Abstract
Elucidating Alzheimer's disease (AD) prodromal symptoms can resolve the outstanding challenge of early diagnosis. Based on intrinsically related substrates of olfaction and spatial navigation, we propose a novel experimental framework for their conjoint study. Artificial intelligence-driven multimodal study combining self-collected olfactory and motion data with available big clinical datasets can potentially promote high-precision early clinical screenings to facilitate timely interventions targeting neurodegenerative progression.
Keywords
Introduction
First described by Alois Alzheimer in 1907, 1 Alzheimer's disease (AD) is a complex, progressive neurodegenerative disease without remission, 2 difficult to differentiate from alternate causes of cognitive decline (e.g., healthy aging or mild cognitive impairment (MCI)). 3 Addressing the increasingly recognized need for accessible preclinical assessments, we present a novel framework for studying olfactory and motion-based navigational decline as proxy indicators of AD neurodegeneration.
The first part of the review outlines correlations between AD-related olfactory, spatial, and motor functional decline, which reflects a unique functional and anatomical relatedness of their brain substrates.4–8 We highlight these structures’ mutual evolutionary origin,5,9 central role in the adult neurogenesis,10–12 and early and severe AD-related neurodegeneration 8 (Figures 1 and 2). The second part outlines olfactory identification (OI), global positioning system (GPS), and accelerometry (ACC) tests—the potential preclinical, accessible AD indicators—in the context of the established AD biomarkers (Figure 3).
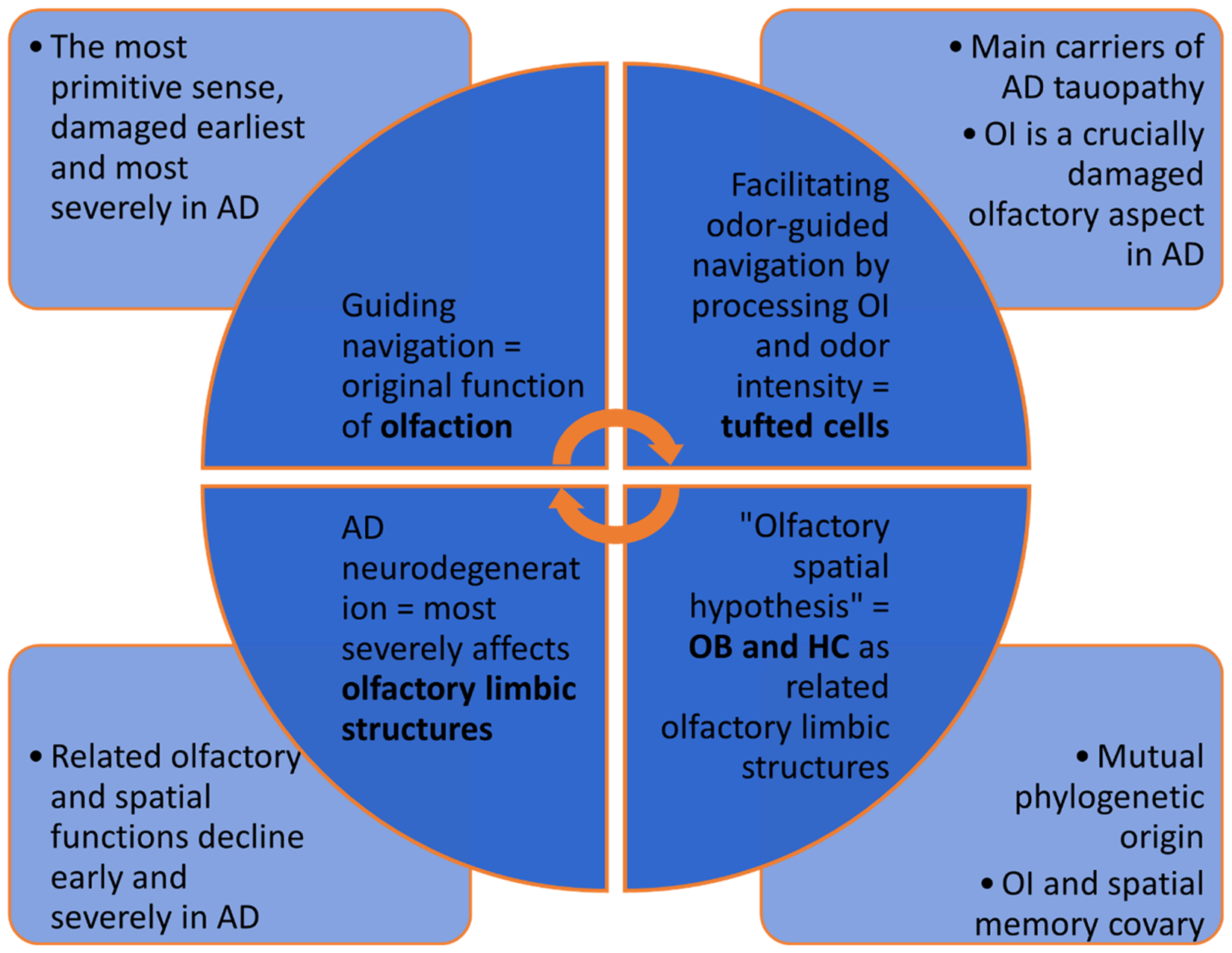
Review-based elaboration of Jacobs’ “olfactory-spatial hypothesis” is the basis of the proposed experimental framework. The evolutionary correlation between spatial and olfactory brain structures, such as OB and HC, and their corresponding functions underlines the proposal for their conjoint studying as early indicators of AD-related neurodegeneration. AD: Alzheimer's disease; HC: hippocampus; OB: olfactory bulb; OI: olfactory identification.
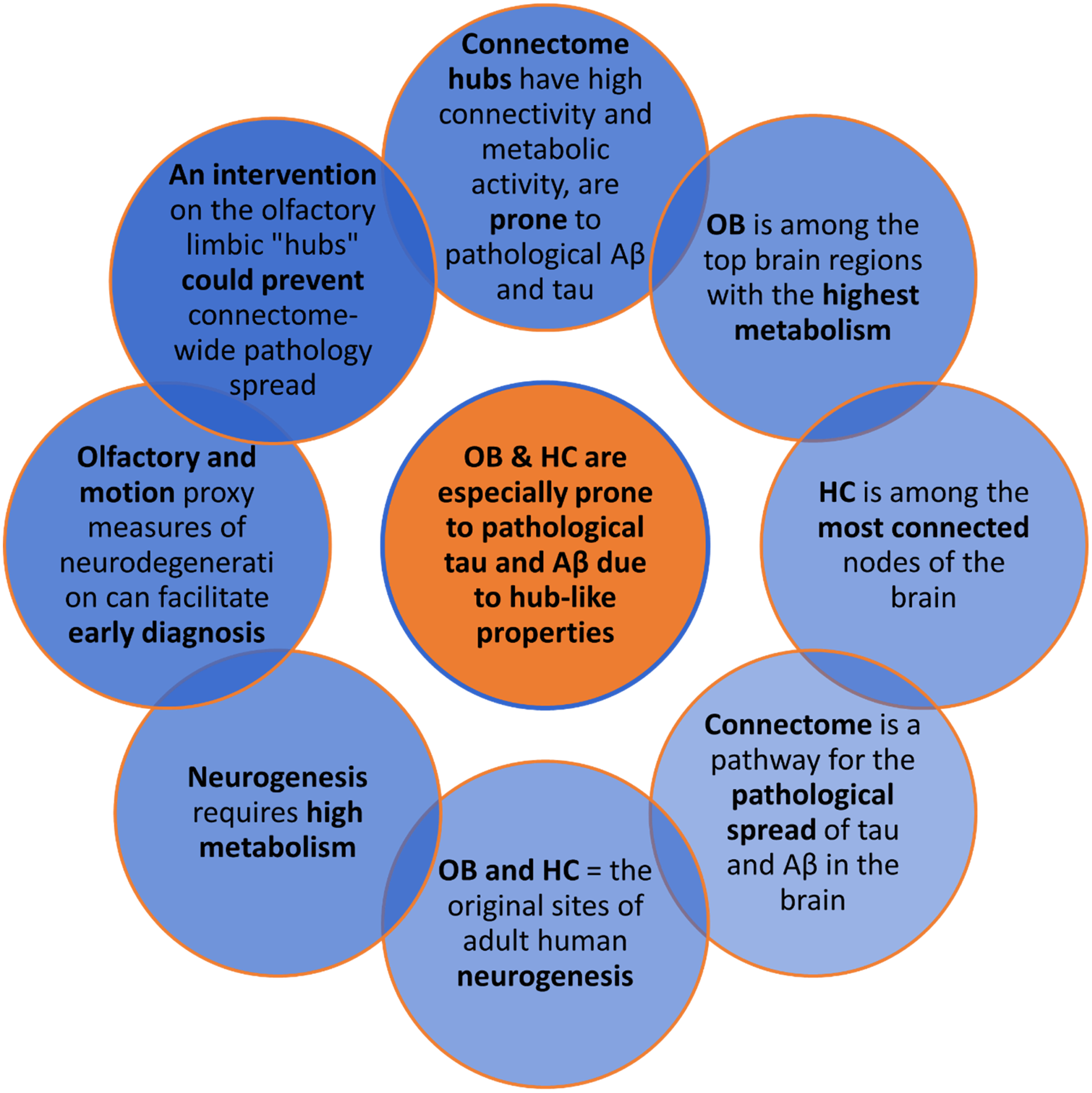
Neurogenesis versus AD neurodegeneration. Olfactory limbic regions are implicated in the two contrasting functions. OB and HC are the olfactory limbic structures prominently involved in adult human neurogenesis and display high metabolism and connectivity. These hub-like characteristics arguably increase their AD neurodegeneration proneness, negatively affecting neurogenesis. Since neurodegeneration spreads across the brain via the connectome, early diagnosis and hypothetical intervention on olfactory limbic “hubs,” such as OB and HC, could prevent further spreading via the connectome. Aβ: amyloid-β; HC: hippocampus; OB: olfactory bulb.
