Abstract
Background
Epidemiological studies have indicated an inverse association between neurodegenerative diseases and cancer. However, the relationship between liver cancer and dementia, particularly Alzheimer's disease (AD), remains underexplored.
Objective
This study aimed to determine the association between dementia, specifically AD, and liver cancer using Mendelian randomization (MR) analysis.
Methods
We conducted a bidirectional, two-sample MR analysis using publicly available genome-wide association studies data. The inverse-variance weighted method was employed as the primary analytical approach. To detect and correct for the effects of horizontal pleiotropy, we applied three complementary methods: MR Egger, weighted median, and Maximum likelihood.
Results
The analysis indicated significant associations between dementia and a reduced risk of hepatocellular carcinoma (HCC) (OR: 0.87; 95%CI: 0.81–0.95; p < 0.001) and intrahepatic cholangiocarcinoma (ICC) (OR: 0.81; 95%CI: 0.72–0.92; p < 0.001). AD was significantly associated with a decreased risk of HCC (OR: 0.94; 95%CI: 0.88–0.99; p = 0.033), ICC (OR: 0.85; 95%CI: 0.78–0.93; p < 0.001), and combined hepatocellular-cholangiocarcinoma (CHC) (OR: 0.64; 95%CI: 0.43–0.93; p = 0.020). Conversely, inverse MR analyses indicated that ICC was associated with increased dementia risk (OR: 1.05; 95%CI: 1.01–1.09; p = 0.019) and CHC with increased AD risk (OR: 1.03; 95%CI: 1.00–1.04; p = 0.014).
Conclusions
This study suggests that dementia, particularly AD, is associated with a reduced risk of liver cancer. Conversely, liver cancer may be associated with a slightly increased risk of developing dementia and AD, although some observational studies have reported a lower risk of these conditions among cancer survivors.
Introduction
With the demographic aging of the population, the prevalence of age-related disorders, including dementia and cancer, has steadily increased over the past decade. Currently, approximately 50 million people are living with dementia, a number projected to rise to 152 million by 2050. 1 Alzheimer's disease (AD) is one of the most common neurodegenerative disorders in the elderly and the most prevalent form of dementia, accounting for 60–70% of all dementia cases. 2 Cancer also represents a major public health challenge and is one of the leading causes of death globally. 3 Both dementia and cancer are heterogeneous diseases of aging, imposing substantial burdens on individuals, their families, and society. Numerous epidemiological studies have indicated that patients with dementia or AD are at a lower risk of developing certain types of cancers,4–6 and inversely, cancer survivors are at a reduced risk of developing subsequent dementia and AD.7–9 However, not all studies support this inverse association, and findings vary depending on cancer types being studied.10,11
Liver cancer, which encompasses hepatocellular carcinoma (HCC), intrahepatic cholangiocarcinoma (ICC), and combined hepatocellular-cholangiocarcinoma (CHC), ranks as the sixth most prevalent cancer across the world. 12 A recent retrospective study indicate that the risk of liver cancer is significantly lower in patients with AD, 13 corroborating findings from a previous study. 14 However, the association between dementia and various liver cancers is still unclear due to a scarcity of relevant research. Liver diseases have been suggested to play a crucial role in AD progression.15,16 Despite this, epidemiological investigations into the risk of dementia or AD among liver cancer patients are absent, and it remains unclear whether the risk is similarly reduced in these patients as it is in those with other specific types of cancer. Considering the crucial role of the liver in metabolism, elucidating the association between liver cancer and dementia or AD could improve our comprehension of the underlying disease mechanisms, potentially leading to identify novel predictive factors and therapeutic targets. However, conducting high-quality epidemiological studies is challenging due to the generally poor prognosis of liver cancer patients. Moreover, observational studies are particularly susceptible to confounding factors and reverse causation, making it difficult to draw reliable causal inferences.
Mendelian randomization (MR) leverages genetic variants as instrumental variables (IVs) to investigate causal relationships between exposures and outcomes. 17 This method has gained widespread adoption in epidemiologic research and is regarded as an important adjunct to randomized controlled trials, as it can minimize biases from confounding factors and reverse causation.18,19 In this study, we conducted a bidirectional, two-sample MR approach to investigate the potential connections between dementia, particularly AD, and liver cancer.
Methods
Study design and data sources
This study employs a bidirectional, two-sample MR approach to assess potential association between dementia, particularly AD, and liver cancer. Genetic summary statistics were sourced from diverse genome-wide association study (GWAS) datasets. Selected genetic variants must fulfill three essential criteria to qualify as instrumental variables. 20 All participants provided informed consent, and ethical approval was obtained from the ethical review boards for all studies. Detailed information on the studies incorporated in our MR analysis is available in Supplemental Table 1.
