also proposes that successful dissolution of arsenopyrite in deep eutectic solvents can occur at atmospheric pressure and relatively low temperatures through direct leaching.
This is a visual representation of the abstract.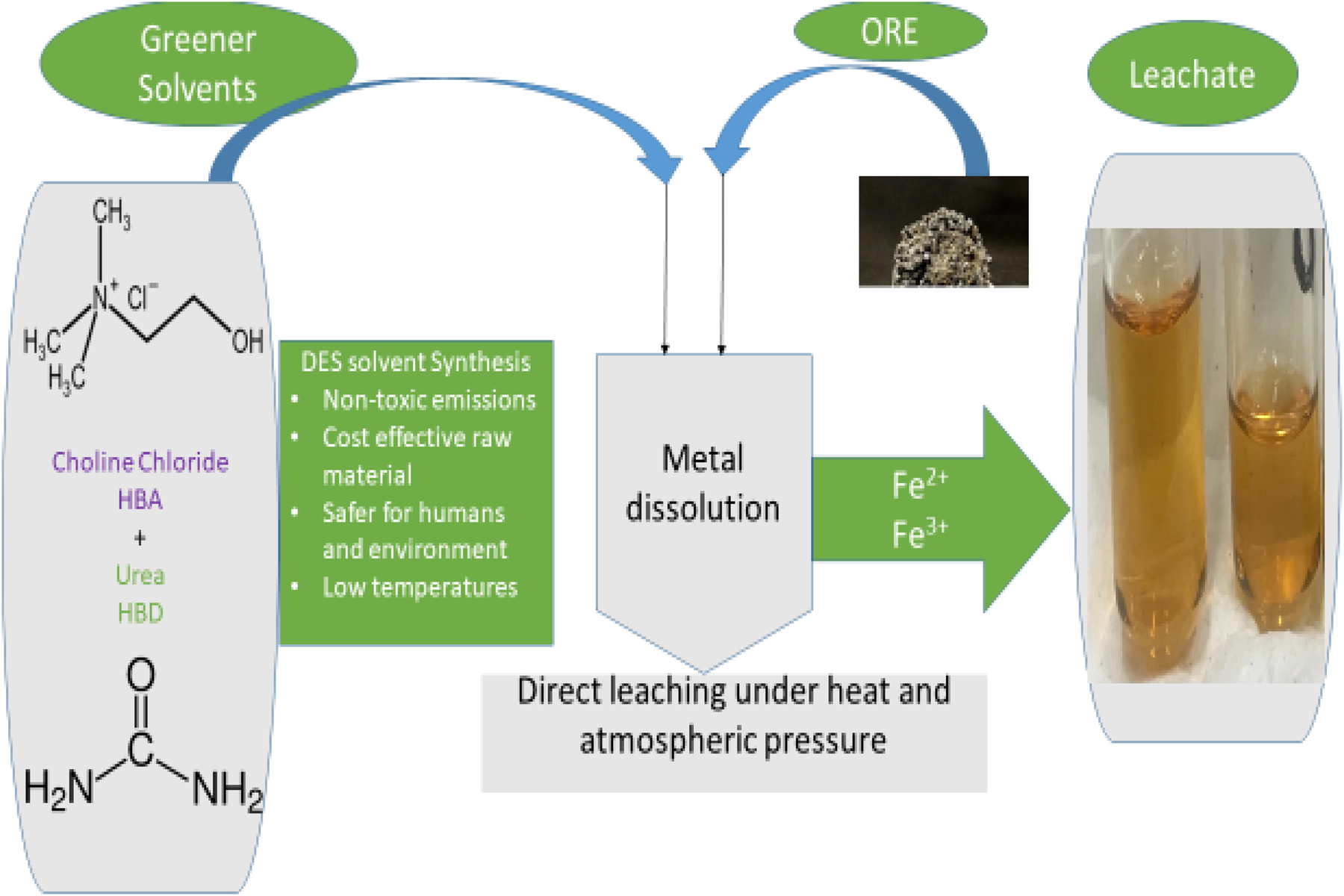
Introduction
Precious metals such as gold, silver, platinum group metals and non-precious metals such as copper, lead, nickel and zinc are indispensable industrial raw materials due to their special properties, such as corrosion resistance, high density and excellent ductility. The demand for these metals continues to rise despite the near depletion of the high-grade ore sources globally for applications in technology, the medical industry, construction and many more. 1 Consequently, secondary-bearing ore sources are gradually becoming the alternative to primary-bearing ore sources, which contain reasonably higher concentrations that are easily processed. Additionally, low-grade ore reserves are abundant; however, they are characterised by complex mineral compositions and the metals in the sulphide ores are finely dispersed and locked up in the minerals with other precious metal associations, leading to the need for traditional methods.2,3 Conventional techniques often give better recovery yields and are effective at concentrating low-grade ores. They do, however, present serious environmental problems, such as high energy prices, acid mine water drainage and SO₂ emissions. Additionally, aqueous solution hydrometallurgy is a more ecologically benign option, but it has slow leaching rates. 4
Techniques currently employed include pressure oxidation, bioleaching, heap leaching, electrochemical dissolution and bench leaching, among many. However, these techniques are associated with the release of toxic gas emissions, acidic water and high energy consumption. Therefore, it is of paramount importance for researchers to explore or bring innovations to greener non-toxic processes which are non-toxic, energy efficient and harmless to both humans and the environment. 5
Common secondary sources for precious and non-precious metals are pyrite, arsenopyrite and chalcopyrite. Mostly these ores are associated with gold, silver, platinum group metals and non-precious metals as finely dispersed particles within the matrix of the ore. Previously, a substantial number of researchers have invested in studying the dissolution of these ores in aqueous, alkaline and acidic solutions in the quest to understand the mechanism of dissolution. It has been reported that pyrite and arsenopyrite have similar thermodynamic properties and values of stability and solubility in aqueous solutions. However, arsenopyrite has been successfully leached through roasting, smelting, pressure oxidation and bioleaching. Moreover, this type of ore releases arsenic as its byproduct at higher temperatures. 1 6–8
Among these techniques, autoclaves for pressure oxidation and bioleaching are relatively safer compared to others, but their disadvantages are higher pressures and energies for the former and relatively lower recoveries for the latter.1,8
Researchers have shown great interest recently, in exploring deep eutectic solvents as alternative solvents in dissolution of ores. These types of solvents emanate from a combination of quaternary salts of hydrogen bond donors and acceptors which are natural, non-toxic and biocompatible sources, including choline chloride, glycerol, lactic acid, sugars, urea and amino acids. Furthermore, it is reported that they can be reusable, resulting in a deep eutectic solvent solution that is biodegradable. Additionally, these solvents have very little volatility, which lowers air pollution, making them safer for the environment and people.9–12
Studies have reported successful results in metal extraction from e-waste and wastewater and ores using deep eutectic solvents through different techniques such as electrodeposition, electropolishing and leaching. However, some techniques have been applied substantially relative to others, triggering the need for their optimisation.
A choline chloride–oxalic acid-based deep eutectic solvent was employed by Reyers et al. and Oke et al. in separate works. They both reported successful results, with the former achieving substantial gold recovery from a refractory gold ore, and the latter reported a successful 89.2% copper and 92.4% cobalt yield, respectively. Furthermore, Aragon-Tobar et al. (2024) carried out a solubility investigation on a variety of metal sulphates, oxides and sulphides, culminating in a successful dissolution from sulphides and recommending the inclusion of iodine. Similar conditions were followed by Karen et al. (2024), with the inclusion of iodine at different pH levels to ethylene glycol and choline chloride to dissolve sulphidic ores in DESs. The study discovered that neutral conditions yielded better results, followed by basic and acidic media. Moreover, S. Karimi and colleagues (2025) examined the electrochemical breakdown of chalcocite in a DES with the development of a sulphur passivation layer. Bidari et al. (2024) effectively investigated the leaching of sulphide flotation concentrate in oxidative and non-oxidative ethaline conditions, finding that chalcocite exhibited high oxidation rates, whereas pyrite oxidation was relatively low. However, the kind of oxidant utilised had a big impact on the rate of dissolution.13–17
Consequently, the novel solvents have shown potential as an effective and sustainable replacement for conventional extraction techniques in the gold mining sector. Several studies indicate that the high solubility, low volatility, good conductivity, as well as the thermal and chemical stability of deep eutectic solvents have made them beneficial in a variety of applications, including hydrometallurgy processes. Furthermore, these solvents have been proven promising in removing valuable metals from mine and industrial waste, despite their primary disadvantages of high viscosity, poor chemical stability, challenges with recycling and reuse.18–20 Although there is still much to learn about the development of deep eutectic solvents in gold extraction, the initial findings are encouraging. However, to completely comprehend the potential and constraints of deep eutectic solvents in gold extraction, more research and developments are necessary as this field of study develops. In addition, determining whether they are viable for extensive industrial application will require investigating their environmental impact and economic viability. Moreover, this review aims to address the possibility of solubilising arsenopyrite in deep eutectic solvents at atmospheric pressure and low temperatures.
