Abstract
Background
Alzheimer's disease (AD) is a progressive neurodegenerative disorder, which subdues over 55 million people and finding a cure, still remains disenchanting. Indian medicinal herbs notably, Withania somnifera, Bacopa monnieri, Curcuma longa, and Clitoria ternatea are traditionally utilized for their memory-enhancing properties.
Objective
We computationally investigated the therapeutic potential of four nootropic herbs by uncovering the molecular mechanisms underlying their treatment for AD.
Methods
Cheminformatics, pharmacokinetics, and system pharmacology studies were carried out to predict the phytocompounds drug-like properties, protein targets, targets functional association and enrichment analysis. A comparative study was performed with phytocompounds and FDA-approved drugs. Investigation on the expression of protein targets in the hippocampus and entorhinal cortex of the AD brain was performed. Network was constructed to depict the interaction between phytocompounds, drugs, and molecular targets.
Results
Through comparative analysis, we found that the phytocompounds shared common targets with both FDA drugs and drugs under clinical trials. We identified potential active compounds notably, Withaferin A, Withanolide-D, Withanolide-E, Withanolide-G, and Humulene epoxide II, that can combat AD. Interestingly, the enzyme inhibition scores of the identified drugs were much higher than FDA-approved drugs. In addition, regulatory proteins such as AβPP, acetylcholinesterase, BACE1, and PTPN1 were the targets of 8, 16, 9, and 22 phytocompounds, respectively. Nonetheless, AR and CYP19A, were the primary targets of most phytocompounds.
Conclusions
Herbal medicines can synergistically stimulate multiple protein targets, rendering a holistic and integrative treatment, encouraging a promising avenue to treat AD.
Keywords
Introduction
Alzheimer's disease (AD) is the most common cause of dementia, and it represents 70% of neurodegenerative diseases. 1 Though the origin is unclear, the most widely accepted theory is the amyloid cascade hypothesis. 2
Encompassing amyloid-β (Aβ) aggregates, elevated oxidative, and inflammatory stress and genetic mutations as the predominant risk factor, AD is one among the top 7 causes of death, according to the World Health Organization. Plants have been the essential source of food and medicine in all cultures and tradition since antiquity. Indian traditional medicine is one of the most ancient practices of health care that kindled plant medicine and pharmacy to its zenith.3–5 When herbs which are pre-eminent in managing these attributes, act in conjunction, it would indubitably engender a novel class of therapeutics. As their multi-targeted mode of action would be much superior to the individual mode of action of each herb. Their synergistic activity can even outshine the presently used drug entities that impart a uniform pattern of target inhibition. Employing phytocompounds with different patterns of target inhibitory rates can help induce a noticeable impact in advance. World Health Organization states that, more than 60% of the population rely on traditional medicine. 6 Manifestation of such an anti-AD approach in AD drug discovery would be a paramount contribution that hastens the process of recovery.
Withania somnifera (Ashwagandha) roots are known for their reputed memory enhancing and anti-inflammatory properties. Their aqueous extract inhibits the formation of mature Aβ fibrils form Aβ plaques, in vitro. 7 Their methanolic extracts, normalize in vivo Aβ plaque deposition. 8 According to reports, the aqueous root extracts of Ashwagandha show significant protective effects on differentiated PC12 cells against H2O2-Aβ1–42 and Aβ1–42 induced toxicity through mitigating oxidative stress. 9 It renders neuroprotection by its ability to suppress oxidative and inflammatory response in microglial cells by simultaneously downregulating the Nuclear Factor Kappa B and upregulating the Nuclear Factor Erythroid 2-related Factor 2 pathways. 10
Bacopa monnieri (Brahmi) is used in diet to improve intelligence and memory for several centuries. 11 The extract of Brahmi is a potential memory enhancer that could reduce the memory impairment via neuro-modulation of acetylcholinesterase, brain-derived neurotrophic factor, MUS-1, CAMP-responsive element binding protein, and altered the neurotransmitters levels in the hippocampus of rat brain. 12 The mode of neuroprotective action of Brahmi is by its ability to diminish neuronal death induced by amyloid peptide, modulation of mitochondrial dysfunction, restoration of Wingless-related integration site/β-catenin signaling, elimination of amyloid plaques and tau. 13
Curcuma longa (Turmeric) is a native Indian spice that contributes to botanical therapies for AD. It strongly inhibits tau phosphorylation, microglial inflammation, and Aβ aggregation produced by amyloid-β protein precursor (AβPP) in vitro. 14 It is competent in regulating insulin signaling pathways, 15 and prevents the deterioration of bone structure that co-occurs with AD. 16 In vivo reports notify that the major component curcumin naturally possesses anti-AD attributes. It attenuated Aβ42-induced neuronal metabolic dysfunction by upregulating Thrb and SIRT3 activity, 17 reduced Aβ production by downregulating BACE1, 18 prevented synaptic degradation, improved learning, and inactivated glycogen synthase kinase 3β and inhibited tau phosphorylation. 19
Clitoria ternatea (Butterfly pea) commonly known as Aparajita, is used in Medhya Rasayana in Ayurveda and is also referred to as anti-depressant flower and embodies colossal anti-oxidative properties that combat age-related disease. 20 The cyclotide-rich fraction of the flowers delay Aβ-induced paralysis in vivo models. 21 The hydroalcoholic extract possessed more antioxidant and enzyme inhibitory activities than aqueous extract and prevented cognitive impairments by reducing oxidative stress, cholinesterase activity and Rho associated coiled-coil containing protein kinase 2 (ROCK II) expression. 22
These four nootropic herbs are much revered in Indian traditional medicine formulations for various ailments, especially for brain rejuvenation and memory that are blemished in AD.23–25 The holistic impact of these four herbs could combat AD and can also promote the overall well-being.
The hippocampus and entorhinal cortex are the most affected regions of a human AD brain.26,27 During the initial stages of AD, the neurons encountered connectional damage in these regions, which subsequently impacts the cerebral cortex responsible for visibly generating the symptoms. 28 The mis-regulation of genes in these areas during AD condition should be contemplated to discover novel targets for anti-AD drugs. The ever-increasing prevalence of AD makes it as one of the most significant global healthcare problems that demands immediate drug development. This computational approach comprehends the combined effect of four key herbs in combating AD through a multi-targeted approach.
Methods
Collection of phytochemicals
IMPPAT (Indian Medicinal Plants, Phytochemistry And Therapeutics, IMPPAT 2.0) (https://cb.imsc.res.in/imppat/), is the largest digital database on phytochemicals of Indian medicinal plants up-to-date. 29 Totally, 213 phytochemicals from Bacopa monnieri leaves, Clitoria ternatea flowers, Curcuma longa rhizoids, and Withania somnifera roots were collected from IMPPAT database.
Retrieval of phytochemical information
Simplified Molecular Input Line Entry System (SMILES) are the most commonly used molecular representation that are defined to be meaningful as a whole. 30 The Canonical SMILIES of the corresponding 213 phytochemicals were derived from PubChem database (https://pubchem.ncbi.nlm.nih.gov/) 31 and IMPPAT database. 29
Identification of active compounds properties
The canonical SMILIES of each phytocompound were input individually in Molinspiration, (https://molinspiration.com/), which is a free online tool that aids in calculating molecular properties and the bioactive scores of drugs targets. 32 Targets such as, G Protein-Coupled Receptor Ligand Activity (GPCR), Kinase inhibitor activity (Ki), Protease inhibitor activity (Pi), enzyme inhibition (Ei), nuclear receptors (Ncr), and number of violations (nvio) were considered. Molecules with bioactivity score greater than 0.00 are likely to have significant biological activities, and those scores less than −0.50 are considered to be inactive. 33
ADME (Absorption, Distribution, Metabolism and Excretion) analysis
The Swiss ADME (http://www.swissadme.ch/) 34 software was accessed, and individual ADME properties of phytocompounds were deduced. The canonical SMILIES were uploaded to the web tool to compute the physicochemical descriptors (molecular weight, number of H-bond acceptors, and number of H-bond donors), lipophilicity (WLOGP), solubility (Log S), pharmacokinetics (GI absorption, blood-brain-barrier (BBB) permeability, and p-glycoprotein Substrate) and drug likeness (Lipinski Drug Likeness and Bioavailability) of the phytocompounds.
Human target imputation
Swiss Target Prediction (http://www.swisstargetprediction.ch/), is a web tool that aims to predict the most probable protein targets. They work by searching for similar molecules like the input query, in 2D and 3D, within a larger collection of 376,342 compounds that are known to be experimentally active. They show high level of predictive performance as they predict at least one correct human target among the top 15 targets. Predictions are based on similarity principle via reverse screening. 35 The canonical SMILIES of the phytocompounds were provided as the input in the tool to derive the protein targets of phytocompounds. NCBI was used to identify the gene information including chromosome number and orthologs (https://www.ncbi.nlm.nih.gov/gene/).
