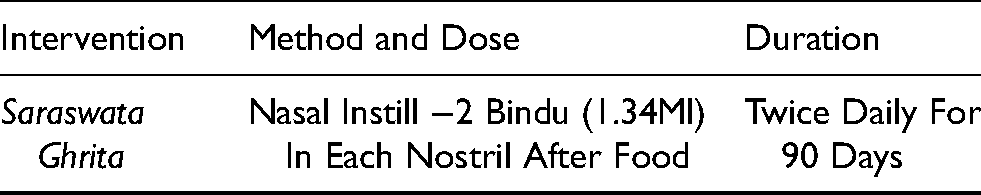
Abstract
Background
Alzheimer's disease (AD) is a progressive neurodegenerative disorder that impairs memory, language, and cognitive functions and currently has no definitive cure. Saraswata Ghrita (SG), a traditional Ayurvedic remedy administered nasally, offers a holistic approach and is believed to directly affect brain functions through its unique delivery route.
Objective
This study aimed to evaluate the effectiveness of SG in improving cognitive function and neurochemical biomarkers in a patient with AD. Key outcomes included electroencephalography-based excitation/inhibition (fE/I) ratio, and levels of phosphorylated Tau-181 (pTau-181), serotonin, dopamine, acetylcholine, and dehydroepiandrosterone (DHEA).
Methods
A 90-day proof-of-concept clinical trial was conducted with one AD patient. Nasal administration of SG was performed twice daily. Measurements included EEG spectral power analysis across 1–48 Hz, cognitive function assessed by Alzheimer's Disease Assessment Scale-Cognitive Subscale (ADAS-Cog), Mini-Mental State Examination (MMSE), Montreal Cognitive Assessment (MoCA), and Quality of Life in Alzheimer's Disease (QoL-AD) scales, and biochemical analyses of pTau-181, serotonin, dopamine, acetylcholine, and DHEA.
Results
Notable improvements were observed: ADAS-Cog score decreased from 40 to 36, QoL-AD score increased from 23 to 31, MMSE score improved from 13 to 18, and MoCA score increased from 8 to 13. Biochemical markers showed a decrease in pTau-181 (12.50 pg/ml to 6.28 pg/ml), an increase in acetylcholine (13.73 pg/ml to 31.83 pg/ml), while serotonin and DHEA levels rose, and dopamine levels decreased (39.14 pg/ml to 36.21 pg/ml).
Conclusions
SG demonstrated potential in enhancing cognitive functions and neurochemical markers in AD, with the nasal route proving safe and effective. These findings suggest the value of traditional Ayurvedic treatments in contemporary AD management.
Introduction
Alzheimer's disease (AD) is a progressive neurodegenerative disorder characterized by the degeneration of nerve cells in brain regions critical for memory, language, and cognition. Early manifestations often include difficulties in memory retention and cognitive processing. The disease is marked by the accumulation of amyloid-β plaques and hyperphosphorylated tau proteins, leading to neuronal damage and loss, making AD a leading cause of dementia, affecting over 55 million individuals globally. 1 Currently, there is no definitive cure for AD; however, several multi-target therapeutic strategies, including amyloid peptide aggregation inhibitors and tau aggregation inhibitors, are under investigation. 2 Eight drugs have received approval for AD treatment, with two addressing the underlying biology of the disease to potentially slow cognitive and functional decline, while six focus on alleviating the symptoms of AD dementia. 3
In India, increasing lifespan and decreasing fertility rates have resulted in a growing number of older persons. 4 By 2050, people over 60 years of age are predicted to constitute 19.1% of the total population. Dementia is currently the seventh leading cause of death among all diseases and one of the major causes of disability and dependency among older people globally. The World Health Organization recognizes dementia as a public health priority. Ayurveda, the ancient system of medicine, provides comprehensive wellness solutions by harmonizing the intricate interplay between physical, mental, and spiritual facets of human existence. In Ayurveda, AD can be conceptualized as a manifestation of dhee (intellect), Dhruti (ability to control impulses), and smruti (memory recall) and raja (agitation) and tama (inertia) mansika dosha. 5 The general line of treatment of mental disorders put forward by seers was non-pharmacological viz. through Daivavyapashraya Chikitsa (Spiritual healing or mantra therapy) and Satvavajaya Chikitsa (psychotherapy/counselling/yoga/meditation), and pharmacological approach through Yuktivyapashraya Chikitsa (rationale therapy/medicinal treatment). These could be instrumental in reducing the progression of neurodegenerative diseases and thus improve overall quality of life for such patients. When we consider the clinical features of AD, we found a formulation named as Saraswata Ghrita (SG) is indicated to induce Vaka (speech), Medha (intellect), Smriti (cognizance), and Vahnikrit (appetizer), quoted in the ancient text of Ayurveda Astanga Hridya. 6 It includes eight ingredients and it is processed with goat's milk and cow's ghee, the details of the SG have been provided in Table 1. The formulation was prepared in the Rasa Shastra and Bhaishajya Kalpana Department laboratory at AIIA, New Delhi, following Ayurvedic Pharmacopoeia of India (API) standards under sterile conditions. 7
Formulation composition of SaraswataGhrita.

