Abstract
This study introduces a multistage hydrometallurgical process designed for the recovery and individual separation of Nd and Pr from NdFeB magnet scrap. Following demagnetization, the magnet underwent crushing, grinding, and leaching using a sulfuric acid solution. The pH of the resultant leachate was subsequently adjusted to 1.2 using a NaOH solution, leading to the precipitation of Nd and Pr as sodium double sulfates. The produced double sulfate was then mixed with a saturated NaOH solution, resulting in the formation of rare earth hydroxide. The hydroxide was further decomposed into oxides (REO) after calcination at 500°C. The REO was dissolved in hydrochloric acid and nitric acid solutions to investigate the individual separation of Nd and Pr using the D2EHPA–TOA extractant system and compare the results with D2EHPA-only system. It was concluded that the extraction mechanism of D2EHPA–TOA system is highly dependent on the acidity of the solution, with TOA having a synergistic effect on the REE extraction when pH > 3, and an antagonistic effect when pH ≤ 3. In terms of extraction efficiency, both extractant systems demonstrated efficiency levels exceeding 99% when the pH was ≥2, with similar behavior when extraction was performed in HCl and HNO3 media. Although both extractant systems yielded unsatisfactory outcomes in the separation of Nd and Pr, a marginal improvement was observed with the incorporation of TOA into the extractant system. This observation implies the potential of exploring analogous combinations in future studies.
Keywords
Introduction
The demand for rare earth elements (REEs) is on rise owing to their growing utilization in a range of high-tech applications. In reaction to this trajectory, the recycling of REEs from secondary sources has garnered significant attention and has undergone substantial development in recent years.1–3 This approach enables the recovery and reuse of valuable REEs, effectively addressing the escalating demands of various industries, while simultaneously reducing dependence on primary sources. NdFeB permanent magnets stand as a substantial secondary source of REEs, owed to their exceptional properties. These magnets have been extensively used across various products, including wind turbines, cell phones, computer hard drives, and electric vehicles. Given their exceptional strength and durability, NdFeB magnets play a critical role in the development of high-performance and energy-efficient technologies.4,5 Given the high concentration of REEs present in NdFeB magnets, the recycling of these magnets is poised to have a crucial role in the future supply chain of these valuable elements. 3 The primary constituent elements of NdFeB magnets comprise Nd, Pr, and Fe. However, the presence of additional elements such as Dy, Gd, Ce, Al, Cu, Co, and Ni can vary based on the desired properties and intended applications. 6 These supplementary elements are meticulously chosen to finely adjust the magnetic and mechanical properties of the magnet, making it appropriate for various high-performance applications. Extensive research has been dedicated to pyrometallurgical processes for extracting REEs from magnet scrap, with molten slag extraction standing out as a widely employed technique. 7 Hydrometallurgical processes currently stand as the most extensively employed techniques for recovering REEs from end-of-life permanent magnets, owing to their effectiveness and versatility.3,7–10
Among the different methods introduced for leaching NdFeB magnets, the utilization of mineral acids such as sulfuric acid and hydrochloric acid is widely reported in the literature.11–14 The significant challenge at this stage arises from the substantial iron content within the leachate, which hinders the effective separation of REEs from other elements. To address this issue, a proposed solution involves a single-step selective precipitation process to isolate the REEs from other elements.11,13 This method enables the selective precipitation of REEs, leaving iron and other impurities in the solution. This simplifies the separation process and enhances the efficiency of REE recovery. The precipitation of double sulfate salt of REEs by introducing NaOH, KOH, or NH4OH into the leachate has proven to be an efficient and effective recovery technique. Nevertheless, precise control of the solution's pH is crucial for achieving high recovery efficiency while minimizing the co-precipitation of other elements. Another documented recovery approach involves the precipitation of REEs as oxalate, but this method can incur significant costs due to the substantial consumption of oxalic acid and may result in substantial co-precipitation of Ni if it is present in the solution.11,15 Direct precipitation of REE fluorides by adding hydrofluoric acid represents an alternative method for separating REEs from iron. Nevertheless, this approach comes with primary challenges, including the demanding filtration conditions required for NdF3 and the susceptibility of process to iron contamination.11,16
In contrast to selective precipitation methods, some researchers7,17 have directed their attention toward eliminating iron via a selective leaching process. This approach involves subjecting the material to high-temperature treatment, known as oxidative roasting, before proceeding with the leaching phase. In the course of this process, REOs are selectively leached, but it is imperative to exercise precise control over temperature and pH in order to attain high recovery efficiency for REEs. Despite the need for extra processing steps, this method has demonstrated its potential for effectively separating REEs from iron in NdFeB magnets.
Solvent extraction is a widely employed method for recovering REEs from NdFeB magnet leachates as it facilitates the separation of REEs, either as a group or individually, from aqueous solutions. 18 Various organic extractants have been utilized for this purpose, including D2EHPA (di-(2-ethylhexyl) phosphoric acid), 19 PC 88A (2-ethylhexyl phosphonic acid mono-2-ethylhexyl ester),19,20 Cyanex 302 (bis(24,4-trimethylpentyl)monothiophosphinic acid), 21 and Cyanex 272 (bis-(24,4-trimethylpentyl) phosphinic acid).22,23 Among these extractants, D2EHPA stands out as the most frequently employed option, due to its functional versatility and high recovery efficiency. 24 Numerous studies have reported on the effectiveness of D2EHPA-based solvent extractant systems for the recovery of REEs from NdFeB magnet leachates. Yoon et al. 19 conducted a study examining the recovery process of Nd and Dy from NdFeB magnet scrap leachate. They conducted a comparative analysis of two organophosphorus extractants, D2EHPA and PC 88A, for the separation of REEs, concluding that both extractants are highly effective, demonstrating significant efficiency. Padhan and colleagues 21 investigated the leaching process of NdFeB magnet scrap in an HCl solution, followed by solvent extraction of REEs using the Cyanex 302 organic phase system. They reported a high separation factor for Nd over Dy at a low equilibrium pH. In another study, Padhan and colleagues 22 performed a comparative analysis of various extractants for the recovery of Nd and Pr from the chloride leachate of NdFeB magnet scrap. They ranked the extraction efficiency of different extractants as follows: Cyanex 272 + Alamine 336 IL > D2EHPA + Alamine 336 IL > Cyanex 272 > D2EHPA > Aliquat 336. Banda and colleagues 23 conducted a study focused on the separation of Nd and Pr from a chloride solution that also contained La. Although they noted relatively low extraction efficiencies, Cyanex 272 exhibited a notably high separation factor between Pr and Nd. Liu and co-workers 25 enhanced the separation of Nd and Pr by introducing TBP (tri-n-butylphosphate) into the mixture of Cyanex 272 and Alamine 336, resulting in increased separation factors for these two elements.25,26
The recovery efficiency of REEs in precipitation and solvent extraction from aqueous media can be influenced by a variety of factors. This study explores a multistage hydrometallurgical process aimed at recovering REEs from NdFeB magnet scrap and studying the individual separation of the elements. The procedure entails leaching the demagnetized and ground magnet in an H2SO4 solution, followed by the precipitation of REEs double sulfate salt. During the precipitation, iron and other impurities remain in the leachate, while the precipitate undergoes treatment with a saturated NaOH solution to yield a mixture of Nd–Pr hydroxides. A straightforward calcination process is employed to convert the hydroxide mixture into oxide, with the study also delving into the chemical reactions and thermodynamic aspects of this process. Subsequently, the work assesses the individual separation of Nd and Pr through solvent extraction from the leachate of their produced mixed oxide. A novel extractant system, comprising a combination of D2EHPA and TOA is employed, and the effect of adding TOA to the extractant system on the Nd–Pr separation is thoroughly examined and discussed.