Expression of human targets
The Human Protein Atlas (HPA) version 23.0 (https://www.proteinatlas.org/) is one of the world's largest and most visited databases. 36 All the data of the HPA project, which is a combination of transcriptomics and antibody proteomics used to map all human proteins at a single-cell and spatial resolution, is made publicly available. Most of the efforts focuses on the sc-RNA sequence. Of the 10 sections in the database, the part of brain with maximum expression of predicted target proteins, were derived from Brain section. 37
Compound comparison studies
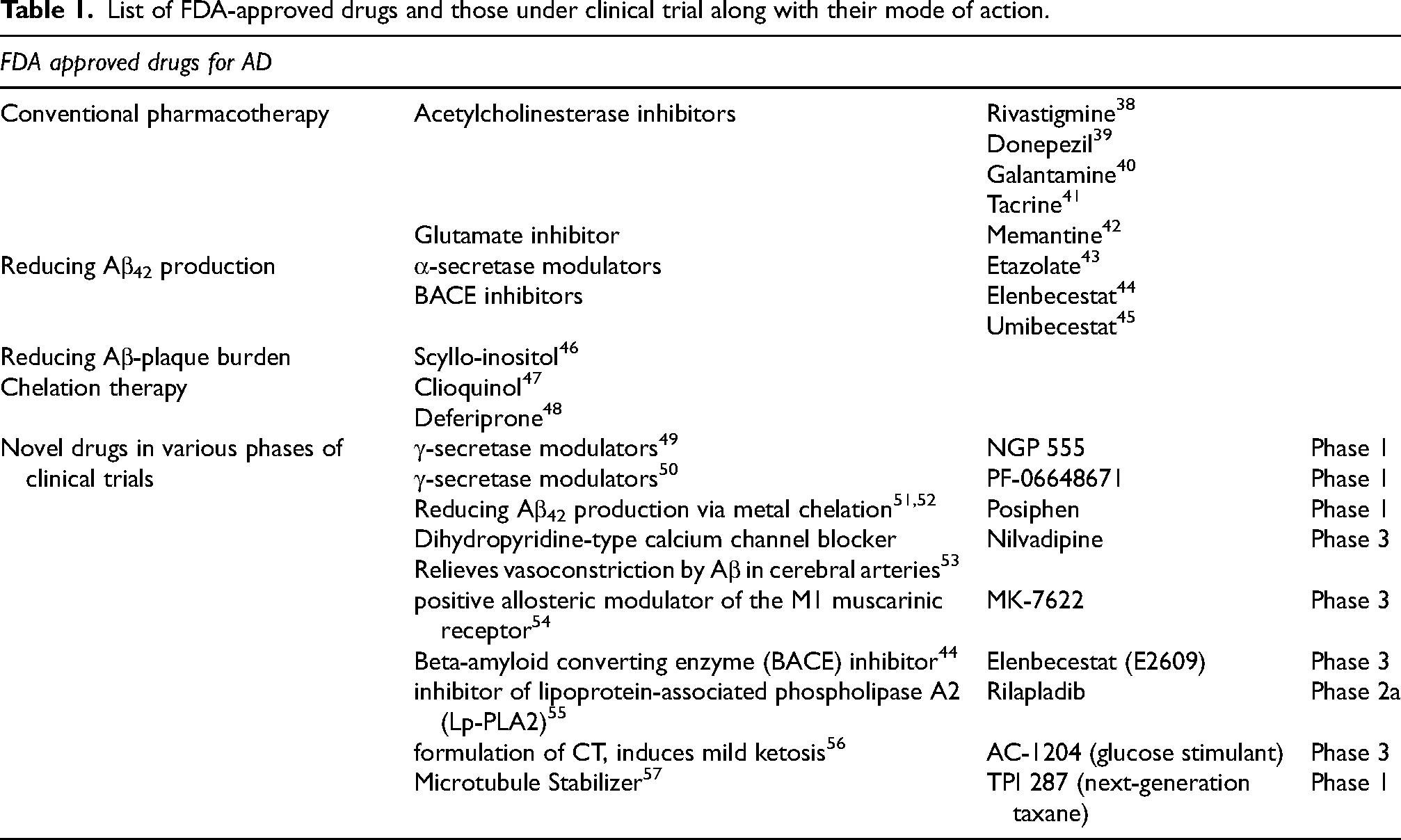
The FDA-approved AD drugs and the novel drugs that are presently under clinical trials for AD were identified and the corresponding canonical SMILIES were derived from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/). 31 All the SMILIES were tabulated in Supplemental Material 1. The SMILES were subjected to Molinspiration (https://molinspiration.com/) 32 and their molecular and bioactive properties like GPCR, Ki, Pi, Ncr, and nvio were retrieved. Swiss Target Prediction tool (http://www.swisstargetprediction.ch/) 35 was used to deduce the corresponding human targets. These attributes were compared with the phytocompounds to identify novel candidate compounds. Table 1 enlists the FDA-approved drugs and those under clinical trial along with their mode of action.
List of FDA-approved drugs and those under clinical trial along with their mode of action.
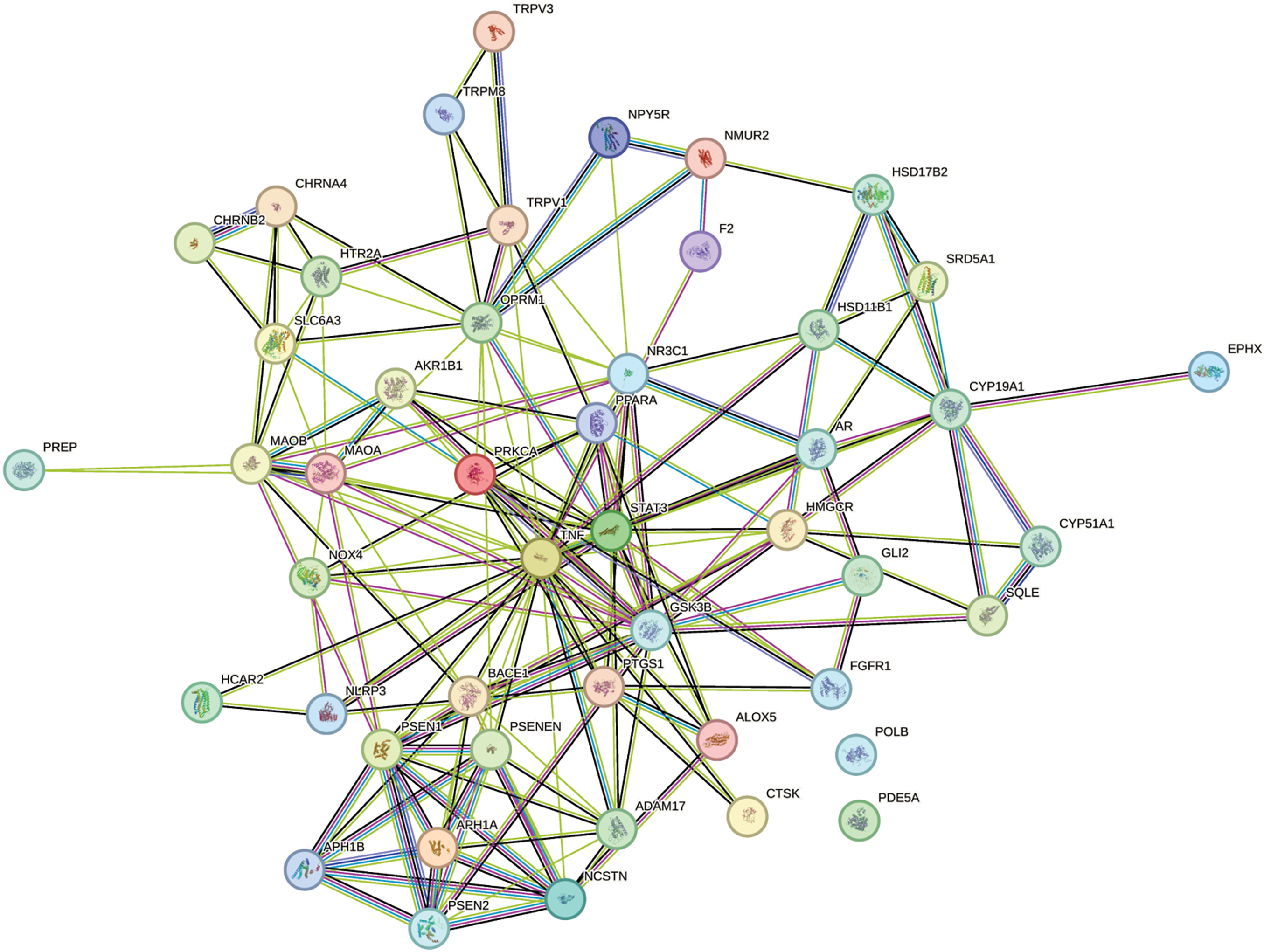
Functional protein- protein association network
The STRING database (https://string-db.org/) systematically collects and integrates protein-protein interactions, both physical and function. 58 The data originate from automated text mining of scientific literature, computational interaction prediction from co-expression, conserved genomic context, databases of interaction experiments, and known pathways from curated sources. The predicted targets of the phytocompounds were uploaded in the String tool to deduce the functional protein association.