Deep eutectic solvent background
Deep eutectic solvents (DESs) started with the innovative works of Abbott and his group and are now widely accepted as a revolutionary class of ionic liquids (ILs). They are the results of a eutectic mixture of Lewis or Brønsted acids and bases able to contain diverse species of anions and cations. DESs are systems including non-symmetric ions with a large size, low lattice energy, and low melting point. They are generally formed by a composite including a quaternary ammonium salt with a hydrogen bond donor (HBD), which can be a metal salt, while the melting point of the system must be lower than that of the individual components, as shown in Figure 1. The decrease in the melting point in DES systems is the result of charge delocalisation happening through hydrogen bonding. 12
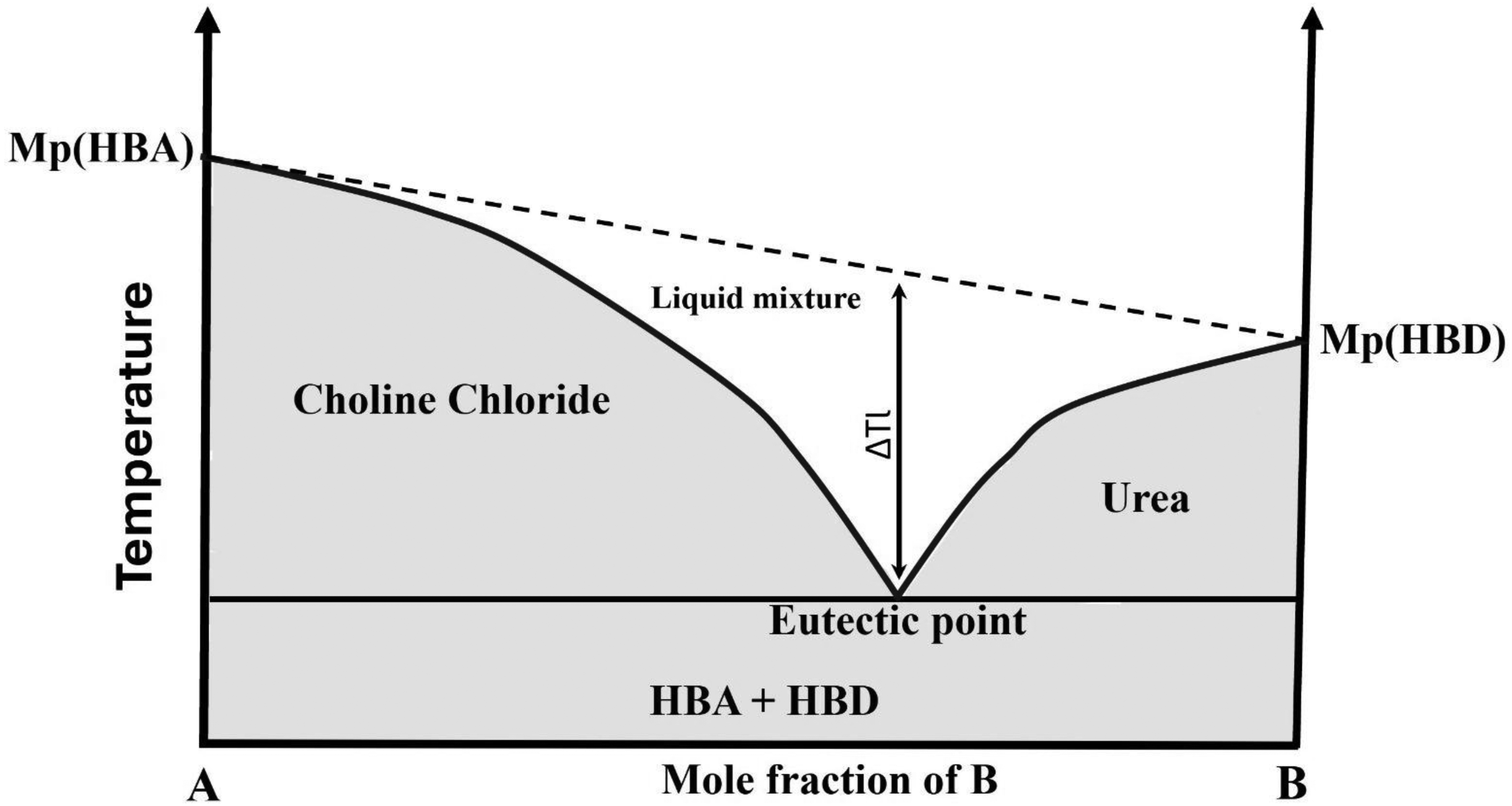
Schematic representation of a eutectic point on a binary mixture for choline chloride–urea DESs (A + B).
DESs exhibit many properties compared to Ionic Liquid (IL) solvents and share several characteristics. However, they were named deep eutectic solvents to distinct them from ILs, because DESs refer to the liquid phase at room temperature of the eutectic composite caused by the mixtures, whereas ILs consist of the liquid phase at room temperature of solvent-free molten salts caused by their low melting temperatures. Several species of anions and cations can be used in DES systems; however, in IL systems, only discrete anions and cations are allowed, confirming that DESs and ILs are two different kinds of solvents.21–23
Synthesis of deep eutectic solvents
The synthesis of DESs is done by mixing two or three cost-effective and safe components that can be self-associated through hydrogen bond interaction, followed by moderate heating with constant stirring. The components frequently used in the preparation of DESs are cheap and non-toxic, e.g., hydrogen bond acceptors (HBAs), such as choline chloride (2-hydroxyethyltrimethylammonium), which is vitamin B4, and diverse hydrogen bond donors (HBDs), such as urea, ethylene glycol and glycerol.22,24
The viscosity and conductivity of DESs are strongly dependent on the mixture and the nature of the HBD. That is why their properties can easily be designed for a specific application. In 2014, Abbott et al. described the general formula for DESs:22,24,25
where Cat+ is any ammonium, phosphonium or sulphonium cation and X is a Lewis base (halide anion). The interaction between X− and either a Lewis or Brønsted acid Y forms the complex anionic species (z is related to the number of Y molecules interacting with the anion).
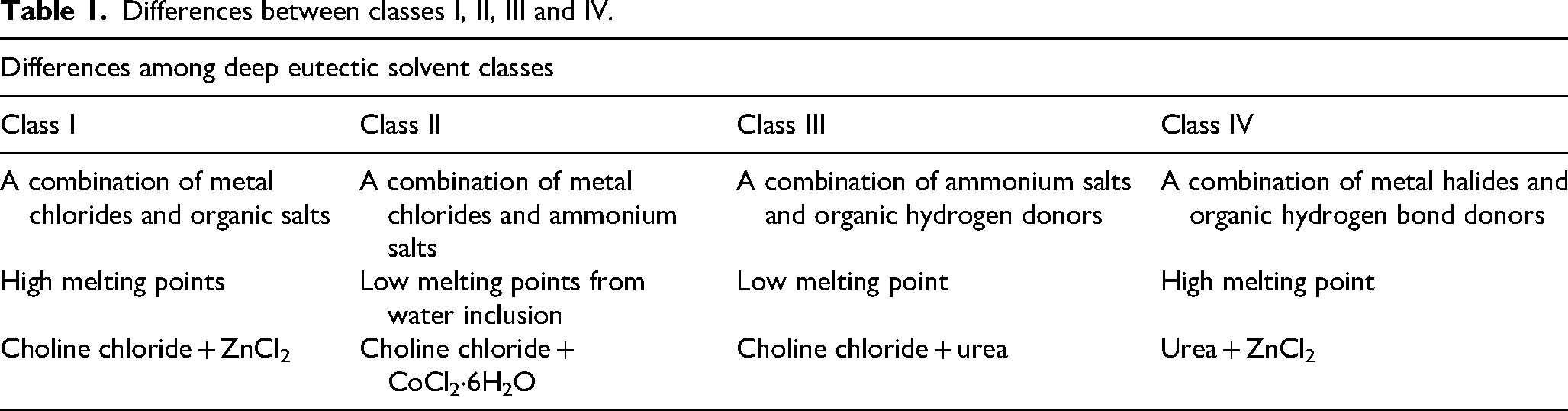
The general formula described by Abbott et al. revealed that DESs are categorised into four classes, which are according to the nature of components used. 25 Additionally, Table 1 shows the distinction of the classes given below.
Differences between classes I, II, III and IV.
v
v
v
The difference between
Most of the IL solvents in the liquid form at room temperature are prepared using organic cations, among which are ammonium, phosphonium and sulphonium moieties. The use of inorganic cations was unsuccessful due to the high charge density of the majority of inorganic cations, causing them not to form low-melting-point eutectics.
25
Moreover, it was unbelievable for them to normally ionise in non-aqueous media. Surprisingly, certain studies have reported that it is feasible to form eutectic melting points of <150 °C from the complex of metal halides with urea. On the other hand, it was shown that ZnCl2 could form eutectics with urea, acetamide, ethylene glycol and 1,6-hexanediol. Relying on these studies, Abbott et al. have confirmed that certain transition metals can be blended into room-temperature eutectics. Thus, they termed this DES system as
Properties of deep eutectic solvents
Generally, it is highly desired that DESs be in a liquid form at ambient temperature so they can be used as solvents for various applications; nevertheless, this is not always the case. The liquid phase of DESs is the result of their freezing point. 22
Freezing point (ΔTf)
The eutectic point of many synthesised DESs is identified by the freezing point (ΔTf). The freezing point (ΔTf) is known as the temperature at which the liquid becomes a solid at ambient atmospheric pressure. It is the most crucial factor for the expansion of the usage of DESs in various applications. However, it can be measured by the use of Differential Scanning Calorimetry (DSC). 27 The freezing point (ΔTf) of DESs is lower than that of the components they are made of. They are strongly linked to the magnitude of the interactions between components. For instance, for a complex made of a binary mixture (A + B); the stronger the interaction between A and B, the better the freezing point. However, those interactions occur differently depending on the DES classes. This section discusses only three of them (classes I, II and III).
In Class I, the interactions are affected by the melting point of the metal halide used. It was observed that, in this system, the eutectic can be produced at room temperature if the metal halide possesses a melting point of about 300 °C or less. In such cases, interactions occur between the quaternary ammonium salts (especially metal halides and halide anions), which will all yield the same halometallate species with the same enthalpies of formation. That is why most of the metal halides able to produce room temperature eutectics in this system have a melting point of approximately or less than 300 °C. In addition, when the quaternary ammonium salts possess a lower melting point, they yield a lower eutectic point as well. For instance, imidazolium halides C2mimCl (mp = 87 °C) and C4mimCl (mp = 65 °C) exhibit a superior phase behaviour and mass transport when they are used with ChCl (301 °C). 23
The metal halide hydrates are used to synthesise
In
Density
Density is a substantial physical property that permits the identification of the nature of elements in the environment. It is one of the most crucial physical property of solvents. However, most of the DESs display higher densities than water, which can be attributed to several factors, among which are the molar ratio used to make the complex, the molecular organisation of the DES system, and the hole theory (the hole theory is discussed in detail below). The DES density is usually measured by the use of a special gravity meter. Moreover, the experimental determination of the density of DESs as a function of temperature is sometimes a difficult goal to achieve. Therefore, some other methods were developed to record the measured results with more accuracy, among which the Rackett equation was used to determine the density of DESs with an error of ±1.9%.