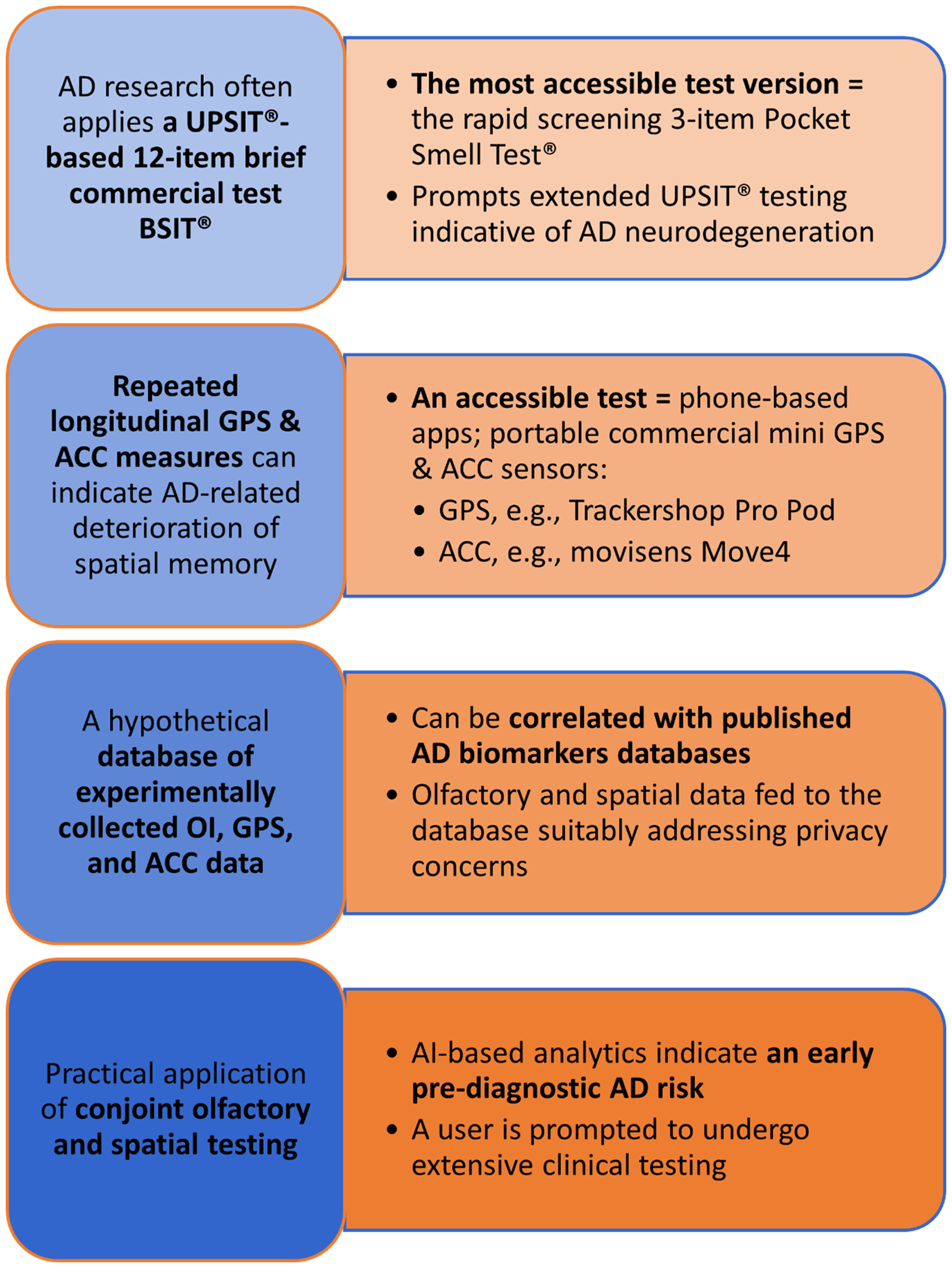
Proposed practical application of the experimental framework for conjoint study of olfactory and spatial disorders. Self-use of commercially available and affordable OI, ACC, and GPS tests facilitates broad, cross-cultural data collection. An AI-driven analytic tool cross-references the data with OI, gait speed, navigation patterns, and biomarker datasets in a privacy-protective manner, indicating potential AD risk. Non-invasive blood-based biomarkers can further strengthen the preclinical indications. Focusing costly, invasive clinical biomarker testing and prospective treatment administration on at-risk candidates could reduce economic costs and improve patients’ quality of life. The early AD diagnosis could prepare patients and their carers for the disease course. ACC: accelerometry; AD: Alzheimer's disease; AI: artificial intelligence; BSIT®: Brief Smell Identification Test®; GPS: Global Positioning System; UPSIT®: University of Pennsylvania Smell Identification TestTM.
Recent multimodal artificial intelligence (AI)-driven tools’ high precision and early detection capability suggest they can effectively inform clinical research and practice: Multimodal models, especially the Transformer, are significantly more precise than the single-modality models in combining different data modalities and sources (e.g., clinical, neuroimaging, genetic, and biochemical biomarkers). 13 We hypothesize that a multimodal AI-driven approach combining self-collected OI, ACC, and GPS data with the big datasets of OI, gait speed, navigational patterns, and clinical biomarkers could effectively indicate prodromal AD. Self-use OI tests14–16 and wearable digital sensors,17–19 combined with emerging non-invasive blood-based biomarkers,20–25 can potentially become widely accessible, preclinical AD screening tools.
Methods
Upon introducing key AD notions, we establish the framework's functional-anatomical basis: We give a sense of the olfactory limbic system's broad multifunctional implications (focusing on the interlaced olfactory, motor, and spatial functions) and highlight the evolutionary perspective on olfactory and navigational functional relatedness and high metabolic expense of the adult neurogenesis in the key olfactory and spatial brain structures, a potentially amplifying factor in their susceptibility to AD pathology. Next, we define a methodology based on AD studies that applied commercial OI tests and ecologically valid GPS or ACC measures.