Functional enrichment analysis
The STRING database (https://string-db.org/) performs both functional protein-protein association and functional enrichment analysis. 58 The same proteins were input in string and functional enrichment was performed. Enrichment pathways like gene ontology analysis KEGG, Reactome, Wiki were performed and Disease-gene Associations (DISEASES), Tissue Expression (TISSUES) and Subcellular Localization (COMPARTMENTS) of the target proteins were deduced.
Network construction
Compound-Target-Network (C-T-N). Cytoscape.js (http://js.cytoscape.org), is an open-source JavaScript-based graph library and is a visualization software component. It provides interactive graphs in a web browser that enables users to interact with networks for analysis. 59 The C-T-N was constructed using Cytoscape 3.10.1 to combat AD by expounding the multi-target therapeutic capabilities of the phytocompounds. An interaction between protein targets and the phytocompound would be made possible only if the protein is targeted by the respective phytocompounds. Also, one protein may be targeted by more than one candidate compound. The network visualization was accomplished by Cytoscape 3.10.1. 59
Target-Disease-Network (T-D-N). The predicted human targets of each phytocompounds were subjected to gene expression analysis using AlzData database (http://www.alzdata.org/) 60 to identify the differential expression of genes in normal and AD condition, in the entorhinal cortex and hippocampus regions of AD brain. AlzData is a one-stop database to make full collection of current high-throughput omics data. It is an in-depth integrating system and covers high-throughput omic data like genomics, transcriptomics, proteomics, and functional genomics and high-confident functional data like neuroimaging screening, population based longitudinal studies and transgenic mouse phenotyping. 60 Network was constructed, and visualization was accomplished by Cytoscape 3.10.1. 59
Drug-Target-Network (D-T-N). The top 15 most probable targets of 20 FDA-approved drugs and those under clinical trials were identified, and network with their corresponding targets were constructed and visualized using Cytoscape 3.10.1. 59
Phytocompound and drug target network (P-DT-N). The phytocompounds which had common targets with the anti-AD drugs were identified and a network was constructed, and visualized using Cytoscape 3.10.1. 59
Results
The flowchart of the entire process followed in the manuscript is depicted in Figure 1.

The flowchart of the methodologies employed in the research.
Collection of phytochemicals
IMPPAT 2.0 database was used to retrieve 213 phytocompounds for the four herbs, and they are listed out in the Table 2.
List of 213 phytocompounds from four herbs.

Retrieval of phytochemical information
We derived the canonical SMILIES of the phytocompounds through PubChem database. IMPPAT database was used for those phytocompounds whose canonical SMILIES were not available in PubChem. The canonical SMILIES of phytocompounds compounds are listed out in Supplemental Material 1.
Identification of properties of active compound
The molecular properties and bioactivity with drug targets like, GPCR, Ki, Pi, Ei, Ncr, and nVio were derived for the 213 compounds and are tabulated in Supplemental Material 2. These targets have therapeutic implications in AD as GPCR signaling pathway regulate the cascades of tau and Aβ in AD, 61 ion channels are involved in the neuronal homeostasis, and pathways underlying AD, 58 likewise, the targets like ki, 62 Ncr, 63 pi 64 and Ei 65 are role players in the modulation of the disease progression. Thereby, assimilation of the bioactivity scores of such important targets were essential. Based on the enzyme inhibitor activity scores (>0.00), the phytocompounds were classified and chosen for further examination. A total of 118 phytocompounds were considered for further evaluation and are tabulated in Supplemental Material 3.
ADME analysis
The pharmacokinetic and drug-likeness properties of the selected 118 phytocompounds were calculated by the SwissADME online version tool. It comprises of molecular and physicochemical characteristics like molecular weight, number of hydrogen bond acceptors and number of hydrogen bond donors. The BBB, is a natural shield to most of the therapeutic drugs that enter the brain as it restricts the permeation and diminishes the efficiency of the medications. 66 Molecules with molecular weight less than 500 Da and which are lipophilic in nature can cross BBB and enter brain. 67 Thereby, inspecting these imperative characteristics like molecular weight, BBB permeation, high solubility (logS) and P-glycoprotein substrate are essential, as they are one of the important properties for a drug to be termed as a neuroprotectant. The ADME results for 118 compounds are tabulated in Supplemental Material 4.
Molecular weight (mw)
Lipinski's rule states that the limit of MW of drugs should be within 500 g/mol as it increases their probability of being absorbed orally in the gastrointestinal tract. 68 In this study all the active compounds had MW lesser than 500 g/mol except Sitoindoside IX, Withanolide-S, and Withanolide-C. The results are highlighted, and depicted in Supplemental Material 4. The analysis of MW shows that, almost all these molecules due to their small size can act as drugs, according to Lipinski's rule.
Lipophilicity
Lipophilicity is a principal parameter in the process of drug discovery and design. SwissADME supports 5 freely assessable models for lipophilicity, and WLOGP is used in this study. WLOGP is the application of purely atomistic method stationed on fragmental system. 69 Log Po/w is an arithmetic mean of the values predicted by all 5 proposed methods. 32 All the studied compounds possess Po/w values within the range −0.18 to 3.50. In the present analysis of lipophilicity, 3 the phytocompounds of Bacopa monnieri were within the limits and 6 were not. All phytocompounds of Clitroia ternatea and 9 phytocompounds of Withania somnifera were withing the range, and 6 were not. 33 phytocompounds of Curcuma longa were within the range, and 57 were not. The pattern of lipophilicity for all phytocompounds are listed out in Supplemental Material 4.
Solubility (log s)
Log S refers to the aqueous solubility of phytocompounds that have direct impact on oral absorption and should be within the specified range of −6.5 to 0.5. 70 Swiss ADME provides three topological approaches to predict water solubility. The one used in this study is developed by SILICOS-IT (solubility class: Log S Scale: Insoluble < -10, poorly < −6, moderately < −4, soluble < −2, very < 0 < highly) where the linear coefficient is corrected by molecular weight (R2 = 0.75). The predicted values are decimal logarithm of molar solubility in water (log S). 33 Solubility of Bacopa monnieri phytocompounds were from moderately (−6.20) to highly soluble (1.49). All the compounds of Clitroia ternatea ranged from soluble (−3.24) to very soluble (−1.51). Compounds of Withania somnifera ranged from moderately (−6.19) to very soluble (−1.85), and those of Curcuma longa were from moderately (−6.19) to very soluble (−1.46). None of the compounds were poorly soluble or insoluble. Log S values of the phytocompounds are depicted in Supplemental Material 4.
Pharmacokinetics: gi absorption
Egan egg is used to assess the predictive power of the model for GI passive absorption and prediction for brain access by passive diffusion. 71 The BOILED-Egg (Brain or Intestinal Estimated D permeation predictive model) has 2 regions the white and yellow. White region is space for molecules with greater extend of absorption by GI tract while yellow is the region with highest probability to permeate the brain.72,73 The GI absorption of all compounds was collected and tabulated in Supplemental Material 4.
Pharmacokinetics: blood-brain-barrier (BBB) permeability
BBB, the superlative natural barrier prevents the entry of harmful toxins, and infections in the blood stream from entering the brain. More than 98% of small drugs do not cross the BBB as it allows the permeation of only selected molecules into the brain. 67 Not all phytocompounds listed could cross BBB. The nature of permeability patterns for the compounds are listed out in Supplemental Material 4.
P-glycoprotein substrate (p-gp)
P-gp is localized in tissues like columnar epithelial cells, lower gastrointestinal tract, capillary endothelial cells of brain and testis, canalicular surface of hepatocytes, and apical surface of proximal tubules in kidney. 74 Such selective distribution at the site of drug absorption and elimination changes the pharmacokinetic properties of the drug. 75 The efflux pump of the P-gp limits the entry of drugs into systemic circulation from intestine, prevents its distribution into brain, and also limits reabsorption of drug into systemic circulation from renal tubules. 76 Therefore, the P-gp affinity screening is an integral part of drug discovery program. The P-glycoprotein Substrate for phytocompounds are tabulated in Supplemental Material 4.
Human target imputation
Swiss Target Prediction predicted the targets of the 118 phytocompounds along with their respective Uniprot ID. The target displaying the highest probability scores were obtained, and the associated chromosome number and ortholog information were retrieved from NCBI. Out of the total 118 phytocompounds, 11 were found to have no predicted targets. Supplemental Material 5 has information of all these data. Of the 107 phytocompounds, information on in vivo, and in vitro AD studies were available only for 30 phytocompounds and is collectively listed out in Supplemental Material 5. These unique qualities contribute to the comprehension of the intricate molecular functionality of the target protein.