Materials and methods
Materials
This study employed a NdFeB magnet scrap sourced from household electrical appliances as the primary raw material. The organic extractants utilized consisted of di-(2-ethylhexyl) phosphoric acid (D2EHPA, Farapouyan Isatis, Iran) and Trioctylamine (TOA, Wako-Japan, purity: 97%). Additionally, 1-Decanol (Merck, purity: 99%) and Kerosene (Tehran refinery, Iran) were incorporated as phase modifier and diluent, respectively. Analytical grade sodium hydroxide with a purity of 99.9% from Merck was utilized. Furthermore, the sulfuric acid, nitric acid, and hydrochloric acid employed within this study held analytical grades and were obtained from a domestic company. Solid materials were characterized using X-ray diffraction analysis (Bourevestnik DRON-8, Russia, Cu Kα radiation, 40 kV, 30 mA, step size = 0.04) and scanning electron microscopy (TESCAN VEGA//XMU) equipped with energy-dispersive X-ray spectroscopy (SEM-EDS). For the quantification of metal concentrations in the solution, inductively coupled plasma optical emission spectrometry (ICP-OES, Varian Vista-PRO) was employed.
Methods
Production of REEs mixed oxides
The NdFeB magnet scrap underwent an initial demagnetization process within a muffle furnace at 450°C for a duration of 1 hour. Following demagnetization, the magnet was subjected to crushing using a metallic pounder, and subsequently ground using a planetary ball mill equipped with tungsten carbide-coated balls and a grinding jar. This grinding procedure effectively reduced the average particle size of the magnet to below 100 μm. The chemical composition of the magnet was determined through XRF analysis, with the outcome detailed in Table 1.
Chemical composition of the used NdFeB magnet.

The magnet, once ground, was subjected to leaching in a 2 M sulfuric acid solution, maintaining a solid-to-liquid ratio of 1:20, for a duration of 1 hour at room temperature. To induce the precipitation of Nd and Pr as sodium double sulfate, sodium hydroxide solution was gradually added to the leachate, thereby adjusting the pH to 1.2. The resultant double salt underwent filtration, followed by rinsing with distilled water. Subsequently, it was combined with a saturated sodium hydroxide solution at twice the stoichiometric quantity of REEs, allowing it to react for one hour at 70°C with a solid-to-liquid ratio of 1:20. This process yielded Nd–Pr mixed hydroxides. The resultant product was washed with distilled water, dried in an oven for mass loss calculations, and subjected to heating at 500°C for 1 hour to decomposition into oxides (REO).
Solvent extraction of REEs
To assess and compare the extraction behavior of the employed extractant systems across different acidic environments, stock solutions were prepared by dissolving REO in 0.5 M solutions of HCl and HNO3. Initially, attempts were made to extract REEs from the chloride stock solution using organic phases containing D2EHPA at concentrations ranging from 5 to 40 v/v% at an A/O (Vaqueous/Vorganic) ratio of 1, and an initial pH of 3. This aimed to identify the optimal D2EHPA concentration. Subsequently, the A/O ratio was adjusted from 1 to 10 to identify the optimal value. The extraction behavior of Nd and Pr in HCl and HNO3 media within a pH range of 0–6 was investigated using the optimized values of A/O and D2EHPA concentration, for two different extractant systems of D2EHPA and D2EHPA-5 v/v% TOA-5 v/v% Decanol for comparison. The influence of TOA addition on the Nd–Pr separation was assessed and discussed and the extraction mechanism was analyzed in depth. All experiments were conducted at room temperature for a duration of 15 min, and following each experiment, the characterization of the aqueous phase was performed to calculate the extraction efficiency of the elements.
Results and discussion
Production of REEs mixed oxides
Table 2 presents the chemical analysis of the magnet leachate, demonstrating leaching efficiency of over 99% for REEs, and 97.4% for iron as the major impurity. To adjust the pH, a sodium hydroxide solution was gradually added to the leachate while stirring, resulting in the formation of a faintly pink precipitate. As provided in Table 2, nearly all the Nd and Pr were precipitated in a single-step process. However, according to the concentration balance, the precipitate contained approximately 1 wt.% iron, with negligible co-precipitation of other elements. X-ray diffraction (XRD) analysis confirmed that the precipitate comprised a mixture of neodymium and praseodymium sodium double sulfates with no other impurities detected.
The results of leaching and precipitation experiments
Figure 1 illustrates the XRD pattern of the synthesized Nd–Pr sodium double sulfate, displaying the characteristic peaks of NaNd(SO4)2 (Ref. code: 01-083-2167) and NaPr(SO4)2 (Ref. code: 00-021-1154).
