Abstract
Background
Behavioral variant frontotemporal dementia usually presents with behavioral and personality changes, social disinhibition, apathy, and lack of empathy, and is characterized by atrophy of the frontal and temporal lobes. Corticobasal syndrome is characterized by asymmetrical involuntary movements, rigidity, apraxia, tremor, dystonia, and cortical sensory deficits.
Objective
We present the case of a 59-year-old patient with a frontotemporal presentation and parkinsonism linked to progranulin gene deletion. We also report the clinical workup needed to reach the diagnosis.
Methods
Clinical, neuropsychological, computed tomography, magnetic resonance imaging, 18F-fluorodeoxyglucose and 18F-Flutemetamol positron emission tomography (PET), dopamine-transporter-single-photon emission computed tomography imaging, electroencephalography, and genetic evaluations were conducted.
Results
Our patient presented initially with executive and mnesic deficits along with the presence of apathy and loss of autonomy. Subsequently the cognitive deficits became associated with parkinsonian-like movement disorders and apraxia. Structural images showed right onset temporal and insular atrophy, and the PET images demonstrated right frontotemporal hypometabolism and the absence of amyloid in the cortex. The molecular analysis revealed a heterozygous deletion c.813_816delCACT on the GRN gene. This variant has been reported in the literature as pathogenic and associated with autosomal dominant frontotemporal dementia and corticobasal degeneration. Our patient presented different clinical features than those of the members of the families already described. In these families, some patients either presented immediately with motor syndrome with extrapyramidal features, or never developed extrapyramidal signs. Some subjects presented prevalent language dysfunction while others never presented memory disorders.
Conclusions
The clinical case highlights the phenotypic variability of this entity.
Keywords
Introduction
Frontotemporal lobar degeneration (FTLD) encompasses highly diverse clinical manifestations characterized by atrophy of the frontal and temporal lobes. 1 It is associated with different neuropathological substrates which affect these same areas and produce neuronal cell death and gliosis. 2 The current neuropathological classification is based on the predominant protein abnormality and allows most cases of frontotemporal dementia (FTD) to be placed into one of three broad molecular subgroups: frontotemporal lobar degeneration with tau-positive inclusions (FTLD-tau), frontotemporal lobar degeneration with TDP-43-positive inclusions (FTLD-TDP), and frontotemporal lobar degeneration with FET-positive inclusions (FTLD-FET). 2
FTLD clinical presentation includes: behavioral variant frontotemporal dementia (bvFTD), nonfluent/agrammatic variant primary progressive aphasia (nfvPPA), and semantic variant primary progressive aphasia (svPPA). 2 bvFTD usually presents with changes in behavior and personality, social disinhibition, apathy, and lack of empathy.2,3 PPA variants, instead, consist of various language disorders. 3 FTLD represents the third most common form of neurodegenerative dementia after Alzheimer's disease (AD) and dementia with Lewy body (DLB), and affects men and women equally. 4 The typical age of onset is between 45 and 65 years.4,5
In magnetic resonance imaging (MRI), FTD usually presents with atrophy in frontal and temporal lobes, insula, and anterior cingulate cortex. 6 Positron emission tomography with 18F-fluorodeoxyglucose tracer (18F-FDG PET) typically shows hypometabolism in the orbitofrontal cortex, anterior temporal poles, and basal ganglia. 6
Over half of FTD cases are sporadic, but up to 40% of cases have a family history of dementia, with at least 10% of cases having an autosomal dominant pattern. The three most common genes associated with FTD are a hexanucleotide repeat, GGGGCC, in chromosome 9 (C9ORF72), Microtubule Associated Protein Tau (MAPT) and Progranulin (GRN) gene. 2
Corticobasal syndrome (CBS) is characterized by asymmetrical involuntary movements, rigidity, apraxia, tremor, dystonia, alien limb phenomena, myoclonus, and cortical sensory deficits. CBS is associated with several distinct histopathologies, which share the presence of tauopathy, including corticobasal degeneration (CBD), AD, and other forms of FTLD-tau. 7
CBS was first described in 1967 and 1968, when Rebeiz et al.8,9 reported the clinical and neuropathological features of three patients with a syndrome they called “corticodentatonigral degeneration with neuronal acromasia”. Gibb and Marsden coined the term CBD in 1989. 10 The clinical features described by Rebeiz et al. were associated with different neuropathological phenotypes. Consequently, two distinct terms were adopted: CBD, in which the pathological findings described by Rebeiz et al. are present, and CBS, which refers to the clinical phenotype independent of the underlying histopathology. 7
The present paper reports the case of a patient with a frontotemporal presentation and parkinsonism linked to a deletion in progranulin gene, along with the clinical workup needed to reach the diagnosis.
Case presentation
A 59-year-old right-handed man, a retired paper mill worker, was referred to the Treviso Cognitive Impairment Center (TCIC) in October 2018 complaining of amnesic deficits, such as not remembering where he stored objects, and of temporal disorientation, over the course of a couple of years. He had completed 8 years of schooling. Currently living with his wife, he had a son and a daughter. He consumed wine occasionally and had never smoked.
A diagnosis of dementia was reported in a brother (FTD), in the mother (AD), in two maternal aunts and in two cousins, that is, the sons of one of the aunts affected. He suffered from arterial hypertension, glycemic intolerance, mild hypercholesterolemia, hypertensive heart disease and obstructive sleep apnea for which he benefited from a continuous positive airway pressure (CPAP) machine at night. He was on therapy with Irbesartan/hydrochlorothiazide 150/12.5 mg 1 tablet a day and with a hypoglycemic diet.
His wife also reported that the patient appeared listless, and often complained of feeling tired over the last two years; he also appeared more distracted than in the past and showed some difficulties in judgment. Changes in routine activities, abulia, inactivity, asthenia, were reported. He also needed assistance with some activities of daily living.
A brain MRI, performed on December 29, 2017, before the patient was referred to the TCIC, showed a substantially normal brain morphology (Figure 1).