Expression analysis of human target
The 107 targets of the 107 phytocompounds were utilized as query in the ‘The Human Protein Atlas database’. Through this process, the specific region of the human brain with the highest RNA expression for each target was deduced and recorded in Supplemental Material 5.
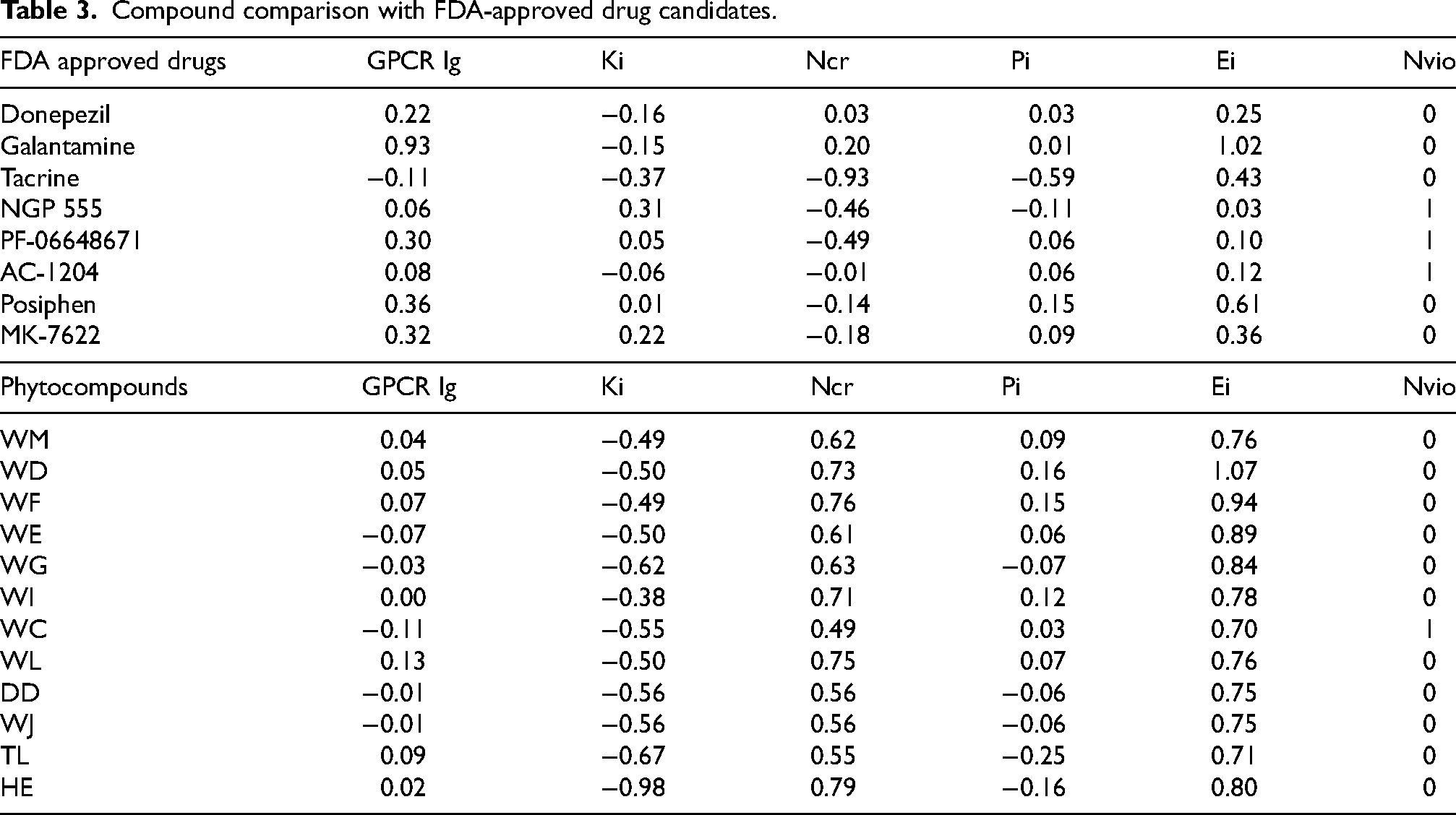
Comparison analysis with FDA-approved drugs and their targets
The molecular properties and predicted targets of FDA-approved drugs were identified and tabulated in Supplemental Material 6. We identified phytocompounds that targeted the same protein targets as FDA-approved drugs and compared their molecular and bioactive properties. Among the 20 drugs considered in the study, only donepezil, galantamine, tacrine, NGP 555, PF-06648671, AC-1204, Posiphen, and MK-7622 had positive Ei scores. Table 3 depicts that phytocompounds like Withanolide-D (WD), Withaferin-A (WF), Withanolide-E (WE), Withanolide-G (WG), and Humulene epoxide II (HE) have better Ei scores than the FDA-approved drugs and are identified as the novel compounds that could combat AD. Table 3 enlists the compound-comparison with FDA-approved drug candidates.
Compound comparison with FDA-approved drug candidates.
Functional protein association network
The string tool was used to determine the interaction between the top 40 targets of 107 phytocompounds. Figure 2 shows the functional protein association network. The query proteins are represented by colored nodes, with filled structures indicating known or predicted 3D structures. The edges in the graph represent protein-protein associations and indicates that they have a shared function. This does not mean that they are binding to each other. Blue-colored edges in the graph represent known interactions from the curated database, while violet-colored edges indicate determined interactions. Predicted interactions are denoted by three colors: green implies gene neighborhood, red shows gene fusion and dark blue denotes gene co-occurrence. Others include text-mining, which is in light green color, co-expression in black and protein homology in light blue. Protein targets like tumor necrosis factor (TNF), 77 GSK3B, 78 monoamine oxidase B (MAOB), 79 and monoamine oxidase A (MAOA), 80 known to regulate AD, exhibit interactions with other targets, whereas DNA Polymerase Beta (POLB) does not. All proteins, except DNA polymerase β (POLB), Carbonic Anhydrase 4 (CA4), and Carbonic Anhydrase 12 (CA12), contribute to a joint function. The network denoted that the 40 proteins have more interactions among themselves than what would have been expected for a random set of proteins of the same size.
Functional protein association network.
Functional enrichments in network
Using string software, the functional enrichments in the network were carried out for the 40 proteins. Enrichment pathways like gene ontology analysis KEGG, Reactome, Wiki, DISEASES, Tissue Expression and Subcellular Localization data were obtained and are listed out in Supplemental Material 7. The enrichment data is indicated in 3 columns namely, count in network, strength, false discovery rate.
Count in network: The first number indicates how many proteins in the network are annotated with a particular term. The second number indicates how many proteins in total (in your network and in the background) have this term assigned.
Strength: Log10 (observed / expected). This measure describes how large the enrichment effect is. It's the ratio between i) the number of proteins in your network that are annotated with a term and ii) the number of proteins that we expect to be annotated with this term in a random network of the same size.
False discovery rate: This measure describes how significant the enrichment is. Shown are p-values corrected for multiple testing within each category using the Benjamini–Hochberg procedure.
Gene ontology analysis: The phytocompound-targeted proteins were identified to be involved in carrying out biological processes like negative regulation of Aβ clearance, dopamine catabolic process, Aβ formation, notch receptor processing, regulation of chronic inflammatory response, AβPP catabolic process, neurotransmitter catabolic process, negative regulation of acute inflammatory response, estrogen biosynthetic process, reactive oxygen species biosynthetic process, negative regulation of inflammatory response, regulation of neurotransmitter levels, regulation of reactive oxygen species metabolic process, regulation of MAP kinase activity, regulation of extrinsic apoptotic signaling pathway etc. The other processes are listed out in Supplemental Material 7. These processes are listed based on their descending order of strength (2.69 −1.1) and ascending order of false discovery rate (0.0048–0.0244).
The target proteins were involved in molecular functions like monoamine oxidase activity, phenethylamine-oxygen oxidoreductase activity, aliphatic-amine oxidase activity, androgen binding, primary amine, transition metal ion binding, cofactor binding, coenzyme binding, heme binding, tetrapyrrole binding, steroid dehydrogenase activity, oxidoreductase activity, nuclear receptor activity, retinal dehydrogenase activity, primary amine oxidase activity, steroid dehydrogenase activity, flavin adenine dinucleotide binding activity, electron transfer activity, oxidoreductase activity, acting on the CH-OH group of donor, oxidoreductase activity, transition metal ion binding activity, catalytic activity, ion binding activity, heterocyclic compound binding and organic cyclic compound binding activity.