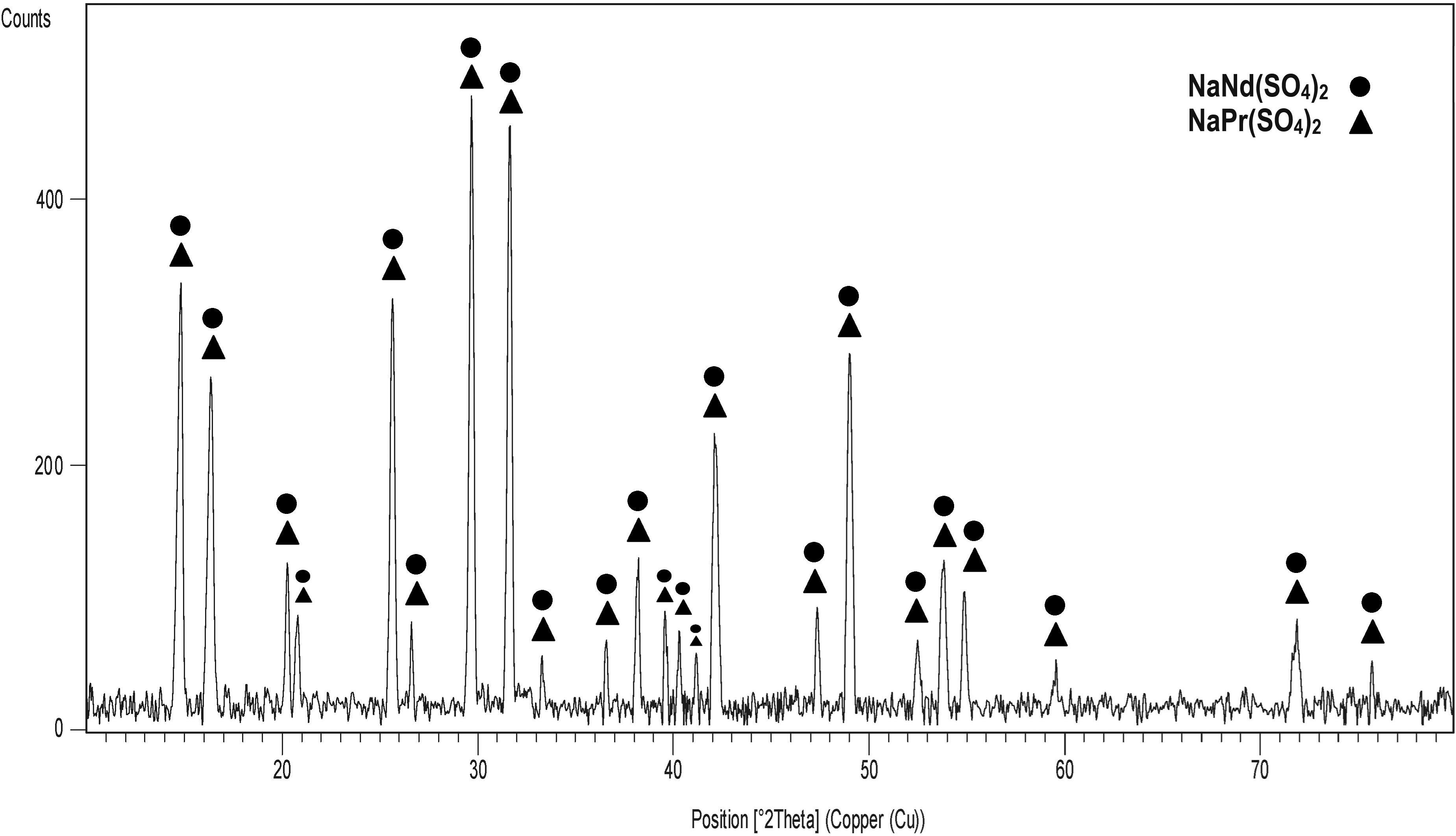
X-ray diffraction pattern of the produced REEs double sulfate salt.
Based on the metathetic reaction provided in Equation (1), subsequent treatment of the Nd–Pr mixed sodium double sulfates with a saturated solution of sodium hydroxide results in the formation of Nd and Pr hydroxides.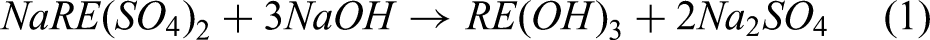
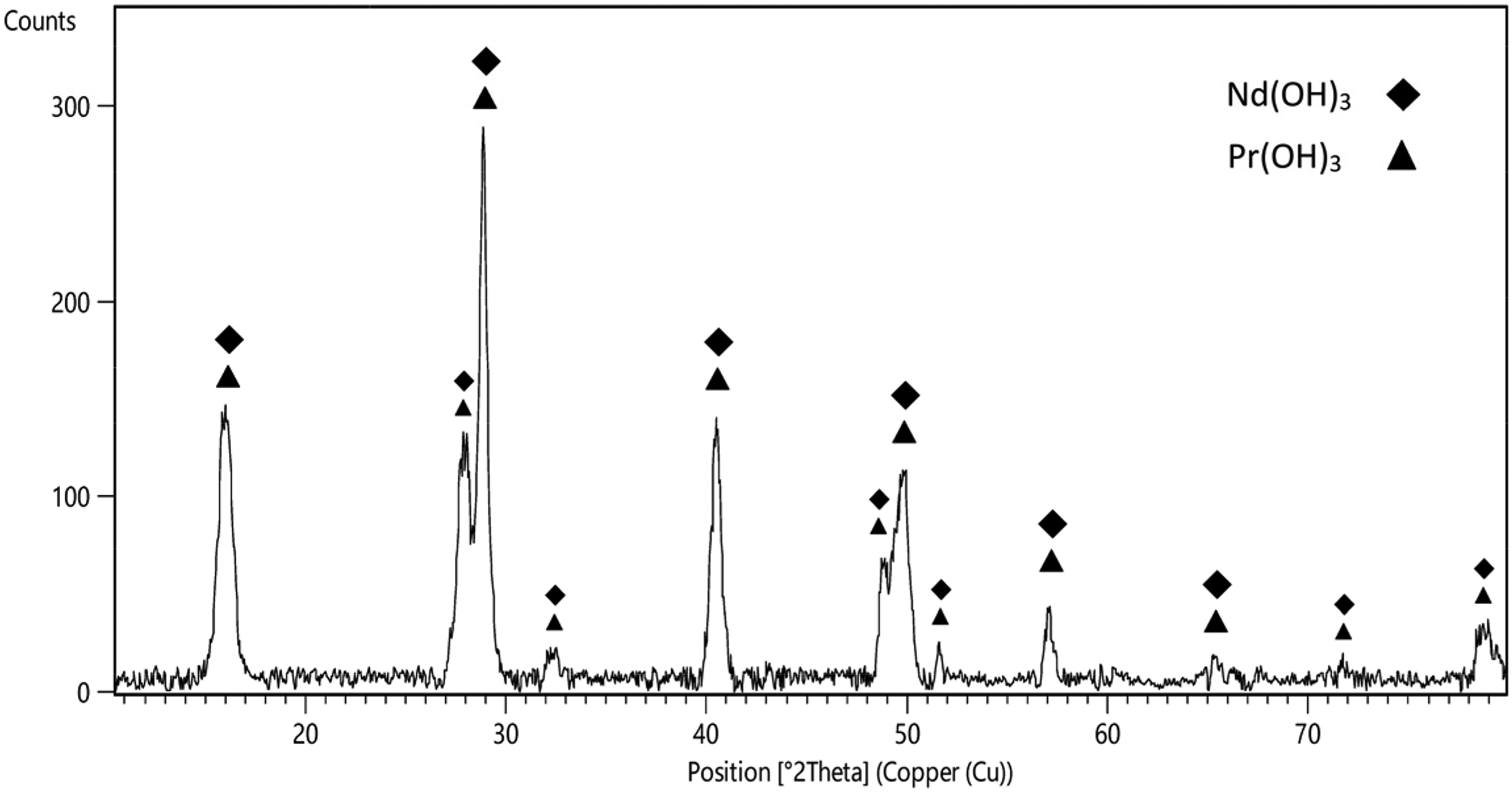
XRD diffraction pattern of the produced Nd–Pr mixed hydroxides.