We base the novel experimental framework on the hypothesis that the combined or interchanged OI, GPS, and ACC measurements and blood-based biomarkers could indicate AD risk upon cross-referencing the OI, gait speed, navigational patterns, and biomarker datasets within an open-access AI platform. After discussing the framework's limitations and external validity, we conclude that it could facilitate timely interventions to improve at-risk individuals and their carers’ quality of life and reduce economic costs. 26
Alzheimer's disease as the framework context
Several key AD facts outline the framework's context: The rapidly growing aging population and a significant AD frequency increase27,28 affect society at the individual and family levels and the country-wide economies in terms of decreased quality of life and high economic cost, including the commonly unpaid caregiving by family or other caregivers (see report). 26 Interventions concerning risk and protective etiologic factors, early detection, and patient care and therapy28–30 (e.g., the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability – FINGER program 31 and PREVENT Dementia Study) 32 can help postpone dementia onset.
Following criticism of the term “dementia” (i.e., “absence of mind” in the translation of the original Latin word), deemed as pejorative, stigmatizing, and insufficiently indicative of its heterogeneity, the American Psychiatric Association included the term in the broader category of neurocognitive disorders (NCD), proposing its further use in parenthesis33,34 (we will specifically refer to the dementia aspect of the broader term “major NCD,” using the general term “NCD” for conciseness unless specified otherwise).
As “the most common neurodegenerative disorder,” 23 AD is a significant cause of NCDs 35 (major and mild NCDs due to AD are typically amnestic, with impaired memory and learning abilities, 35 behavioral (neuropsychiatric) symptoms mostly precede cognitive symptoms;36–38 mild NCD criteria overlap with MCI,35,39 episodic memory impairment and preserved functional independence characterize the amnestic MCI (aMCI)). 39 Although age is a considerable risk factor, AD is an “age-related” rather than an “age-dependent” disease. 2 Most cases are sporadic or inherited non-Mendelian (with a late onset, ≥ 65 years); the rest are familial, with an earlier-onset Mendelian transmission.23,40,41
Much is known about AD pathological staging 42 in the olfactory limbic brain structures, whose multifunctionality 43 and interconnectivity7,44,45 imply a wide-scale functional decline. Our review emphasizes the relevance of studying these regions’ corresponding functions that consequently decline early in the disease.46–49 To substantiate this further, we outline these structures’ interconnectivity 44 (including an evolutionary perspective)5,7 and roles in metabolically expensive neurogenesis11,12 (potentially relevant factors in early AD diagnostics and prevention due to connectome-wide pathology spread50,51 and the possible predisposition of high-activity/connectivity connectome hubs to AD neuropathology).52–54
Olfactory limbic structures and multifunctional implications of their early Alzheimer's disease neuropathology
An outline of the olfactory limbic system
An early notion of the olfactory cortex as “a non-specific activator for all cortical activities” 55 resonates with modern findings of the human olfactory system's early-processing cortical targets and a unique parallel organization. 56 The primary olfactory cortex (POC) comprises the “secondary olfactory structures” that directly connect with the olfactory bulb (OB), circumventing the thalamus,56–58 and project to the tertiary olfactory structures44,45,57,58 (see centripetal projections from OB and centrifugal and associational projections from secondary olfactory structures). 44
The functionally multifaceted “olfactory limbic system”5,43 (we use this term synonymously with the “olfactory system” to denote the limbic system's “olfactory components” 43 relative to the “olfactory spatial hypothesis”) 5 comprises structures of the limbic system (see the limbic system functional-anatomical organization and the definition history) 45 —“the anatomic substrate of memory, emotion, and learning” as the functions that are “central to the clinical expression of AD”. 59 The amygdala and the posterior orbitofrontal cortex (OFC) are the olfactory limbic structures associated with the hedonic aspect of olfactory processing44,60,61 (which conditions survival behaviors relative to odor sources62,63 and was suggested as the key olfactory function); 64 the basal ganglia loops (the “parallel cortico-subcortical reentrant circuits”) comprise the limbic structures severely degenerating in AD and exemplify the motor, cognitive-executive, and emotional-motivational overlap; 45 the medial temporal lobe 65 facilitates olfaction, 8 gait speed as a mobility measure, 66 and spatial navigation and memory67,68 (the most severe AD pathology in the hippocampus (HC) and temporal lobe 69 possibly underlines these functions’ decline).8,18,47,70–75
This brief overview of the olfactory limbic system implies multifunctional outcomes of its AD neurodegeneration. The following sections detail some of its structures and their roles in olfactory-guided navigation, including the high-level concept of olfactory-spatial cognitive maps.
Olfactory limbic structures in olfactory, motor, and spatial processing
Olfactory and spatial structures have a mutual phylogenetic origin 9 and have remained closely evolutionarily related:4,5,7 the olfactory cortex, HC, and entorhinal cortex (ERC) form the allocortex, the phylogenetically oldest cerebral cortex part;4,9 the direct connection between the human POC and the HC cortical network (preserved in humans through evolution) remains analog to the rodent network organization instead of “rerouting” to include the association cortices before reaching the HC (as the other human sensory systems). 7 This similarity arguably justifies studying rodent models. 7 The OB and HC cross-species size covariation,5,43 olfactory-spatial mapping in the piriform cortex 4 and HC 76 (predominantly olfactory and spatial structures, respectively), and OI and spatial memory's common neural substrates (medial OFC and HC), 6 additionally suggest an “intrinsic association” between olfaction and spatial navigation, 6 conceptualized as the “olfactory spatial hypothesis” of navigation as the original olfactory function 5 (Figure 1).
Tufted cells (the leading carriers of AD-related OB tauopathy) 77 are critical to OI and odor intensity processing, 78 which suggests their relevance to navigation (odor intensity changes indicate the distance to an odor source).78–80 Being predominantly related to motor function and associated with the olfactory limbic system44,81–83 (including sniffing regulation 83 and olfactory discrimination and memory), 82 the cerebellum was implicated in various aspects of spatial navigation, 84 including facilitating the HC spatial representation via the processing of self-motion signals in path integration (a navigation aspect).84,85 The cerebellum and basal ganglia (associated with the olfactory limbic system) facilitate motion elements, such as gait (see in reviews);17,86,87 the cerebellum's and OFC's connection to the POC44,83 possibly underlines a correlation between gait and olfactory function.81,88 Cerebellum might also relate to impaired neurobehavioral modulation. 89 Being strongly functionally connected to the HC 7 and subject to AD-related neuropathology (see, e.g., in a meta-analysis comment), 90 the olfactory tubercle (a multifunctional structure receiving direct olfactory input from the OB44,63,91,92 and associated with odor-motivated behaviors 93 and the basal ganglia)45,94 is a site of multifunctional “convergence” of the olfactory and the limbic-motor inputs. 95 HC (an olfactory limbic structure vital to the OI, 96 adult neurogenesis, 97 learning and memory, 98 and cognitive maps,68,99,100 and critically impaired in AD) 42 has a key role in the medial temporal lobe's automatic processing of internal spatial representations during self-motion (see review). 68
In addition to being considered a biomarker for conversion from MCI to AD, 101 neurodegeneration of HC and ERC (“AD signature regions”) is associated with declining olfaction, mobility, gait, cognition, and spatial navigation:8,70–72,102 impaired path integration ability (a function integrating vestibular, motor, and proprioceptive information that dynamically interacts with visual information to facilitate navigation, see in reviews)67,103 reflects early ERC degeneration and the related disrupted grid cell functioning, a potential biomarker of MCI-to-AD transition;102,104–107 an HC-ERC coherent neuronal firing pattern circuit evolves during the encoding and retrieval of olfactory-spatial associative information; 108 the OB-ERC-dorsal HC circuit is essential to recognition memory (impaired in prodromal AD) and its altered activity is potentially a better early AD biomarker than the volumetric or architectonic brain changes. 109 Besides interactive cognitive mapping between OFC and HC, 110 critical involvement of the medial OFC and the HC in both OI and spatial memory suggests that olfaction initially mediated spatial memory and cognitive maps - 6 the focus of the next section.