The target proteins were identified to be part of cellular components like cell body fiber, integral component of presynaptic membrane, membrane raft, external side of plasma membrane, neuronal cell body, cell surface, endoplasmic reticulum membrane, organelle sub-compartment, somato-dendritic compartment, endoplasmic reticulum, intrinsic component of plasma membrane, integral component of plasma membrane, neuron projection, organelle membrane, integral component of membrane, endomembrane system, membrane and cytoplasm.
KEGG pathway: According to this analysis, the 40 target proteins were found to be involved in various metabolic pathways of nitrogen, phenylalanine, histidine, tyrosine, glycine, serine and threonine, tryptophan, arginine and proline. Biosynthesis of steroid, steroid hormone, amphetamine and cocaine addiction. They showed involvement in ovarian steroidogenesis, serotonergic synapse, inflammatory mediator regulation of TRP channels, Fc epsilon RI signaling pathway, hedgehog signaling pathway, dopaminergic synapse, mTOR signaling pathway, neuroactive ligand-receptor interaction, pathways in cancer and metabolic pathways. Also, they were involved in diseases pathways like African trypanosomiasis, prostate cancer, insulin resistance, yersinia infection, alcoholism, non-alcoholic fatty liver disease, hepatitis C, influenza A and Alzheimer disease.
Reactome pathway: According to this analysis the 40 target proteins were involved in pathways like, biogenic amines are oxidatively deaminated to aldehydes by MAOA and MAOB, dopamine clearance from the synaptic cleft, estrogen biosynthesis, fructose metabolism, regulated proteolysis of p75NTR, reversible hydration of carbon dioxide, metabolism of steroid hormones, cholesterol biosynthesis, SUMOylation of intracellular receptors, trp channels, activation of gene expression by SREBF, phase I - functionalization of compounds, metabolism of steroids, nuclear receptor transcription pathway, transcriptional regulation by RUNX2, metabolism of lipids, metabolism, disease and signal transduction.
Wiki pathway: According to this analysis the 40 target proteins were involved in pathways of neurotransmitter disorders, dopamine metabolism, dopaminergic neurogenesis, vitamin D in inflammatory diseases, NRF2-ARE regulation, oxidative stress response, notch signaling pathway, chronic hyperglycemia impairment of neuron function, notch signaling, Alzheimer's disease and miRNA effects. These are a few essential processes involved in Alzheimer's disease regulation. The other pathways are listed out in Supplemental Material 7.
Disease-gene associations (DISEASES): the analysis reviles that the 40 target proteins were involved in various diseases like Tinea pedis, atypical depressive disorder, dysthymic disorder, social phobia, diabetic neuropathy, obsessive-compulsive disorder, pseudohermaphroditism, anxiety disorder, dementia, mood disorder, Alzheimer's disease, cognitive disorder, disease of mental health, endocrine system disease, central nervous system disease, nervous system disease, disease of anatomical entity and disease.
Tissue expression (TISSUES): The 40 target proteins were identified to be involved in primary cell line, amyloid plaque, neurofibrillary tangle, trigeminal nerve, hepatocyte, peritoneum, glial cell, nerve, spinal cord, bone marrow cancer cell, colon, liver, blood, lung, digestive gland, viscus, endocrine gland, gland, brain, nervous system, head and whole body.
Subcellular localization (COMPARTMENTS): The subcellular localization of the 40 proteins were identified, and they were involved in c-fiber, Aβ complex, gamma-secretase complex, endoplasmic reticulum, plasma membrane protein complex, organelle sub-compartment, extracellular space, intrinsic component of plasma membrane, intrinsic component of membrane, integral component of membrane, membrane protein complex, endomembrane system, organelle membrane, plasma membrane, membrane, cytoplasm, intracellular membrane-bounded organelle, membrane-bounded organelle and cellular anatomical entity.
Based on the result of enrichment analysis, we infer that, 40 proteins are involved in Aβ clearance, formation, inflammatory, dopaminergic, neurotransmitter disorders, Alzheimer's disease regulation pathways, etc. The disease-gene association indicates that they are involved in disorders like, dementia, AD, cognitive disorder, disease of mental health, central nervous system disease, nervous system disease, etc. The tissue and sub cellular localization describes their involvement in amyloid plaque, neurofibrillary tangle, Aβ complex, gamma-secretase, etc., which are the main regulatory elements of AD. Having involvement with the chief disease regulatory proteins, function and pathways, the phytocompounds can directly modify the AD condition.
C-T-N analysis
The C-T-N represents 107 phytocompounds and their top 15 predicted targets in humans. By analyzing this network, we identified that the candidate compounds exhibited multi-target properties, illustrating the holistic mode of action. Each protein was targeted by more than one phytocompound summing up 332 targets for 107 phytocompounds as represented in Figure 3. There were 115 proteins targeted by individual phytocompounds, and 217 proteins targeted by multiple phytocompounds. Either by modulating, inhibiting, enhancing, or transducing signals of target proteins, these compounds may exhibit properties that can effectively combat AD. The top 9 proteins that had maximum predicted interactions with phytocompounds are illustrated in Table 4. The function of these proteins and their use in AD is depicted in Supplemental Material 8, which identifies their regulation in cognitive function, neurotransmitter systems, biomarkers of AD, regulate memory and AD risk genes. Also, regulatory proteins that are the main drivers of AD, such as AβPP, acetylcholinesterase, BACE1, and PTPN1 were targeted by 8, 16, 9, and 22 phytocompounds respectively.

Compound-target-Network (C-T-N).
Top 9 proteins which had maximum interactions with phytocompounds. (CHRM2: Cholinergic Receptor Muscarinic 2; SIRT3: Sirtuin 3; SLC6A2: Sirtuin Solute Carrier Family 6 Member 2; SHBG: Sex hormone binding globulin; HSD11B1: Hydroxysteroid 11-Beta Dehydrogenase 1; PTPN1: Protein Tyrosine Phosphatase Non-Receptor Type 1; ESR1: Estrogen Receptor 1; NR1H3: Nuclear Receptor Subfamily 1 Group H Member 3; CYP19A1: Cytochrome P450 Family 19 Subfamily A Member 1; AR: androgen receptor.

T-D-N
The T-D-N illustrates the differential expression of protein targets in the entorhinal cortex and hippocampus regions of an AD brain based on their log2 Fold change values. These are the primary regions affected in an AD brain. We extracted the gene expression data from the microarray data set GSE48350, in the AlzData database. The networks for the upregulation and downregulation of targets in the hippocampus and entorhinal cortex regions are depicted in Figures 4 and 5. The corresponding log2 fold change values are tabulated in Supplemental Material 9. This indicated that most of the phytocompound targeted proteins were found to be mis regulated in an AD brain.
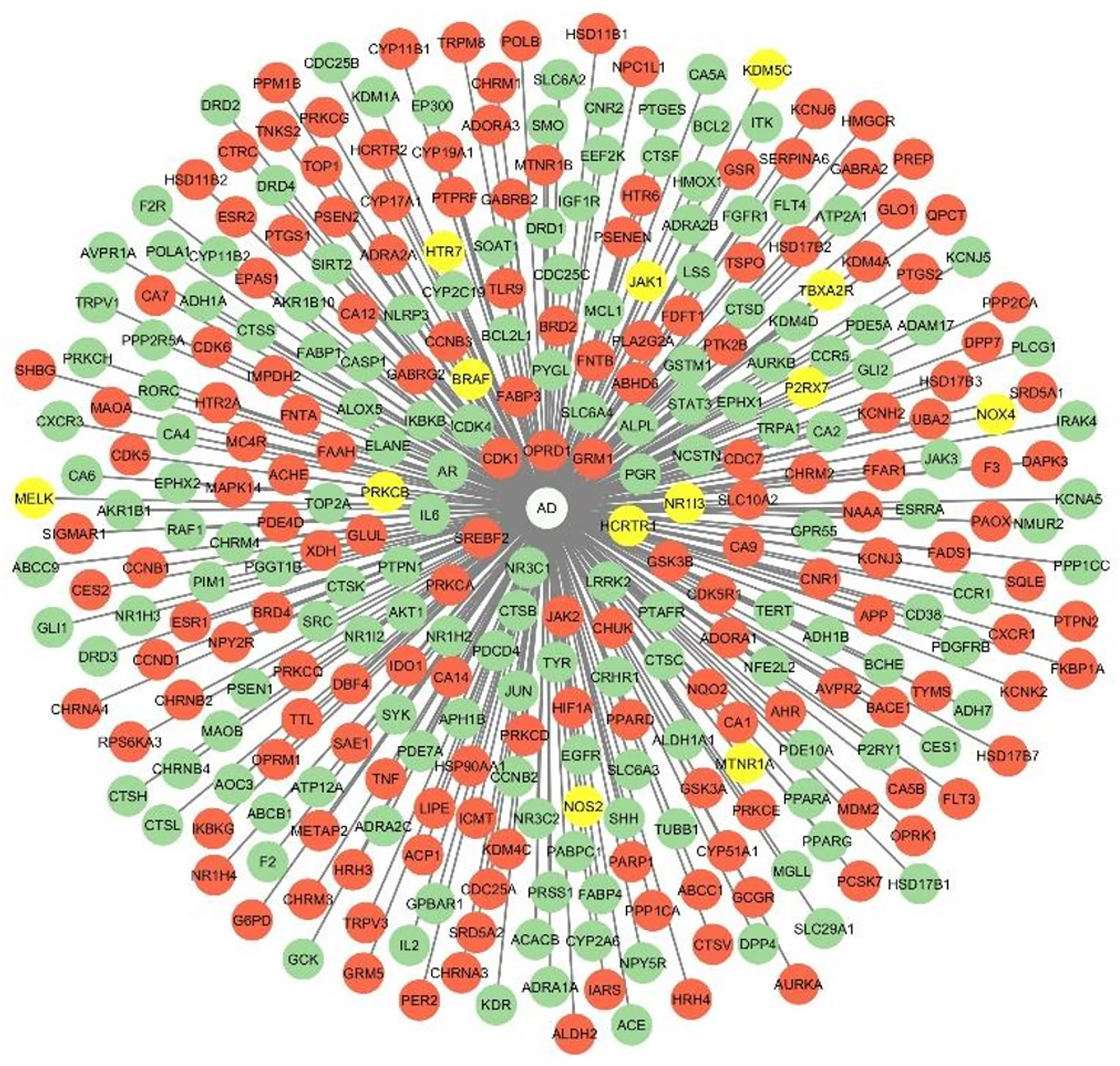
Target-Disease-Network-1. Targets that are upregulated in the hippocampal region are shown in green, downregulated in red, and no change is represented in yellow.