29
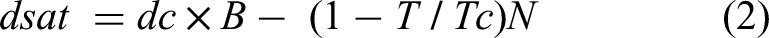
where dsat is the liquid phase for the Rackett equation and Tc and dc are the critical temperature and critical density, respectively.
Viscosity
Viscosity is the most important property of a solvent, which describes its fluidity and its resistance to flow. DESs exhibiting low viscosities are highly desired. However, apart from DESs made of ChCl–ethylene glycol (EG), the viscosities of most of the DESs are relatively high (>100 cP) at ambient temperature, limiting their use in some cases. Generally, these high viscosities are attributed to many factors, among which are: the existence of many hydrogen bond networks between components within DESs reducing the mobility of free species, the electrostatic forces or van der Waals interactions, the chemical nature of the DES complex (the types of ammonium salts and hydrogen bond donors, organic salt/hydrogen bond donor's molar ratio, etc.), the water content and the temperature. The viscosities of many DESs are functions of the temperature; when it increases, the viscosity decreases. The viscosity–temperature relationship can be explicated by using the Arrhenius model (see equation (3)).
28
Nevertheless, DESs with low viscosity can also be designed by the use of small cations or fluorinated HBD, the molar ratio of the complex and by the use of the hole theory.
25
Moreover, the viscosity of the binary eutectic complex is principally affected by the HBD and the electrostatic and van der Waals interactions.
22
The viscosity of DESs can be measured by using a Brookfield R/S rheometer.
27

where
Ionic conductivity
Ionic conductivity is a crucial parameter for the choice of solvents for diverse applications. It indicates the existing chemicals dissolved in the solution, which means, the higher the conductivity is, the more dissolved ionic species are present and moving freely within the solution.
28
In the DES system, the ionic conductivity is proportional to the inverse of the viscosity, which means that when one increases, one decreases, and vice versa. However, most of the DESs display lower ionic conductivities at ambient temperature (2 mS cm−1), since they exhibit higher viscosities at room temperature. Nevertheless, considering the great impact that certain factors discussed above, such as the molar ratio of the complex and the temperature, might have to decrease the viscosities, it can also be concluded that the ionic conductivity can also be influenced at the same time. For instance, it was reported that the increase in the ionic conductivity of DESs increases as the ChCl content increases in a complex.
30
Moreover, the ionic conductivity behaviour as a function of temperature can also be determined by using an Arrhenius-like equation (see equation (4)).
23

Hole theory
The main drawback of a lot of DESs is their viscosity, which is relatively higher at ambient temperature, causing lower ionic conductivities. This situation limits their usage in certain cases. However, among several factors discussed above that can be applied to decrease the viscosity, the hole theory is also recognised as one of the important factors due to the ability to design low viscosity DESs by applying it. Some studies have been done to understand the fluid properties of DESs. Thus, it was assumed that the ionic material includes empty spaces (holes) which emerge due to the temperature during the melting and allow the ions to move effortlessly, passing each other. However, those holes comprise random sizes and locations and undergo constant flux. Moreover, the sizes of holes in the molten salts possess the same dimensions as those of the corresponding ions, making it difficult for the ions to enter the vacant holes, impeding the mobility of the ions. That is why high viscosity is recorded (101–103 Pa).30–32 It is generally easy for ions with small sizes to get access into the holes, resulting in DESs with low viscosities. Nevertheless, the size of the hole can also be influenced by the thermal temperature, which means, a lower temperature system produces smaller holes. In addition, the radius of the average-sized hole is linked to the surface tension of the liquid (see equation (5)).33,34 Moreover, the use of small ions can also help to design DESs with low viscosities.
35

Surface tension
Surface tension is an important physical factor to be addressed in diverse types of industrial and chemical applications, especially when those applications depend on the wettability of their systems.
36
The surface tension can reveal the intermolecular forces between molecules in the complex.
37
However, studies related to the surface tension of DESs are still scarce. Nevertheless, Abbott et al. have shown that the surface tension of DESs follows the direction of viscosity because it depends on the strength of intermolecular interactions, which favours the formation. In addition, the authors confirmed that in certain cases, the surface tension displays a linear correlation with the temperature, which can be verified through equation (6), and can also be measured by the use of an automated tensiometer.23,30
where σ is the surface tension, T is the temperature (K) and a and b are constants.
DESs were previously used as alternative reaction media in chemical synthesis (especially in organic preparation). 38 Thereafter, they were used for electrochemical applications, whereby they were aimed at studying the physico-chemical properties. Then, in electroplating, electrodeposition, environmentally hazardous processes, electrocatalysis, batteries, supercapacitors and hydrometallurgy.20,39–45
Application of deep eutectic solvents in hydrometallurgy
The application of deep eutectic solvents started with the works of Abbott and his group with the solubility of metal oxides and electropolishing of stainless steels in choline chloride-based DESs. 31 Consequently, these solvents gained attention in applications and have since been investigated in various metal oxides under different technologies for the extraction and purification of metals, particularly non-ferrous and rare earth metals.46,47 Their low melting points and great solubility make them effective solvents for the extraction of metals, offering a more sustainable and environmentally benign alternative to traditional methods. 45
Electrochemical dissolution
Electrochemical dissolution is a process in which an ore or material is disassociated in a solvent due to an electrochemical reaction. The process emanates from an applied current into the solvent, resulting in the material breaking down and dissolving. The application can be either electrodeposition, electroplating or electropolishing. Several investigations have been conducted on the solubility of metal oxides in a range of deep eutectic solvents, and a growing number of groups are currently adopting DESs and ILs for solvometallurgy processing. Type III DESs have been shown to dissolve several metal oxides; ligands that can be utilised in these DESs include urea, thiourea and oxalate, which are well-known complexants for a range of metals.48,49
The original matrix determines the extraction technique for metal oxides, which are generally only soluble in an aqueous acid or alkali and insoluble in most molecular solvents, requiring high purity as well as the final product's worth. Precipitation, ion exchange, electrodeposition, and cementation are the four basic techniques used to recover metals from solution. Under the current environmental protection requirements, all solutes must be eliminated before a solvent – typically water – is released into the environment. The most widely used technology for metals like copper, silver and gold is electrowinning or electrodeposition. 50 However, the disadvantage of the deposition process is the requirement of pH maintenance; complexing agents, like cyanide or ammonia, are used to regulate the solubility and properties of the deposit, and hydrogen gas and oxygen evolution may cause the metal deposit to become embrittled, reducing its quality and resulting in low current efficiencies. Although precipitation is an additional option, it typically yields deposits of salts such as hydroxides, sulphides or carbonates rather than the pure metal. Aluminium and chromium must be recovered through chemical or electrochemical reduction, which requires high temperatures and a lot of energy.50,51
The drawback of these liquids is that they are all completely miscible with water and cannot be utilised for biphasic extraction, even though electrochemistry can be employed to separate metals from a complicated combination. In three DESs based on choline chloride, the solubility of 17 metal oxides in the elemental mass series Ti through to Zn has been reported.49,51 Selectivity for extracting specific metals from complicated matrices is achieved by carefully selecting the hydrogen bond donor. 52 Redox potentials in aqueous solutions can be selectively shifted by carefully selecting an appropriate ligand, and in theory, the same could be done in DESs with the same ligands: the relative strengths of potential metal–anion complexes are largely unknown and continue to be an important parameter to quantify.
Abbott et al. have just constructed the first such electrochemical series in two deep eutectic solvents. Using copper/zinc combinations as a model experiment, the scientists have demonstrated how the differential in redox potentials of the metals may be used to electrowin metals into their composite materials. Since copper has a higher redox potential than zinc, copper must be deposited first before zinc can be deposited, the cathode is modified, and the voltage is increased. The authors also show how to dissolve a variety of metals using iodine as a stable, reversible electrocatalyst. I2/I− in a choline chloride : ethylene glycol DES was found to have a redox potential that is more positive than most known metals, and it was demonstrated to be able to oxidise and recover gold. 51
The extraction of lead and zinc from electric arc furnace (EAF) dust has been done on a wide scale using a hybrid DES consisting of choline chloride, ethylene glycol and urea. Iron and aluminium oxides were insoluble in EAF dust, while only zinc and lead oxides were selectively absorbed by the DES. According to the electrochemical series in DESs, Pb would electrowin first over Zn; Pb was cemented using Zn dust because it was not economically worthwhile to extract it. According to tests, Zn could then be electrodeposited with a 75% current efficiency. The most cost-effective method, therefore, was to precipitate ZnCl using ammonia, filter off the precipitate, boil away the ammonia, and leave the DES behind to be used again. However, this is not a very efficient use of such a cheap metal. To evaluate the effectiveness of employing DESs for large-scale metal extraction, a pilot plant was built. It has been demonstrated that choline chloride : urea and choline chloride : malonic acid are efficient in eliminating copper oxides and fluorides from post-etch residues. Additionally, malonic acid-based liquids are effective in eliminating residues from the CF4/O2 etching of a copper-coated DUV photoresist. The removal of organic sulphides from fuels has also demonstrated potential for DESs.51,53,54
The primary drawbacks of aqueous-based systems are their electrochemical stability, which is caused by constrained potential windows that cause gas evolution and hydrogen embrittlement, as well as passivation of deposits, electrodes, and substrates. DESs offer an advantage over aqueous and organic-based electrolytes because of their high solubility for metal salts, which strangely also includes metal oxides and hydroxides. Since non-soluble oxides and/or hydroxides can accumulate on the electrode surface, obstructing the target metal's ability to deposit and creating issues when thick metal films need to be deposited, passivation is frequently an issue in aqueous solutions. The high solubility of metal oxides and hydroxides in DESs prevents the passivation effect, allowing for easier deposition of thicker metal layers. 54
Electrodeposition application
DES has also shown great promise in electrodeposition procedures, where they can be used as electrolytes to deposit metals and alloys onto a range of substrates. They are suitable for electrodeposition applications due to their excellent conductivity and ability to dissolve a wide range of metal salts, which offer enhanced control over the deposition process and the quality of the materials deposited.45–47
Sulphide ores
Arsenopyrite
Arsenopyrite has the chemical formula FeAsS, and it is rich in iron, sulphur and arsenic and bears gold. In solid solutions including other minerals such as sulphides and other base metal complexes, arsenic and antimony, gold can be found as submicroscopic inclusions or small particles. Arsenopyrite is monoclinic, resembling marcasite, according to Bueger et al. Each iron atom is encircled by a warped octahedron, with a triangle of three sulphur and three arsenic atoms on one side. The structure of arsenopyrite is intricate, made even more so by the mineral's near-constant twin distribution. Arsenopyrite frequently encapsulates gold.55,56 Figure 2 shows a typical structure of arsenopyrite.