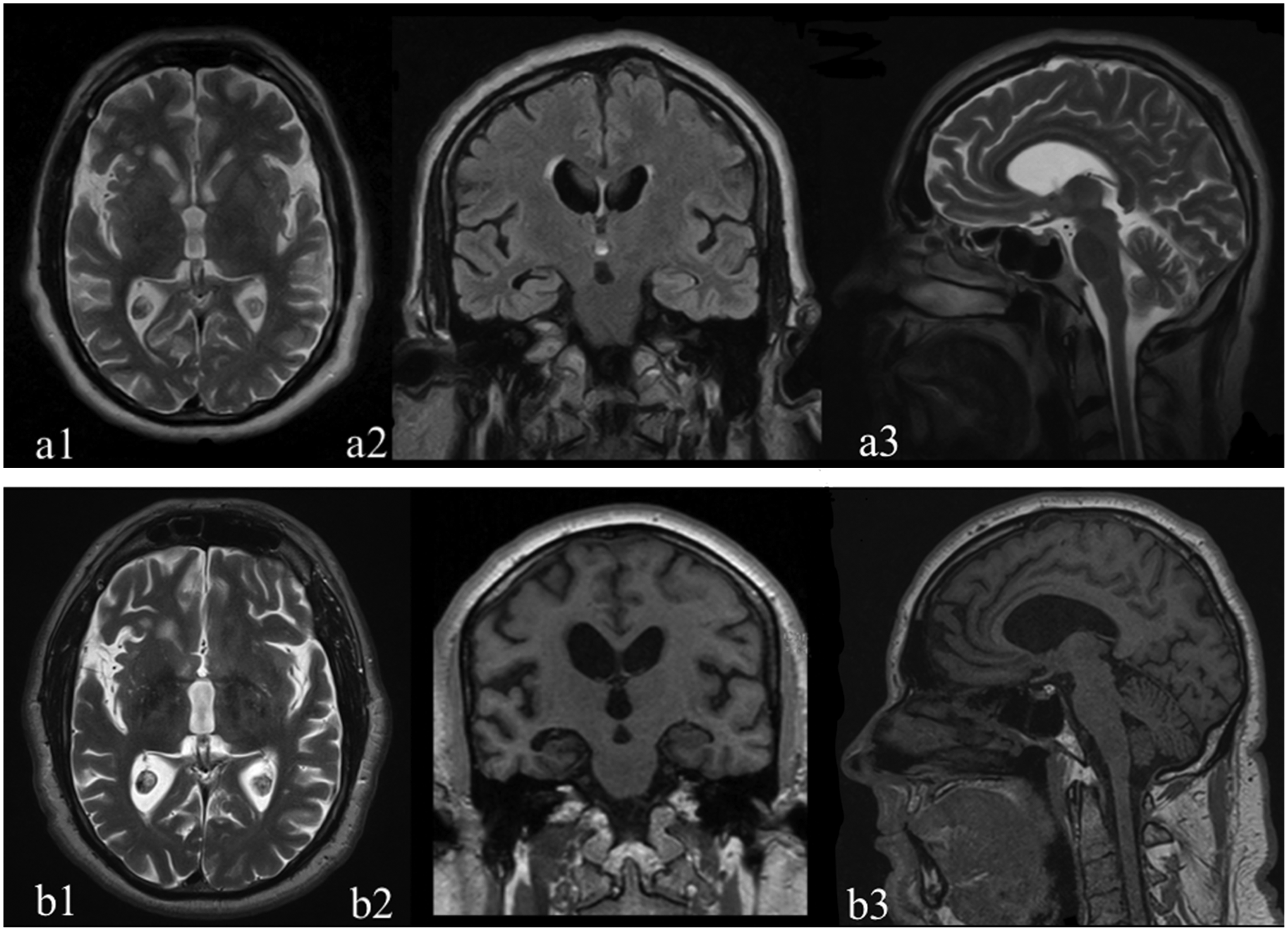
The first MRI study (December 29, 2017) is illustrated by the images above (a1-a3) while the second study (December 17, 2018) produced the images below (b1-b3). (a1, b1, axial sections; a2, b2, coronal sections; a3, b3 sagittal sections). Structural MRI T1 and T2 weighted sequences reported a substantially normal brain morphology in the first study and the appearing of right hippocampus, mesial temporal area, and insular region atrophy (b1, b2) in the second study. Very mild gliotic alterations within the hemispheres’ white matter were also detected.
Upon physical examination, the patient was alert and cooperative. No apparent visual field deficits were detected; his pupils were isocyclic, isochoric; ocular movements were normal. No asymmetries of the face were detectable. The palatal velum was in axis and the tongue midlined and protrusible. Osteo-tendon reflexes and overall neurological examination were normal. The patient was overweight with a body mass index (BMI) of 33.9.
A first neuropsychological evaluation was carried out on October 23, 2018, and showed deficits in executive and mnesic functions (Table 1) along with the presence of apathy and loss of autonomy.
Neuropsychological evaluations. The table shows the patient's performance on tests of memory, attention, language, constructional apraxia, ideomotor apraxia, visual perception, executive functions, and the comprehensive neuropsychiatric profile (raw scores, corrected scores, and cut-off values are reported). While the first neuropsychological assessment showed a deficit only in memory (RAVLT) and executive functions (FAB), the second one highlighted deficit in memory (RAVLT, Visuo-Spatial Span), language (token test and phonemic verbal fluency), constructional apraxia (Design copy test), executive functions (Clock Drawing Test, TMT A, FAB) and reasoning and problem solving (Verbal Judgement Test, Raven CPM).
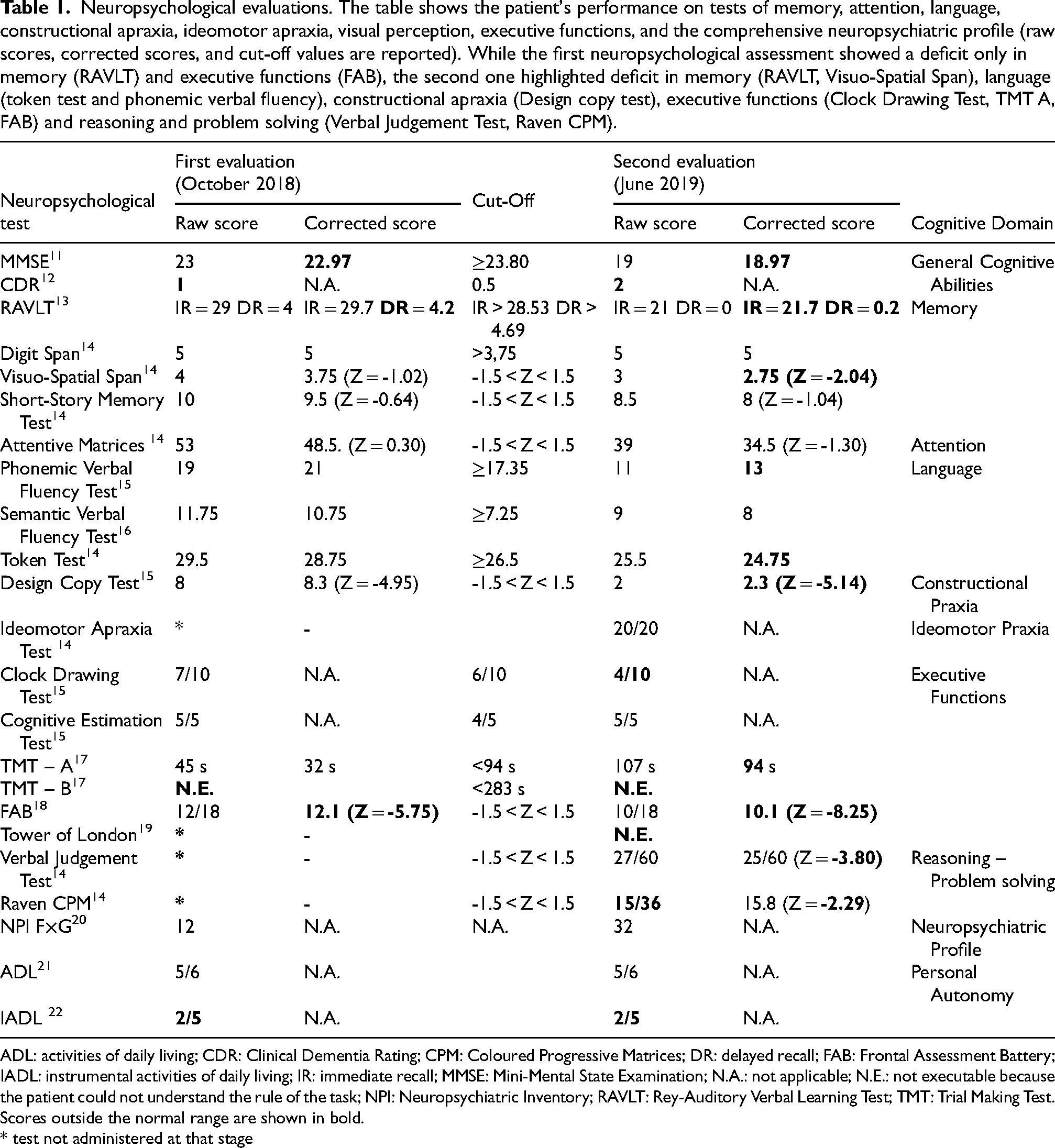