Summary data for AD were acquired from FinnGen (https://www.finngen.fi/en) (n = 218,792) and the UK Biobank (n = 472,868). 21 Summary statistic data for dementia (n = 216,771) and the common specific subtypes of dementia such as Alzheimer's disease dementia (AD-dementia) (n = 211,678), vascular dementia (Va-dementia) (n = 212,159), and Parkinson's disease dementia (PD-dementia) (n = 216,895), were obtained from FinnGen. Data for other dementia subtypes, including Lewy body dementia (LB-dementia) (n = 6618) and frontotemporal dementia (FT-dementia) (n = 3024), were sourced from independent GWAS datasets.22,23 For the inverse MR analysis, summary-level data for CHC (n = 218,792), HCC (n = 475,638), and ICC (n = 476,091) were also retrieved. 24 All participants in the present study were of European descent.
Selection of genetic instrument
Independent single nucleotide polymorphisms (SNPs) with an r2 cutoff of 0.001 and a clumping window of 10,000 kb, achieving genome-wide significance (p < 5 × 10−8), were selected as the IVs. If a trait had fewer than three SNPs meeting this threshold, the GWAS p-value threshold was relaxed to 5 × 10−6. The strength of IVs was evaluated using F-statistics (F = R2/(1-R2) × [(N-K-1)/K], where R2 is the proportion of the exposure explained by the IVs, N is the sample size, and K is the number of IVs. 25 An F-statistic greater than 10 indicates enough power of the instrument. To remove SNPs that were associated with potential confounders and mediators, we looked up each SNP using the PhenoScanner database, which provides SNP phenotypes.26,27 Palindromic alleles were also excluded during data harmonization. The characteristics of the retained SNPs are detailed in the Supplemental Excel 1.
Statistical analysis
The principal MR analysis employed the inverse-variance weighting (IVW) method to examine the potential relationships between dementia, AD, and liver cancer.28,29 A fixed-effects IVW model was used when data exhibited no significant heterogeneity (Cochran's Q test p > 0.05). The MR analyses were performed using the TwoSampleMR R package version 0.5.6. 29 Additional MR analyses included MR Egger, 30 weighted median, 31 and Maximum likelihood 32 methods. Sensitivity analyses tested for heterogeneity using the IVW (Q) methods. The MR-Egger regression (MR-Egger intercept test) intercept was conducted to assess horizontal pleiotropy, and a leave-one-out analysis was performed to evaluate the influence of individual variants on the overall effect estimate. Statistical analyses were performed using the R software (version 4.3.1), with statistical significance set at a two-tailed p-value lower than 0.05.
Results
Effects of dementia and AD on liver cancer
AD was significantly related to a reduced risk of HCC (IVW: odds ratio [OR]: 0.94; 95% confidence interval [CI]: 0.88–0.99; p = 0.033), ICC (IVW: OR: 0.85; 95%CI: 0.78–0.93; p < 0.001), and CHC (IVW: OR: 0.64; 95%CI: 0.43–0.93; p = 0.020), as illustrated in Figure 1. These findings were corroborated by the weighted median (OR: 0.93, p = 0.018; OR: 0.85, p < 0.001; OR: 0.58, p = 0.014), maximum likelihood (OR: 0.94, p = 0.033; OR: 0.85, p < 0.001; OR: 0.64, p = 0.002), and MR-Egger (CHC: OR: 0.50; 95%CI: 0.26–0.95; p = 0.044) methods (Supplemental Table 2). The influence of individual SNPs on these associations is detailed in the scatter plots (Figure 2) and forest plots (Figure 3), with leave-one-out analysis results provided in Supplemental Figures 10–12.