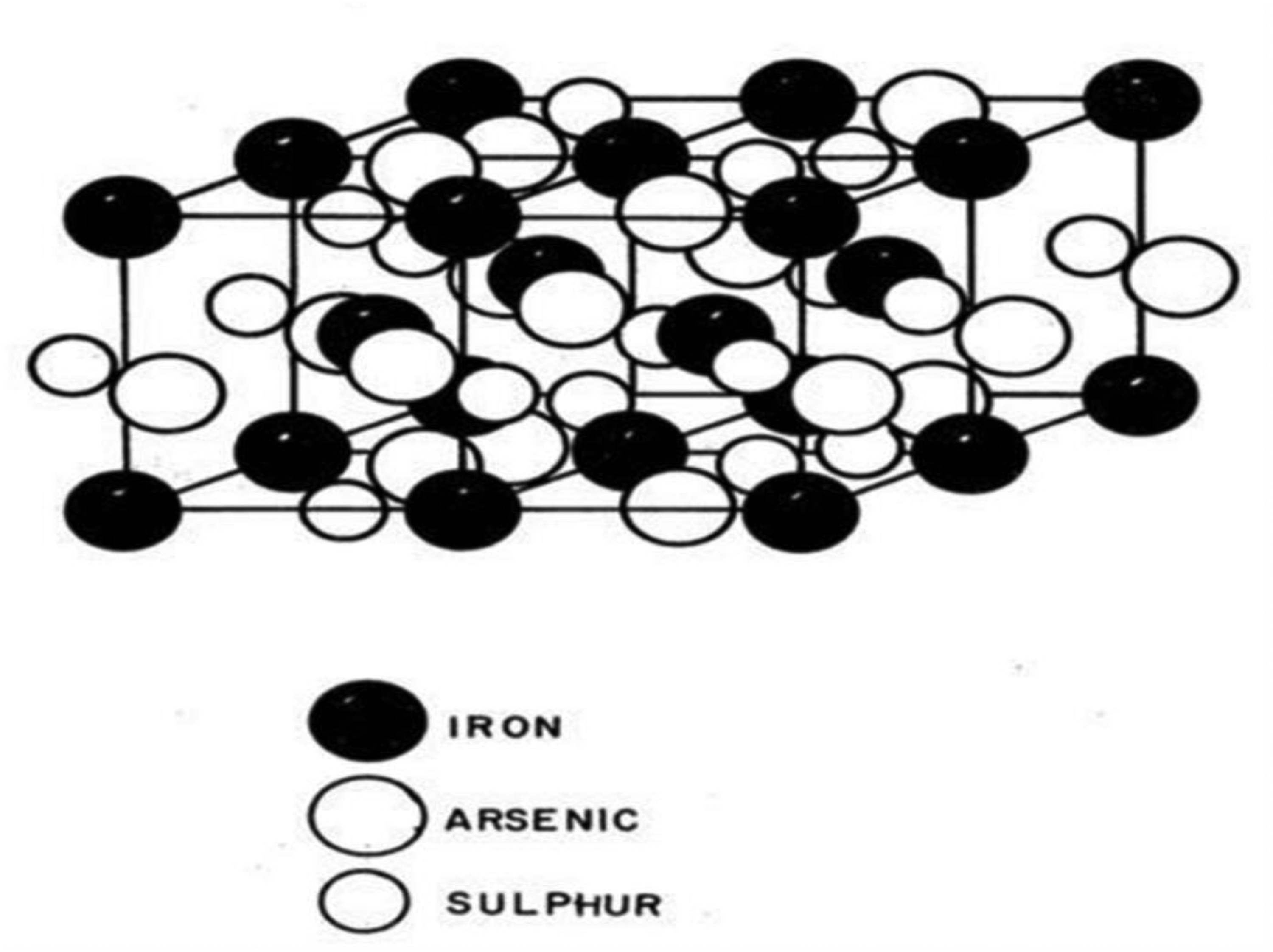
Crystal structure of arsenopyrite. 57
According to Zadra et al., this kind of ore can be leached using cyanide to extract gold; however, recoveries are extremely low, below 70%, and pre-oxidation treatment is necessary to increase recoveries. According to Zadra et al., US Dept. of the Interior Bureau of Mines, R.I. 4843, arsenopyrite is a semiconducting mineral.
Pyrite
Gold is linked to pyrite in three different ways, as opposed to arsenopyrite:
finely distributed metallic gold particles, ranging from micro- to nano-sized, scattered throughout the heterogeneous zones of the pyrite crystal; auriferous arsenopyrite micro-inclusions, primarily concentrated along fractures and inside arsenic-enriched zones; and chemically blended gold with pyrite's crystal structure.
The presence of submicroscopic metallic gold particles (about 200 Å in diameter) in pyrite grains of an unoxidised ore of type ‘Carlin’ was discovered by Bakken et al. (1989) through high-resolution transmission electron microscopy. Additionally, analyses conducted using an electronic microprobe on arsenic-rich gold-bearing zones revealed that the correlation line As [at.-%]–Au [wt-%] coincides well with that of gold-bearing arsenopyrites with invisible gold, which could indicate the presence of only these submicroscopic auriferous arsenopyrite particles within the pyrite grains. Nevertheless, the spectra obtained by Môssbauer spectroscopy of 197 Au revealed a distinct nature of ‘invisible’ gold in pyrite compared to metallic gold or ‘invisible’ gold in arsenopyrite. Therefore, these Môssbauer spectra support the occurrence of coupled gold with pyrite chemically, while the precise nature of the bond type is yet unknown.55,56,58
The presence of arsenic in the sulphide crystal lattice, with a complex and heterogeneous distribution within crystals, is one of the primary properties of pyrite linked to gold mineralisation. The presence of various zonations, most commonly in the form of overgrowth bands created by complex arsenic enrichments, or else in the form of grains with a second phase, sometimes with a collomorphic texture, surrounding a ‘core’ of pyrite that is low in arsenic, reveals this distribution. Arsenic and gold concentrations in pyrite are often positively correlated, regardless of the type of zonation. Figure 3 displays a pyrite's crystal structure.6,56,58
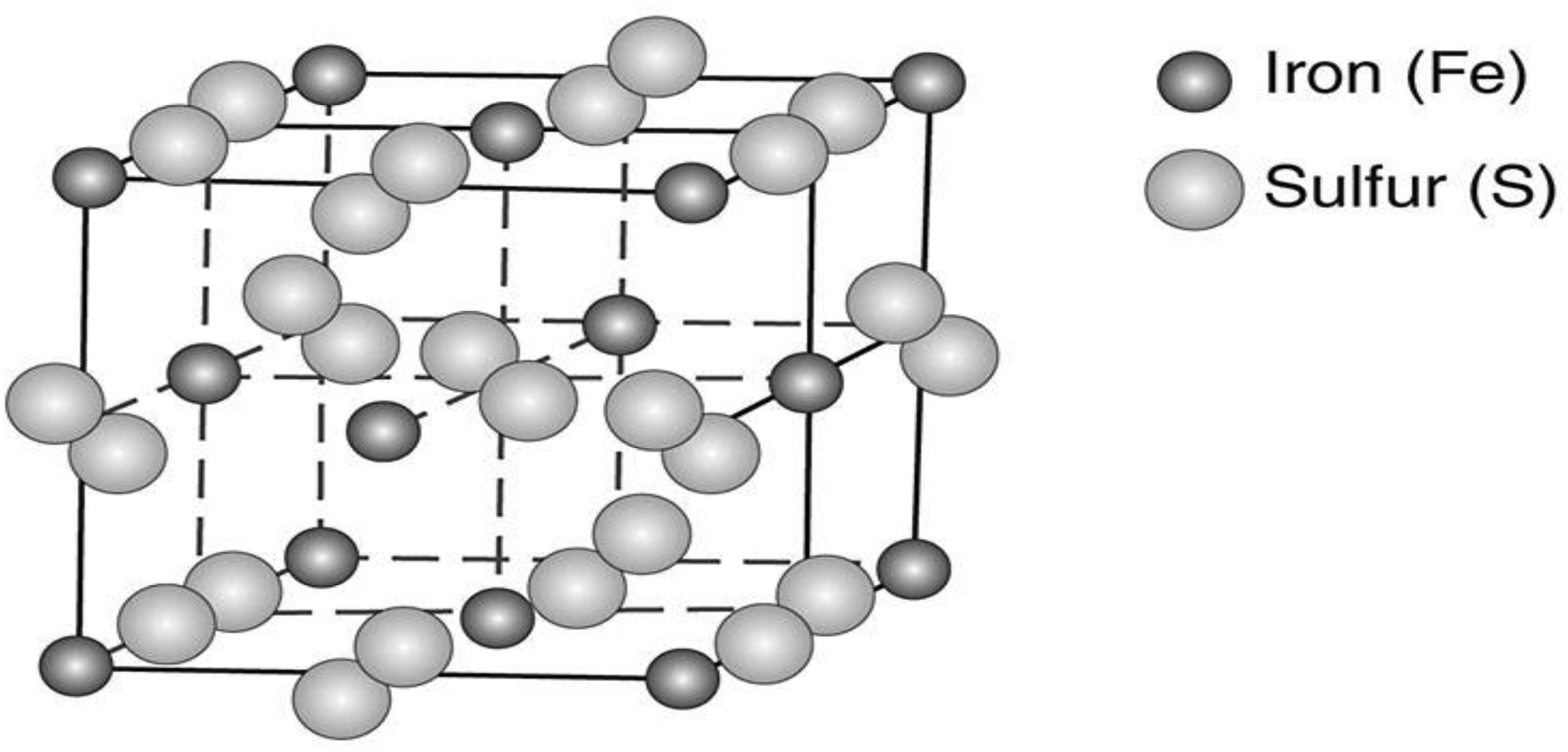
Crystal structure of pyrite. 59
Dissolution
The solid-state reaction kinetics are described by using the shrinking core model. According to this concept, the reaction proceeds inward from the surface of the solid reactant in a sequence of steps as shown in Figure 4. The solid reactant gets smaller as the reaction goes on because reactants are consumed and products are formed. The mass transfer rate of reactants through the product layer determines the reaction's overall rate, which is governed by the rate-limiting step. Solid-state reaction behaviour is frequently analysed and predicted using this model in a variety of industrial processes:
A solvent diffuses from the main fluid to the solid surface via the diffusion layer encircling the particle; Solvent adsorption onto the mineral's (solid) surface and desorption of the product from the mineral surface; A chemical or electrochemical reaction between the reagent and the mineral grains involving dissolution reactions; and Transmission of the product through the diffusion layer or product layer formed as a result of the reaction with the reagent.