ADL: activities of daily living; CDR: Clinical Dementia Rating; CPM: Coloured Progressive Matrices; DR: delayed recall; FAB: Frontal Assessment Battery; IADL: instrumental activities of daily living; IR: immediate recall; MMSE: Mini-Mental State Examination; N.A.: not applicable; N.E.: not executable because the patient could not understand the rule of the task; NPI: Neuropsychiatric Inventory; RAVLT: Rey-Auditory Verbal Learning Test; TMT: Trial Making Test.
Scores outside the normal range are shown in bold.
* test not administered at that stage
On November 8, 2018, the patient underwent a 18F-FDG PET showing moderate-severe reduced glucose metabolism in the right frontal and temporal cortex and slight reduction in the right inferior parietal, precuneus and posterior cingulate cortex (Figure 2). Hypometabolism of the head of the caudate and thalamus on the right side was also noted.
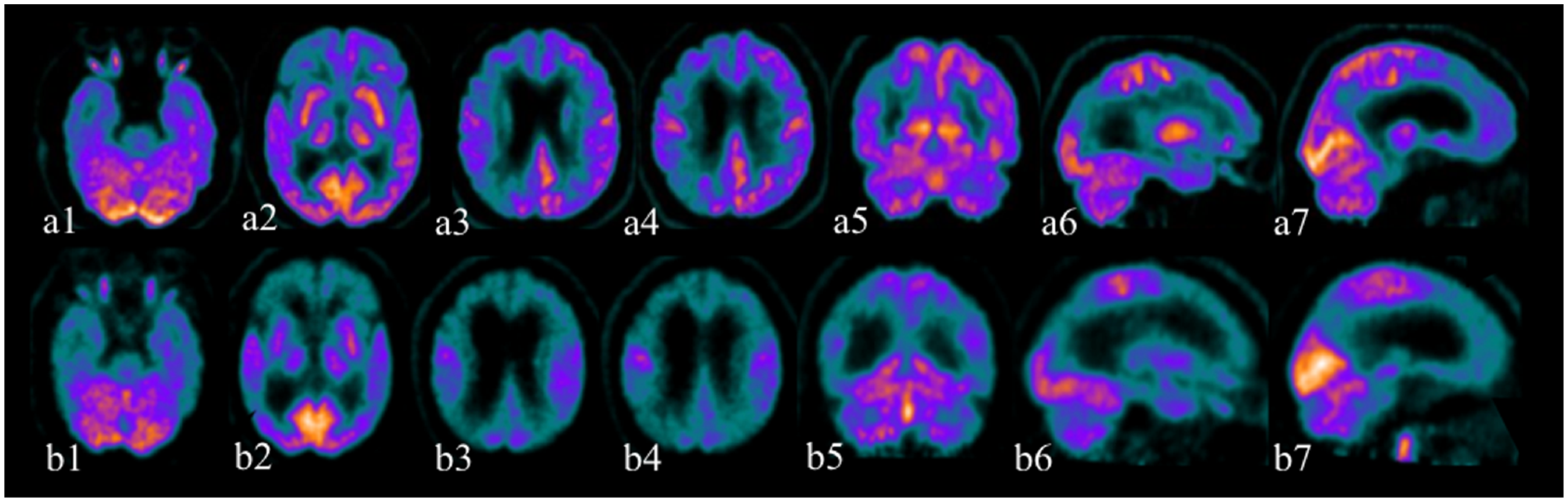
The first 18F-FDG PET study (November 8, 2018), illustrated by the images above (a1-a7) showed a reduction in radiopharmaceutical uptake in the frontal (a2-a4, a6, a7), temporal (a1, a6), and inferior parietal cortex (a4-a6) on the right and in the precuneus and posterior cingulate (a3, a4, a6) always on the right. The head of the right caudate and the right thalamus also showed a reduction in radiopharmaceutical uptake (a2). The second 18F-FDG PET study (December 22, 2020), illustrated by the images below (b1-b7), showed a clear worsening of the radiopharmaceutical uptake in the frontal (b2-b4, b6, b7), temporal (b1, b6), and parietal cortex (b4-b6), as well as in the precuneus and posterior cingulate predominantly on the right (b3, b4, b6, b7). The head of the caudate and the right thalamus also showed a worsening of radiopharmaceutical uptake (b2).
A second brain MRI, performed on December 17, 2018, showed onset of mild right temporal and insular atrophy along with mild gliotic alterations within the hemispheres’ white matter (Figure 1; b1, b2).
As early onset AD was suspected, an amyloid Flutemetamol (18F) PET was performed on April 19, 2019, which did not show an uptake of the amyloid tracer at the level of the cerebral cortex (Figure 3).