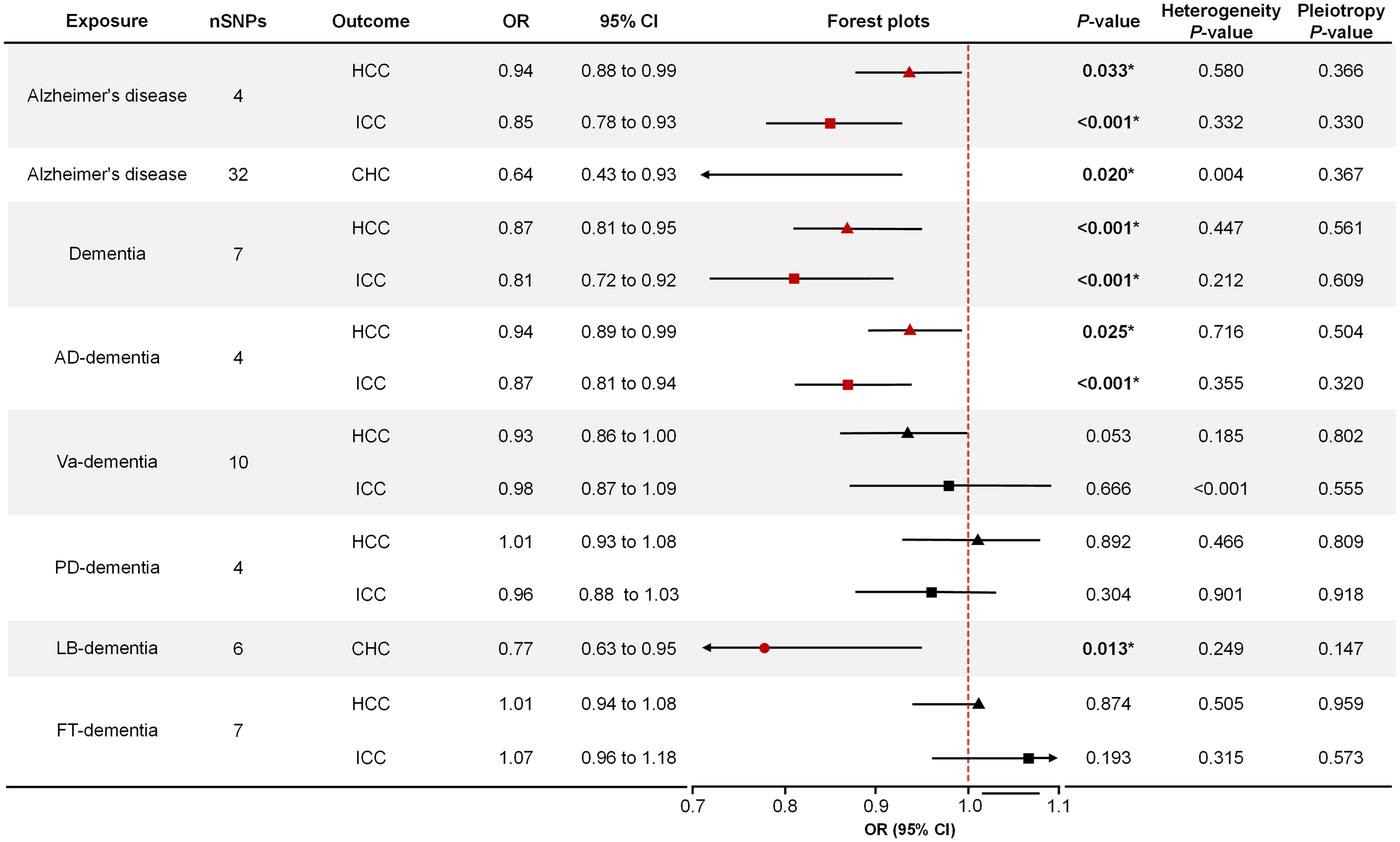
Mendelian randomization estimates of the effects of dementia and AD on liver cancer. AD-dementia: Alzheimer's disease dementia; Va-dementia: vascular dementia; PD-dementia: Parkinson's disease dementia; LB-dementia: Lewy body dementia; FT-dementia: frontotemporal dementia; SNP: single nucleotide polymorphism; OR: odds ratio; CI: confidence interval.
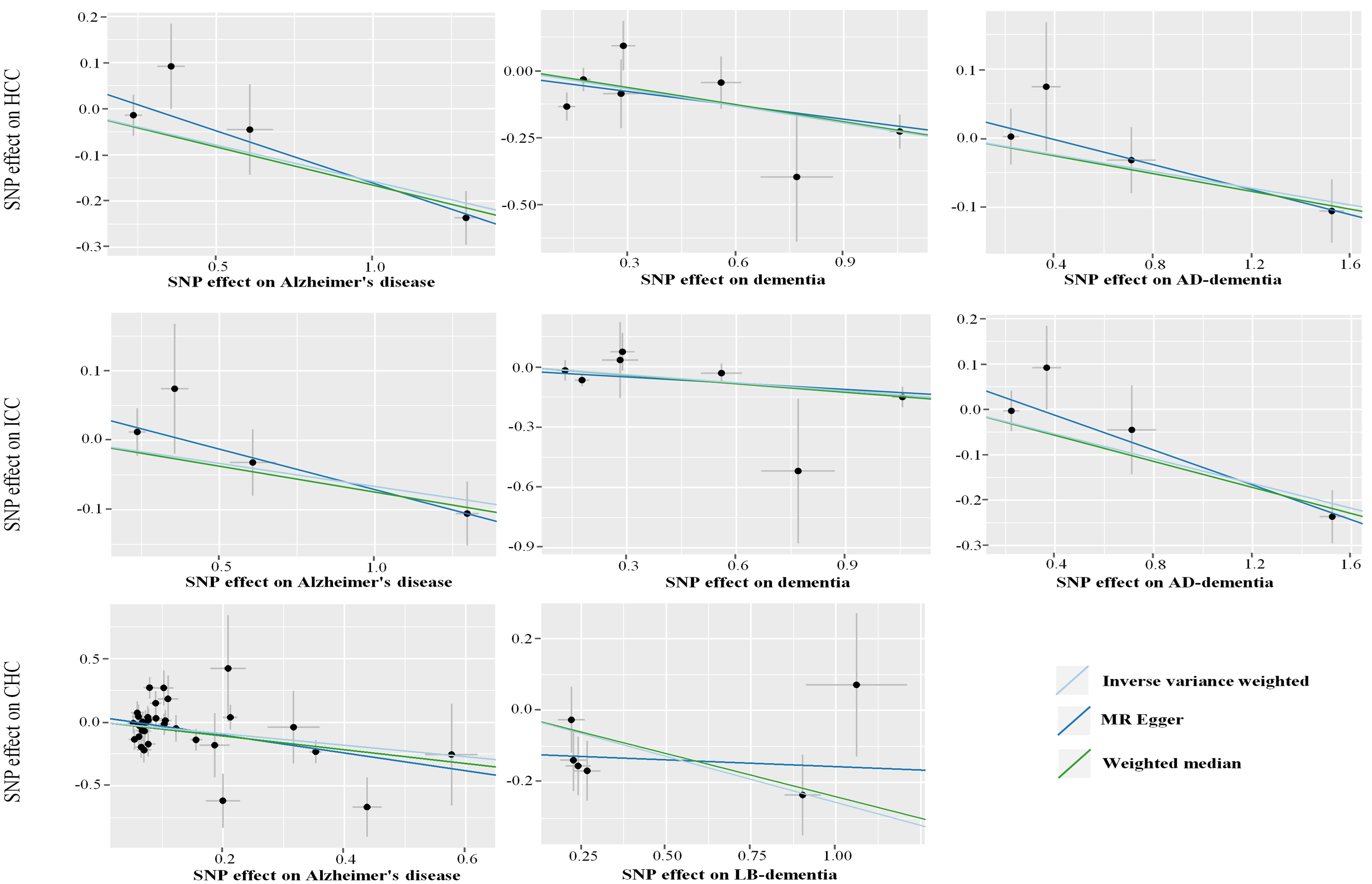
Scatter plots of the effects of dementia and AD-associated SNPs on the risk of liver cancer. AD-dementia: Alzheimer's disease dementia; LB-dementia: Lewy body dementia; SNP: single nucleotide polymorphism.
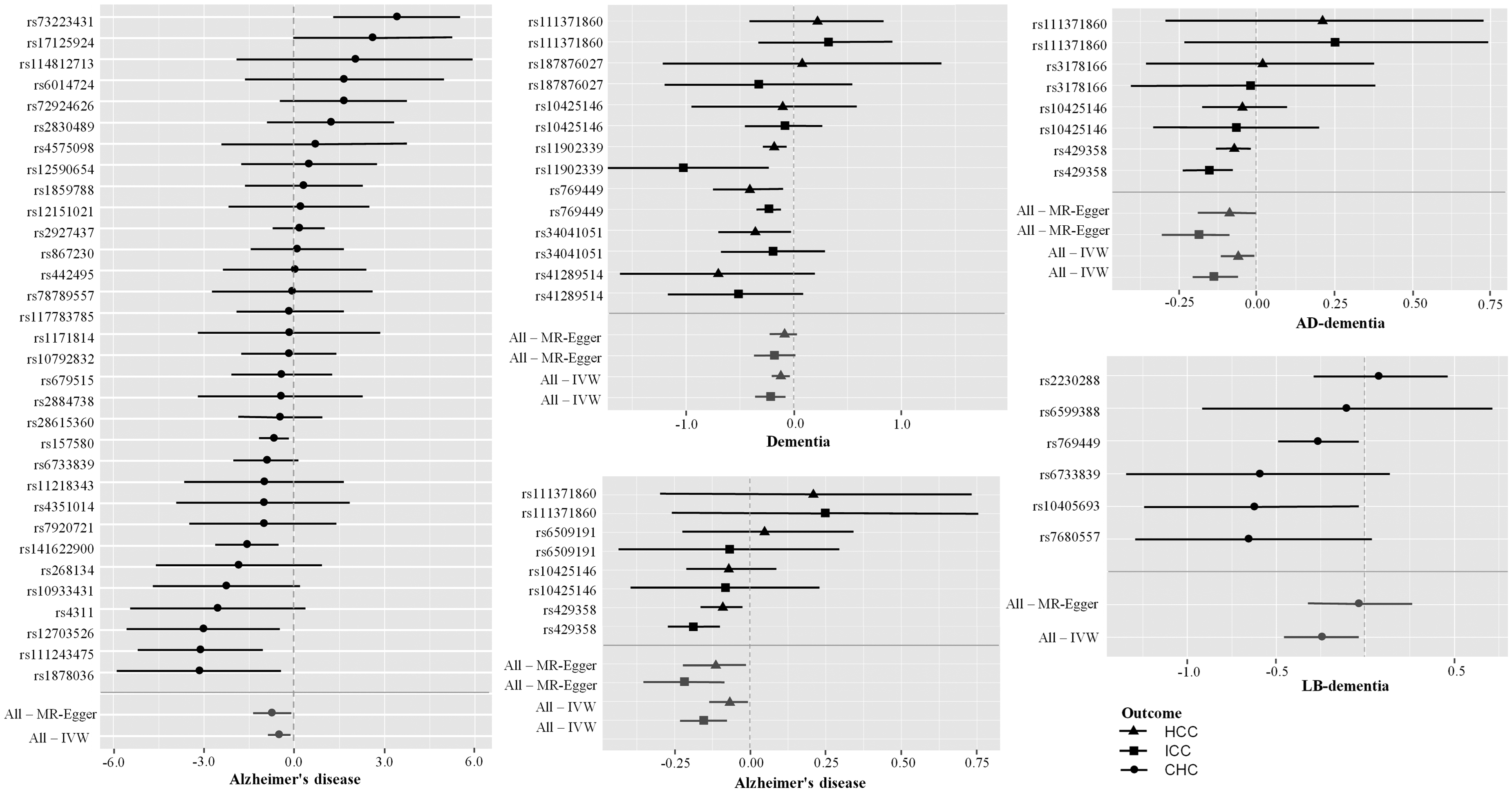
Forest plots of the effects of dementia and AD-associated SNPs on liver cancer risk. AD-dementia: Alzheimer's disease dementia; LB-dementia: Lewy body dementia; IVW: inverse-variance weighted; SNP: single nucleotide polymorphism.