60
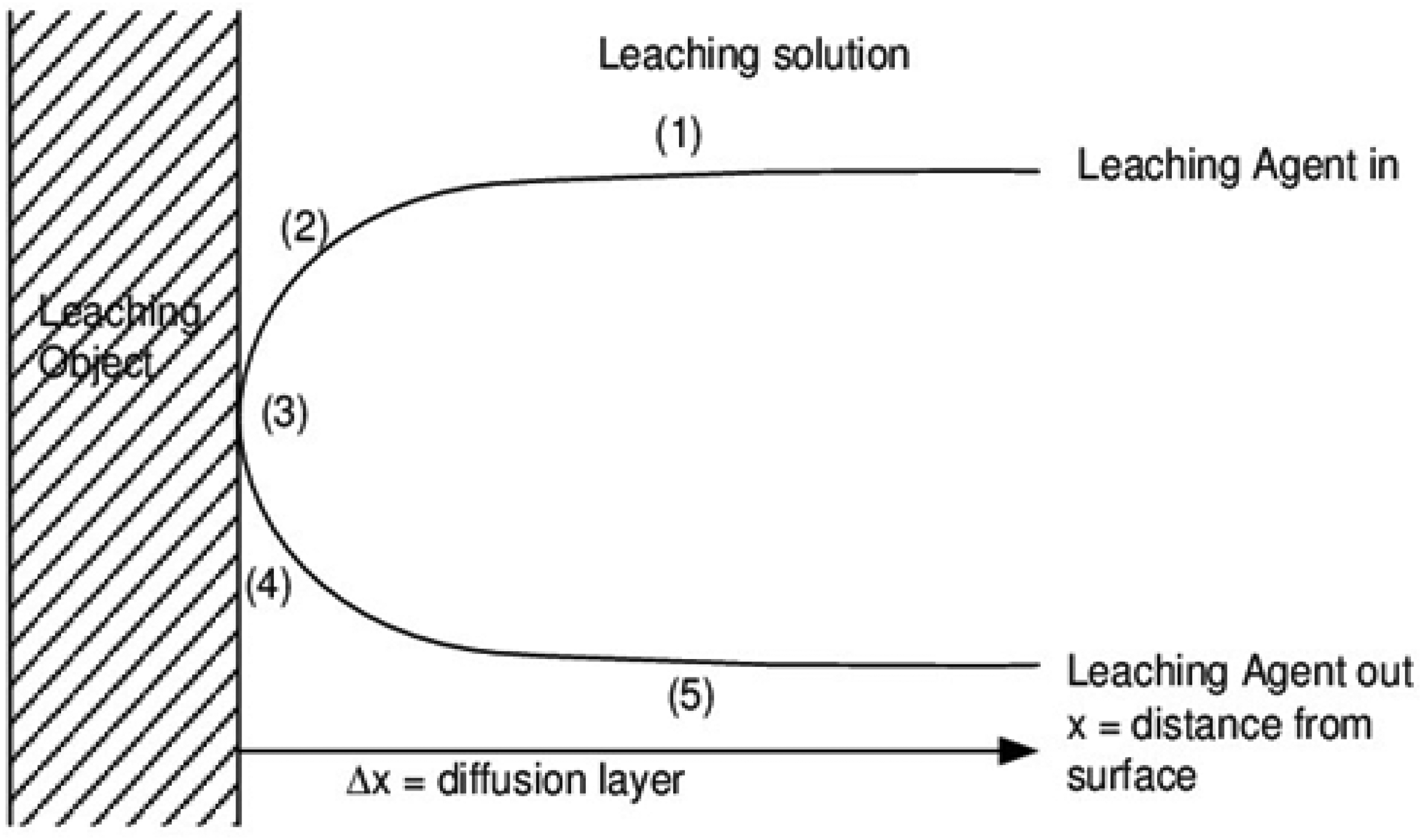
Illustration of the steps occurring in the dissolution of metals from the mineral bearing ore.
The rate of a dissolution reaction can be enhanced through several physicochemical factors such as particle size, agitating speed, solid/liquid ratio, temperature, pH and the leaching solvent. Additionally, the surface area of the particles plays a crucial role in maximising the exposure of ore in the solvent. Reducing the particle size in a well-mixed system could cause the reaction rate to increase. 60 Moreover, the pulp density or the solid/liquid ratio may have an impact on the solution's thickness, which in turn may have an impact on the kinetics of the reaction or gold leaching.
It has been discovered that excessively high or low pulp densities might have an unfavourable effect on the efficacy of the gold leaching process. It is clear that overly thick pulps (solids) impede mass transfer and that diluted pulps cause high reagent addition rates and a loss of ore leaching residence time. The application of the shrinking core model for the solid–liquid systems in hydrometallurgy has been thoroughly studied.61,62 Therefore, prior studies have acknowledged that in leaching trials using concentrated gold ores and gold-bearing ores, the kinetics of the gold leaching match the decreasing core model. Under the assumption that the ore is composed of uniform, spherical solid particles that react isothermally with the solvent, the dissolving rates of a sulphidic gold ore can thus be investigated using the shrinking core model. 63
The fundamental tenets of the shrinking core model are that the rate of reaction can be regulated by a chemical reaction occurring at the solid/solution interface; the rate can be regulated by diffusion of a dissolved reactant or product through the product layer of an inert mineral or the solid product formed during the reaction and the rate can be regulated by some chemical reactions occurring at the solid/solution interface.64,65
Solubility
Solubility is the most crucial parameter in the extraction of precious minerals and metals from ores. The chemical makeup of the ore, the solution's temperature and pressure and the solvent's concentration are some of the variables that affect an ore's solubility in a solvent. Certain ores can dissolve easily in a specific solvent; however, others need specific solvents or circumstances to dissolve. 66
An ore's solubility plays a critical role in deciding how profitable and efficient the extraction process is. Greater extraction yields and faster dissolving are typically associated with higher solubility, whereas low solubility may call for extra processing steps or more aggressive solvents to obtain a good extraction process. For the purpose of creating efficient extraction procedures and maximising the recovery of important metals and minerals from natural deposits, it is imperative to comprehend the solubility of ores.66,67
Moreover, it affects environmental and regulatory issues since some the solubility of metals and minerals may affect how likely it is that they are to leach into the nearby soil and water. Furthermore, very little research has attempted to simulate the solubility of metal oxides. A modified solution theory was employed by Shin et al.