Previous work has been reported on SG in relation to AD, demonstrating its potential therapeutic effects. The ingredients of SG exhibit neuroprotective properties, anti-acetylcholinesterase activity, antioxidant effects, and free radical scavenging activity. 8 The medicine is administered through the nasal route (nasya), which Ayurveda describes as the direct and shortest route for the drug to reach the brain (head), depicted as ‘Nasa hi shirso dwaram. 9 (Nasa is the portal (gateway) of Shira (Head). An in vitro study has shown that SG significantly enhances neurotransmitter levels, particularly dopamine (DA) and serotonin (5-HT), by approximately 20%. This enhancement suggests its potential in improving cognitive function and mitigating neurodegenerative processes associated with AD. 10 In vivo research has further validated the neuroprotective properties of SG. A study conducted in 2018 revealed that SG effectively reduced memory impairment in amyloid-β-induced rats, enhancing spatial memory and exhibiting antioxidant activity by modulating oxidative stress markers. 11 A 2021 study utilizing a Drosophila model indicated that SG improved survival rates and reduced amyloid-β plaque accumulation, highlighting its potential to alleviate neurodegenerative damage. 12 Recent findings from 2023 confirmed significant cognitive improvements in mice administered SG, demonstrating its ability to mitigate oxidative stress and inflammation induced by aluminum chloride and d-galactose. 13 A clinical study conducted between 2018 and 2021 reported that SG significantly improved symptoms in AD patients, as measured by the Alzheimer's Disease Assessment Scale-Cognitive Subscale (ADAS-Cog) and Quality of Life in Alzheimer's Disease (QoL-AD) scales. Although this study relied primarily on subjective parameters, it lays the groundwork for future research to incorporate biochemical measures to further validate SG's therapeutic efficacy. 14
In this report, we document a case of AD treated with SG. Our objective was to provide empirical evidence to the scientific community concerning electroencephalography (EEG)-based functional excitation-inhibition (fE/I) ratio and the biochemical parameters specifically pTau-181 protein. EEG is a non-invasive, cost-effective method used to record the brain's electrical activity through scalp electrodes, providing high-resolution, real-time data. 15 It plays a crucial role in AD research and diagnosis by detecting brain wave anomalies. 16 and monitoring changes in the cerebral cortex before symptoms appear. 17 EEG-based biomarkers evaluate neuronal degeneration in AD by reflecting the balance between excitation and inhibition (E-I) in brain networks, essential for understanding brain dysfunction and cognitive impairment. EEG recordings capture these activity shifts, offering insights into AD progression and underlying brain changes, which are crucial for analyzing treatment efficiency. The spectral slowing observed in AD patients, typically associated with reduced E–I balance, has been alternatively linked to increased excitatory neuronal activity in computational modeling studies.18,19 Plasma p-tau181, phosphorylated at threonine 181, is emerging as a specific, accessible, and scalable biomarker for AD, serving both diagnostic and prognostic purposes. 20 We have tried to generate evidence pertaining to efficacy of SG in AD through changes in EEG based fE/I ratio and pTau181 protein.
Case report
Patient information
A 60-year-old right-handed woman was found on the roadside, and she was taken up to the old age home by a rescue team member, native of Uttar Pradesh. She could read and understand Hindi language, with 8 years of school education. Her marital history includes her husband remarrying and divorcing her, the first wife being the patient herself. According to caregivers at the old-age home, the patient began exhibiting signs of forgetfulness and declining pronunciation around the age of 55–56. She had a 4–5-year history of memory impairment, apraxia, agnosia, slow response, apathy, and disturbance in executive functions. The patient underwent F-FDG brain PET-CT scan to ascertain the cause of dementia. She was unable to do daily activities for 2–3 years as per a Katz Index of Independence in Activities of Daily Living (ADL) score of 5/6 [14] and reported disturbance of sleep for 2 years. 21 The patient was assessed for depression, which found a mild mood disturbance. This suggests that while mild depression was present, it is unlikely to have significantly contributed to the observed cognitive and neurophysiological improvements during the study. Furthermore, the patient's kidney function tests were within the normal range both before and after the treatment period, ruling out any significant creatinine fluctuations as a factor influencing changes in pTau-181 levels. The patient's body mass index (BMI) was recorded at 26.4, classifying them as slightly overweight, and remained consistent throughout the duration of the study.
Ethical considerations
Approval from an institutional review board or ethics committee was obtained with Ref. No. IEC-331/27.06.2023/Rp(E)-12/2023, dated: 17/08/2023.
Before the start of the clinical trials, informed consent was obtained from the participant.
Clinical Trials Registry-India (CTRI) Number: CTRI/2023/10/058237, registered on 04/01/2023.
Clinical findings
Neurological examination
Her short-term memory had gradually declined. Her speech was monosyllabic. She was disoriented to time, place, and person; her sleep disturbance became prominent, and frequently woke up in middle of the night. She was emotionally unstable, grumpy, and easily tearful, which was alleviated when the caregiver comforted her. The patient exhibited a lack of initiative such as reduced interest in previously enjoyed activities. Also, her writing was slower, messier, and more difficult (dysgraphia). The neurological examination revealed normal muscle tone and strength, with the exception of mild ideomotor apraxia observed during the execution of motor commands. Gait was assessed as normal. Frontal release signs were not observed. The patient exhibited notable difficulties in word retrieval, though without phonemic or semantic paraphrasing errors. She had significant difficulty naming pictured objects (anomia); she would often be able to describe the object but unable to say the correct name. Her general physical examination was unremarkable, with no signs of presenile cataracts, and normal hearing was confirmed. She was the case of early onset of AD.
Medical history
The patient was unable to provide her family history or her own past medical and surgical conditions. Currently, she is taking multi-vitamins but is not on any medication for neuro-psychiatric conditions.
Social history
She had completed 8 years of school education. She was a homemaker.
Laboratory testing
Her routine laboratory findings viz complete blood count, erythrocyte sedimentation rate, liver function test, and kidney function test did not reveal any abnormalities.
Timeline
The timeline of the case, divided into objective and subjective parameters, before and after the intervention, is depicted in Tables 2 and 3.
Assessment based on subjective criteria.