Cognitive maps in olfactory-guided navigation
Spatial representations are generated bilaterally in olfactory, visual, and auditory modalities (e.g., birhinal odorant localization, binocular visual depth cues, and binaural sound source localization),111–115 but olfactory chemical cues (effectively circumventing thalamic transmission on the way to POC)56–58 may be the “original stimuli”, universal to all organisms.5,116 The main olfactory functions include OI and spatial localization of odorant sources. 114 A positive correlation between an odorant's concentration and proximity to its source can optimize behavior 117 (different species’ odor-based navigation strategies include a detailed use of an odor plume's temporal structure and the olfactory spatial maps).5,79,118,119 Although humans likely do not intuitively perceive olfaction in the spatial context 111 (disproportionately less research on olfactory-guided navigation in humans versus other species has reflected this so far), 118 they are aware of their odor-based navigation ability. 120 Beyond olfactory-guided navigation in a controlled laboratory environment, 120 humans can track scents outdoors 114 (from the neuroimaging perspective, the greater thickness of medial OFC positively correlates to better OI and fewer errors during the HC-based spatial learning strategy in humans). 6
Odors were suggested as a component of multisensory cognitive spatial maps across species. 118 This aligns with Tolman's concept 121 (“a multi-dimensional map of the statistical relations between environmental elements“), 122 which arguably spans beyond spatial representations 110 to abstract domains (e.g., social). 122 Animal model studies elucidated olfactory limbic structures’ processing of olfactory-spatial cognitive maps and novel research prospects are emerging from human studies: Neural processing of odors is spatially organized by the odorotopic mapping in the mammalian brain (the pattern's spatial map-like arrangement reflects the association between a multi-odorant combination representing each odor and an olfactory epithelium and OB activity pattern);58,62,123,124 the HC generates olfactory-based spatial representations 76 and piriform cortex facilitates odor-spatial cognitive maps in rats; 4 unique ERC grid-like codes facilitate mental navigation of odiferous environments and the olfactory cognitive maps in humans. 117
In a mouse model of olfactory navigation, odors can function as spatial navigation landmarks, enable their future representations, and iteratively interact with path integration to facilitate the olfactory-spatial cognitive maps in the HC;125,126 in the human virtual olfactory domain, temporally stable odors can function as spatially stable landmarks (within cognitive maps supported by the ERC and the anterior piriform cortex grid-like codes); 127 of note, in the human virtual visual domain, allothetic landmarks combine or compete with idiothetic path integration in estimating the heading direction (measured as a turning angle). 128 While memory is essential to navigation 67 and OI, 96 future predictions are no less critical:5,129 Olfaction is “fundamentally a predictive sense.” 117 Cognitive maps encompass OFC and HC, 110 the structures central to olfaction, spatial navigation, 6 and goal-directed tasks. 129 HC facilitates context-dependent, goal-oriented representations 130 and imagination of hypothetical scenarios, 131 while the OFC was suggested as “a cognitive map of task state.”110,132
Alzheimer's disease neuropathology of the olfactory limbic system
AD-related neuropathology 42 reflects the reviewed intrinsic connectivity of the olfactory limbic regions. Pathological amyloid-β (Aβ) and tubulin-associated unit (tau) (AD “hallmarks”)2,51,133–135 affect the olfactory limbic areas59,77,136–141 earlier and significantly more severely than the primary visual and auditory areas.138,142,143 The intracellular tau protein abnormally accumulates in AD 144 according to the six Braak stages of distribution. 42 In contrast, the extracellular Aβ plaques are insufficiently indicative of the neuropathological stages due to within-brain and between-individual variations. 42 The early OB degeneration due to tauopathy precedes Aβ accumulation, 143 which correlates to the Thal and Braak stages 135 (also see somatic mutations driving OB pathology145,146 and OB neuroproteomics). 147
Early AD olfactory decline (in Braak stages 0 or 1)138,143 may relate to tau accumulation in HC and ERC 148 (HC and ERC activity 96 and left HC atrophy 73 relate to OI), the OB (containing the leading cellular tau tangle carriers), 77 and the anterior olfactory nucleus.77,139,140,143 Aβ alters olfactory structures’ activity and functional connectivity.149,150 Olfactory decline factors include reduced levels of choline acetyltransferase and dopamine in the olfactory tubercle and other brain regions,90,141,151 such as locus coeruleus (the first AD tauopathy subcortical site),152,153 which densely innervates the olfactory tubercle91,154,155 and supplies norepinephrine to olfactory structures. 151
The causality between AD pathology and olfactory decline 46 is highlighted by familial AD patients’ OB and the olfactory tract viral infection (the herpes simplex virus type-1 or varicella zoster virus possibly initiates or accelerates AD), 156 gray-matter atrophy in pathology-free anosmic patients (which may suggest various causality directions between AD neurodegeneration, brain atrophy, and olfactory loss), 157 and olfactory recovery. While cell renewal in the olfactory epithelium or OB may repair the postinfectious olfactory loss,158–161 damage to these structures160,162 may impair adult neurogenesis—the focus of the next section.
Neurogenesis and neurodegeneration in the olfactory bulb and the hippocampus as prominent olfactory limbic structures
Adult mammalian neurogenesis associates OB and HC (see review). 10 This highly-metabolic function,46,163 a potentially exacerbating factor of neurodegeneration (Figure 2),50,53,164–170 originates in the subventricular zone of the lateral ventricles and the subgranular zone of the HC dentate gyrus, where the progenitors are produced, migrating further to the OB and the HC and becoming the OB granule and periglomerular neurons and the HC dentate granule cells, respectively. 171 Most newly generated OB neurons possibly originate in the basal neural stem cell type in the basal ventricular-subventricular zone; 172 the adult human olfactory epithelium (provider of the olfactory information to the OB) is a site of robust neurogenesis 173 and subject to AD-related neuropathology.174,175
Adult human HC neurogenesis97,176 continues both through physiological and pathological aging, with severe impairments since the earliest stages of AD. 177 It directly relates to memory loss in AD (via positive effects of neurogenesis augmentation on the restoration of memory and vice versa, via adverse effects of the inactivation of the immature neurons in AD on diminishing memory). 178 OB and HC neurogenesis is implicated in animal olfactory-guided social behavior, see reviews,179,180 and pattern separation (discriminating “between similar contexts and similar odors”). 181 Adult OB neurogenesis positively affects difficult discrimination learning of odors and olfactory memory; 182 conversely, odor discrimination and learning increase the survival of new OB neurons. 183 Motor activity boosts HC neurogenesis184,185 and spatial navigation; 186 conversely, reduced HC neurogenesis impairs spatial relational memory (an “allocentric mapping strategy” for navigating under novel conditions). 12
Future studies in bats (the species comparable to humans due to their longevity and low HC neurogenesis rate) 187 may inform research on human navigation relative to neurogenesis in the cases of sensory deprivation (see, e.g., rats’ use of spatial olfactory cues without audiovisual cues), 76 including AD-related olfactory loss: According to the “olfactory spatial hypothesis,” 5 microbats’ replacing of the “olfactory spatial” system with auditory spatial detection may explain their low or absent HC neurogenesis rates 187 while megabats’ reliance on olfactory predictions could explain persistent HC neurogenesis 188 and a positive correlation between OB mass and wing area 189 (also see in review). 5
The critical role of OB and HC in adult human neurogenesis emphasizes their high metabolism. This hub-like property potentially exacerbates AD neurodegeneration, which spreads via connectome, as outlined further.
The connectome role in Alzheimer's disease pathological spread
The connectome hubs support “integrative information processing and adaptive behavior”.52,165,190–192 They are distinct by the highest neuronal activity in the brain, 166 longer-distance connections, and higher metabolic requirements (in terms of topological value and biological cost tradeoff52,164 and increased connectivity). 165 High metabolism is a possible factor in increased proneness to AD pathology,52,53,164–166,169,170,193 which precedes other brain regions (in the case of Aβ); 50 high hub connectivity is an alternate factor (pathological tau accumulation correlates with a node's connectivity strength and the cortex volume which it connects to). 54 Both in healthy aging and AD, the tau tangles spread across the connecting fibers of the brain138,142 (the connectome), accelerated by Aβ.50,51 Various hypotheses suggest that the olfactory system is a pathological seeding and spreading hub.194–197
Olfactory and motion tests in the context of Alzheimer's disease biomarkers
The first part of the review suggests an intricate relatedness of olfactory, motor, and spatial functions in the AD context (Figures 1 and 2). The second part outlines the corresponding optimal testing methods in conjunction with the established AD biomarkers (Figure 3).