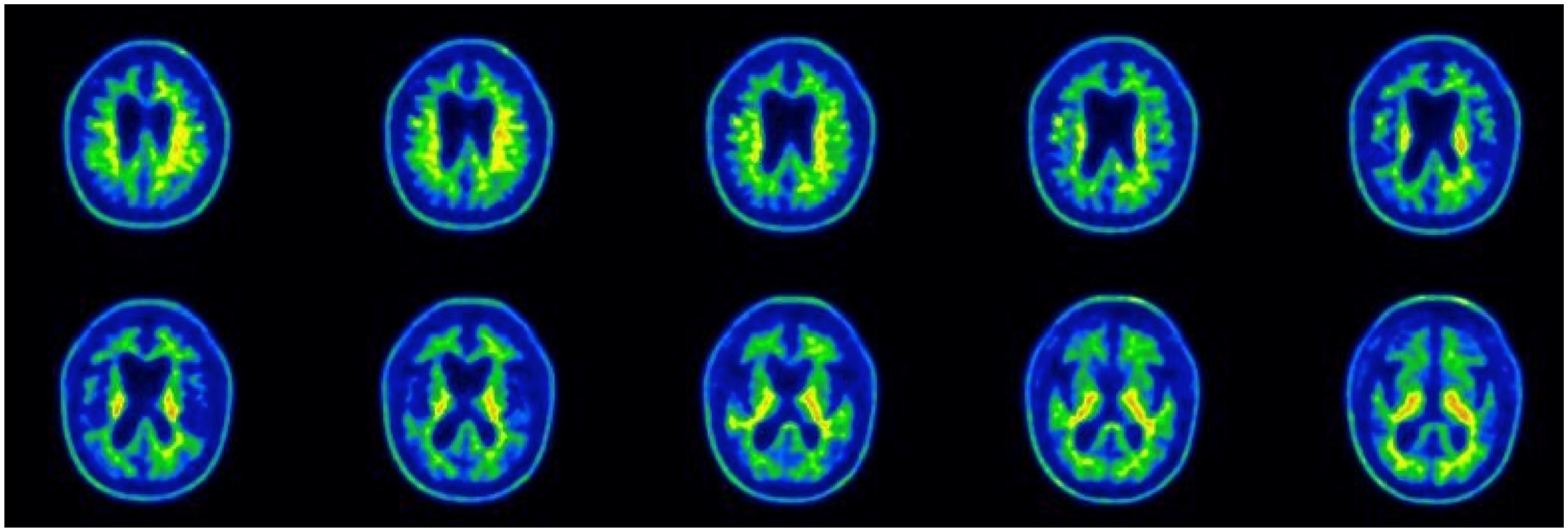
18F-Flutemetamol PET scan did not disclose any amyloid tracer uptake at the level of the cerebral cortex.
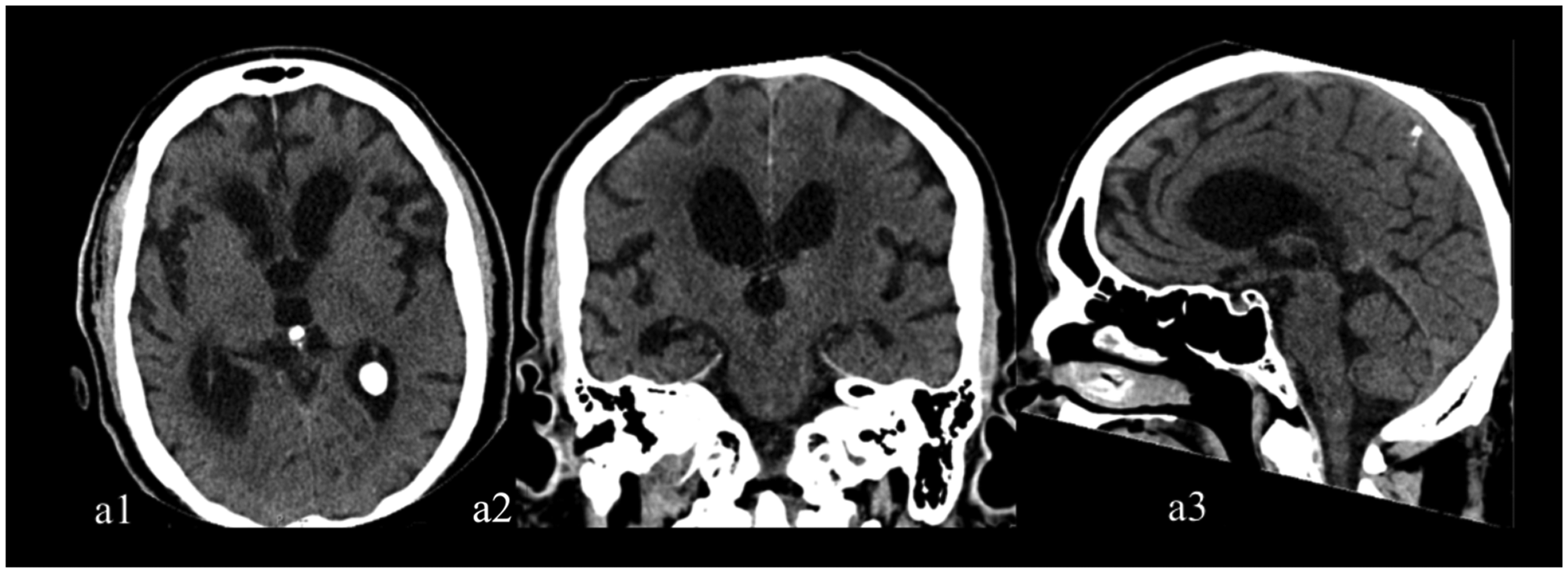
The CT scan (June 11, 2021), with no contrast, showed frontal (a1), mesial temporal (a1, a2), and insular region atrophy. The choroidal fissure was wide on the right due to hippocampal atrophy (a2).
The patient refused lumbar puncture for assay of CSF markers for AD.
The patient performed a second neuropsychological examination on June 28, 2019 after worsening mnesic and behavioral disorders had been reported. There were deficits in memory, language, constructional apraxia, executive functions, and reasoning-problem solving (Table 1), as well as significant alterations in behavior such as abulia, apathy, hyperorality, perseverative attitudes and verbal disinhibition.
The temporal and insular atrophy on MRI, the right frontal and temporal cortex hypometabolism on 18F-FDG PET, the cognitive deficits and behavioral disorders on neuropsychological assessments and the absence of the amyloid tracer at the level of the cerebral cortex suggested, in June 2019, a phenotype belonging to the FTLD spectrum.
The physical examination was still normal in January 2019, while in June 2019 there was the onset of slight left hand tremor, and in September 2019 there was mild extrapyramidal hypertonus in the left arm.
At the follow-up evaluation in December 2020, the patient showed very impoverished speech, almost mute, a slow and insecure gait, loss of facial expression, slowness and rigidity in movements especially in the left limbs, cogwheeling in the left arm, left hand tremor at rest and sometimes in the right hand. The patient also appeared unable to understand and perform some maneuvers that the examiner suggested to him, such as eye movements and cerebellar tests, most probably due to apraxia. Global cognitive performance appeared to worsen (Mini-Mental State Examination score 12/30). Levodopa/Carbidopa 100/25 d three times a day was initiated with minimal benefit in terms of rigidity.