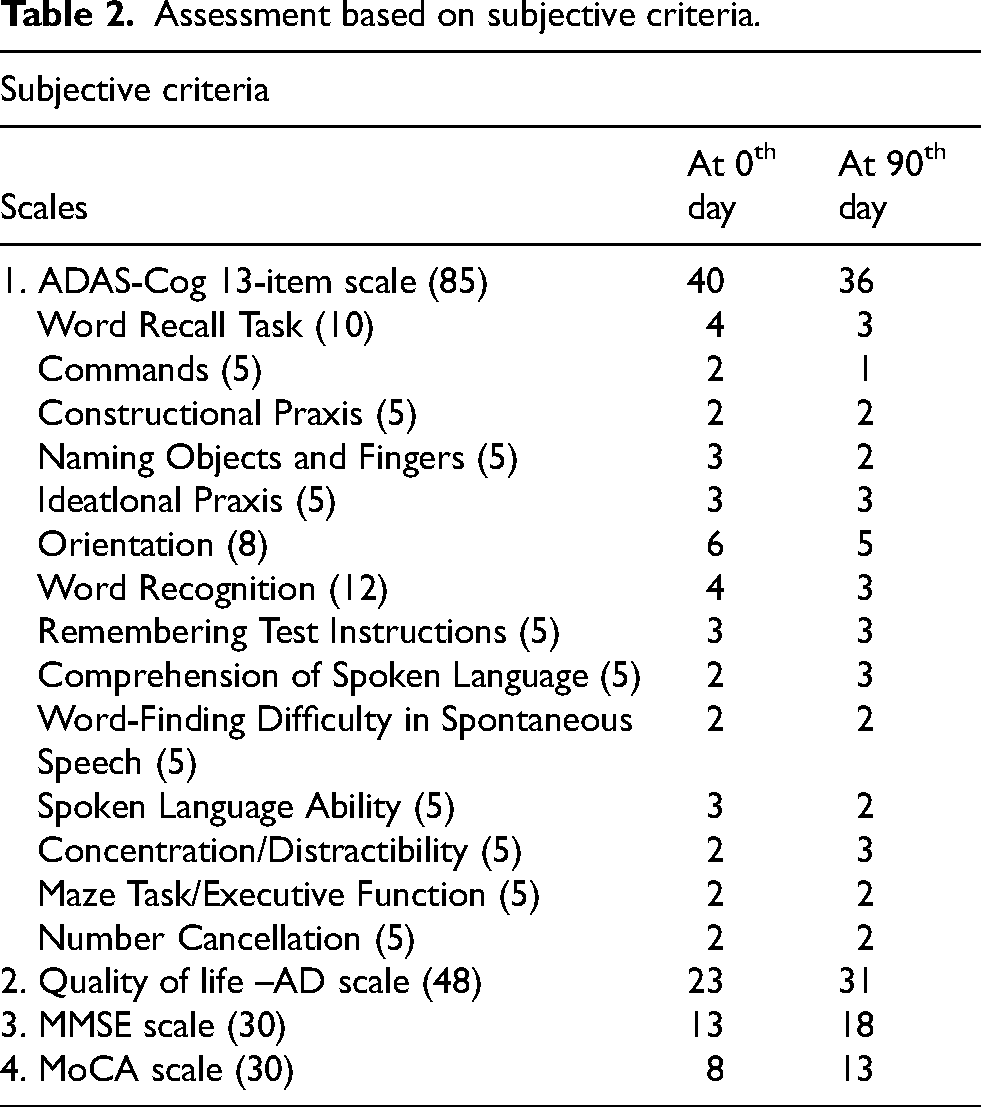
Assessment based on subjective criteria.
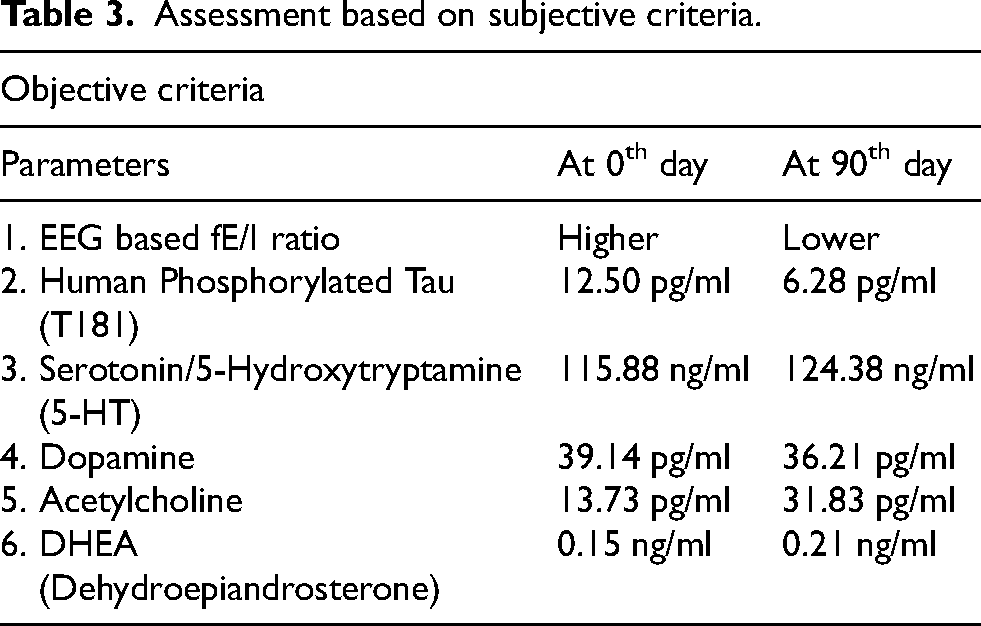
Diagnostic assessment
Upon her psychiatric evaluation, the neuropsychological test was implemented to evaluate the patient's cognitive status. She scored 13 out of 30 in the Mini-Mental Status Examination (MMSE) and 8 out of 30 in Montreal Cognitive Assessment (MoCA). 22 The patient exhibited severe impairments in orientation (2/10), Registration (2/3), attention and calculation (0/5), recall (1/3), language (8/8) and visual construction (0/1) during MMSE. The patient exhibited impairment in visuospatial/executive (0/5), naming (2/3), attention (0/6), language (2/3), abstraction (0/2), delayed recall (3/5) and orientation (1/6) during MoCA. Diagnosis was made as per DSM-IV criteria. Analysis of her brain activity using EEG 64-channels revealed lower detrended fluctuation analysis values in the extended alpha band (6–13 Hz) in the parieto-occipital and hippocampal regions. A fE/I (excitation to inhibition) ratio of ∼ 1.0 indicates balanced brain activity, but her fE/I ratio was < 1.0, suggesting more inhibition-dominated activity. The patient's history, F-FDG brain PET-CT scan, along with EEG data and cognitive test results all were indicative for AD diagnosis.
Therapeutic intervention
Assessment criteria
Subjective criteria for assessing cognitive function and quality of life in AD at the 0th and 90th day intervals included the ADAS-Cog, QoL-AD, and cognitive screening tools like the MMSE and MoCA scales depicted in the Table 2. These instruments provide a comprehensive understanding of cognitive decline, daily functioning, and overall quality of life, guiding personalized care and intervention strategies for individuals with AD.