Similarly, dementia exhibited a significant association with the risks of HCC (IVW: OR: 0.87; 95%CI: 0.81–0.95; p < 0.001) and ICC (IVW: OR: 0.81; 95%CI, 0.72–0.92; p < 0.001) (Figure 1), confirmed by both the weighted median (OR: 0.87, p < 0.001; OR: 0.81, p < 0.001) and maximum likelihood (OR: 0.87, p < 0.001; OR: 0.81, p < 0.001) methods (Supplemental Table 2). The effects of individual dementia SNPs on CHC are displayed through the scatter plots (Figure 2) and forest plots (Figure 3), with corresponding leave-one-out analysis in Supplemental Figures 13 and14.
The associations between common dementia subtypes and liver cancer are shown in Figure 1. AD-dementia was found to be inversely correlated with HCC (IVW: OR: 0.94; 95%CI: 0.89–0.99; p = 0.025) and ICC (IVW: OR: 0.87; 95%CI: 0.81–0.94; p < 0.001) (Figure 1). LB-dementia similarly showed an inverse correlation with CHC risk (IVW: OR: 0.77; 95%CI: 0.63–0.95; p = 0.013) (Figure 1), with findings confirmed by both weighted median and maximum likelihood (Supplemental Table 2). The scatter and forest plots for AD-dementia and LB-dementia SNPs are displayed in Figures 2 and 3. However, Va-dementia, PD-dementia, and FT-dementia showed insufficient evidence of association with liver cancers (HCC, ICC, or CHC) as indicated in Figure 1, Supplemental Table 2, and Supplemental Figures 1–6. of Leave-one-out analysis plots are detailed in Supplemental Figures 15–23.
Effects of liver cancer on dementia and AD
In the inverse MR analyses, ICC was weakly associated with a marginally increased risk of dementia (IVW: OR: 1.05; 95%CI: 1.01–1.09; p = 0.019) (Figure 4), which was supported by the maximum likelihood method (OR: 1.05, p = 0.023) (Supplemental Table 3). Similarly, CHC showed a slight increase in the risk of AD (IVW: OR: 1.03; 95%CI: 1.00–1.04; p = 0.014) (Figure 4), confirmed by both the weighted median (OR: 1.02, p = 0.009) and maximum likelihood methods (OR: 1.02, p = 0.006) (Supplemental Table 3). The impact of individual SNPs from liver cancer on these risks was illustrated via the scatter plots and forest plots in Figure 5, with further details in Supplemental Figures 7–9. Results of the leave-one-out analysis, which assesses the influence of individual variants, are shown in Supplemental Figures 24–28.