On December 22, 2020, the patient underwent a second control 18F-FDG PET showing an accentuation of the reduced glucose metabolism in the frontal and temporal cortex and inferior parietal, precuneus and posterior cingulate cortex bilaterally with a prevalence on the right (Figure 1). Hypometabolism of the basal ganglia and of the thalami, with a predominance on the right side, was also noted.
In April 2021, the patient displayed further deterioration including muteness, ideomotor apraxia, a slower gait conducted with small steps and slightly dragging the left leg.
Given the reported family history of dementia in the patient's blood relatives, a deep molecular analysis was undertaken in May 2021 by next-generation sequencing to investigate the presence of genetic lesions associated with most common mendelian degenerative and vascular dementias.
In order to acquire control structural brain images three years following the 2018 MRI, a brain computed tomography (CT) was performed on June 11, 2021, as the patient was no longer able to collaborate for a longer-running instrumental examination such as MRI. The brain CT showed a progression of subcortical atrophy, with ex vacuo dilatation of the ventricular system. A cortical atrophy, especially in the fronto-temporal area bilaterally, was also noted (Figure 4).
In October 2021, the patient appeared to have further deteriorated from a motor and praxis point of view. He also presented clear weight loss.
A routine EEG analysis, performed on November 4, 2021, showed a tracing substantially in the normal range.
On November 11, 2021 the patient performed a 123I-ioflupane dopamine transporter (DAT) single-photon emission computed tomography (SPECT) which showed a decreased concentration of the presynaptic tracer for the dopaminergic system in the right caudate head and right putamen (Figure 5); a DaTQUANT (see methods) semi-quantitative evaluation was also performed (not shown).
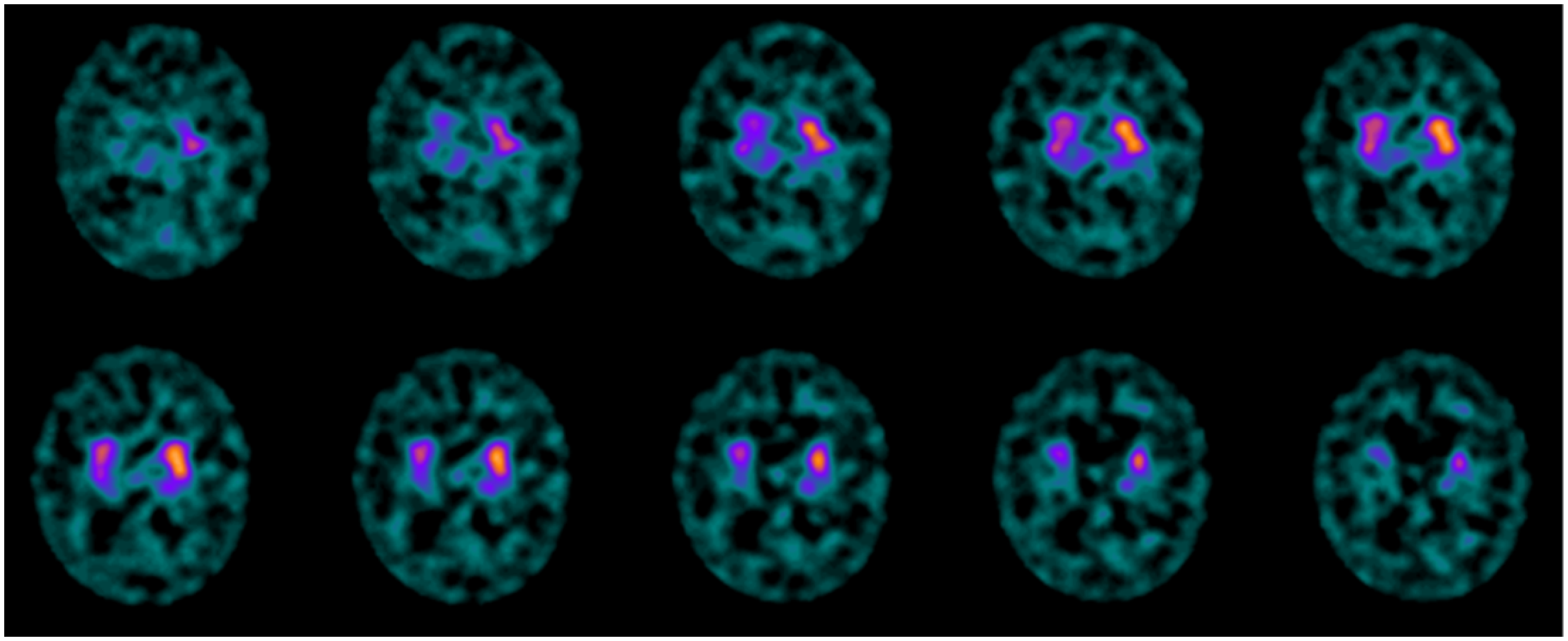
The DaT-SPECT images (November 11, 2021) showed a decreased concentration of the presynaptic tracer for the dopaminergic system in the right caudate head and right putamen. A DaTQUANT semi-quantitative evaluation was also performed (not shown).
The molecular analysis, performed in the meantime, showed the presence of a heterozygous deletion c.813_816delCACT on the GRN gene that causes the replacement of Threonine with Serine at codon 272 and the introduction of premature stop codon after ten amino acids (p.272SfsX10).
This variant is reported in the literature as a pathogenic autosomal dominant variant associated with frontotemporal dementia and corticobasal degeneration.23,24 The presence in heterozygosity of the c.1093T > G variant on the PSEN1 gene, which is predicted to result in the substitution of the serine amino acid for alanine at codon 365 (p.S365A), was also detected. This variant is reported as a rare variant of uncertain pathogenetic significance (VUS). C9ORF72 analysis disclosed the absence of an expanded allele.
Following the results of the molecular analyses, a clinical genetics consultation was performed on December 2, 2021 to offer information, assistance and support regarding the severe implications of an autosomal dominant neurodegenerative disease. Availability was offered to submit the couple's sons to genetic analysis to look for paternal variant of GRN.
It was also communicated that, if they were to consent to genetic testing, a psychological evaluation would be required to assess the sons’ motivation and to ensure they possessed the psychological resources necessary to manage any potential pathological findings. A pedigree was also performed (Figure 6).