Objective criteria for assessing AD progression at the 0th and 90th day intervals encompass a range of biomarkers and physiological parameters. These include EEG-based fE/I ratio reflecting neural network balance, levels of tau phosphorylated at threonine 181 (pTau181) indicative of neurofibrillary tangle formation, and neurotransmitter levels such as serotonin, dopamine, and acetylcholine associated with cognitive function and synaptic transmission. Additionally, biomarker like DHEA (dehydroepiandrosterone) was also evaluated depicted in the Table 3. ELISA-based kits were procured from their respective laboratories, and the assays were conducted according to the manufacturers’ guidelines. The human phosphorylated tau (T181) ELISA Kit (CAT#HR2E1001) used to measure phosphorylated tau levels was purchased from Bioelsa Lab, USA. Dopamine (DA) levels were assessed using the Dopamine ELISA Kit (CAT#E-EL-0046) from Elabscience Biotechnology Inc., USA. Acetylcholine levels were measured with the Acetylcholine ELISA Kit (CAT#E-EL-0081), also from Elabscience Biotechnology Inc., USA. Additionally, the Human DHEA (Dehydroepiandrosterone) ELISA Kit (CAT#E-EL-0115) and the Serotonin/5-Hydroxytryptamine (5-HT) ELISA Kit (CAT#E-EL-0033), both from Elabscience Biotechnology Inc., USA, were used to determine DHEA and serotonin levels, respectively. The findings are depicted in Table 3, showing the values before and after the intervention. The spectral power across frequencies from 1–48 Hz was evaluated in EEG study. Mean power of each frequency bin for 0th and 90th day was compared during eye-closed and eye-open resting state EEGs in the parieto-occipital cortex and hippocampus regions.
Follow-up and outcomes
On the ADAS-Cog scale, the initial score stood at 40 out of 85, indicating a significant level of cognitive impairment. Following a 3-month intervention period, there was a notable improvement, with the score decreasing to 36. This improvement represents a 4-point reduction in cognitive impairment. Among the 13 domains assessed by the ADAS-Cog scale, 6 demonstrated improvement, including word recall task, commands, naming objects and fingers, orientation, word recognition, and spoken language ability. These improvements suggest positive responses to the intervention in various cognitive functions. However, despite the overall improvement, deterioration was observed in two domains: comprehension of spoken language and concentration/distractibility.
In the QoL-AD scale, the initial score was 23 out of 48, indicating a baseline assessment of quality of life. Following treatment, the score improved to 31, reflecting a significant 7-point improvement. This improvement was observed across various domains of the scale, including physical health, energy levels, mood, memory, social interactions with friends, self-perception as a whole, and the ability to perform chores around the house. These positive changes in quality of life domains suggest a beneficial response to the intervention, with improvements observed in both physical and psychosocial aspects of daily functioning in individuals with AD.
The patient initially scored 13 out of 30 on the MMSE. Subsequent improvements were noted in multiple domains: orientation (5/10), registration (2/3), attention and calculation (1/5), recall (2/3), language (8/8), and visual construction (0/1).
On the MoCA scale, the patient demonstrated improvement in various cognitive domains. Specifically, enhancements were observed in visuospatial/executive function (1/5), naming abilities (2/3), attention (2/6), language skills (2/3), delayed recall (3/5), and orientation (3/6). The details of the scores on various scales are presented in Table 2. The changes illustrated in Figure 1(a)-(d) show the variations in the respective scales.
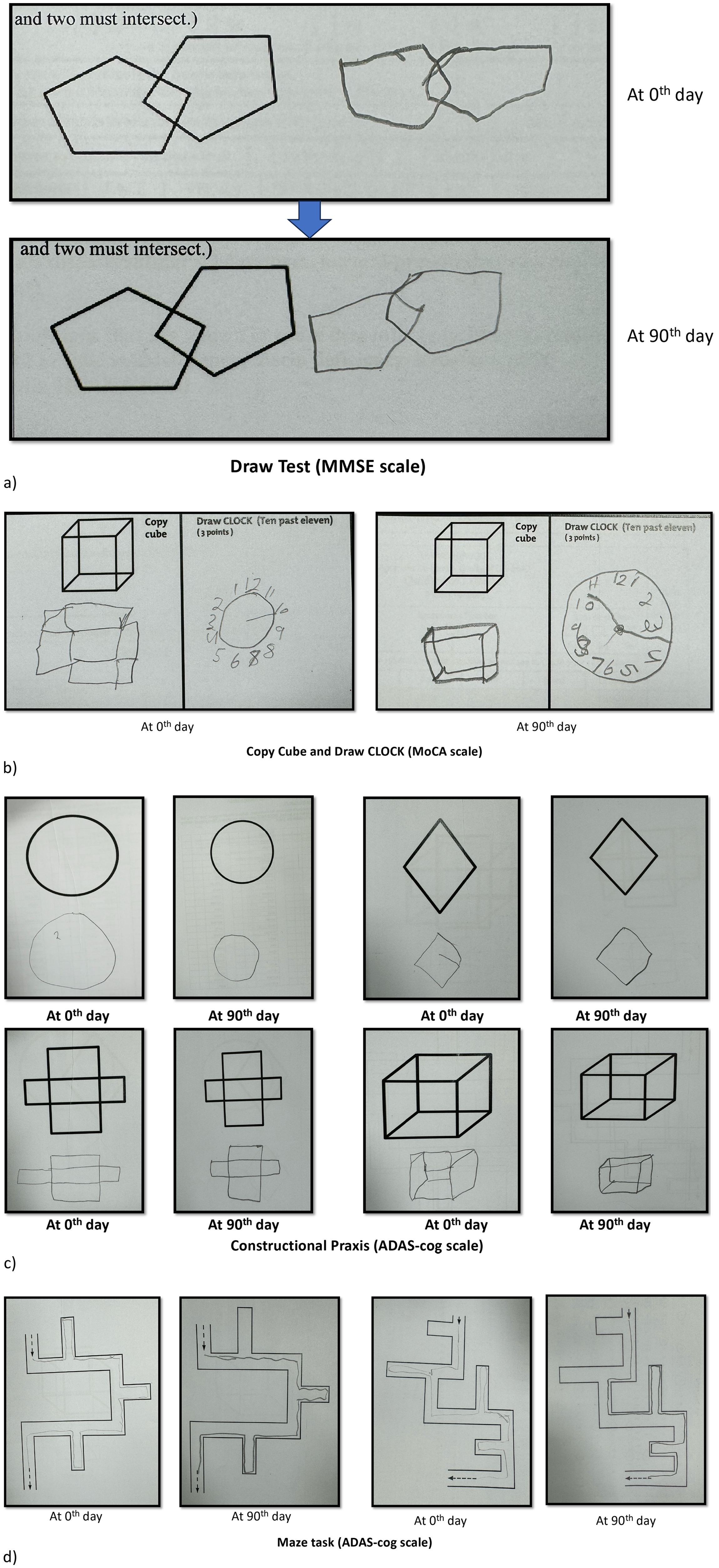
Panels (a-d) illustrate the variations observed in the respective scales.