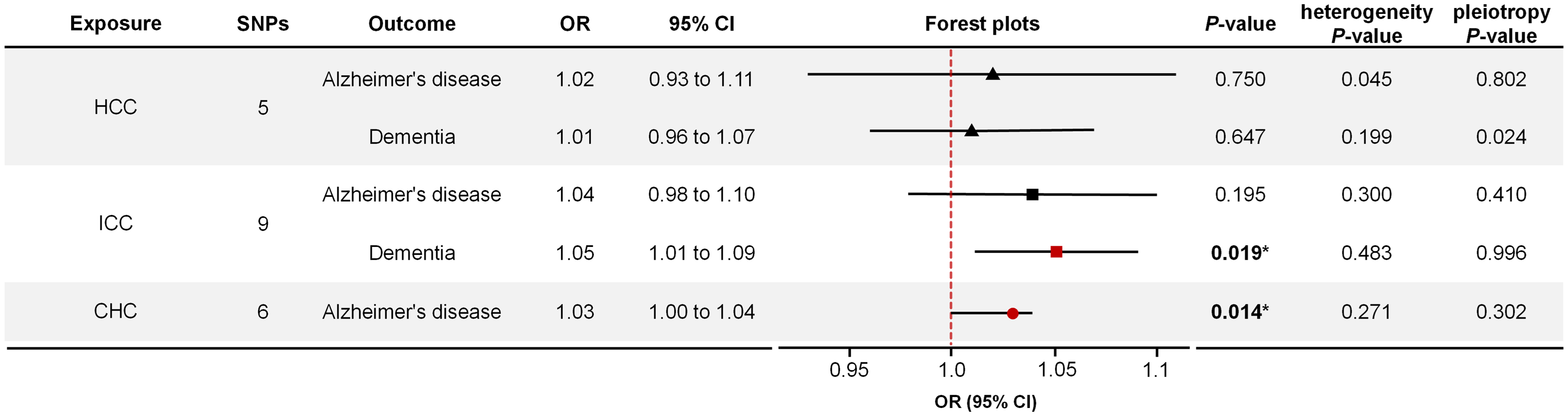
Mendelian randomization estimates of the effects of liver cancer on the risk of dementia and Alzheimer's disease. SNP: single nucleotide polymorphism; OR: odds ratio; CI: confidence interval.
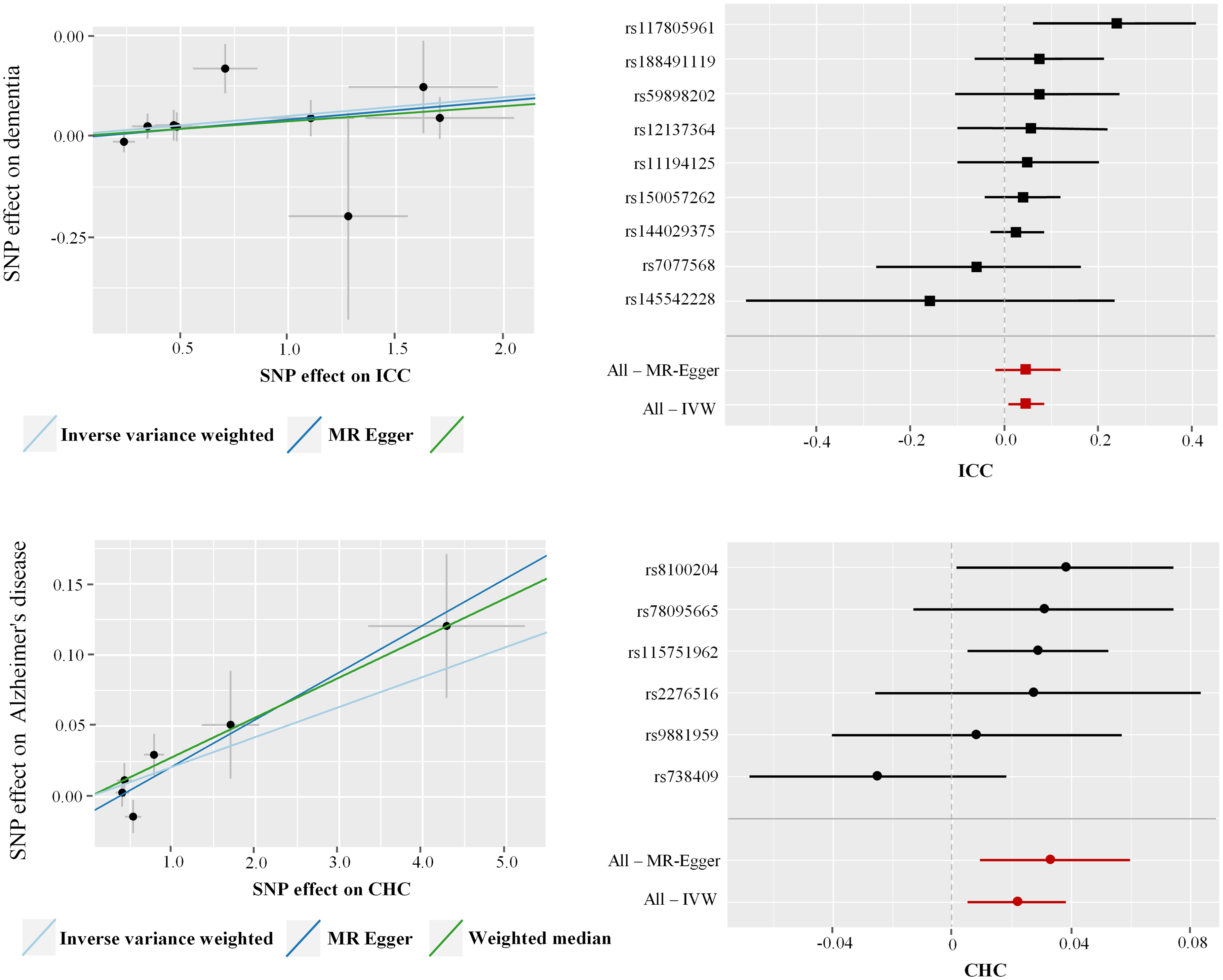
Scatter plots and forest plots of the effects of liver cancer-associated SNPs on the risk of dementia and Alzheimer's disease. IVW: inverse-variance weighted; SNP: single nucleotide polymorphism.
Discussion
To our knowledge, this study is the first to employ MR to investigate the associations between dementia, particularly AD, and liver cancer. Our findings suggest causal relationships wherein dementia and AD appear to inversely influence liver cancer risk. Specifically, only AD-dementia and LB-dementia demonstrated significant inverse associations with liver cancer. Conversely, in the inverse analyses, liver cancer did not exhibit a protective effect against dementia or AD but was instead weakly associated with an increased risk of of both conditions.
Numerous studies have highlighted the inverse relationship between dementia, particularly AD, and various cancers. A meta-analysis reported a significant association between AD and a reduced risk of cancer (RR: 0.55; 95%CI: 0.41–0.75). 33 Large-scale cohort studies utilizing the Korean National Health Insurance database have also suggested that individuals with AD have a lower incidence of liver cancer compared to control groups.13,14 In addition, inverse transcriptomic and genetic links between AD and liver cancer have been identified in recent studies.34,35 However, despite these findings, the evidence remains predominantly observational and limited, leaving the association between dementia, particularly AD, and liver cancer unclear. Our results not only align with these previous findings but also extend the understanding of how dementia and AD may inversely correlate with liver cancer risk.
Importantly, we evaluated different subtypes of dementia, revealing that only AD-dementia and LB-dementia exhibited significant inverse associations with liver cancer. This finding highlights the need to consider the genetic and metabolic heterogeneity among dementia subtypes. Generalizing across all forms of dementia could introduce biases that obscure critical subtype-specific associations. Although the inclusion of diverse dementia subtypes allowed for a comprehensive evaluation, it is crucial to recognize that such an approach may mask distinct pathways that are specific to each subtype. Future research should aim to explore these subtypes independently to mitigate potential confounding factors.