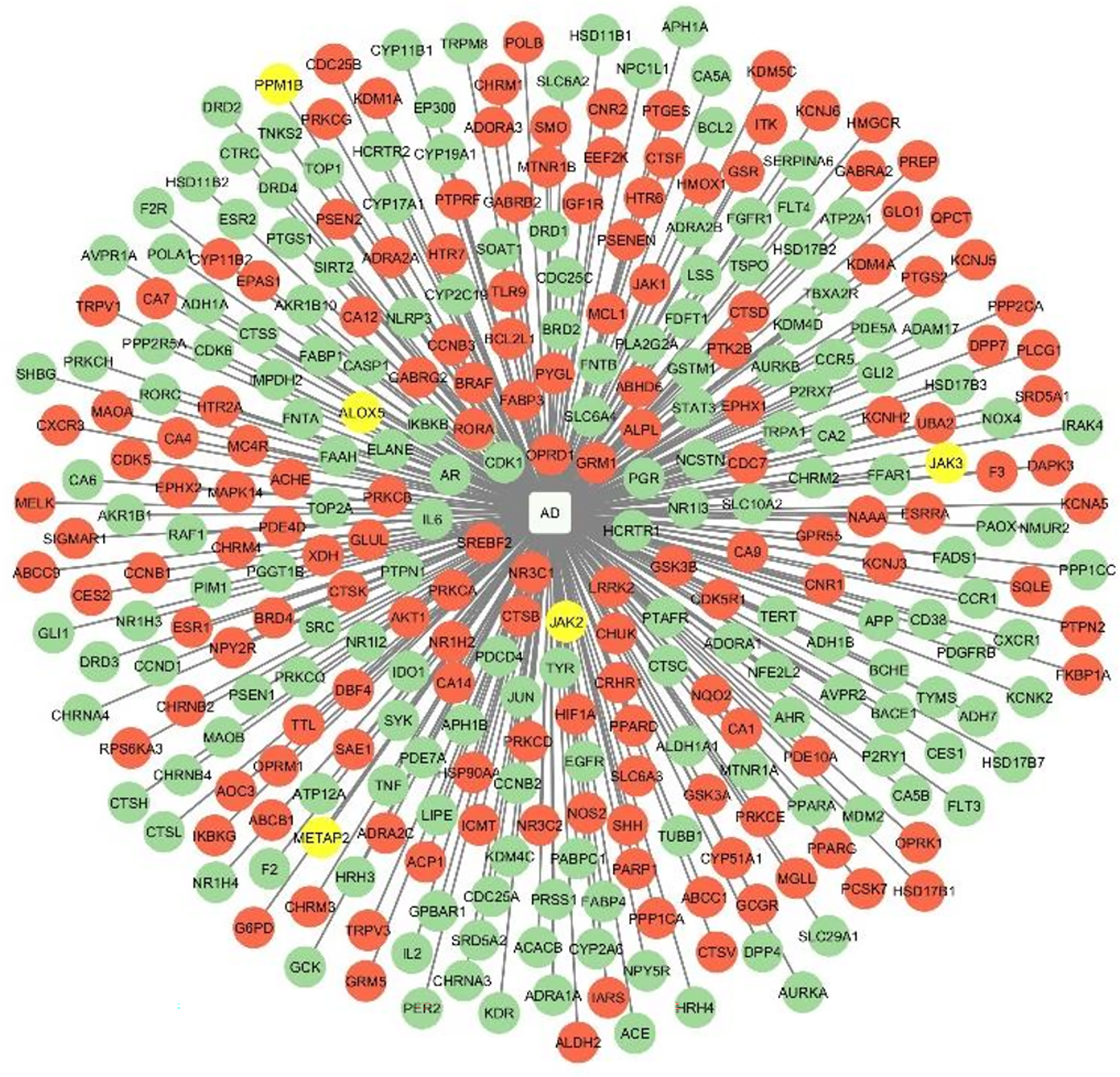
Target-Disease-Network-2. Targets that are upregulated in the entorhinal cortex region are colored green, while downregulated targets are colored red, and those with no change are depicted in yellow.
D-T-N
The D-T-N network represents the top 15 targets of FDA-approved drugs and those under clinical trial against AD. The network revels 190 targets for 19 drugs and is depicted in Figure 6.
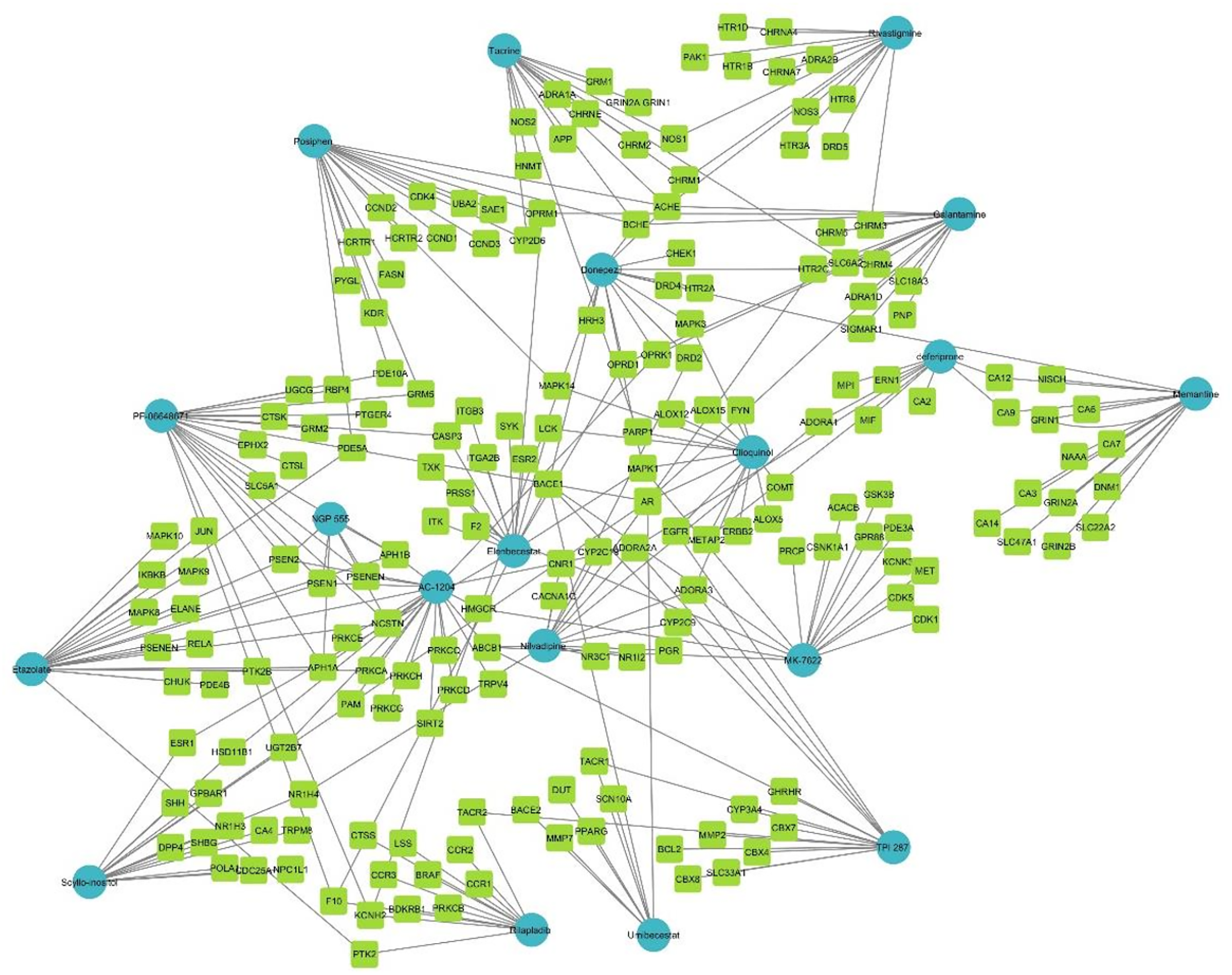
Drug-Target-Network (D-T-N). Blue represents drugs, while light green represents their targets.