Levels of phosphorylated Tau181 (pTau181) also decreased from 12.50 ng/ml at the start to 6.28 pg/ml after 90 days, suggesting a potential improvement in AD pathology. Serotonin levels increased slightly from 115.88 ng/ml to 124.38 ng/ml, while dopamine levels decreased from 39.14 pg/ml to 36.21 pg/ml. Acetylcholine levels notably increased from 13.73 pg/ml to 31.83 pg/ml, indicating enhanced cholinergic activity. Additionally, DHEA levels rose from 0.15 ng/ml to 0.21 ng/ml, suggesting a beneficial hormonal response to the intervention. These findings collectively suggest a positive impact on both neurochemical and neurophysiological parameters following the intervention period.
The EEG analysis demonstrated improved neurophysiological measures, particularly focusing on the fE/I (excitation to inhibition) ratio. After 90 days of medication, there was a relative decrease in the fE/I balance, indicating a trend towards hypoactive and silent neurons. This change suggests a shift in brain activity towards inhibition-dominated patterns.
Patient perspective
From the perspective of the old age home caretaker, the patient exhibited noticeable improvements over time. Initially, she was resistant to nasal administration, but after 4–5 days, she became more cooperative with the treatment. Over the course of 90 days, she gradually became more engaged and interactive with the staff, showing a significant increase in her willingness to participate in daily activities. The caretaker observed that she appeared more relaxed and seemed to enjoy her daily routine much more than before.
Discussion
In AD, the progression of cognitive decline can be metaphorically related to the Bhagavad Gita, Chapter 2, Verse 63: “From anger follows delusion; from delusion, failure of memory; from failure of memory, the loss of understanding; from the loss of understanding, he perishes.” This sloka captures the sequential cognitive deterioration in AD. Initially, AD patients often experience emotional disturbances like irritability and anger, leading to confusion and memory loss. As the disease progresses, memory failure becomes more pronounced, leading to significant cognitive decline and loss of understanding. 23 The Gita's wisdom provides a profound framework for understanding the interconnectedness of emotional, cognitive, and existential decline in AD.
The main pathological hallmarks of AD include not only amyloid-β plaques and neurofibrillary tangles but also neuroinflammation, altered insulin resistance, oxidative stress, and cerebrovascular abnormalities. Targeting plasma pTau-181 in AD involves multiple therapeutic strategies aimed at reducing tau phosphorylation and aggregation, as well as promoting its clearance. 24 As we observe decrement of the Tau-181 value from 12.50 to 6.28 pg/ml, protein in AD after the intervention of medication is a critical indicator of treatment efficacy. Tau-181, a phosphorylated form of tau protein, is closely associated with neurofibrillary tangles found in AAD patients. Elevated levels of tau-181 are correlated with disease severity and progression. Successful therapeutic interventions aim to reduce these levels, thereby potentially slowing or halting disease progression. Studies have shown that certain medications can significantly lower plasma tau-181 concentrations, reflecting a decrease in neuronal damage and improved clinical outcomes.25–27 Terminalia chebula (TC) primarily focused on acetylcholine modulation, its ability to reduce amyloid beta production may indirectly influence tau pathology by alleviating upstream amyloidogenic stress. 28 Moringa oleifera by reducing amyloid-β levels and enhancing synaptic plasticity, it potentially impacts tau pathology indirectly, although direct effects on Tau-181 are less documented. 29
After the intervention, acetylcholine levels increased significantly from 13.73 pg/ml to 31.83 pg/ml. Acetylcholine is a key neurotransmitter in the brain, with significant activity in the cortex, basal ganglia, and basal forebrain. 30 Cholinesterase inhibitors increase the availability of acetylcholine at synapses in the brain. This substantial rise indicates a potential improvement in cholinergic function, which is crucial for cognitive processes. Such findings are supported by studies demonstrating the role of acetylcholine in enhancing neural communication and cognitive function. 31 The TC, known for its acetylcholinesterase (AChE) inhibitory effects, further reinforces this point. TC contains compounds like chebulinic acid and punicalagin that have demonstrated strong AChE inhibition in both in silico and in vitro studies, potentially contributing to the preservation of acetylcholine levels and cognitive improvements.28,32,33 Cissampelos pareira and Acorus calamus both show significant AChE inhibitory potential, enhancing cholinergic transmission.34,35
The neurotransmitter serotonin (5-hydroxytryptamine, 5-HT) is crucial for regulating mood, cognition, and overall neural health. In our study, a noteworthy elevation in serum 5-HT levels was observed, rising from 115.88 ng/mL before intervention to 124.38 ng/mL afterward. This significant increase suggests that the intervention may have positively influenced serotoninergic activity. Enhanced serotonin levels are known to be associated with improved neuropsychiatric outcomes, which could imply that the intervention not only aids in symptom management but also holds potential for neuroprotective effects.36,37 Piper nigrum:-Piperine acts on presynaptic 5-HT1A receptors, linking it to serotonin signaling alterations, thereby potentially impacting mood and cognition in AD. 38 Zingiber officinale enhances serotonin receptor activity, which may help modulate mood and behavior aspects of AD. 39
DA levels in AD patients decreased from 39.14 pg/mL before the intervention to 36.21 pg/mL afterward. This decline suggests a possible reduction in dopaminergic function, which plays a critical role in cognitive and motor processes. While the decrease in dopamine levels indicates that the intervention may not have had the desired positive impact on dopaminergic pathways. 40 In the brain, DHEA acts as a neurosteroid, modulating neurotransmitter receptors and influencing neuronal growth and survival. Acorus calamus is known for enhancing DA receptor binding in neuroprotective studies, potentially benefiting DA-related cognitive functions (protective effect of Acorus calamus against acrylamide). The baseline level of DHEA was 0.15 ng/ml, which increased to 0.21 ng/ml post-intervention. It has been shown to have both neuroprotective and neurotrophic effects, DHEA is believed to exert neuroprotective effects by modulating neurotransmitter receptors such as NMDA (N-methyl-D-aspartate) and GABA (gamma-aminobutyric acid) receptors. This modulation can influence neuronal excitability and has been associated with improved mood and cognitive functions. 41
EEG study revealed that on the 0th day, the parieto-occipital cortex showed significantly higher power in the extended alpha frequencies (∼6–13 Hz). In higher frequency ranges, the parieto-occipital cortex exhibited lower power, contrasting with observations in the hippocampus. The extended alpha frequency range was thus used for the excitation to inhibition (E-I) ratio analysis. (Figure 2(a), (c), (e) and (g)). Figure 3 shows the cortex map of relative power alterations in the extended alpha range for the 0th and 90th day. Higher power in this frequency range was observed in both the parieto-occipital region and hippocampus. Spectral increment is intuitively associated with reduced E–I balance. Several computational modelling studies have linked this with the decreased excitatory neuronal activity instead.18,42 With prolonged medication use, there was less disruption in the E-I ratio, indicating a shift towards lower excitation or higher inhibition patterns. This is reflected by a lower fE/I during rest. An fE/I < 1.0 is associated with pathological network imbalance and an inhibition-dominated state.43,44 On the 0thday, the median fE/I was relatively higher compared to the 90th day, suggesting less inhibition and more balanced networks initially.