Neuroimaging and biomarker research has focused on defining the early AD pathophysiology, e.g., neurodegeneration, neuroinflammation, and synaptic dysfunction, via minimally invasive methodologies that could prompt potential therapies. 198 Integrating the biochemical and imaging biomarkers with cognitive and behavioral assessments could enhance accuracy and accessibility in predicting AD.47,133 Biomarkers including genetic disposition (e.g., apolipoprotein E (APOE) ε4 genotype), Aβ, phosphorylated tau, neurofilament light chain, glial fibrillary acidic protein (GFAP), and inflammatory factors, are measured in cerebrospinal fluid (CSF), blood or brain matter by genotyping, the Pittsburgh compound B for positron emission tomography (PET) (for example), magnetic resonance imaging (MRI), and immunochemical assays for early disease detection.24,199 Easily applicable, affordable tests have been frequently called for, e.g.,21,137,200–202 in this sense, serial olfactory testing has been proposed as “a pragmatic, low-cost alternative to invasive AD biomarkers” (the olfactory decline rate was reported as “the second strongest predictor” of incident MCI or NCD after the genetic risk factor APOE ε4); 203 GPS and ACC wearable sensors can capture longitudinal motion data in ecologically valid settings as a measure of declining motor and spatial functions in AD, in review; 17 advances in AI research suggest a prospective application of multimodal analyses in AD diagnostics.13,204 We briefly review the AD biomarkers commonly applied with OI testing and introduce the blood-based biomarkers (as prospective proxy indicators of AD neuropathology) 21 to provide the clinical context for the framework.
Postmortem morphological brain studies
Alzheimer made the first observations of the Aβ plaques, the tau neurofibrillary tangles, and the cellular loss in the context of dementia in the early twentieth century by using Bielschowsky's silver staining method in the postmortem brain analysis. 1 Starting with the earliest subsequent postmortem analyses of AD patients’ brains, which demonstrated that AD neurodegeneration affects the olfactory brain areas,77,139,140,143 extensive studies of these neurodegenerative changes further expanded Alzheimer's research throughout the century, e.g.42,69,205 A 1980s pioneering histological brain analysis study reported that AD-related tau tangles most severely affected the HC and the temporal lobe cortex. 69 A study in line with Braak staging 42 attributed olfactory dysfunction in AD to pathological tau neurofibrillary tangles in the OB, anterior olfactory nucleus, periamygdaloid cortex, and amygdala. 143 The impaired OI was correlated with the AD-related tau tangles in HC and ERC. 148
In vivo positron emission tomography imaging of amyloid-β and tubulin-associated unit deposits
The relatively recent application of PET has facilitated studies in living AD patients: PET enables spatial quantification of the pathological protein deposits in the brain long before the manifestation of the clinical symptoms, facilitating in vivo monitoring of the disease progression and increasing diagnostic accuracy.35,51,133,206–210 A recent study applied a subnanomolar PET tau tracer to demonstrate the longitudinal accumulation of tau according to Braak stages. 207 Other studies applied the epidemic spreading model to PET scans, suggesting an Aβ and tau spread via the connectome (neuronal communication pathways).50,51 Dynamic Aβ-tau interactions may explain accelerated pathological tau spread from the medial temporal lobe into the isocortex. 51 Longitudinal PET imaging combined with OI testing with the initially cognitively normal (CN) older adults demonstrated associations between declining olfactory function and Aβ and tau accumulation in the olfactory regions, prompting further studies of olfaction as a potential predictor of AD neurodegeneration. 209
Cerebrospinal fluid
A study evaluating neuroimaging and clinical biomarkers longitudinally and cross-sectionally with CN, MCI, and AD subjects confirmed the predicted 12-month progression rate of the MCI and reliability of the CSF as a predictor of the 12-month progression; 211 a cross-sectional in vivo study established a correlation between the CSF total tau and phosphorylated tau to Aβ in CN adults at risk due to family history and OI dysfunction. 201
Magnetic resonance imaging of olfactory limbic regions in Alzheimer's disease
MRI scanning successfully detected volume changes in the olfactory regions affected by early AD tauopathy (e.g., the HC, ERC, and locus coeruleus).74,101,212–215 Volume reduction of the HC and the ERC has been considered an early marker of AD pathology (see review) 216 and a clear differentiating marker of MCI and AD,213,215 predictive of MCI-to-AD conversion. 101 The ERC (a “gateway” for the sequential transmission of olfactory input to the HC, upon its transmission from the olfactory receptors to the OB and the POC)216,217 deteriorates severely during NCD due to AD and less severely in normal aging.218,219 Due to the greater AD-related volume reduction of ERC than HC, ERC was a better means of distinction between MCI and AD. 213 The contrasting findings reported a greater HC than ERC volume loss in AD versus MCI patients and implied that the ERC measurements better differentiated MCI from CN subjects. 215
In healthy female participants, specific HC and ERC activations accompanied correct spontaneous OI; 96 in CN participants, impaired OI was associated with lower volumes of the brain areas typically implied in AD, including the ERC and HC 75 (the latter's size reduction indicates the loss of OI ability rather than memory loss). 74 OI function correlated with the ERC and HC atrophy both retrospectively and prospectively relative to a performed OI test, as well as other temporal and frontal gray matter areas and, prospectively, the white matter temporal areas 72 (more fibrous connections in the ERC in MCI versus CN participants indicated a compensatory olfactory mechanism). 220
Apolipoprotein E ε4
Multiple candidate gene association studies cross-validated strong associations of AD with the ε4 allele of the polymorphic gene encoding the APOE. Being expressed in the liver, blood, and central nervous system and having a role in multiple metabolic processes and inflammation (see review), 23 it has been considered the most significant genetic risk factor for late-onset AD (see reviews).221,222 Recent research on APOE ε4 microglia reported that APOE ε4 induces accumulation of lipids, causing AD-typical neuroinflammation (the decreased metabolism of lipids disrupts coordination of the neuronal ensembles). 223 The first genome-wide association meta-analysis study of the brain changes throughout a lifespan determined the role of APOE gene mutation rs429358 in the age-related volume reductions of the HC and the amygdala 224 (the two limbic structures significant for olfactory processing). 44 The first documented findings demonstrated significantly lower olfactory sensitivity in MCI APOE ε4 carriers than in non-carriers. 225 OI decline rate was doubled in middle-aged APOE ε4/4 homozygotes carriers (compared to non-carriers and heterozygotes). 226 Comparisons with age-related changes in cognitive performance suggested that olfaction can predict cognitive decline. 226
Blood-based biomarkers
Blood-based biomarkers, such as plasma neurofilament light chain, phosphorylated tau, and Aβ,21,25 activity-dependent neuroprotective protein (ADNP),22,145,227–229 and GFAP24,230 (whose expressing progenitors are the principal source of constitutive neurogenesis in the adult mouse forebrain), 231 are promising, non-invasive AD biomarkers; see reviews20,25 and proposed guidelines. 21
Olfactory identification testing in correlation with established Alzheimer's disease biomarkers and cognitive tests
The first systematic study of OI across the entire age span established impairment in more than half of participants above 65, correlating the olfactory dysfunction to normal aging.232,233 These findings aligned with previously reported age-related deterioration in visual and auditory sensory functions (interpreted relative to a view of the senses as “interactive systems for extracting environmental information”).232,234 Olfaction is the first sensory function to deteriorate in AD (neurodegeneration of the central, rather than the peripheral olfactory system, arguably contributes to its impairment). 235 The deteriorating olfactory function in AD refers to the sensory, perceptual, and cognitive levels of the sense of olfaction, including odor threshold detection, odor memory, and OI.90,208,236 OI comprises detecting and recognizing an odor and recalling the associated objects, sources, and names 137 and implicates semantic and episodic memory. 96 Stronger correlations between the episodic and semantic olfactory memory components (measured as odor recognition and OI) than between the olfactory and episodic and semantic memory suggest that olfactory memory is a category distinct from visual and auditory memory. 237
AD studies frequently measured OI as a profoundly impaired function in a majority of patients (with deficits manifesting in the early stages of cognitive impairment, 236 beginning earlier than cognitive decline, 220 and predicting future cognitive decline).137,238 An early study suggested olfactory function decline as “an additional criterion on which to base a diagnosis of dementia.” 239 Olfactory limbic system neurodegeneration and OI deterioration have been considered as early AD markers, e.g.,16,46,72,75,137,138,143,174,200–203,220,235,236,240–249 (as well as a marker for other neurodegenerative diseases, e.g., Parkinson's disease, Down's syndrome, and multiple sclerosis, and schizophrenia).3,46,90,96,174,195,250–253