17
to forecast the solubility of a variety of inorganic compounds in supercritical water. It was demonstrated that the solubility of a solid solute may be expressed as follows: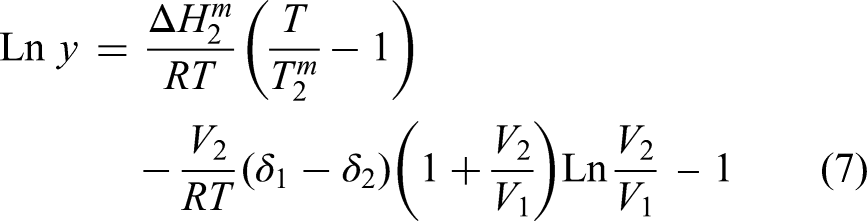
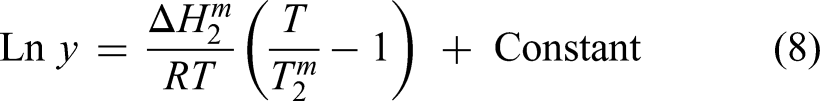
The dissolution mechanism of pyrite
The dissolution of pyrite in deep eutectic solvents is a multifaceted process involving both physical interactions between the solvent and the mineral surface and chemical reactions. To completely comprehend the mechanisms at work, more research is required. The process of pyrite dissolving in nitric acid is made up of both cathodic and anodic processes. 58 On the surface of the pyrite particle, anodic pyrite dissolution and cathodic oxidant reduction occur simultaneously. The potential difference across the interface between pyrite and the solution is what propels these reactions. The assumed general equation in a nitric acid dissolution process is shown in the equation below:58,68
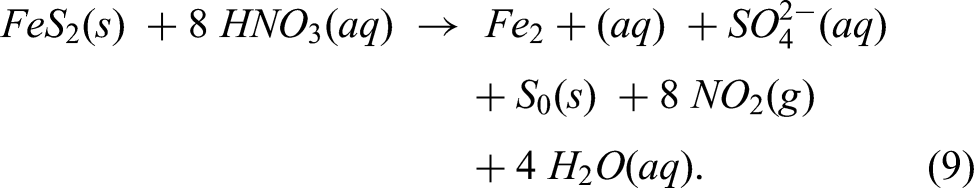
Oxidation of pyrite is expressed as:
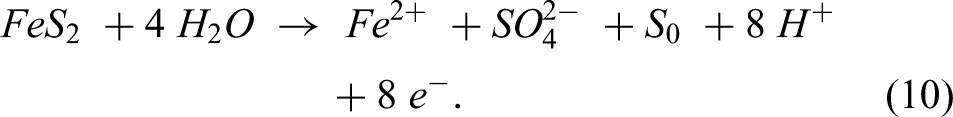
Reduction in the oxidant is expressed as:
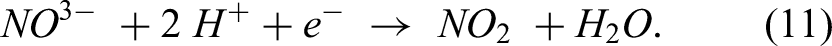
Figure 5(a) and (b) depicts the proposed mechanism and thermodynamic dissolution of pyrite in an acid solution. It shows the ions at the pyrite mineral's surface, the species Fe2+ (aq.), SO (s), and SO42− (aq.) being released into the bulk solution, and the oxidant (NO3−) absorbing the electrons released by S to form NOx(g) or NO2(g). Pyrite undergoes a redox process during the thermodynamic dissolution of pyrite in nitric acid, losing electrons and generating ferrous Fe2+ and ferric Fe3+ at a lower pH level. Sulphur is released as elemental sulphur, which eventually oxidises into sulphate (SO42−) at a higher potential. 68
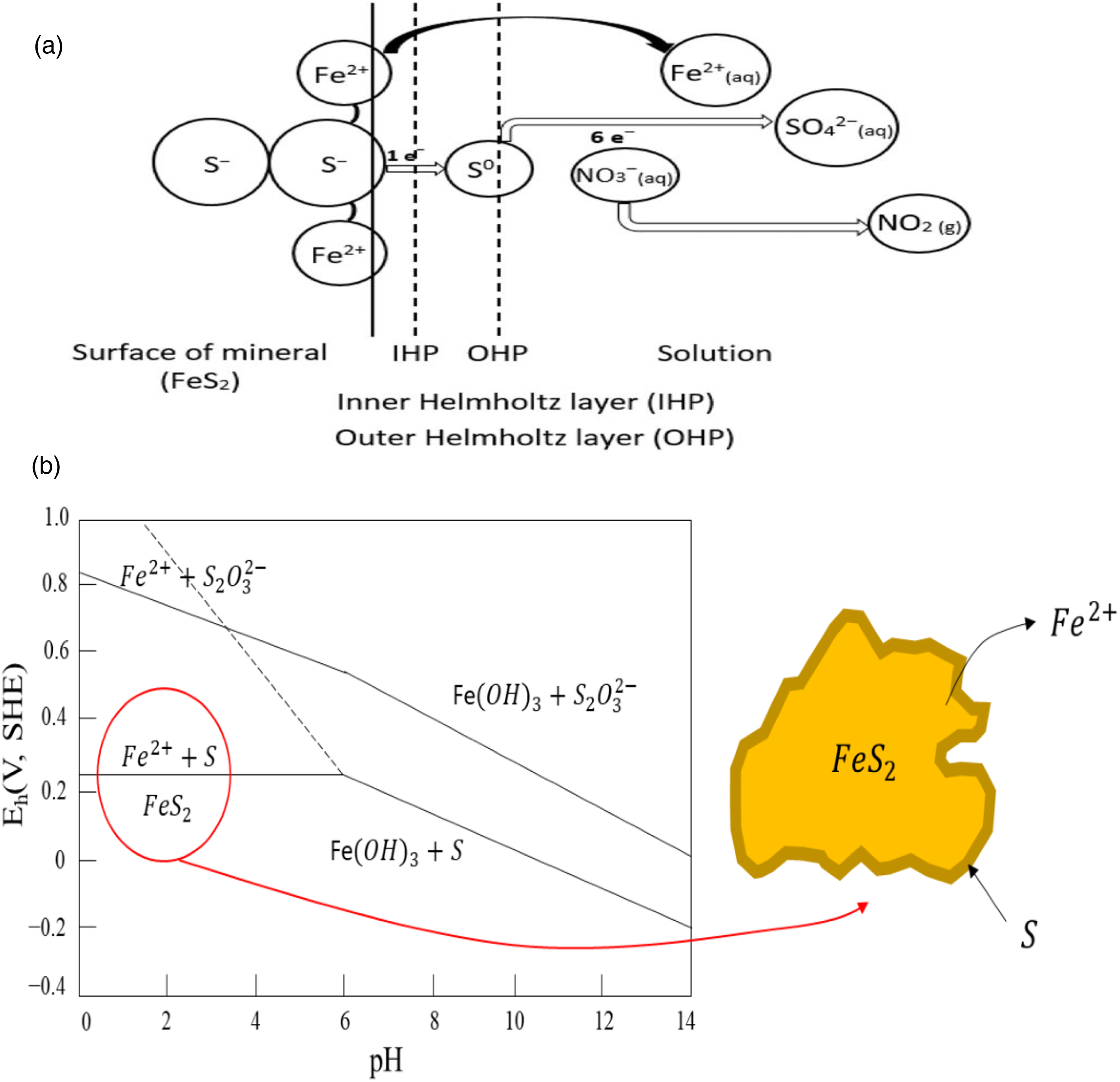
(a) Proposed mechanism for pyrite dissolution in a nitric acid solution. 68 (b) Eh versus pH diagram of the dissolution of pyrite.
The oxidation number of iron (Fe2+) remains unchanged in pyrite upon breaking of the connection between Fe2+ and S22−. Iron is the electropositive element in FeS2, which explains why it transfers to the solution in its original form (without releasing an electron). Consequently, the pyrite lattice's iron oxidation number was +2, and the iron species that were liberated into the solution (Fe2+(aq.)) were also +2. In an oxidative medium, it can, nevertheless, further oxidise to (Fe3+(aq)). However, the electronegative element sulphur experiences recombining processes after losing one or more electrons. 55
Moreover, S. Temouri studied the kinetics of dissolving pyrite in nitric acid at atmospheric pressure. Changes in significant leaching parameters were used to study the oxidative dissolution of pyrite in a nitric acid solution. Pyrite dissolved between 72% and 95% in 3 M HNO3 after 120 min of leaching at temperatures above 45 °C and up to 85 °C, according to the study. The strong oxidising capacity of HNO3 produced a sufficient pyrite dissolution between 74% and 95% at nitric acid concentrations of 2.5 M to 4 M. Increased iron extraction from 75% to roughly 92% resulted from pyrite fraction size reduction from +150–212 µm to +53–75 µm. Fe extraction of 80% to 98% was obtained by increasing the stirring speed from 1000 rpm to 4000 rpm. Moreover, in deep eutectic solvents, certain DESs, like oxalic acid and choline chloride, have acidic characteristics that can cause the production of hydronium ions (H3O+), which react with the pyrite surface to release sulphur and iron ions into the solvent. The chemical reaction of the dissolution of pyrite (FeS2) in deep eutectic solvents (DESs) can be expressed as follows:49,51,68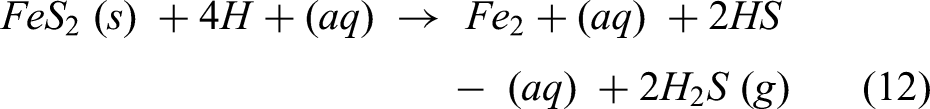