The subsequent step involved the conversion of the hydroxides into oxides through a calcination process. Figure 3 displays the predominance diagrams for the Nd(OH)3–Nd2O3 and Pr(OH)3–Pr2O3 systems, with the data obtained from HSC Chemistry 9.9. The diagrams provide thermodynamic insights, indicating that under conditions where the partial pressure of oxygen (P(O2)) is 0.21 atm (air) and the temperature (T) exceeds 308°C, Nd(OH)3 should thermodynamically decompose into Nd2O3. Similarly, Pr(OH)3 is expected to decompose into Pr2O3 when the temperature exceeds 304°C. Consequently, a calcination temperature of 500°C was selected to ensure the complete conversion of both hydroxides, according to Equation (2).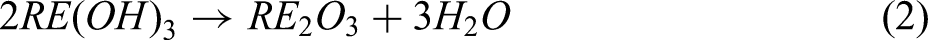
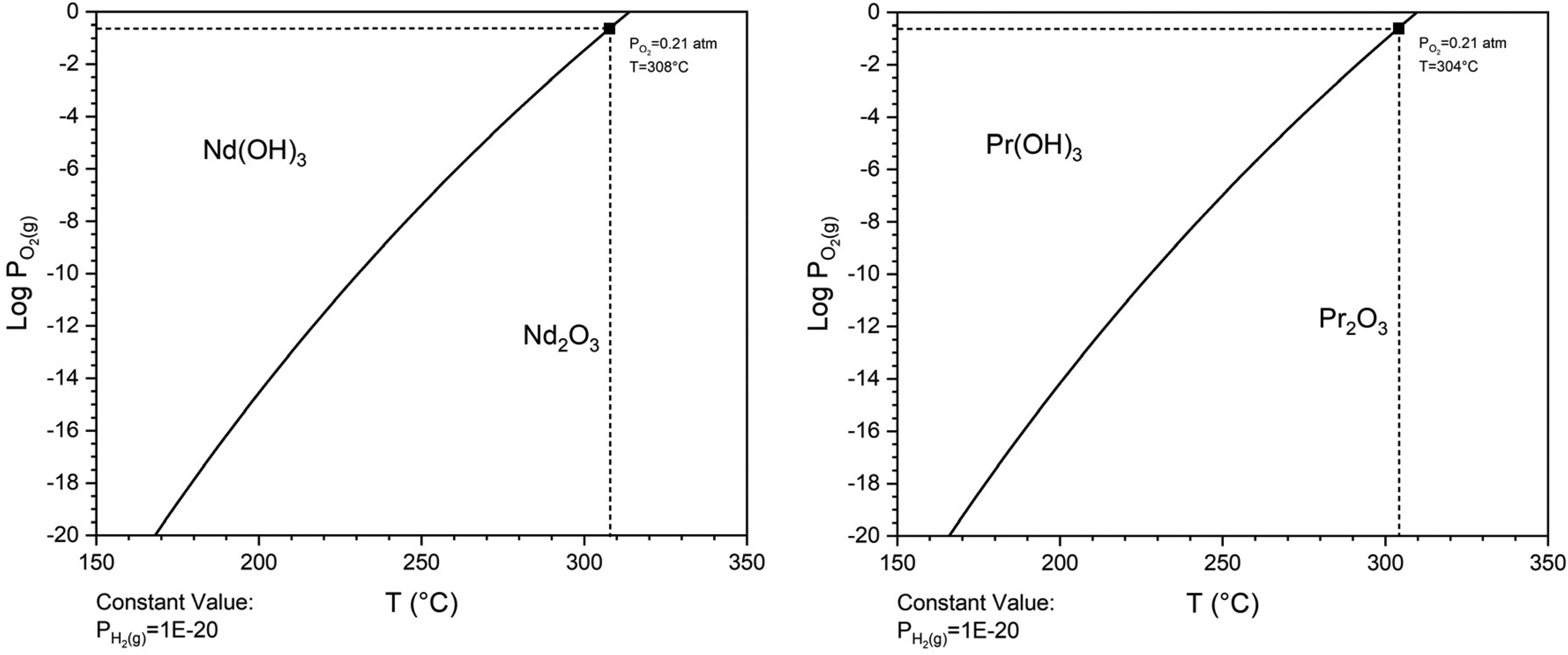
Predominance diagrams for Nd(OH)3–Nd2O3 and Pr(OH)3–Pr2O3 systems (data from HSC chemistry 9.9).
Figure 4 presents the X-ray diffraction pattern of the calcined product, offering proof of the thorough conversion of Nd and Pr hydroxides into Nd2O3 (Ref. code: 00-045-0087) and Pr2O3 (00-042-1121), respectively.