The OI assessment corresponded to other biomarkers in most studies, e.g.,73,137,148,201,203,254 with few contrasting exceptions, e.g.,208,255 and indicated a transition from a healthy state to MCI and AD and from the aMCI to AD.203,240,248,256 Being superior to memory as a predictor of cognitive decline 220 and occurring before AD-related memory loss, OI testing has been considered a non-invasive and inexpensive, non-clinical biomarking tool for early assessment of MCI or AD onset,137,220,257 with the strength of its predictability second to APOE ε4 as the most reliable known genetic predictor of AD. 203 Due to these qualities, the decline of olfactory function has been suggested as the most promising non-invasive, early biomarker of AD, acting as a practical proxy for AD-related neurodegeneration in the brain137,203 (olfactory decline can indicate the MCI and the pathological tau or Aβ levels). 209
A study on odor detection thresholds, quality discrimination, and OI with the elderly population demonstrated impaired olfaction in all three domains in MCI patients (controlling for cognitive deficit) and significantly worse olfactory discrimination and OI in AD patients (as well as a higher correlation of odor discrimination and OI than the odor detection thresholds with the neuropsychological tests). 258 An earlier meta-analysis of olfactory deficits in AD and Parkinson's disease patients showed extremely large effect sizes between the patients and controls in three olfactory domains (olfactory detection, OI, and recognition), although there were no differences between the AD and Parkinson's disease groups nor between the olfactory domains. 90 A more recent meta-analysis revealed that OI is significantly more impaired than olfactory detection in AD patients (this possibly results from the impairment of the high-level cognitive functions related to OI). 259 Olfactory discrimination testing is a distinct indicator of CN, MCI due to AD, and mild NCD due to AD; 260 nevertheless, most AD studies of olfaction have tested OI ability (despite its potential proneness to personal or cultural bias).202,260
Olfactory test administration is necessary since AD patients may be unaware of impaired olfactory function.247,261–264 Rapidly administered OI tests have been widely applied as an objective assessment of olfactory function (at clinics, in studies, and outside of a laboratory environment), suitable for self-use and indicating the need for further olfactory or cognitive testing. 15 The demonstrated diagnostic quality of OI testing supported the initiative for its clinical use in the early diagnosis of AD. 202 Widely applied commercially available tests 137 include the scratch-and-sniff Sensonics’ University of Pennsylvania Smell Identification TestTM (UPSIT®)14,15 and its shorter version, Brief Smell Identification Test® (BSIT®) (with Version A being indicative of an increased risk of developing AD) 16 and the felt-tip pens-based ODOFINTM Sniffin’ Sticks Smell Identification Test (SSSIT).265,266
UPSIT® and a derived 10-item scale strongly predicted conversion from MCI to AD; 16 the subsequent combination of UPSIT® with MRI of ERC and HC volumes, a neuropsychological, and a social measure yielded a strong prediction of MCI-AD conversion, superior to the combination of age and Mini-Mental State Exam and exceeding the consensus sensitivity and specificity threshold criteria for AD markers (>80%).244,267,268 As measured by UPSIT® cross-culturally, OI impairment was a better predictor of AD than a verbal episodic memory test with the initially CN participants. 246 Lower scores on the cross-cultural version of BSIT® 269 were indicative of a higher NCD risk over 12 years in a biracial cohort, with a greater effect observed with the white study population. 245
A pilot study with healthy, aMCI, and AD participants used both BSIT® and SSSIT, demonstrating that the BSIT® scores indicated aMCI-to-AD conversion. 74 Another study applied the BSIT® (Version A) OI test and a range of cognitive assessments and found an association between the decreasing OI and the increased risk of aMCI but not of the non-aMCI: In line with the previously mentioned study, BSIT® scores predicted the progression of aMCI to NCD due to AD. 240 Recent longitudinal data analyses correlated a one-unit BSIT® score increase with ∼14% reduced risk of CN-to-MCI transition and ∼11% risk of MCI-to-NCD transition, as well as increased prospects for MCI-to-CN reversal (several covariates had detrimental effects on the cognitive state, such as APOE ε4, which increased these risks and decreased prospects for MCI reversal). 242
Following the observed correlations between the impaired OI ability and the MCI 270 and postmortem AD pathology (tau tangles-OI inverse relation was robust in the ERC and CA1/subiculum area of the HC), 148 a longitudinal study correlated lower BSIT® scores with a higher rate of episodic memory loss and MCI risk, and with postmortem AD pathology in the individuals who exhibited no clinical manifestations of AD or MCI in vivo: These results suggested that olfactory dysfunction can indicate AD pathology in asymptomatic people and assess the risk of developing prodromal AD symptoms. 200 An extended study (involving longer follow-up time and MRI scans of CN participants) associated OI decline with faster global cognitive decline and lower volumes of “AD signature areas”, such as the HC, ERC, middle temporal cortex, and fusiform gyrus, regardless of the APOE ε4 status. 75 More recently, a study combining the serial BSIT® olfactory testing (at ≥3 annual intervals during periods of normal cognition) with cognitive testing, medical histories, and MRI demonstrated a correlation between the rapid olfactory function deterioration and the subsequent MCI and AD incidence: The predictability of the olfactory decline rate was analog to that of the APOE ε4 allele (the best known AD genetic risk factor), indicating that further serial olfactory measurements could establish correlations with the progressing neuropathology. 203
OI reportedly increasingly declines with age.262,271–273 SSSIT-16, in combination with demographic, genetic, health, mobility, memory, verbal, neuropsychiatric, and behavioral measures, established correlations with the deteriorating OI in an initially CN aging population.237,274,275 Within the same population-based study series, OI impairment and APOE ε4 individually doubled the risk of NCD, while their combination quadrupled it. 264
Each item with a lower score on the SSSIT-16 OI test corresponded to a higher (22%) risk of developing MCI and an increased Aβ in the OFC and tau in the ERC (measured longitudinally by in vivo PET). 209 Subsequent retrospective and prospective MRI and neuropsychological data analyses (relative to the first performed OI test) associated better OI performance with slower rates of brain atrophy in AD signature regions and slower decline of neuropsychological and motor functions; 72 earlier analyses of a CN subsample yielded strong correlations between OI and motor function. 81
Applying SSSIT with cognitive measures demonstrated significant differences and progressive worsening of OI function between CN, subjective cognitive decline, MCI, and AD subjects. OI was associated the most with memory (particularly immediate verbal recall) across subjects and positively correlated with global cognition in AD and MCI. 276 Structural equation modeling revealed associations between SSSIT and cognition relative to affective and psychosis symptoms in AD and MCI patients. 277
Global positioning system and accelerometry measurements of spatial navigation and motion
Previous studies demonstrated functional and anatomical overlaps between olfaction, movement, spatial navigation, and spatial memory, e.g.6,81,84 The sensory and motor decline can precede the cognitive manifestations of AD and indicate an increased risk.278,279 Declining gait pace differentiated between CN, MCI, and AD subjects, 280 and predicted cognitive decline 279 (longitudinal studies suggested it as a reliable predictor of NCD coupled with cognitive decline48,49 and olfactory loss).81,281 Its correlation with postmortem AD pathology can indicate the risk of developing the motoric cognitive risk syndrome (conjunction of slow gait and subjective cognitive complaint was associated with AD and other NCD risks). 88
Motion (in the sense of locomotion) is intrinsic to spatial navigation.282,283 Self-motion clues include “vestibular and proprioceptive signals, motor efference copies, and optic flow” with specialized neural substrates 100 that are reflected in spatial memory 284 and its AD-related decline.47,285–287 The missing vestibular and proprioceptive signals due to lack of motion in virtual reality (VR) studies (see reviews)68,100 and a positive effect of locomotion on episodic memory in augmented reality 288 exemplify this. Nevertheless, VR (both immersive and desktop versions) could facilitate spatial learning (although it is surpassed by real-world spatial learning) 289 and predict real-world spatial disorientation in AD patients (provided suitable task design), 290 while immersive VR (including locomotion) 104 revealed overestimated angular turns (a path integration component) as a prospective AD risk marker.102,105 VR is a promising research avenue for olfactory-guided spatial navigation and memory: Delivering odorized air to participants inside the functional MRI (fMRI) during odor-based VR spatial navigation demonstrated the human ability to navigate based on the relative positioning of the odors. 127
While a stopwatch 291 or an electronic walkway71,292 are standard measures of gait speed in a controlled setting, wearable sensors and smartphone apps offer unprecedented opportunities for longitudinal measures of navigation and gait as non-invasive, passive biomarkers17,19,292–294 (e.g., in terms of AD-specific patterns of navigation, 18 motion, 19 and life space). 295 Given the essential role of motor function in spatial navigation,84,85,125,282,283,296–299 movement measurements could define AD-related spatial cognition decline.47,286,300–302 The amounts of outdoor and indoor (measured in terms of mobility within the levels of the “life space” concept) 303 indicate health and quality of life293,304 and can differentiate between AD patients and CN controls. 295 We review GPS and ACC studies of AD-related changing movement patterns in everyday settings.