P-DT-N
The P-DT-N focuses on phytocompounds that have similar targets to anti-AD drugs. The phytocompounds targeted 109 proteins out of the 190 drug targets, which are represented in Figure 7. The presence of such common targets suggests that these phytocompounds may have a similar mode of action as the currently used drugs.
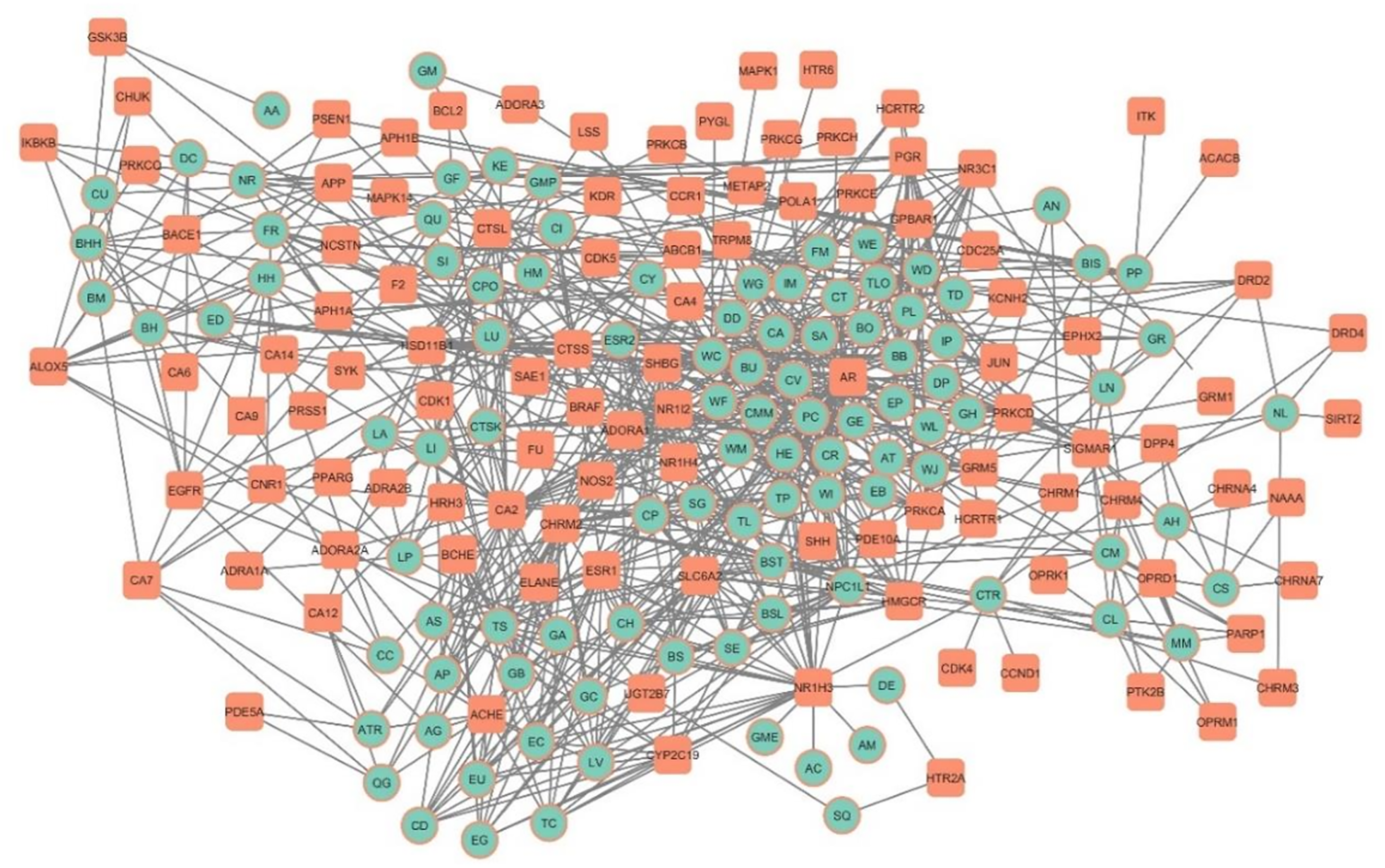
Phytocompounds and drug target network (P-DT-N). Red represents phytocompounds while green represents targets.
Discussion
The current research on phytomedicines extends the knowledge on the potential use of plant extracts to combat multifactorial disorders, giving new insights into the therapeutic avenues for AD. 81 Research in the present decade is focused on the conviction that, plants and phytocompounds separately or in combination can effectively treat AD, with lesser side effects than synthetic drugs. 82 AD is a non-curable brain disorder, that has become a major public health challenge in recent years. 83 Current therapeutics only slows down disease progression, preventing further damage. 84 AD drugs are inefficient in eradicating the disease due to the involvement of heterogeneous genes in AD development. 85 Targeting and developing a cure for AD is both difficult and time consuming. This statement emphasizes the importance of finding novel drugs that interact with multiple targets. Although neuroprotective medicinal plants have great potential in AD treatment, concerns about their usage and safety are being raised. The insufficient scientific evidence regarding their physiochemical properties, efficacy, and molecular targets may be the cause for it. The mechanism by which herbal drugs affect molecular targets and their pathways remains unclear. In order to tackle this constraint, we identified bioactive compounds from four plants and with the aid of computational methods predicted their pharmacological effects in AD treatment at the molecular level.
Previous reports document that, drugs with individual main target and uniform mode of action are not successful in bringing about a desired result that can modify the disease condition. 86 Drug entities imparting a uniform pattern of target inhibition, may be one of the primary reasons to why they cannot induce a prominent impact. Bringing the current hybrid therapeutic research into the limelight, combination of drugs like donepezil and rivastigmine are being developed to treat complex AD with their synergism. 87 Thereby, it is wise to introduce a “group” of candidate compounds that act on multiple targets, with different rates of target inhibition, so that an advanced noticeable change in AD progression is observed.
The C-T-N delineates the multi target functional modules of individual phytocompounds. The present-day AD drugs work by suppressing targets such as butyrylcholinesterase or acetylcholinesterase or AβPP. In such a scenario, they are targeted only by one particular drug entity. But in the present study 12, 16, and 8 phytocompounds are found to target butyrylcholinesterase, acetylcholinesterase and AβPP, respectively. This may induce a prolonged suppression of targets, generating a faster, colossal and long-lasting impact on the disorder. This may be much reliable and better than the drugs that work by uniform suppression of targets. Also, AR, which is targeted by 49 compounds, is upregulated in both hippocampus and entorhinal cortex in AD condition. Research with 150 AD cases apprises that polymorphism in exon 1 of AR is associated with AD in men. 88 Even though AR is an important target and is reported to be dysregulated in AD, 89 it is not targeted by many of the FDA-approved drugs, considered in this study. Upon investigation of the top 15 targets of the 20 FDA-approved drugs, we found that out of 20, only 6 have AR as their target, and none have it as their main target. But among the 49 phytocompounds that target AR, 10 had AR as their most probable target. Also, CYP19A, which is targeted by 47 compounds, is downregulated in hippocampus and upregulated in the entorhinal cortex. It has been documented that Cytochrome P450 aromatase activity (CYP19A) is found to be increased in the nucleus basalis of Meynert during AD, making it a potential risk factor, 90 CYP19A is not targeted by the 20 FDA-approved drugs, but out of the 47 phytocompounds that target CYP19A, 15 had CYP19A as their most probable target.