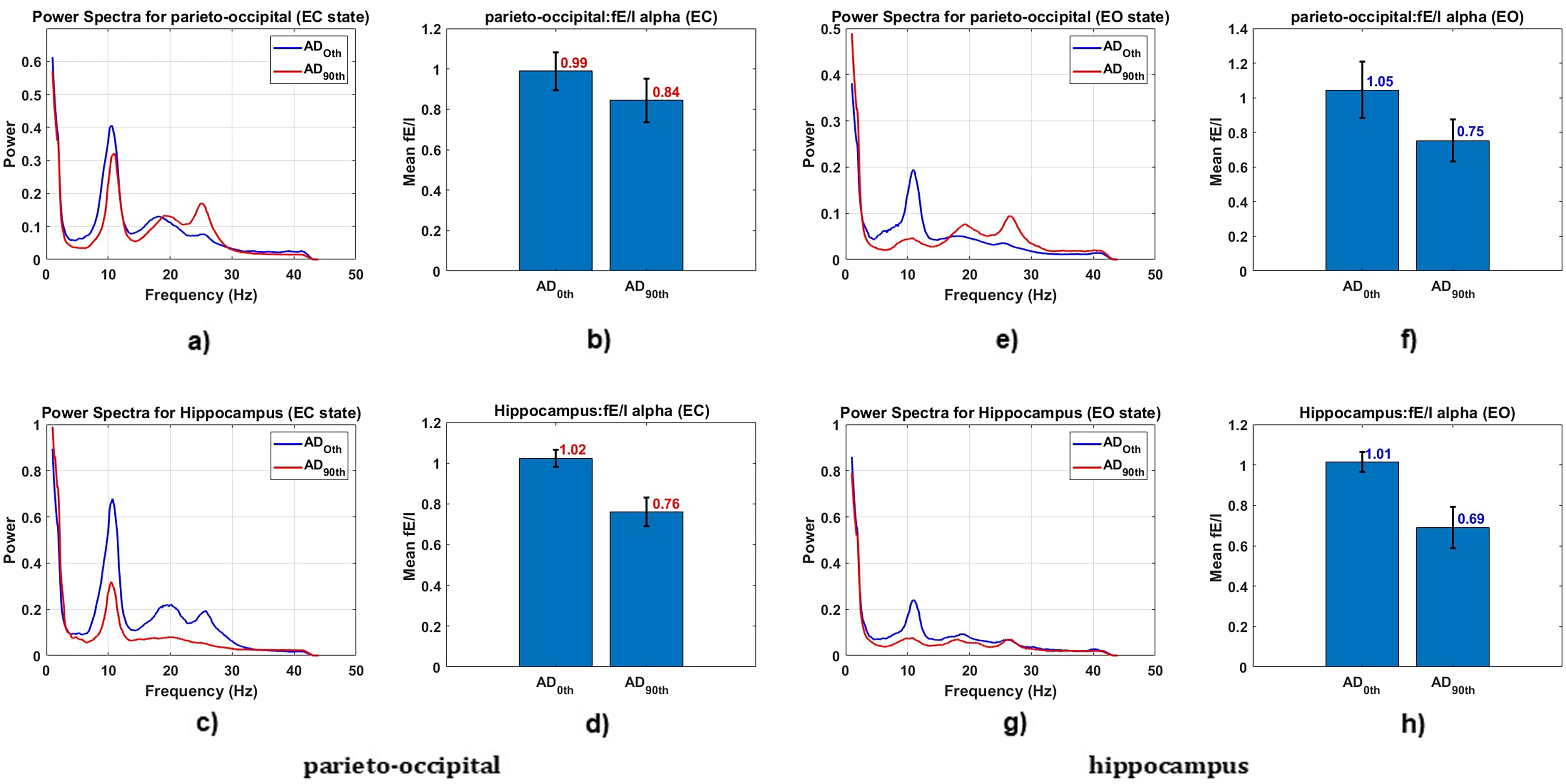
Mean power spectral and fE/I ratio of parieto-occipital and hippocampus regions during eye-closed and eye-open conditions. (a) and (e) show mean power spectral for parieto-occipital and hippocampus during eye-closed conditions. (b) and (f) depict mean fE/I ratio for parieto-occipital and hippocampus during eye-closed conditions. (c) and (g) display mean power spectral for parieto-occipital and hippocampus during eye-open conditions. (d) and (h) illustrate mean fE/I ratio for parieto-occipital and hippocampus during eye-open conditions.