Ecologically valid navigation and motion studies in neurodegenerative diseases are scarce: a two-week outdoor study with GPS trackers (Trackershop Pro Pod 5)18,305 discriminated AD patients’ specific navigation patterns (lower segment entropy, higher segment similarity, and moving less far from home) using machine learning. 18 Two circadian cycles-long (48 h) study with continuous ACC recording (ankle-mounted Shimmer accelerometers) discriminated AD and CN subjects’ motion patterns at the group and individual levels, controlling for lifestyle (the study also determined the best-performing time window and filter). 19 A study of sleep behavior disorder as a Parkinson's disease biomarker monitored gait continuously 306 with a validated ACC sensor (Axivity AX3). 307
Application of an artificial intelligence-driven approach in global positioning system and accelerometry studies
A systematic review of wearable GPS and ACC studies of neurodegenerative diseases suggested AI's effectiveness in longitudinal data analyses (cross-domain transfer, machine learning, or deep learning networks could account for data heterogeneity, sampling rate, and the devices’ sensor biases). 293 A meta-analysis suggested combined machine learning and neuropsychological assessments in automatic MCI classification and AD-conversion prediction 308 (support vector machine algorithm best predicted NCD-risk in aMCI individuals). 309 A prospective aMCI study suggested the extreme gradient boost model (comprising demographic, memory, neuropsychological, and biomarker variables) as an effective individual AD-conversion predictor. 310
Multimodal artificial intelligence-driven approach for Alzheimer's disease prediction
The study first mentioning olfactory dysfunction in the context of “senile dementia” combined the olfactory and gustatory cranial nerves’ impairment with the psychiatric, psychometric, neurophysiologic, cerebral-circulatory, biochemical, and neuropathologic studies within a longitudinal study. 311 More recently, a review of olfactory, auditory, and visual impairments in AD emphasized multisensory assessments as indicators of a sensory function's relative effectiveness as an AD biomarker. 137 Longitudinal multivariable analyses can determine the olfactory decline rate (an AD-risk predictor);202,203,244 its combination with behavioral and biological biomarkers could facilitate early AD diagnosis. 200 AI can technologically facilitate such multimodal big data analyses. An AI-driven multimodal diagnosis model with high classification accuracy identified brain regions associated with AD neurodegeneration. 13 A proposed AI-driven causal model based on large longitudinal datasets could hypothetically facilitate the identification of early AD genetic and cognitive biomarkers. 204
Conclusions of the review and discussion of the novel experimental framework
Based on the reviewed olfactory limbic structures’ relatedness in the context of evolution, neurogenesis, and multifunctional implications of their AD-related neurodegeneration (including olfactory, motor, and spatial functions), we hypothesize that the serial longitudinal OI, ACC, and GPS testing in prodromal AD could reveal these functions’ synchronous decline rates relative to AD neurodegeneration, e.g.17,18,72,102,203,244 Next, we discuss the review findings as the basis for the first experimental framework for the conjoint study of olfaction and motion-based spatial navigation in AD (effectively, an elaboration of Jacobs’ “olfactory spatial hypothesis” in AD context). 5
We expect that the longitudinal olfactory decline (BSIT® scores), spatial navigation (GPS recordings of navigation patterns), and motion (ACC gait) measures will indicate a lower neurogenesis rate in line with correlations between neurogenesis and olfaction,182,183 spatial navigation and memory,5,10,12,186,283 and motor function.184–186 Early AD neurodegeneration in the OB77,143 (a key adult human neurogenesis site)171–173 suggests that impaired adult neurogenesis is a factor in OI and the functionally and anatomically related spatial navigation and memory decline4–6,10 (Figure 2; see links between neurogenesis and AD-related memory loss 178 and olfaction).179,182,183 Early AD neurodegeneration in the HC (the second key adult neurogenesis site, essential to spatial navigation and memory)68,99,283,312 suggests that its impaired neurogenesis is a factor in the early spatial navigation decline; AD-related degeneration of motor regions and the subsequent motor function decline (the most significant enriched-environment booster of the mouse HC neurogenesis184,185 and spatial learning) 186 suggests that impaired HC neurogenesis is a factor in motion decline (an essential navigation component).67,282–284,297
We hypothetically attribute the OB and HC neurodegeneration to their high metabolic requirements and connectivity44,46,138 (the neurodegeneration risk factors associated with connectome hubs).50–54,164–167,169,170 In this context, hubs’ absence from the primary sensory and motor areas 165 could explain the lack of tau neurofibrillary tangles and Aβ plaques in the motor, somatic sensory, and primary visual areas. 138 Connectome-wide neurodegeneration spread51,138 could progressively weaken the hub connectivity due to the tau burden 54 (considering OB as a possible hub for early pathological tau aggregation, in review, 195 and direct functional connectivity between POC and HC). 7 This could result in progressive deterioration of the olfactory and spatial functions (see correlation between tau-driven medial temporal lobe degeneration and allocentric spatial processing; 313 recent high-precision fMRI findings of the medial temporal lobe connectivity 65 may inform future studies of connectome-wide pathological spread).
The newly demonstrated role of the tufted cells–anterior olfactory nucleus loop in OI and odor intensity processing 78 (the essential aspects of olfactory-based navigation, in review) 79 may underline olfactory and navigation decline, in line with the specific proneness of the tufted neurons and the anterior olfactory nucleus (as their “cortical target”) 78 to AD tauopathy 77 and the tufted cells’ projections to the olfactory tubercle91,92 (the structure integrating olfactory and limbic-motor processing). 95 The deteriorating olfactory function due to piriform cortex, HC, and ERC (OI-associated structures) neurodegeneration may affect odor landmarks’ detection.125,127 HC and ERC degeneration may negatively affect path integration102,105–107 and egocentric and allocentric spatial orientation and memory.17,285–287,313 AD-related degeneration of the brain substrates of motion 87 (a navigation component, see in review) 283 could be a factor in the navigational decline (damage to ERC, 117 OFC, and HC—the structures facilitating cognitive maps and predictions—could contribute to deterioration).5,6,110,117,129,130 Prospective behavioral and physiological studies may determine the effects of viral-induced olfactory loss 156 on spatial navigation and memory.