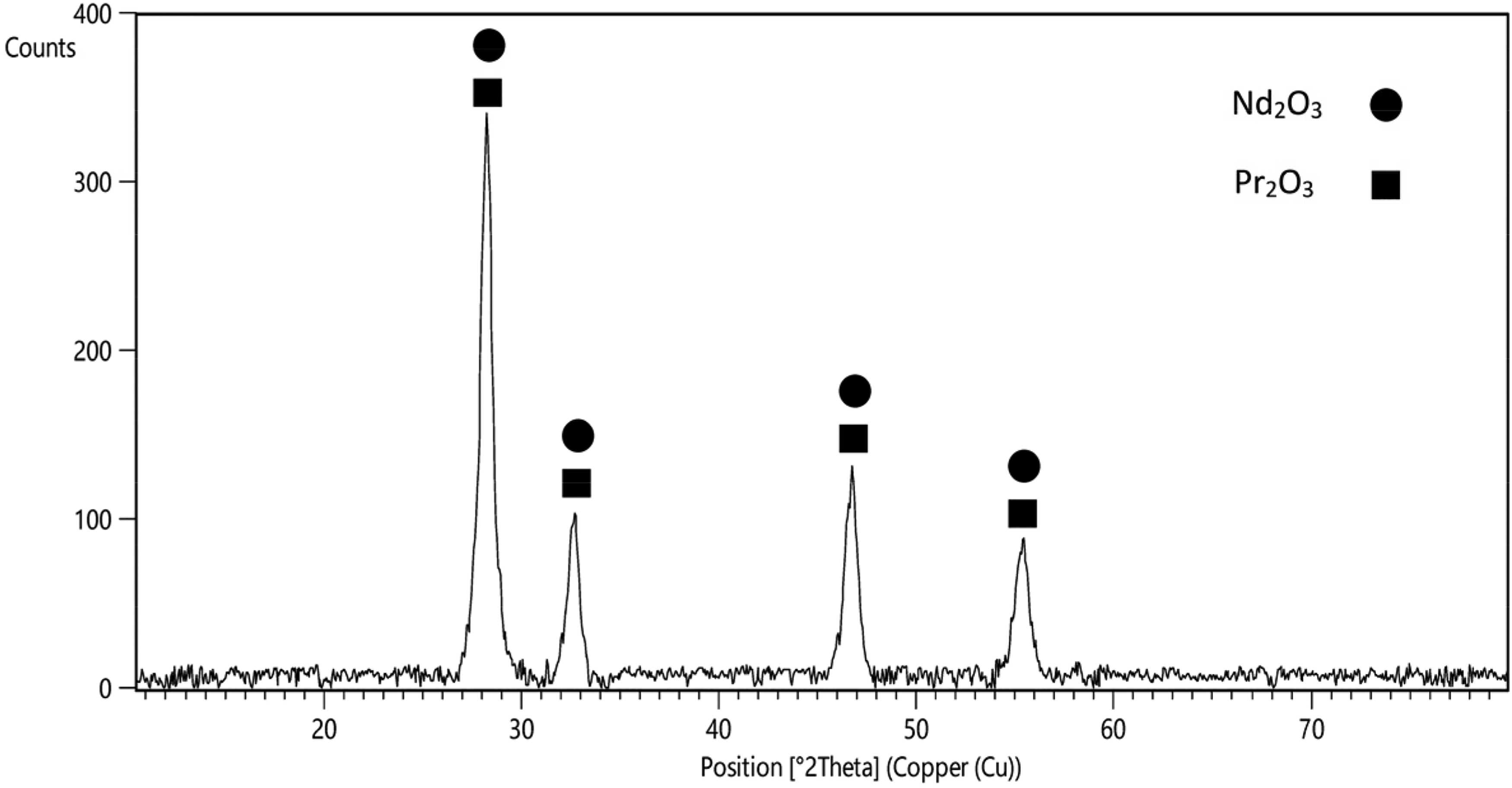
XRD analysis of the produced REO.
The SEM analysis of REO in Figure 5 depicts intermingled rod-shaped oxide particles characterized by an average particle size of 4 µm and a uniform structure. The close chemical and physical resemblance between Nd2O3 and Pr2O3 led to the creation of a mixed compound, making individual distinction impossible. Moreover, the EDS analysis of these particles showcases the mixture's high purity, as no other elements were detected.
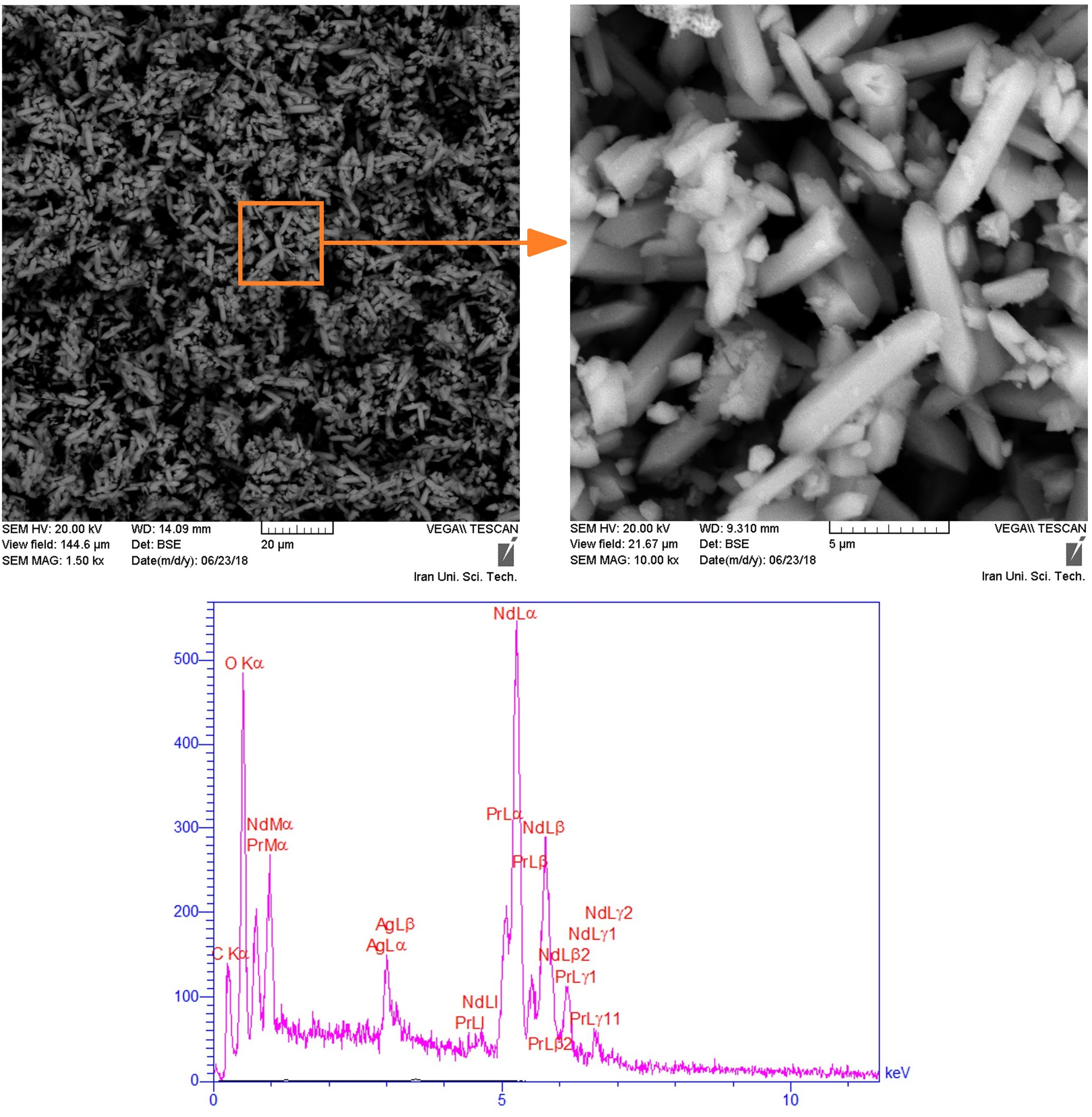
SEM and EDS analysis of the produced REO (Ag coating).