The association between cancer and a reduced risk of dementia or AD remains controversial. While several studies have reported an inverse relationship between dementia and the risk of cancers,36–38 others have reported neutral or positive associations.10,39–41 A recent meta-analysis highlighted that cancer survivors exhibit a significantly lower risk of dementia and AD compared to non-cancer controls (RR: 0.89; 95%CI: 0.82–0.97). 42 However, limited studies has focused specifically on the risk of dementia or AD in patients with liver cancer, resulting in a gap for the relationship between liver cancer and dementia or AD. Previous research indicated that liver function is significantly associated with the development of AD,43,44 with liver fibrosis and fatty liver disease also correlated with dementia and AD;45–48 however, the mechanisms underlying the connection between the liver diseases and AD are not fully understood. As an essential metabolic organ, the liver plays a crucial role in clearing peripheral metabolites. For instance, it has been suggested that the liver physiologically regulates the metabolism of amyloid-β, a key protein in the pathology of dementia and AD.49,50 Moreover, metabolic impairment and subsequent chronic inflammation may contribute to the development and progression of AD.51–53 The observed inverse association between dementia and liver cancer in some studies could stem from competing risks, where liver cancer patients may not live long enough to develop dementia. On the other hand, the weak positive association between liver cancer and an increased risk of dementia and AD could indicate shared inflammatory or metabolic pathways, but this hypothesis warrants further investigation. This study is the first to report a potential association between liver cancer and an increased risk of dementia or AD. Given the weak positive correlation observed, these findings should be interpreted with caution, and more extensive studies are necessary to clarify this relationship.