Combined findings from VR fMRI100,282 and real-world navigation studies18,305 could delineate specific brain regions’ contributions to spatial navigation decline (since motion is an intrinsic real-world navigation component282,283—see HC spatial updating during self-motion 68 and the cerebellar integration of motor and spatial processing).84,85 A declining navigational function, identified in a real-life setting with maximal ecological validity, can be replicated in a VR design and tested within an MRI paradigm: For example, in the case of lower entropy as a proposed compensation for AD patients’ deteriorating navigation ability, 18 insights from complementary MRI could indicate possible neurophysiological causes of the disrupted spatial behavior (e.g., by associating the ERC degeneration with the impaired angle estimation102,104,105 or an HC-supported allocentric-to-parietal-supported egocentric navigational strategy shift;17,47,285,287 a correlation between the medial temporal lobe tau pathology and early AD cognitive changes, as opposed to medial parietal Aβ pathology, 313 may highlight this further).
Although VR findings can translate to real-world navigation,289,290 ecological validity is relevant in terms of multimodal inputs to spatial processing, e.g.;125,298,299 studies outside the lab can increase the ecological validity by accounting for locomotion as an intrinsic outdoor navigation component282,283 (data acquisition in everyday settings would maximize external validity by capturing real-life motion behaviors, such as life space 292 or getting lost)18,305 and the specific environmental odor mixtures (including the potentially detrimental effects of pollution,314,315 a recognized NCD risk factor). 30
Outline of the novel experimental framework as a preclinical screening paradigm for improving health-related quality of life
Integrating feasible, self-use OI tests and wearable or smartphone ACC and GPS sensors into daily routines could produce higher enrollment rates in hypothesis-testing studies and a broader prospective application in the general population: serial longitudinal OI testing with rapid Pocket Smell Test® ($4.75, one or more missing items prompt the extended UPSIT®) 316 can indicate potential early-AD neuropathology underlying OI decline, e.g.,;73,74 daily outdoor navigation recording with the Pro Pod personal GPS devices (up to 15 h-long battery life)18,317 or another portable GPS sensor can indicate AD-specific navigation patterns 18 and the underlying neurodegeneration (e.g., ERC-dependent path integration17,102,104–107 or the suggested substitute of HC-dependent allocentric navigation with parietal-mediated egocentric strategy relative to AD progression).17,285–287,313 Daily gait measuring with Shimmer,19,318 Move4,319,320 Axivity AX3306,307,321 or another wearable ACC device can indicate future AD-related cognitive decline48,49,70,71,279,309 and spatial navigation disorder (gait measurements reflect motion17,86,87—an integral navigation component).282,283 Nighttime movement could additionally indicate circadian cycle disruption19,306 as a possible prodromal-NCD symptom – see in report); 30 other commercially available sensors, e.g., 295 or smartphone GPS 292 and ACC applications could be a low- or no-added-cost alternative, pending validation (see review). 17
An initial small-scale, hypothesis-testing study may precede a longitudinal experimental paradigm for monitoring olfactory and motion changes as the proxy measures of the disease progression (within and between the age-matched CN, MCI, and AD subjects); pending the hypothesis acceptance, implementing AI (with suitable identity-protection mechanisms) may facilitate the accessible preclinical screening. A future AI-based analytic tool, based on models such as13,204 could perform two-fold cross-referencing of the self-collected data: 1) with the biomarker datasets (including blood and CSF markers, volumetric MRI, PET, or postmortem brain pathology, which indicated AD neuropathology in correlation with cognitive measures and OI, motor, or spatial functions in the reviewed studies, e.g.)72–75,88,102,148,203,209,244,264,274,313 and 2) with normative OI, 16 gait speed, 291 and prospective navigation patterns datasets, e.g. 18 Should the initial hypothesis-testing study establish a correlation between the olfactory, spatial, and motor decline rates, either measurement could be self-collected to indicate the AD risk. A potential combination of this method with blood-based biomarkers20–25 could increase the preclinical testing specificity and strengthen the clinical testing indications.
The external validity of such a study is in validating the novel framework for preclinical self-assessment of AD risk. This is in line with current research initiatives for accessible, non-invasive biomarkers prompting a formal medical diagnosis (Figure 3). Following AD risk indications, undergoing clinical examinations would be vital: Neuropsychiatric assessments (such as the Mini-Mental State Examination, see, e.g., application in OI studies,72–75,209,244,264,270 and the Clinical Dementia Rating, see, e.g., an application in a gait study) 279 and composite measures of midlife dementia risk (such as the cardiovascular risk factors, aging, and dementia study (CAIDE) scores,313,322 which correlated with ERC-based path integration impairment, 102 and the ADCS Preclinical Alzheimer Cognitive Composite (ADCS-PACC), which indicated early signs of cognitive decline in CN elderly individuals with AD pathology) 323 could complement biomarker screenings.
In addition to providing an optimal window for effective medication administration (future pharmacological therapies may target the olfactory limbic hubs especially prone to AD pathology such as HC 52 and, possibly, a highly active 46 and connected 44 OB, to restore adult neurogenesis vastly performed in these two structures178,182 and prevent connectome-wide pathological spread in early disease stages, 51 Figure 2), an effective preclinical risk indication could alleviate the economic burden by focusing expensive biomarker testing on at-risk candidates and enabling timely financial planning.
Limitations of framework implementation
The potential framework implementation may face initial challenges related to collaboration with medical institutions: For example, in the case of the Pocket Smell Test® (a brief indicator for further testing with the UPSIT®), the minimum order amount (N = 50) 316 may limit individual purchases. While it would be more feasible for a local healthcare provider to order high test quantities, this intermediate step (involving access to healthcare) might deter some users; engaging local or country-wide dementia societies could mitigate this issue (a local dementia society could promote information or facilitate individual test distribution as a part of raising the NCD risk awareness).
Access to motion sensors and smartphone applications may yield fewer issues—over half (53%) of the global population accesses the internet via personal smartphone (according to the latest report) 324 but correlating a smartphone user's identity with the data may raise privacy issues; device positioning may bias smartphone ACC applications, which are pending validation, e.g., 325 (see in review). 17
Ultimately, embedding technology in the experimental framework may be advantageous in facilitating broad access to the international populations, with technological advances ensuring a continuously improved user experience; vice-versa, such dependence might highlight a currently uneven distribution of socioeconomic groups’ access to devices and internet. 324 Future healthcare policies may increasingly consider technological advancements: Capitalizing on self-use wearable digital sensors and AI could facilitate non-invasive AD risk assessment and focused clinical screenings for at-risk candidates.
Conclusion
Upon establishing the relatedness of olfaction and motion-based spatial navigation from perspectives of evolution, adult neurogenesis, and early AD neurodegeneration, we outlined a novel framework for accessible AD risk detection. A multimodal AI-driven approach, trained on self-use OI tests, ACC and GPS digital wearable sensors, blood-based biomarkers, and big multimodal clinical datasets is a prospective tool that could prompt timely clinical screenings for early AD diagnosis and treatment.
Footnotes
Author contributions
Katarina Biljman (Conceptualization; Investigation; Methodology; Writing – original draft; Writing – review & editing); Illana Gozes (Investigation; Methodology; Validation; Writing – review & editing); Jacqueline CK Lam (Conceptualization; Funding acquisition; Investigation; Methodology; Validation; Writing – review & editing); Victor OK Li (Conceptualization; Funding acquisition; Investigation; Methodology; Validation; Writing – review & editing).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the United States National Academy of Medicine Healthy Longevity Catalyst Award (Grant No. HLCA/E-705/24), administered by the Research Grants Council of Hong Kong, awarded to V.O.K.L. and J.C.K.L, and by The Hong Kong University Seed Funding for Collaborative Research 2023 (Grant No. 109000447), awarded to V.O.K.L.and J.C.K.L.
Declaration of conflicting interests
I.G. serves as Vice President for Research and Development at ExoNavis Therapeutics Ltd developing davunetide (boosting spatial memory in a sex-dependent manner) under patent protection (e.g., Gozes I, Blatt J and Lobyntseva A. Davunetide sex-dependently boosts memory in prodromal Alzheimer's disease. Transl Psychiatry 2024; 14: 412. DOI: 10.1038/s41398-024-03118-0).
The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