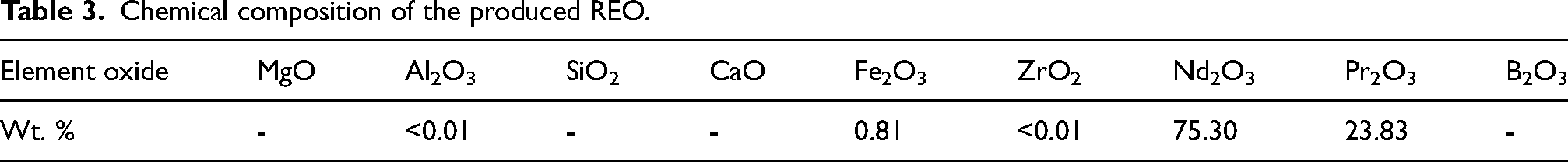
The determination of the chemical composition of the REO involved dissolving a sample in a 0.5 M HCl solution, and the corresponding data is presented in Table 3. The analysis affirms the purity of Nd and Pr in the REO to be over 99%, with Fe identified as the primary impurity. This outcome indicates the effective achievement of recovery and purification of REEs from the magnet.
Chemical composition of the produced REO.
Solvent extraction of REEs
Optimizing extraction parameters for the reference system
Figure 6 depicts the influence of D2EHPA concentration in the organic phase on the extraction efficiency of Nd and Pr from the chloride leachate. It is evident that elevating the D2EHPA concentration enhanced the extraction efficiency of Nd and Pr. The utilization of a 30 v/v% D2EHPA system yielded 100% efficiency for both elements, which remained consistent at higher concentrations. Consequently, the 30 v/v% D2EHPA system was chosen as the reference for subsequent experiments.
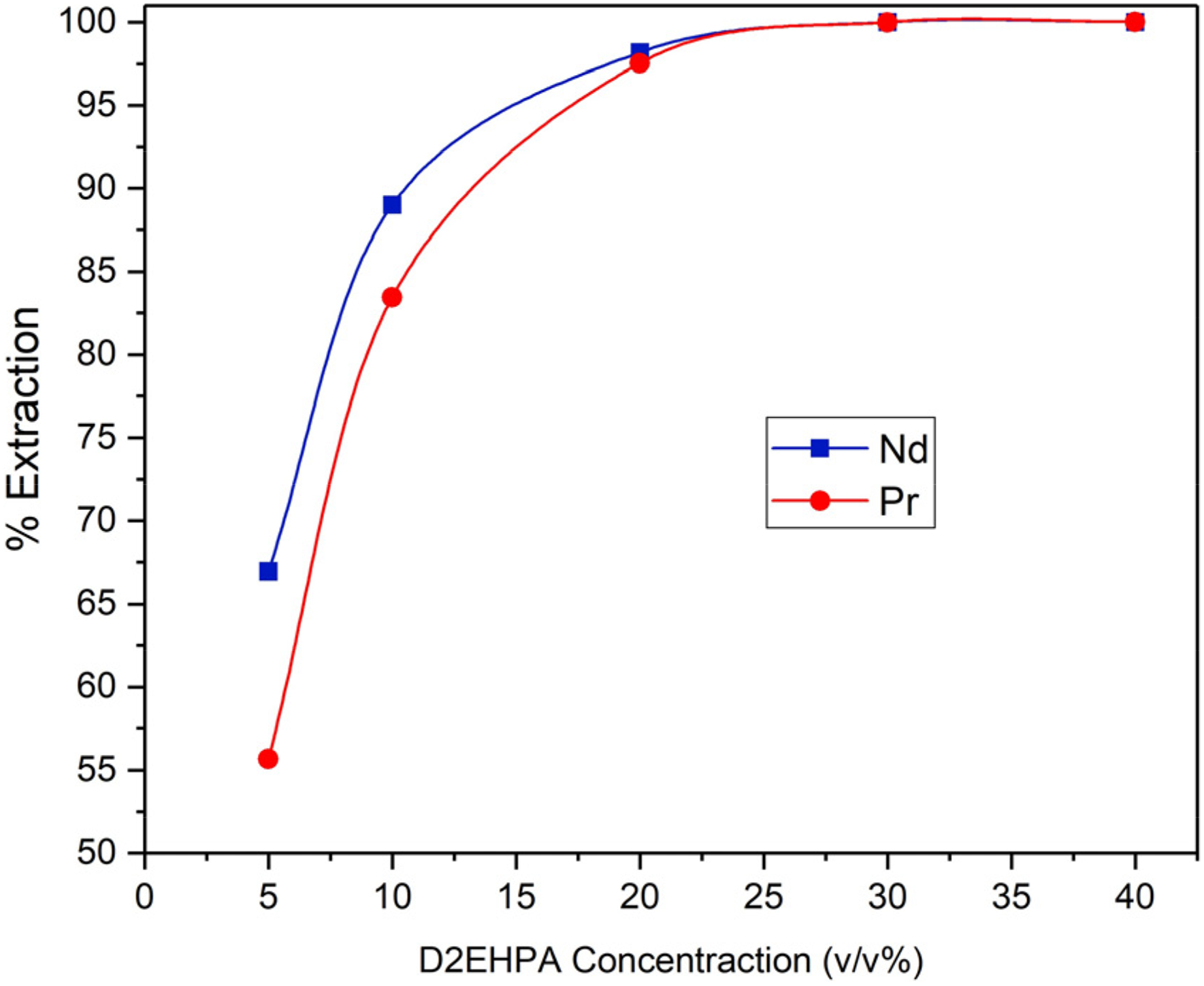
The effect of D2EHPA concentration on Nd and Pr extraction yield (A/O = 1, pH = 3).