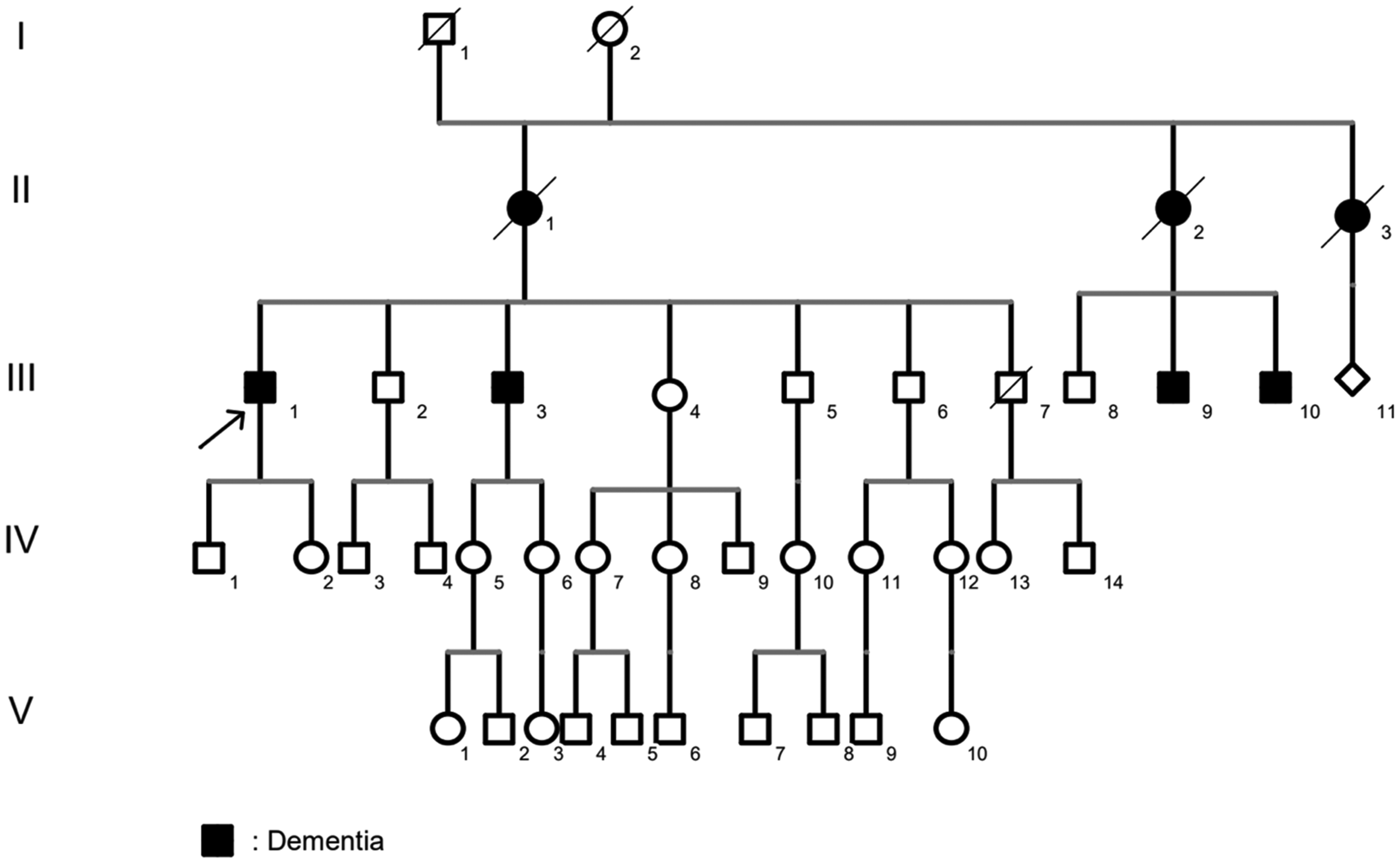
Family history revealed the diagnosis of dementia in the patient's brother, in the mother, in two maternal aunts and two cousins, both sons of one of the affected aunts (squares, males; circles, females; rhombus, gender unknown; oblique lines identify deceased subjects).
In January 2022, the patient stopped walking and spent his time either in bed or in an armchair. He needed continuous assistance day and night. The care burden fell on the wife and daughter and on a nurse who took care of complete intimate hygiene once a week.
In December 2022, an interview was held with the patient's wife, as he was no longer able to be transported.
The patient was raised from bed and seated in a chair three times a day. He was mute and had to be fed. He had urinary and fecal incontinence. He no longer took any medicine. He benefited from financial aid provided by the Italian State for situations of serious disability.
The patient's sons preferred not to undergo genetic analysis.
Methods
Clinical, neuropsychological, CT, MRI, 18F-FDG PET, DaT-SPECT imaging, electroencephalography (EEG), and genetics evaluations, were conducted. The case presented was a participant in the TREDEM Study and Registry 25 which was approved by the Treviso Province Ethics Committee (protocol no. 42326 of April 10, 2015).
Neuropsychological evaluation
Neuropsychological evaluations were carried out at admission in October 2018, in June 2019 and then in December 2020. Psychometric test results are included in Table 1 (raw scores, corrected scores, and cut-offs) for the first two, while for the third the patient's conditions did not allow full testing. The evaluation included the following tests: Clinical Dementia Rating, 12 Mini-Mental State Examination, 11 Digit Span, 16 Visuo-Spatial Span, 14 Short Story Memory Test, 14 Rey-Auditory Verbal Learning Test (RAVLT), 13 Attentive Matrices, 14 Semantic and Phonemic Verbal Fluencies,14,15 Token Test, 14 Design Copy Test, 26 Ideomotor Apraxia, 26 Clock Drawing Test, 15 Cognitive Estimation Test, 15 Trial Making Test (TMT) A and B, 17 Frontal Assessment Battery (FAB), 18 Tower of London, 19 Verbal Judgement Test, 14 Raven's Progressive Matrices, 27 and the Neuropsychiatric Inventory (NPI). 20
Structural and nuclear medicine imaging
The CT scans were acquired with the volumetric scanner EMOTION 6 Siemens. Section orientation was parallel to the orbitomeatal plane. Sections on the same plane (time of 2 s, 120 kV, 130 mA, section thickness of 5 mm, no intersection gap) covered the remaining brain from the inferior aspect of the cerebellum to the vertex of the cranium.
The structural MRI scan was performed with 3-T Magnetom 135 mT of gradients with 64 channels (Siemens Magnetom Vida). T1, T2, and T2 FLAIR weighted sequences were extracted.
The patient underwent two 18F-FDG PET brain scans using a PET tomograph Discovery MI (General Electric Healthcare). The CT scan was used for attenuation and scatter correction with set voltage tuned to 120 is. The scan was obtained over 15 min, starting 60 min after i.v. injection of 227 MBq. The images of the two scans were only visually assessed.
The patient also underwent a PET brain scan with 18F-Flutemetamol (Vizamil®), using a PET tomograph Discovery MI (General Electric Healthcare). The 18F-Flutemetamol study (Amyloid-β PET) was obtained over 20 min starting 90 min after intravenous injection of 198 MBq. The images of the scan were visually assessed.