The present study underscores the complexity of deciphering associations between cancer and neurodegenerative diseases. The controversial nature of existing evidence, where some studies reveal an inverse association while others show neutral or even positive associations, suggests that these relationships are likely context-dependent, varying with cancer type, patient demographics, and genetic background. For instance, some cancers may induce systemic effects that protect against neurodegeneration, whereas others may aggravate it through chronic inflammation or metabolic dysfunction.4,36,54 Thus, our findings contribute to this nuanced understanding and also caution against overgeneralization.
This MR study boasts several strengths. Primarily, it is the first to systematically investigate the impact of dementia and its common subtypes on liver cancer risk and it is also the first to report an increased risk of dementia or AD in patients with liver cancer. Furthermore, the MR approach effectively minimized biases from confounding factors and reverse causation, while the large sample size enhanced statistical power, providing robust evidence for potential causality.
However, the study also has some limitations. Firstly, to avoid population stratification bias, it included only participants of European descent. Secondly, the reliance on summary-level data restricted our ability to perform further stratified analyses on specific factors such as age, gender, and lifestyle. Lastly, potential horizontal pleiotropy, indicated by the leave-one-out plots, suggests that our results should be interpreted with caution.
In conclusion, this study demonstrated that dementia—primarily AD-dementia and LB-dementia—was associated with a decreased risk of liver cancer. However, evidence supporting a similar association with other subtypes of dementia, such as Va-dementia, LB-dementia, PD-dementia, and FT-dementia, was lacking. Additionally, our findings suggest that liver cancer may be associated with an increased risk of dementia and AD. Future research is necessary to validate these findings and to further explore the underlying mechanisms.
Supplemental Material
sj-docx-2-alr-10.1177_25424823241299661 - Supplemental material for Association between dementia, Alzheimer's disease, and liver cancer: A Mendelian randomization analysis
Supplemental material, sj-docx-2-alr-10.1177_25424823241299661 for Association between dementia, Alzheimer's disease, and liver cancer: A Mendelian randomization analysis by Tianze Li, Jianwei Yi, Xuliang Hu, Huajun Wu and Kai Wang, Binghai Zhou in Journal of Alzheimer's Disease Reports
Supplemental Material
sj-docx-3-alr-10.1177_25424823241299661 - Supplemental material for Association between dementia, Alzheimer's disease, and liver cancer: A Mendelian randomization analysis
Supplemental material, sj-docx-3-alr-10.1177_25424823241299661 for Association between dementia, Alzheimer's disease, and liver cancer: A Mendelian randomization analysis by Tianze Li, Jianwei Yi, Xuliang Hu, Huajun Wu and Kai Wang, Binghai Zhou in Journal of Alzheimer's Disease Reports
Supplemental Material
sj-docx-4-alr-10.1177_25424823241299661 - Supplemental material for Association between dementia, Alzheimer's disease, and liver cancer: A Mendelian randomization analysis
Supplemental material, sj-docx-4-alr-10.1177_25424823241299661 for Association between dementia, Alzheimer's disease, and liver cancer: A Mendelian randomization analysis by Tianze Li, Jianwei Yi, Xuliang Hu, Huajun Wu and Kai Wang, Binghai Zhou in Journal of Alzheimer's Disease Reports
Footnotes
Acknowledgments
We acknowledge the GWAS and Finngen databases for providing their platforms and contributors for uploading meaningful datasets.
Author contributions
Tianze Li (Conceptualization; Data curation; Formal analysis; Validation; Writing—original draft); Jianwei Yi (Formal analysis; Validation; Visualization); Xuliang Hu (Data curation; Formal analysis; Writing—original draft); Huajun Wu (Writing—review & editing); Kai Wang (Conceptualization; Funding acquisition; Project administration; Supervision; Writing—review & editing); Binghai Zhou (Conceptualization; Funding acquisition; Project administration; Supervision; Writing—review & editing).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grants 82160576 and 82060454), the Natural Science Foundation of Jiangxi Province of China (grants 20212BAB216052 and 20232ACB206038), the key research and development program of Jiangxi Province of China (grant 20203BBGL73143), and the Jiangxi Province high-level and high-skill leading talent training project (grant G/Y3035).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