The effect of the A/O ratio on the extraction efficiency of Nd and Pr is depicted in Figure 7. The data reveals that when A/O was set at 1, more than 99% of Nd and Pr were extracted. Nevertheless, as the A/O ratio increases, the extraction efficiency declines, with the rate of decrease being more significant for Pr in comparison to Nd.
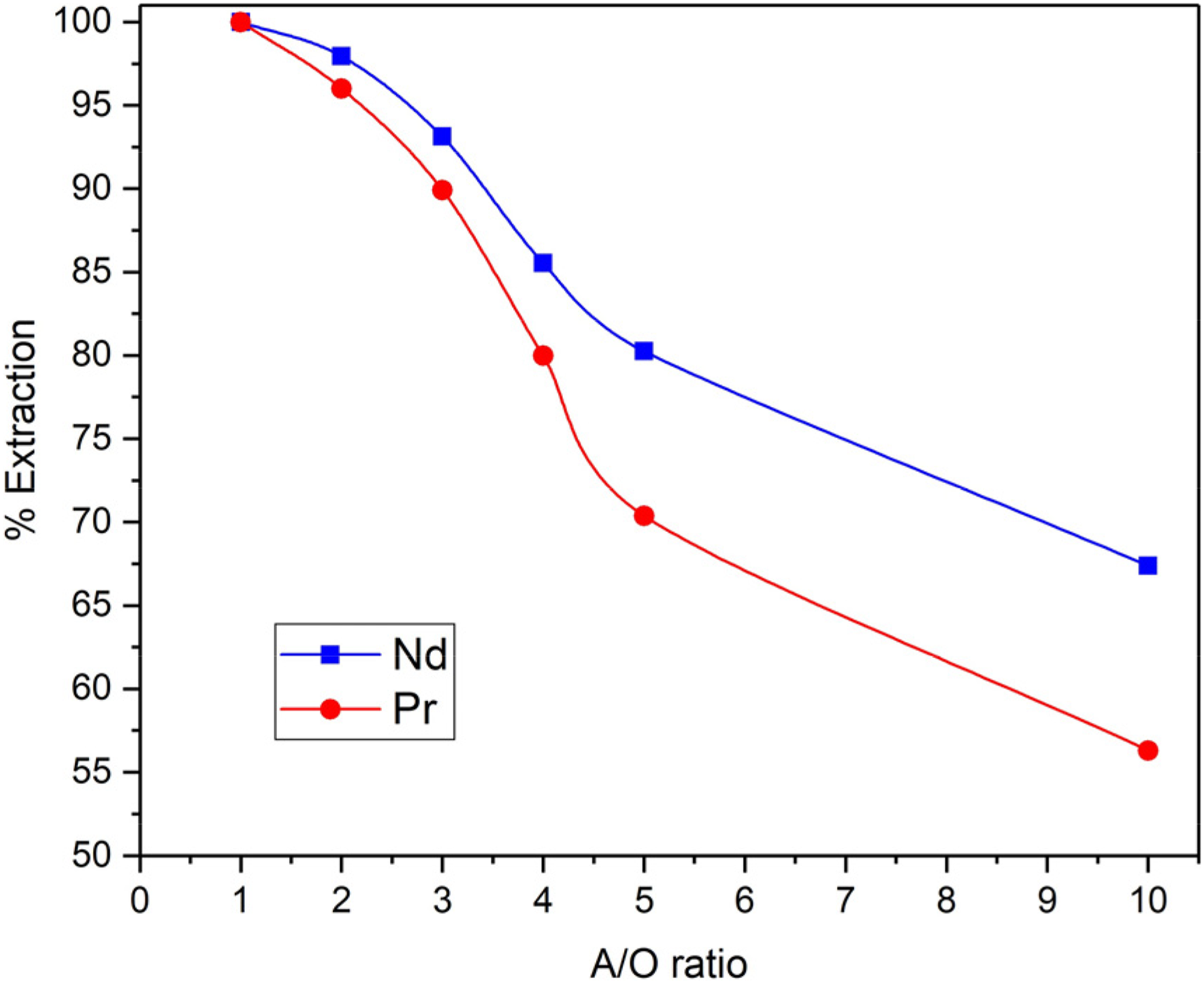
Variation of Nd and Pr extraction yield with A/O ratio (30 v/v% D2EHPA, pH = 3).
Extraction mechanism of D2EHPA–TOA system
According to Zhang and colleagues,
27
the extraction mechanism of D2EHPA undergoes a transition from cation exchange to solvation in highly acidic solutions. In these conditions, D2EHPA molecules extract neutral complexes comprising the metal cation and the conjugate base of the acid (Cl− or NO3− in this instance). The extraction equations for a metallic ion (Mn+) with D2EHPA (HA) in low and high-acidity aqueous solutions are provided as Equation (3) and Equation (4), respectively. In low-acidity environments, Mn+ is extracted by D2EHPA via cation exchange. Nevertheless, in highly acidic media, the elevated acidity level of the solution hinders the dissociation of D2EHPA molecules. Consequently, D2EHPA extracts Mn+ through the solvation mechanism, where the conjugate base of the acid (B) and the metallic ion combine with a D2EHPA molecule to form a neutral salt.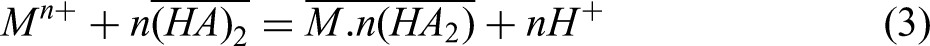
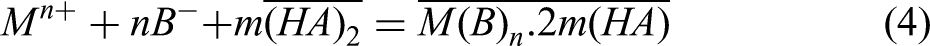
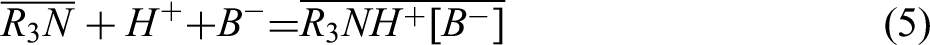
Effect of TOA addition on Nd and Pr extraction
Figure 8 illustrates the extraction performance observed in the cases of the D2EHPA and D2EHPA-TOA systems concerning the extraction of Nd and Pr from chloride and nitrate solutions. The outcomes demonstrate that when the pH is equal to or greater than 2, both metals exhibit analogous extraction behaviors, achieving extraction efficiencies of over 98%. Nonetheless, for the pH values lower than 2, the extraction efficiencies drop to less than 1% when the pH reaches 0. Furthermore, the introduction of TOA into the organic phase exhibited an antagonistic effect on the extraction of Nd and Pr at pH values below 3, where the extraction mechanism shifts toward solvation. This drop could potentially be attributed to TOA extracting Cl− or NO3− ions, as elaborated on previously in Extraction mechanism of D2EHPA-TOA system section. The separation between Nd and Pr slightly enhanced at pH < 2 for both extractant systems, with a higher efficiency for Pr compared to Nd due to its heightened electric charge density. It should be noted that the gap between the curves for the D2EHPA-TOA system was slightly broader than that of the reference system.