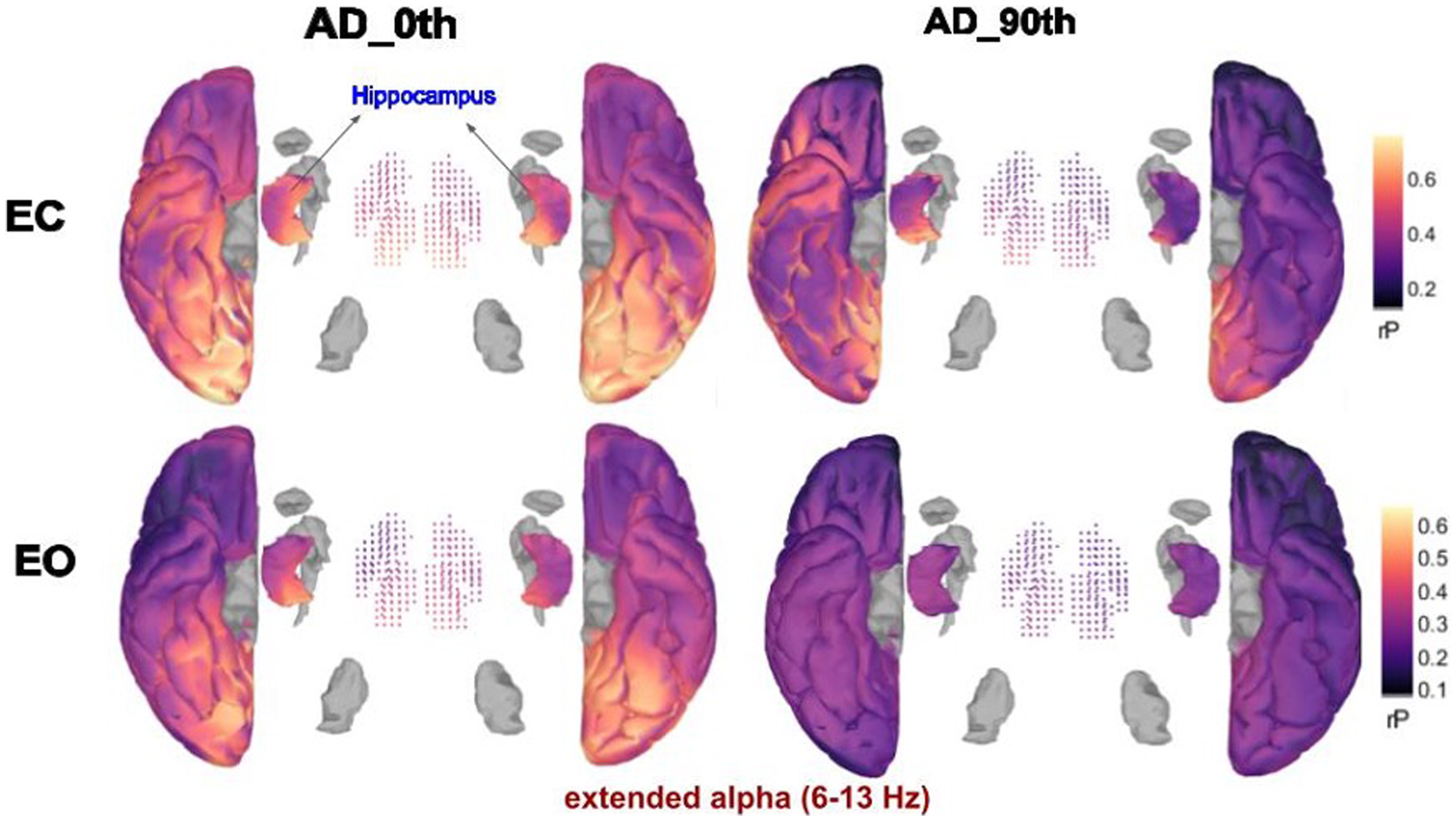
Cortex and hippocampus map representation showing relative power alterations in the extended alpha frequency range observed on the 0th and 90th days. The color gradient represents the rP, with warmer colors indicating higher power and cooler colors indicating lower power. These changes suggest a shift in brain activity, potentially reflecting the progression or response to treatment in AD.
In this case report, significant improvements were observed across multiple cognitive and quality of life domains following a three-month intervention in a patient with AD. The initial ADAS-Cog score of 40, indicative of substantial cognitive impairment, decreased to 36 post-treatment, demonstrating efficacy in enhancing cognitive function. 45 Specifically, improvements were noted in tasks such as word recall, orientation, and language abilities, aligning with previous studies highlighting targeted cognitive gains in AD interventions. Despite overall improvement, challenges persisted in comprehension of spoken language and concentration, underscoring the complexity of cognitive responses to treatment. Quality of life, assessed using the QoL-AD scale, improved significantly from a baseline score of 23 to 31, with enhancements observed in physical health, mood, memory, and social interactions post-intervention. 46 EEG neurophysiological analysis revealed a shift towards hypoactive and inhibition-dominated neuronal patterns, corroborating findings that AD interventions can influence underlying brain activity.
These findings highlight the comprehensive benefits of integrated treatment approaches in AD, leveraging both cognitive assessments and neurophysiological measures to optimize therapeutic outcomes effectively. This collectively suggests SG's positive impact on neurochemical and neurophysiological parameters following intervention (summary depicted in Figure 4). An in vivo study reported that SG (3 ml/kg, orally) showed significant neuroprotective effects in aluminum chloride and d-galactose-administered mice. SG improved cognitive functions by reducing escape latency time, enhancing target quadrant performance, and decreasing trial numbers in reversal learning and executive functioning. It also prevented locomotor decline, reduced oxidative stress, and lowered brain inflammatory markers, including TBARS and MPO, while increasing GSH levels compared to the disease control group. 13 Several studies have demonstrated the ingredients of SG act as neuroprotective effect against AD. In an animal study, ginger (500 mg/kg) effectively mitigated monosodium glutamate-induced neurotoxicity in rats by reducing lipid peroxidation, nitric oxide, and amyloid-β accumulation, while restoring neurotransmitter levels and antioxidant markers like glutathione and superoxide dismutase. 47 In SH-SY5Y cells, [6]-gingerol from ginger significantly suppressed amyloid-β-induced oxidative stress and apoptosis through Nrf2 activation, enhancing γ-glutamylcysteine ligase and heme oxygenase-1 expression. 39 In clinical studies, 800 mg/day of Zingiber officinale improved cognitive function, increasing P300 amplitude and accuracy in choice reaction tasks, alongside improvements in working memory. 48 Another study on rats with AD induced by aluminum chloride found that ginger (216 mg/kg/day) significantly improved behavioral function, increased brain acetylcholine, and decreased AChE levels, similar to rivastigmine. 49 Piper nigrum extract reduced cholinesterase levels by 25.69% at 200 mg/kg in AD rats and improved cognitive function in the Morris water maze test, reducing escape latency by 67%. 50 Piperine (2.5 mg/kg) improved memory and reduced oxidative stress in an AD model, as shown by decreased malonaldehyde levels and increased antioxidant activity. 51 Moringa oleifera's active compound, niazimicin, reduced amyloid-β levels by 59% and increased glutathione by 54% in a dementia model, suggesting its neuroprotective potential through caspase-3 inhibition. 52