Hydrosulphide ions (HS−), hydrogen sulphide gas (H2S) and ferrous ions (Fe2+) are the products of this interaction between the pyrite (FeS2) solid and hydrogen ions (H+) in the deep eutectic solvent. Teimouri et al. (2023) investigated the dissolution of pyrite in a deep eutectic solvent from choline chloride and glycerin glycol at various pH and solid/liquid ratios to determine Fe extraction as a sign of any pyrite dissolution. Both oxidative and reductive principles are seen in the electrochemical behaviour of sulphide ore dissolving in DESs. The structure decomposes into sulphate and ferric ions. Furthermore, iron in pyrite undergoes oxidation to Fe3+, while sulphur undergoes oxidation to elemental sulphur (S⁰) or, at higher potentials, to sulphate (SO₄2−). Additionally, using density functional theory, Teimouri et al. looked into whether either of the two ligands supplied by DESs (Cl– and/or [C2H4O2]2−) could combine with Fe2+ or Fe3+ to produce the most stable combination. Consequently, the most likely and stable combination was determined to be the tetrahedral complex [Fe(C2H4O2)2]− with the ligand [C2H4O2]2− by O-donor chelating with Fe3+. 68
Moreover, the Abbott group conducted research on pyrite in an ethaline DES; however, their main focus was on the feasibility of dissolving it using the electrolysis method with iodine acting as the oxidant. The other study investigated a unique electrochemical technique for determining the dissolving mechanism of a pyrite–ethaline paste on the Pt electrode. Figure 6 illustrates the dissolution rate from choline chloride and ethylene glycol, which shows the feasibility of deep eutectic solvent application in electrodeposition of metals, metallurgical extraction of ores under atmospheric conditions, and electrochemical techniques.49,51
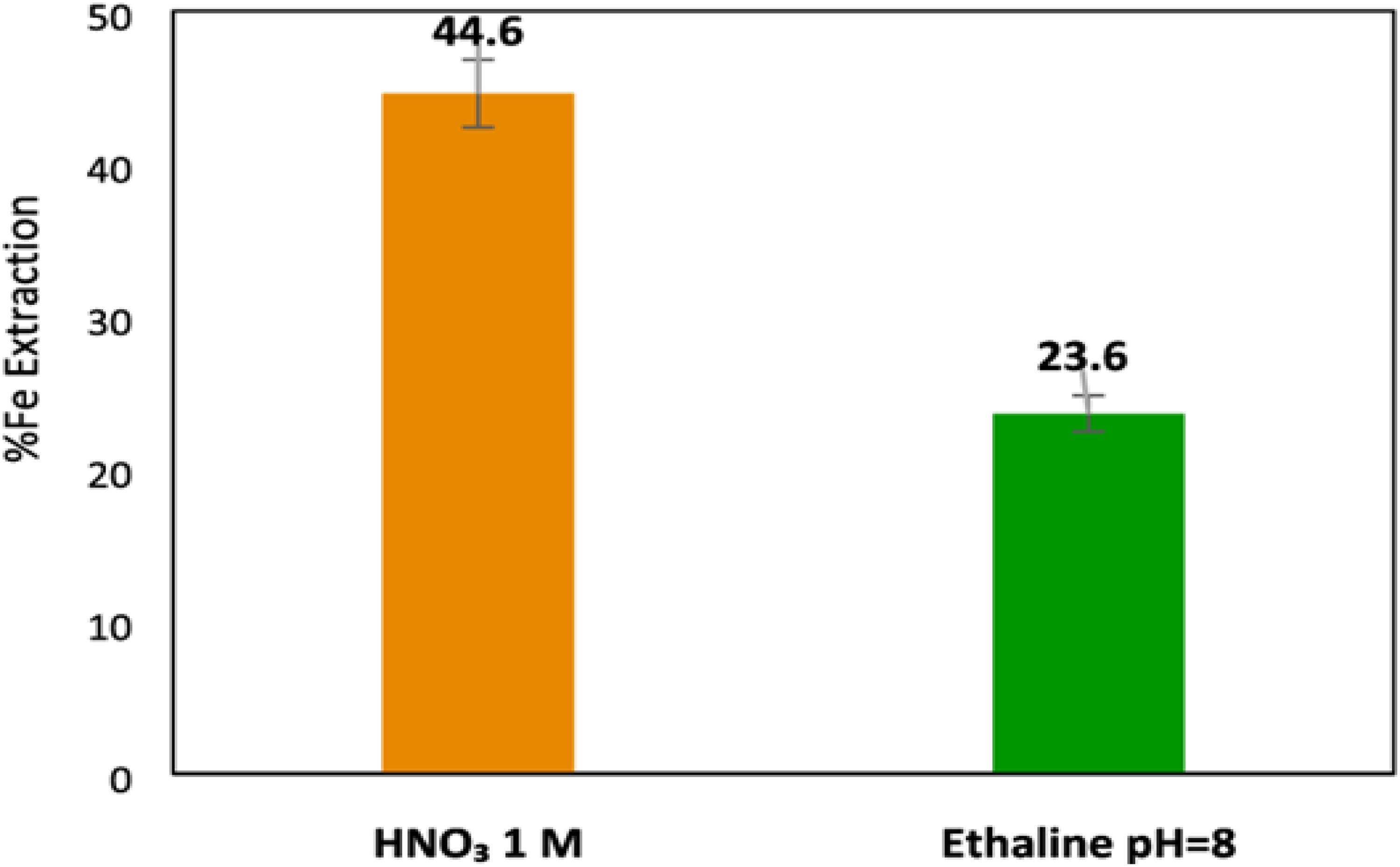
Comparison of pyrite dissolution in 1 M HNO3 and DESs. 69
The dissolution mechanism of arsenopyrite
According to Abbot et al. (2006), the first step in the dissolution of arsenopyrite (FeAsS) occurs when the mineral comes into contact with water or an acidic solution. This can result in the oxidation of Fe2+ in the mineral to form Fe3+ and the release of As3+ ions. The oxidation of Fe2+ to Fe3+ can be represented by using the following equation:
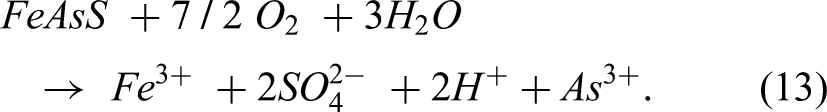
Once the FeAsS mineral has been oxidised, the release of the As3+ ions can occur. This can lead to the formation of arsenic-bearing compounds or the release of arsenic into the surrounding environment. The overall dissolution reaction can be represented by the sum of the individual oxidation reactions according to the equation below:
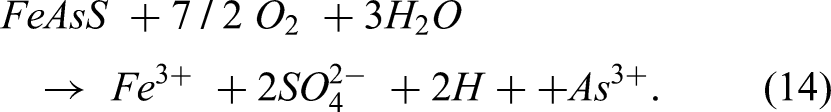
The reaction of dissolution of arsenopyrite can release toxic arsenic compounds into the environment, posing serious environmental and health risks. Therefore, the management of arsenopyrite waste is crucial to prevent the release of arsenic and its contamination of water sources and ecosystems.
Kuzas et al. studied the kinetics of dissolving arsenopyrite in nitric acid utilising the rotating disk method. The increase in temperature from 318 K to 333 K and nitric acid concentration from 4 M to 5 M was shown to significantly accelerate the rate at which arsenopyrite dissolves. Concurrently, the dissolving rates of iron and arsenic in the arsenopyrite composition increased by 30.1–30.4 times, respectively, from 0.26–0.27 × 10–6 mol dm−2 s−1 to 7.83–8.21 × 10–6 mol dm−2 s−1. The temperature is more important than the concentration of nitric acid. There is no discernible impact from the disk rotation frequency. The rate of dissolution is not significantly increased by increasing the temperature to 348 K. 1 The dissolution results are illustrated in Figure 7(a) to (d).
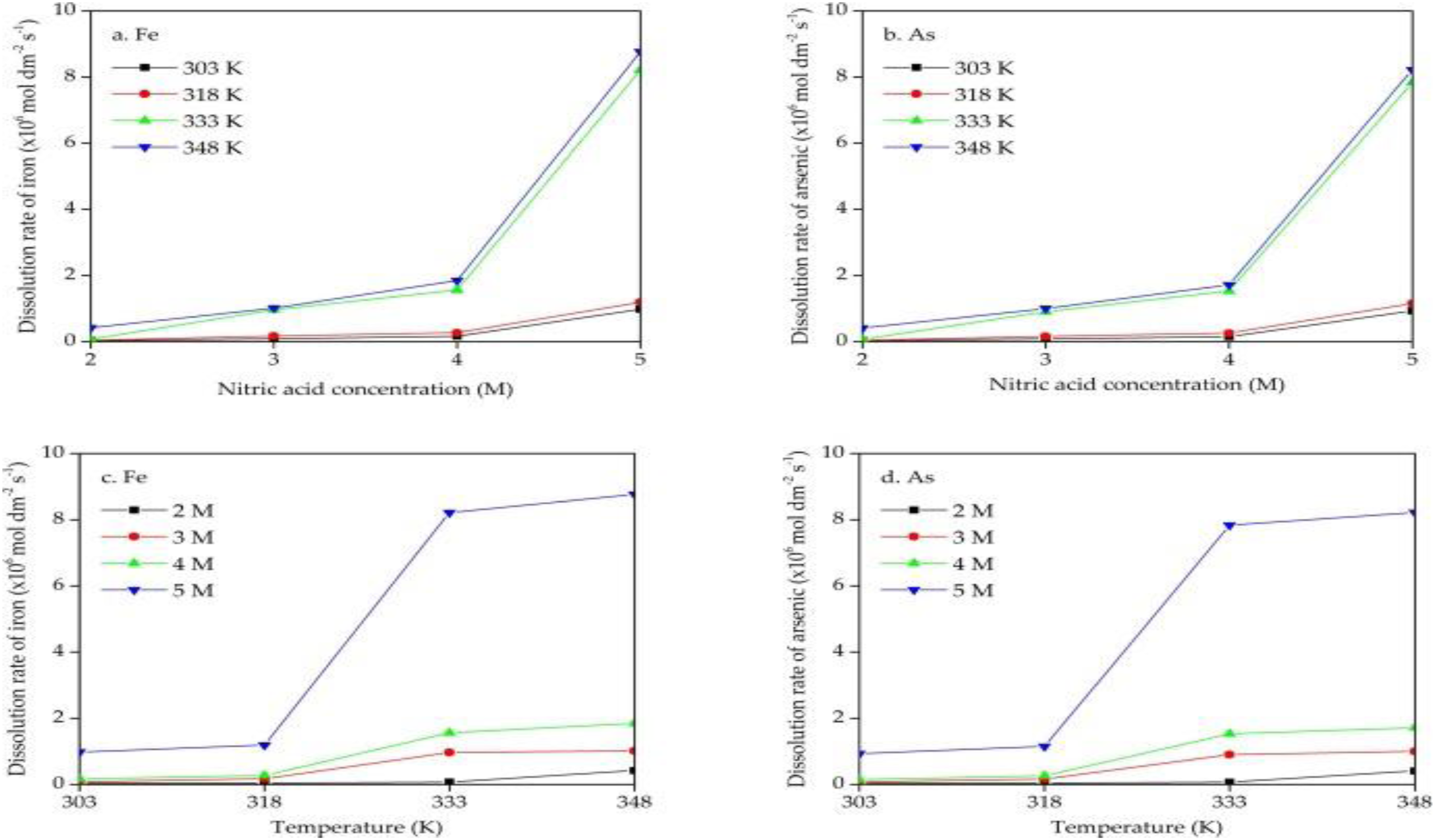
Dependencies of the dissolution rates of iron (a and c) and arsenic (b and d) obtained by graphical differentiation on the nitric acid concentration (from 2 M to 5 M) and temperature (from 303 K to 348 K) at a disk rotation frequency of 10 rps after 3000 s. 1
However, DESs offer advantages relative to acid solvents with the potential of offering a similar mechanism. The chemical reaction for the dissolution of arsenopyrite (FeAsS) in deep eutectic solvents (DESs) can be expressed as follows: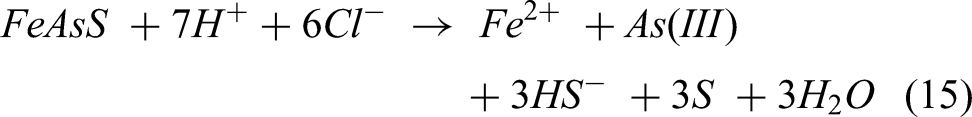
Ferrous ions (Fe2+), trivalent arsenic (As(III)), hydrogen sulphide ions (HS−), elemental sulphur (S) and water (H2O) are formed in this reaction when arsenopyrite (FeAsS) is dissolved in the DES. The precise process and products of the dissolving reaction can be influenced by the particular composition 70 of the deep eutectic solvent as well as the reaction conditions.