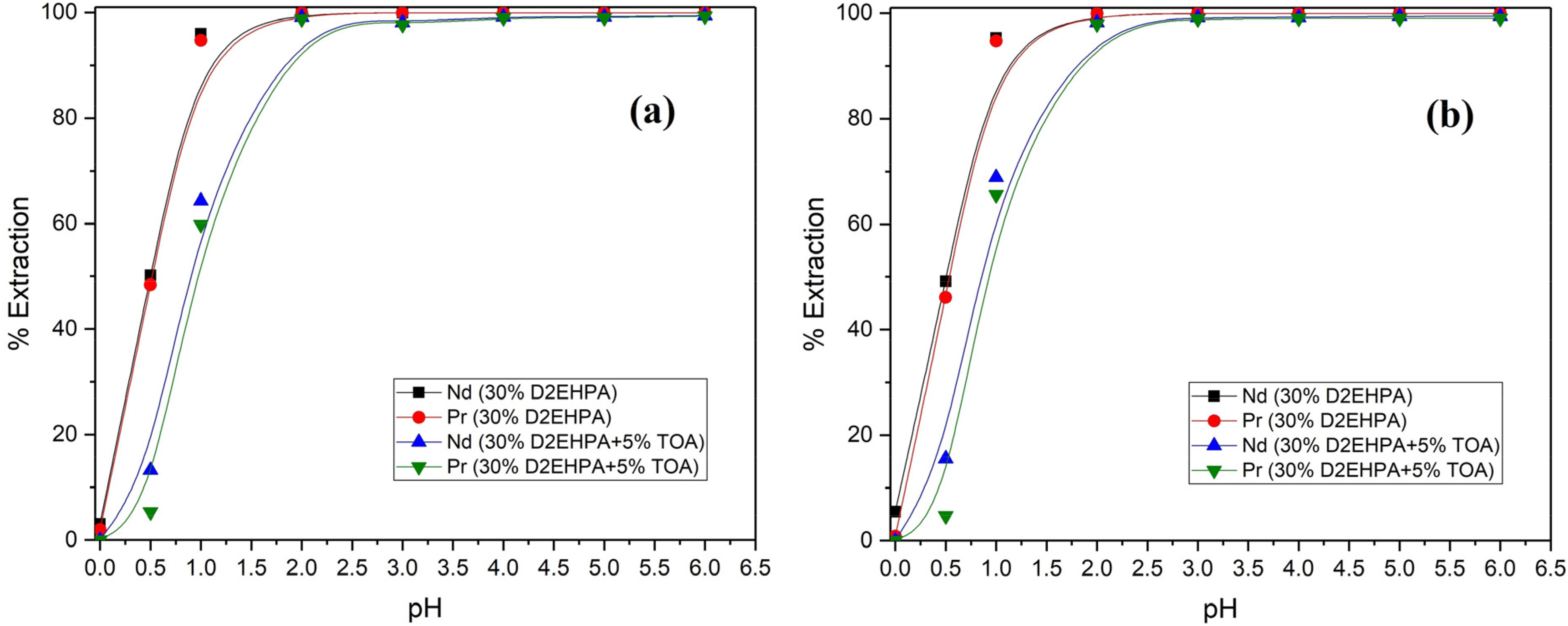
Extraction behavior of Nd and Pr using D2EHPA and D2EHPA-TOA systems in (a) chloride and (b) nitrate medium (A/O = 1).
The extraction behavior of Nd and Pr in both chloride and nitrate solutions exhibited a parallel trend. Nonetheless, under nitrate conditions and at pH values below 3, the extraction efficiency slightly surpassed that of chloride. This phenomenon can be attributed to the greater thermodynamic inclination of nitrate ions to undergo extraction through solvation mechanisms.27,28 Nevertheless, employing nitric acid within the current system is not advisable owing to its notably corrosive character and the significant likelihood of undesirable crud formation during the extraction process. Overall, both extractant systems showcased remarkable efficiency levels surpassing 99% for Nd and Pr when pH ≥ 2, with analogous behavior during extraction in both HCl and HNO3 media. While both systems displayed poor results in the Nd–Pr separation, there was a slight enhancement when TOA was integrated into the extractant system, which suggests the potential of similar combinations for future studies.
Conclusions
This study introduces a hydrometallurgical process aimed at recovering and individual separation of Nd and Pr from NdFeB magnets scrap. The results highlight the following key discoveries:
Almost all the constituent elements, including Fe as the major impurity, were leached out with high efficiencies in a single-step process using 2 M sulfuric acid at room temperature.
A pH of 1.2 yielded complete precipitation of Nd and Pr as double sulfate salt, with less than 0.5 wt.% iron co-precipitation, signifying a highly effective recovery of REEs.
Conversion of Nd and Pr double sulfates to hydroxides occurred via metathetic reaction with a saturated sodium hydroxide solution at 70°C for 1 h. Subsequently, complete decomposition into pure oxides was achieved through calcination at 500°C for 1 hour.
SEM analysis unveiled a uniform rod-shaped morphology of the resulting Nd–Pr mixed oxides. EDS analysis detected no additional impurities.
Despite the reduction in extraction efficiency of Nd and Pr due to the antagonistic effect of TOA when added to D2EHPA, it yielded a slightly higher separation between Nd and Pr at pH < 2 compared to D2EHPA alone. This observation suggests the possibility of further research into selectively separating REEs using a combination of acidic and basic extractants via solvent extraction.
The overall process presents an efficient and successful approach for recovering Nd and Pr from NdFeB magnet scrap. The findings hold substantial implications for the sustainable management of REEs, critical components in numerous modern technologies. Additional investigations are required to optimize the separation and recovery of Nd and Pr and to explore potential applications of their products across various industrial sectors.
Footnotes
Acknowledgments
The authors acknowledge the support provided by the Center for International Research Collaborations of Iran Ministry of Science, Research, and Technology, as well as the Research Council of Iran University of Science and Technology (IUST).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