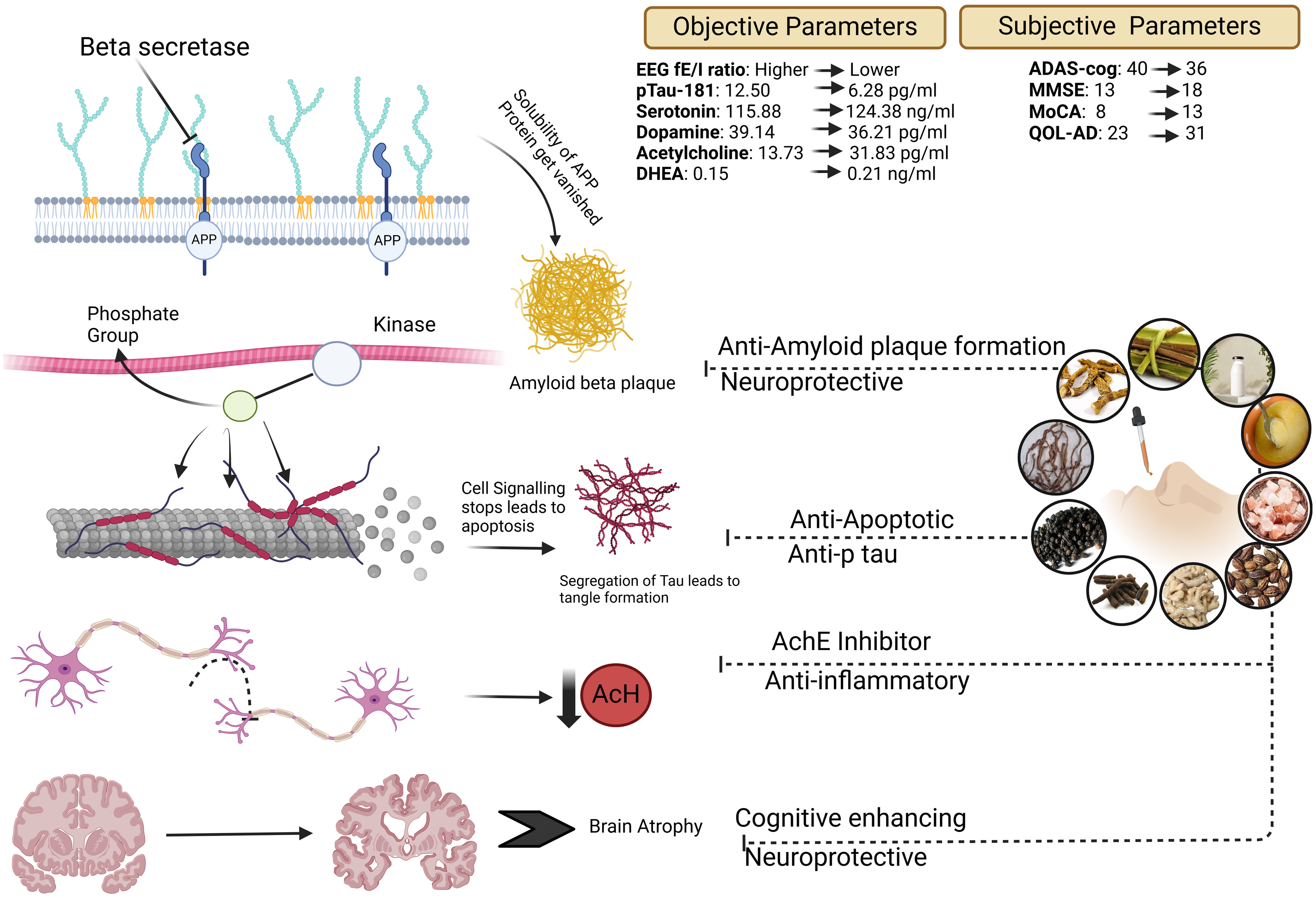
Overview of the study workflow and outcomes.
The drug administered through nostrils, reaches Shringataka which is a composite structure consisting of four Sira (Vessels) in connection with four sense organs i.e., nose, ear, eye and tongue and then spreads in the Murdha (Brain) taking route of Sringataka Marma and scratches the morbid Doshas in supra clavicular region and eliminates them efficiently. 53 This classical concept correlates with Nasal drug delivery operates through the highly vascularized nasal mucosa, allowing for rapid and efficient absorption directly into the systemic circulation, bypassing the gastrointestinal tract and first-pass metabolism. This route offers quick onset of action and direct access to the central nervous system via the olfactory and trigeminal nerves, making it effective for neurological treatments. This method is advantageous for drugs requiring rapid delivery or those unstable in the digestive system. 54
Conclusion
This proof-of-concept clinical study found that Nasya (nasal instillation) therapy was safe, tolerable, and free of any untoward adverse effects for long-term use in adult subjects with same dose as per the classics. Therapeutic interventions showed significant improvements, with reductions in plasma tau-181 levels and increases in acetylcholine and serotonin, indicating improved cognitive and neuropsychiatric outcomes. Despite a slight decrease in dopamine levels, the increase in DHEA demonstrated neuroprotective effects. The case report detailed a patient's cognitive and quality of life improvements post-intervention, supported by EEG analysis, demonstrated improved neurophysiological measures, particularly focusing on the fE/I ratio. After 90 days of medication, there was a relative decrease in the fE/I balance, indicating a trend towards hypoactive and silent neurons. This change suggests a shift in brain activity towards inhibition-dominated patterns. The EEG results highlight the potential of EEG-based biomarkers in monitoring treatment effects in AD. Overall, integrated treatment approaches, including SG, demonstrate promising effects on neurochemical and neurophysiological parameters in AD.
Footnotes
Acknowledgments
We extend our heartfelt gratitude to Mrs. Ritu Bharadwaj, Blood Bank Technical Supervisor, for her indispensable assistance with the ELISA kit assessments. We are also deeply thankful to Dr Ishan Bharti, Radiologist, and Dr Aditya Dixit, Consultant Intensivist (ICU), for their expert guidance in the case diagnosis. We would like to thank Dr Deenadayal Devarajan, Phd scholar, Department of Shalakya Tantra and Dr Arushi Saroch, PG scholar of Department of RS&BK for creating the graphical presentation for this case report. Lastly, we would like to extend special thanks to Dr G.P. Bhagat, Founder of SHEOWS Guru Vishram Vridh Ashram, for his support in facilitating the availability of the patient.
ORCID iDs
Author contributions
Robin Badal (Conceptualization; Data curation; Investigation; Methodology; Writing – original draft); Shivani Ranjan (Data curation; Investigation; Writing – original draft); Lalan Kumar (Formal analysis; Methodology; Supervision; Validation; Visualization); Lokesh Shekhawat (Supervision; Validation); Ashok Kumar Patel (Supervision; Validation); Pramod Yadav (Formal analysis; Methodology; Supervision; Validation; Writing – review & editing); Pradeep Kumar Prajapati (Resources; Supervision; Validation).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to the Researchers Supporting Project number RP04502G by IIT Mandi iHub and HCI Foundation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available within the article.