Deep eutectic solvents have often been favourable for the dissolution of pyrite compared to arsenopyrite. Consequently, a variety of scientific reports demonstrated its dissolution in DESs using various techniques of extraction, and had revealed that the dissolution of all minerals results in the formation of Fe(III); thus, the yellowish colour to orange confirms the presence of ferric iron as shown in Figure 8. The authors stated that for pyrite, it was assumed that both sulphur and Fe(III) were dissolved. However, the dissolution of arsenopyrite only resulted in arsenic and not sulphur or Fe(III). In the same direction, Jennifer et al. disclosed the colours of different solutions obtained after the bulk electrochemical dissolution of 2 g of mineral powder in a choline chloride-based DES. The depicted colours of pyrite and arsenopyrite in this work were those reported (see Figure 9). The work also denoted that the dissolution of arsenopyrite was considerably slower compared to the other oxide minerals.25,70

Image displaying a sample of ethaline before electrolysis (centre), after both reduction of pyrite (left) and oxidation of pyrite (right) at a constant potential of 2 V. 49

Solution colour after bulk anodic dissolution of 2 g of mineral powder in 1ChCl : 2EG, at a constant current of 10 mA (except for loellingite-1, which was 5 mA) over 24 h at 50 °C. 70
The successful dissolution of arsenopyrite in DESs mostly occurred through the electrochemical process; though in many cases, the dissolution was reported to be very slow or did not occur at all. This could probably be attributed to the composition of DESs and the reaction conditions. In contrast, pyrite has been successfully dissolved in choline chloride-based DESs.
We recently started investigating the possibility of dissolving arsenopyrite in DESs through a direct leaching process at atmospheric pressure and low temperature, as revealed in Figure 10. Although the study is still ongoing, and many characterisation techniques are still being carried out to better understand the findings, we are delighted to disclose that the obtained DESs were favourable for the dissolution of arsenopyrite, as displayed in Figure 10(f), showing the photograph of different samples obtained after a direct leaching process of the received ore in DESs. Since the composition of DESs is crucial for a successful dissolution process, various physical parameters were selected and varied during the preparation of DESs. DESs were prepared by varying the molar ratios of the hydrogen donor. The hydrogen acceptor and donor were mixed into a beaker under vigorous stirring and aged for 30 min at 50 °C until a homogeneous liquid was obtained. Consequently, three types of DESs were prepared. Afterwards, a certain amount of arsenopyrite was put into each of the obtained DESs and kept under continuous stirring and at a constant temperature for different time periods ranging from 1 to 3 h. Abbott et al. demonstrated that the formation of Fe(III) occurs during the dissolution of all minerals, resulting in a yellowish to orange colour that confirms the presence of ferric iron. 49 Considering Abbott's statement, it is obvious that some samples shown in Figure 10(f) indicate the presence of ferric iron, which is depicted by the colour. This indicates that the obtained DESs are promising for the dissolution of arsenopyrite. However, more insights will be revealed after completing the characterisation process. Meanwhile, Figure 10(a) displays the mineralogical composition of the received ore recorded using an XRD Rigaku SmartLab X-ray diffractometer with CuKα (λ = 1.5418 Å) radiation. The XRD patterns indicate the presence of arsenopyrite, a high concentration of quartz, and a low concentration of pyrite. 71 Furthermore, Figure 10(b) shows the SEM image captured by using a JEOL JSM-7800F FE-SEM coupled with an EDS Thermo Fisher Scientific, revealing the arsenopyrite spheres layered with crystal particles, presumably quartz. Meanwhile, Figure 10(c), along with Figure 10(d), displays the elemental composition of the received ore, disclosing the chemical elements such as Si, S, Fe, As, Ag and Au, which were identified by the XRD results earlier. However, the presence of carbon is attributed to the sample preparation before analysing to stabilise the particles from scattering. Figure 10(e) shows the mapping images of the received ore showing the elemental distribution within the ore. It was observed that a high concentration of quartz present as silica was seen, which was also witnessed in the XRD results. Moreover, the presence of gold in fine particles spread throughout the ore, iron, arsenic and sulphur was also observed. This proved that the received ore was that of arsenopyrite.
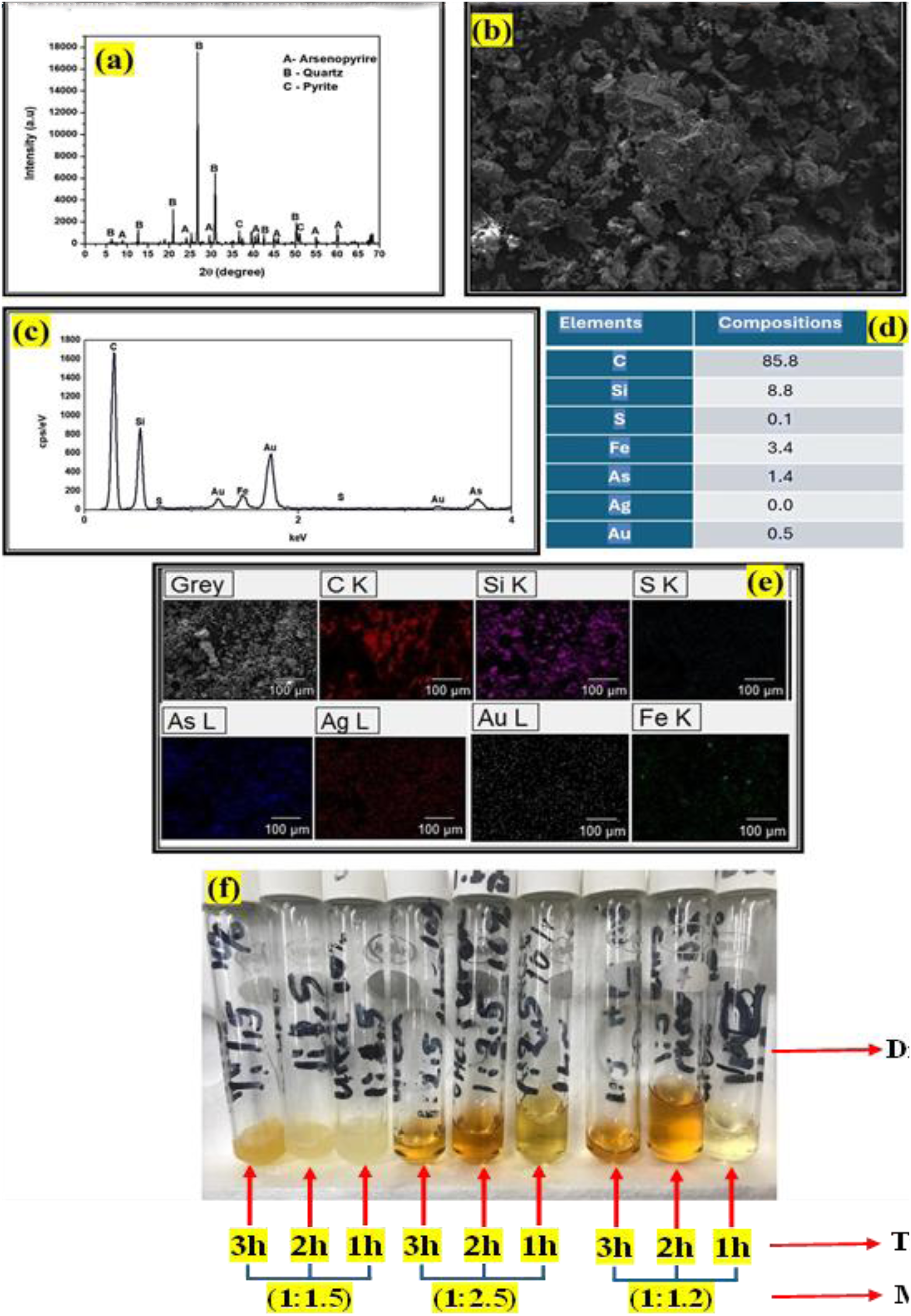
(a) XRD patterns, (b) SEM image, (c) EDS spectrum, (d) mapping images of the received ore, (e) elemental composition and (f) the photograph of different samples obtained after direct leaching of the received ore in DESs.
Conclusion
This work aims to display the possible dissolution of arsenopyrite through direct leaching in deep eutectic solvents. Although arsenopyrite has shown partial to no dissolution in other previous works relative to pyrite, this current work has displayed partial results favouring the possible solubility of the sulphide ore in choline chloride-based DESs. The identity of the ore used in this work was confirmed by XRD and EDS. Although the current study is still underway due to other characterisation processes for a better insight into the findings, the preliminary results ensure that DESs are also promising for the dissolution of arsenopyrite through direct leaching.
Footnotes
Acknowledgements
The authors would like to thank the Tshwane University of Technology, Department of Chemical, Metallurgical and Materials Engineering and the staffs for providing necessary supporting materials during the write-up of this review.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.