Moreover, to obtain the DaT-SPECT images, the patient was administered two capsules of 200 mg of potassium perchlorate orally, approximately 1 h before the injection of 123I-ioflupane. The patient was administered 170 MBq of 123I-ioflupane intravenously, and SPECT imaging was detected 3 h post-injection. Furthermore, a DaTQUANT analysis, an adjunct processing tool with advanced quantitative uptake methods and with a designated normals database, was also performed (not shown) for a more accurate assessment of a patient's case.28,29
Electroencephalography
EEG assessment was performed with nineteen Ag/AgCl disk scalp electrodes placed according to the international 10–20 system, recorded EEG from Fp1, Fp2, Fz, F3, F4, F7, F8, Cz, C3, C4, Pz, P3, P4, T3, T4, T5, T6, O1, and O2. EEG activity was analyzed from single or multiple leads grouped to define the following scalp regions: anterior (Fz, Fp2, F7, Fp1, F3, F4, F8), central (Cz, C3, C4), posterior (Pz, P3, P4, O1, O2), temporal (T3, T4, T5, T6), peripheral (Fp1, Fp2, F8, T4, T6, O1, O2, T3, T5, Fz), and internal (F3, F4, Fz, C3, Cz, C4, P3, Pz, P4). Recordings were obtained with the patient resting comfortably, with eyes closed. Patient's wakefulness was ascertained about every 2 min by inviting him to open his eyes and checking block reactions. Electrodes to record the electrocardiogram were also applied. EEG was acquired as a continuous signal for 20 min and visually inspected for current clinical interpretation or detection of artefacts.
Genetic analysis
Patient DNA was analyzed by massively parallel next-generation sequencing (Next Generation Sequencing) on PGM Ion Torrent platform using an Ampliseq panel including disease genes most frequently associated with some forms of neurodegenerative dementia (AD, FTD, Creutzfeldt-Jakob disease) and vascular dementia (cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy, cerebral autosomal recessive arteriopathy with subcortical infarcts and leukoencephalopathy, cerebral amyloid angiopathy). The sequences obtained were aligned to the Homo Sapiens hg19 human reference sequence and analyzed with reference to coding regions and to the relevant flanking intronic regions. Regions 100% covered with ≥ 20× read depth are considered valid for reporting. The method used allowed the analysis of 100% of the following genes: APP, CHMP2B, FUS, GRN, MAPT, PLD3, PSEN1, PSEN2, TARDBP, TBK1, TUBA4A, VCP, and APOE.
PCR reagents were optimized for the amplification of the C9ORF72 hexanucleotide repeats (AmplideX®PCR/CE C9ORF72 Kit, Asuragen, Inc.). 30 Amplicons were sized using capillary electrophoresis (CE) on a CEQ8800 Genetic Analyzer (Beckman Coulter).
Results
Neuropsychological evaluation
The first evaluation (October 2018) (Table 1) showed a deficit in memory (RAVLT) and in executive functions (FAB). Apathy (NPI) and partial loss of autonomy in instrumental activities of daily living (IADL) were also detected.
The second evaluation (June 2019) (Table 1) highlighted a deficit in memory (RAVLT, Visuo-Spatial Span), language (Token test and Phonemic Verbal Fluency), constructional apraxia (Design copy test), executive functions (Clock Drawing Test, TMT A, FAB) and reasoning-problem solving (Verbal Judgement Test, Raven Coloured Progressive Matrices). Concomitant alterations in behavior such as abulia, apathy, hyperorality, perseverative attitudes, verbal disinhibition and sometimes some visual hallucinations (NPI) were also detected. His autonomy in daily living activities became increasingly reduced.
During a further evaluation (December 2020, not shown) the patient was basically mute and manifested alterations in behavior including increased abulia, poor initiative, apathy, and random non-purposeful activities. A reduction in appetite, with reported weight loss of about twenty kilograms and a tendency to retain food in his mouth for a long time, was also reported by his wife.
At observation, amimic facies and ideomotor slowdown were noted. He needed assistance in most activities involved in personal care and in the instrumental activities of daily living (ADL = 2/6, IADL = 0/5). Due to the difficulties presented, it was not possible to carry out any neuropsychological tests.
Structural and nuclear medicine imaging
The first structural MRI T1 and T2 weighted sequences performed on December 29, 2017, before the patient was referred to the TCIC, showed a substantially normal brain morphology (Figure 1).
The second brain MRI, performed on December 17, 2018, showed right temporal and insular atrophy and mild aspects of cerebral vascular disease. The hippocampus and the mesial temporal area appear fairly atrophic on the right (Figure 1).
The first 18F-FDG PET study, performed on November 8, 2018, showed a reduction in radiopharmaceutical uptake in the frontal, temporal, and inferior parietal cortex on the right and in the precuneus and posterior cingulate always on the right. The head of the caudate and the thalamus on the right side also showed a reduction in radiopharmaceutical uptake (Figure 2).
The second 18F-FDG PET study (December 22, 2020) showed a clear worsening of the reduction in radiopharmaceutical uptake in the frontal, temporal, and parietal cortex, as well as in the precuneus and posterior cingulate, predominantly on the right (Figure 2).
The 18F-Flutemetamol PET (April 19, 2019) scan showed the absence of the amyloid tracer at the level of the cerebral cortex (Figure 3).
The CT scan (June 11, 2021), with no contrast, showed frontal, mesial temporal, and insular region atrophy. The choroidal fissure was wide on the right due to hippocampal atrophy.
The DaT-SPECT study (November 11, 2021) showed a decreased concentration of the presynaptic tracer for the dopaminergic system in the right caudate head and right putamen.
Electroencephalography
The routine EEG analysis, performed on November 4, 2021, showed a tracing substantially in the normal range with a posterior background rhythm around 10 Hz, stable, bilateral, responsive along with bitemporal theta activity.
Genetic analysis
The molecular analysis revealed a heterozygous deletion c.813_816delCACT on the GRN gene that causes the replacement of Threonine with Serine at codon 272 and the introduction of premature stop codon after ten amino acids (p.T272SfsX10) in progranulin (PGRN).
This mutation is foreseen to cause a loss of function, thus adhering to the pathogenicity model validated for FTLS-PGRN; it is reported in the literature as a pathogenic variant associated with frontotemporal dementia and corticobasal degeneration.23,24
The analysis also identified c.1093T > G heterozygous variant of the PSEN1 gene whose molecular lesions represent the main cause of autosomal dominant AD. The detected variant leads to the substitution of Serine at codon 365 (p.Ser365Ala). This variant is reported in gnomAD database (4.1 version) as a rare variant (MAF: 1,7*10-5) of uncertain significance (VUS). 31
C9ORF72 analysis did not disclose the presence of an expanded allele. Considering the possible autosomal dominant transmission of the genetic mutation found, a family tree was built in collaboration with the clinical geneticist (Figure 6).
Analysis of APOE was consistent with a ε3/ε2 genotype.
Discussion
It has been shown that some cases of frontotemporal dementia and parkinsonism linked to chromosome 17 (FTDP-17) are caused by mutations in the MAPT gene, located on chromosome 17 and encoding the microtubule-associated protein tau. The pattern of inheritance suggests autosomal dominant transmission. FTDP-17 caused by mutations in the tau gene has a wide range of age of onset and several distinct clinical presentations. Indeed, the clinical presentation may differ not only between mutations, but also within the same mutation and even within individual families involved.32,33 The identification of mutations in GRN, which cause ubiquitin-positive FTD, explains why large families with FTD had not previously shown MAPT mutations.