The other protein targets that had maximum interactions are identified to regulate cognitive function, neurotransmitter systems, biomarkers of AD, regulate memory and AD risk genes. The present study highlights the targets AR and CYP19A and opens new avenues to investigate their potential for the development of a novel class of herbal therapeutics against AD. Phytocompounds like WD, WF, WE, WG, and HE have good Ei scores than the FDA-approved drugs which clearly shows that phytocompounds may have a stronger impact on enzyme inhibition activities than the commercial drugs. WD, WF, WE, WG are phytocompounds of Withania somnifera and HE is a phytocompound of Curcuma longa. WF is presently under clinical trials phase 1/2 for Combination Therapy for Recurrent Ovarian Cancer (NCT05610735). In acute toxicity study WF was found to be safe up to 2000 mg/kg, LD50 was >2000 mg/kg. Based on repeat doze toxicity 500 m/kg of WF was considered as No-Observed-Adverse-Effect Level (NOAEL) dose. Intravenous route of administration was better than oral administration when tested in mice. 91 WE is employed in in vitro cancer studies with cell lines, 31 but are not yet employed in animal study. WD had the best Ei score, and cancer research in mice termed WD as the lead candidate of antileukemic agent. 92 Much information on WG and HE was not available as it has not been used for both in vitro and in vivo studies.
The phytocompounds also targeted 109 proteins out of the 190 drug targets, indicating that their mode of action may be similar to those of the drugs. This make the phytocompounds more reliable to be used as a drug for palliative care. Regulatory proteins such as AβPP, acetylcholinesterase, BACE1, and PTPN1 were targeted by 8, 16, 9, and 22 phytocompounds respectively. The key drivers of the disease are being targeted by the phytocompounds, with which its undoubtable that disease can be regulated and modified by their action. Also, more than 95% of the proteins targeted by the phytocompounds were found to be dysregulated in an AD brain. This implies that such multitargeted treatments may not only positively modulate the disorder but may also treat other ailments that progress along with AD. As we witnessed a larger group of proteins being mis regulated in AD, there is a possibility that it may trigger the onset of other diseases too. Impaired insulin signaling is a consequence of AD and causes diabetes. 93 For example, AR, which is the most targeted protein in the present study, is also an important target for diabetes. 94
AD is not developed by a single factor each year novel genes are reported to drive the pathogenesis of AD, 95 but drugs which target all the main regulators and newly emerged factors have not yet emerged. The chosen herbs are employed in formulating both Medhya Rasayana 96 and Divya-Medha-Vati 97 (ayurvedic herbal medicines) and are principal herbs for management of AD. Consumption of these nootropic plant on a regular bases may even act as a preventive from various ailments, improving memory. The use of these phytocompounds for palliative care is yet to be unraveled, as it demands validation with animal studies. Though the phytocompounds are validated through Insilco studies, further in vivo and in vitro studies are essential to validate it as a drug for preventive or palliative care. The present work has laid the foundation for further studies on drug discovery. Given the current lack of progress in current medication, it is imperative to redirect our attention towards the disease-free aging and educate the modern medicine industry on the invaluable benefits of utilizing herbs with their remarkable phytocompounds.
Conclusion
In this study, we identified 107 phytocompounds derived from Curcuma longa, Withania somnifera, Bacopa monnieri, and Clitoria ternatea. Upon analysis, these phytocompounds possessed positive Ei values. Their molecular and pharmacokinetic properties were studied, protein targets were determined. The functional association of the protein targets in various pathways were deduced through GO analysis, KEGG, Reactome and Wiki pathways. The target proteins disease gene association, tissue and subcellular localization were also determined. Through the log2 fold change values, from AlzData, the expression of target proteins, in the hippocampus and the entorhinal cortex of AD brain were deduced. A comparison was made between the phytocompounds and FDA-approved drugs. This method identified WD, WF, WE, WG, and HE which have Ei values higher than the currently available drugs. Also, 109 protein targets that were common to both, the phytocompounds and FDA-approved drugs. All the networks were constructed and represented using cytoscape. AR and CYP19A were identified as targets for 49 and 47 phytocompounds respectively, while most of the drugs did not target them. AD regulatory proteins were targeted by many phytocompounds. We also conclude, there is a possibility that these phytocompounds may cease the onset of other ailments too, that are triggered with the progression of AD. This study highlights the need for integration of traditional medicine to the modern therapy, positioning the age old, Indian traditional medicine as a phenomenal cure for the new age disorder, AD. The combination of herbal medicine and its synergistic effects might cause a significant advancement in the field of neuro-medicine for AD. These phytocompounds may require further optimization for the development of drugs targeting AD.
Supplemental Material
sj-docx-1-alr-10.1177_25424823241307019 - Supplemental material for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach
Supplemental material, sj-docx-1-alr-10.1177_25424823241307019 for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach by Reiya Bosco Don Bosco, Johnson Retnaraj Samuel Selvan Christyraj and Beryl Vedha Yesudhason in Journal of Alzheimer's Disease Reports
Supplemental Material
sj-docx-2-alr-10.1177_25424823241307019 - Supplemental material for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach
Supplemental material, sj-docx-2-alr-10.1177_25424823241307019 for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach by Reiya Bosco Don Bosco, Johnson Retnaraj Samuel Selvan Christyraj and Beryl Vedha Yesudhason in Journal of Alzheimer's Disease Reports
Supplemental Material
sj-docx-3-alr-10.1177_25424823241307019 - Supplemental material for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach
Supplemental material, sj-docx-3-alr-10.1177_25424823241307019 for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach by Reiya Bosco Don Bosco, Johnson Retnaraj Samuel Selvan Christyraj and Beryl Vedha Yesudhason in Journal of Alzheimer's Disease Reports
Supplemental Material
sj-docx-4-alr-10.1177_25424823241307019 - Supplemental material for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach
Supplemental material, sj-docx-4-alr-10.1177_25424823241307019 for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach by Reiya Bosco Don Bosco, Johnson Retnaraj Samuel Selvan Christyraj and Beryl Vedha Yesudhason in Journal of Alzheimer's Disease Reports
Supplemental Material
sj-docx-5-alr-10.1177_25424823241307019 - Supplemental material for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach
Supplemental material, sj-docx-5-alr-10.1177_25424823241307019 for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach by Reiya Bosco Don Bosco, Johnson Retnaraj Samuel Selvan Christyraj and Beryl Vedha Yesudhason in Journal of Alzheimer's Disease Reports
Supplemental Material
sj-pdf-6-alr-10.1177_25424823241307019 - Supplemental material for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach
Supplemental material, sj-pdf-6-alr-10.1177_25424823241307019 for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach by Reiya Bosco Don Bosco, Johnson Retnaraj Samuel Selvan Christyraj and Beryl Vedha Yesudhason in Journal of Alzheimer's Disease Reports
Supplemental Material
sj-docx-7-alr-10.1177_25424823241307019 - Supplemental material for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach
Supplemental material, sj-docx-7-alr-10.1177_25424823241307019 for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach by Reiya Bosco Don Bosco, Johnson Retnaraj Samuel Selvan Christyraj and Beryl Vedha Yesudhason in Journal of Alzheimer's Disease Reports
Supplemental Material
sj-docx-8-alr-10.1177_25424823241307019 - Supplemental material for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach
Supplemental material, sj-docx-8-alr-10.1177_25424823241307019 for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach by Reiya Bosco Don Bosco, Johnson Retnaraj Samuel Selvan Christyraj and Beryl Vedha Yesudhason in Journal of Alzheimer's Disease Reports
Supplemental Material
sj-xlsx-9-alr-10.1177_25424823241307019 - Supplemental material for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach
Supplemental material, sj-xlsx-9-alr-10.1177_25424823241307019 for Synergistic activity of nootropic herbs as potent therapeutics for Alzheimer's disease: A cheminformatics, pharmacokinetics, and system pharmacology approach by Reiya Bosco Don Bosco, Johnson Retnaraj Samuel Selvan Christyraj and Beryl Vedha Yesudhason in Journal of Alzheimer's Disease Reports
Footnotes
Acknowledgments
The authors acknowledge ‘Sathyabama Institute of Science and Technology’ for providing funding support and necessary facilities to carry out the research work. In addition, authors acknowledge, PubChem, IMPPAT, Molinspiration, Swiss ADME, Swiss Target Prediction, The Human Protein Atlas database, String and Cytoscape, as they are various online tools and databases that were employed in carrying out this research work. These databases delineate an entire pathway for drug discovery. Upcoming studies in this field are fully dependent on these tools.
Author contributions
Reiya Bosco Don Bosco (Conceptualization; Formal analysis; Methodology; Writing – original draft); Johnson Retnaraj Samuel Selvan Christyraj (Conceptualization; Formal analysis; Investigation; Methodology; Supervision; Writing – original draft; Writing – review & editing); Beryl Vedha Yesudhason (Conceptualization; Writing – review & editing).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research work was funded by ‘Science and Engineering Research Board (SERB), New Delhi, India under SERB-POWER grant (File Number: SPG/2021/002927)’ and ‘Department of Science and Technology (DST) under Science and Heritage Research Initiative (SHRI), File number DST/TDT/SHRI/26/2021’.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available within the article and in its supplemental material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