Progranulin is a growth factor involved in the regulation of multiple processes, such as development and inflammation, tissue remodeling, and wound repair.34–37
A progranulin mutation causing FTDP-17 and CBS has been identified in two independent pedigrees. 24 This is the same variant found in our patient (presence in heterozygosity of the deletion c.811_814delCTCA [alias c.813_816 CACT] on the GRN gene). It is a rare variant accounting for 1.64% (four cases) of 243 FTLD cases analyzed by Borroni et al. 23
This mutation in the gene for progranulin explains the association of bv-FTLD with CBS, an entity known as FTDP-17. 38
Both mutations in the MAPT and GRN genes (17q21.31) are associated with highly variable clinical phenotypes.39–46
The c.1093T > G heterozygous variant on the PSEN1 gene was found in a woman who presented with AD symptoms at age 55, and was not present in her two healthy siblings. 47
In silico algorithms, used to predict the effects of this variant on protein function (SIFT, Polyphen-2, LRT, MutationTaster, MutationAssessor, FATHMM, PROVEAN, CADD, REVEL, and Reve in the VarCards database), yielded conflicting results. 31 It was originally classified as possibly pathogenic. 47 In subsequent studies, it was described as most likely having reduced penetrance 48 and finally classified as of uncertain significance. 31
However, in any case, the diagnosis of AD was excluded because the 18F-Flutemetamol PET scan did not disclose any amyloid tracer uptake at the level of the cerebral cortex (Figure 3).
Two years before being referred to the TCIC, then at the age of 57, our patient presented initially with executive and mnesic deficits along with the presence of apathy and loss of autonomy. Subsequently, about three years after the first prodromal symptoms reported by his wife and one year after the first visit to our center, the cognitive deficits became associated with parkinsonian-like movement disorders and apraxia.
The right-sided caudate head and the right thalamus showed decreased uptake at 18FDG-PET on November 8, 2018, evidence that we interpreted as a sign of nigrostriatal degeneration likely explaining the later onset of parkinsonian-type movement disorders.
In our patient, the diagnostic criteria for bv-FTLD and CBS were both met.49,50
Our patient presented different clinical features than those of the members of the two families described by Benussi. 24 In the FAM047 family (5 affected members), patient III:3 and III:14 immediately presented a motor syndrome with extrapyramidal features leading to a diagnosis of CBS. Patient III:2 never developed extrapyramidal signs. Patients III:11 and III:12 started with prevalent speech dysfunction. The mean age at onset in affected members was 66.4 years.
In the FAM071 family (7 affected female members) patients III:4, IV:7, IV:8, and III:20, never presented with extrapyramidal signs. Patient IV:34 presented with phonemic paraphasias. Patient IV:19 never presented with memory impairment. Patient IV:5 presented important behavioral abnormalities. The mean age at onset in affected members was 62 years.
Our patient presented different clinical features from the four cases described by Borroni 23 : two cases had mainly behavioral disorders and the other two had progressive nonfluent aphasia.
The mean age at onset was 56.2 years.
The age of onset of symptoms in our patient was therefore significantly earlier than the average ages of onset of affected individuals in the two families described by Benussi 24 and instead similar to the average age of onset of disease in the four cases described by Borroni. 23
These results emphasize that additional environmental and/or genetic factors not yet identified might produce important phenotypic variability in the clinical presentation of the disease even in the presence of an identical mutation, thus suggesting a probabilistic presentation of symptoms even in diseases of genetic origin.
In this regard, the co-occurrence of the PSEN1 p.Ser365Ala VUS leads to the speculation that components of non-mendelian inheritance might have modulated the phenotypical expression of the primary GRN mutation in our case. Indeed, digenic or oligogenic inheritance have been reported consistently in the FTLD and FTLD-amyotrophic lateral sclerosis spectrum. 51 Systematic genomic burden analysis by whole genomic sequencing or whole exome sequencing will help to address this issue in larger series. The co-occurrence of two variants in GRN and PSEN1 does not imply obvious convergent molecular pathways. The lack of CSF biomarkers, of a co-segregation analysis of genetic variants within the family and, ultimately, of a pathological autoptic study, hamper deep phenotype-to-genotype correlations and robust physio-pathological hypotheses. Nevertheless, it should be remembered that PGRN is a multifunctional growth factor; its deficiency may cause neurodegeneration leading to increased expression of proinflammatory cytokines, microglial activation and astrogliosis; although GRN-FTLD is associated with TDP-43 pathology, some GRN mutations, especially when co-occurring with APOE ε4, disclosed features of AD pathology (amyloid aggregation and neurofibrillary tangles), in addition to TDP-43-proteinopathy. 52
On the other hand, presenilin-1 forms the catalytic component of γ-secretase which is responsible for the proteolytic cleavage of amyloid-β protein precursor (AβPP); pathogenic variants of PSEN1 trigger the amyloidogenic cascade in autosomal dominant AD. Intriguingly, experimental investigations have demonstrated that PGRN protects against amyloid-β (Aβ) deposition and toxicity in AD mice models and that selective reduction of microglial PGRN increases the amyloid plaque load in AD mice. 52 Here we can only wonder whether the detected PSEN1 variant may have contributed to the clinical presentation (early memory impairment) and MRI findings (atrophy of right hippocampus and mesial temporal area) reminiscent of AD.
Conclusions
We describe the case of a patient who came to our observation at the age of 59 and manifested mnesic and executive deficits, an asymmetrical cerebral atrophy and hypometabolism with a clear predominance on the right side to which corresponded, about three years after the first prodromal amnesic symptoms, the onset of extrapyramidal signs and apraxia prevalent in the left hemisome. Our patient met the diagnostic criteria of both FTLD and CBS. Genetic analysis confirmed and reinforced the diagnosis by showing the presence of a mutation in the gene coding for Progranulin located on chromosome 17 and responsible for the entity known as FTDP-17. 38
The clinical phenotype of our patient appears different from those studied in two families with the same rare mutation 24 and in four other described cases. 23 This evidence underscores the phenotypic variability of these entities, which is probably also conditioned by as yet unknown genetic and environmental factors.
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
Author contributions
Maurizio Gallucci (Conceptualization; Data curation; Investigation; Methodology; Supervision; Validation; Visualization; Writing – original draft); Chiara Da Ronch (Writing – review & editing); Matteo Bendini (Writing – review & editing); Vittorio Fiore (Writing – review & editing); Licia Turolla (Writing – review & editing); Moreno Ferrarini (Writing – review & editing); Gian Maria Fabrizi (Conceptualization; Supervision; Writing – review & editing); Pietro Tiraboschi (Conceptualization; Supervision; Writing – review & editing).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
