Abstract
Background
Patient harm from healthcare is prevalent, serious, and can be long lasting. How healthcare organizations respond has implications for patients, families, healthcare professionals, and organizations. We sought to synthesize the evidence on the effectiveness of communication and resolution programs (CRPs).
Methods
Harm response programs that incorporated communication with patients and families, event review, quality improvement, and in a qualifying subset of events, an apology and an offer of compensation were included. We searched PubMed and the Cochrane Library for systematic reviews and primary studies published from 1 January 2010 to November 2025, supplemented by a review of gray literature.
Results
We retrieved 2801 citations, of which nine primary studies and no systematic reviews were identified as eligible. The studies focused on medical malpractice experience and financial outcomes. The most robust CRPs reduced the rate of claims, and defense legal fees and expenses (strength of evidence: low). Time to resolution, rate of lawsuits, overall costs to healthcare organizations, and settlement amounts decreased or had no significant change. Evidence was lacking or limited for CRPs’ effects on: other aspects of patient, family, or clinician outcomes or experience; patient safety; quality of care; communication and relationships between patients, families, and organizations; and interprofessional communication.
Conclusions
CRP implementation is supported by evidence of positive or neutral effects on organizational liability and cost outcomes, but more research is needed to determine the effects on patient, family, and clinician-oriented outcomes.
Keywords
Background
Patient harm from healthcare is prevalent and serious.1–5 Harms can be physical, emotional, psychological, socio-behavioral, or financial. The effects can last years and may extend to families and clinicians.6–9 How healthcare organizations respond after patients experience harm in their care has implications for the impact on patients, their trust, their willingness to return to the organization,10–12 clinicians’ ability to learn and respond more effectively after future events,13,14 organizations’ culture of safety, 15 medicolegal experiences,16–21 and the safety of future patients. 22
Traditionally, when patients are harmed, healthcare organizations have responded using a risk and malpractice-focused process that limits communication between patients/families and clinicians/organizations and defends against any claim or lawsuit filed. 23 Recognizing that this approach may compound suffering of patients, families, and clinicians who are left without information and support, ethically grounded person-centered approaches for responding have emerged. Communication and resolution programs (CRPs) are proactive and comprehensive organizational responses when patients are harmed by their care, designed to promote healing, learning, and improvement.24,25 Specific examples of CRPs include the Agency for Healthcare Research and Quality's (AHRQ's) CANDOR (Communication and Optimal Resolution) program, CARe (Communication, Apology, and Resolution), 26 the Seven Pillars program, 19 BETA HEART (healing, empathy, accountability, resolution, and trust), 27 and Stanford's PEARL program (Process for Early Assessment and Resolution of Loss). 28
Policymakers expect such responses after patients are harmed by their care. The Joint Commission, the National Quality Forum, the Leapfrog Group, and others emphasize proactive communication with patients and families after certain events.2,29–32 CRPs have been promoted by the 2023 President's Council of Advisors on Science and Technology 33 and the Centers for Medicare & Medicaid Services (CMS) through the Patient Safety Structural Measure (PSSM). 34 Despite extensive stakeholder input shaping CRP design, rigorous evidence of their effectiveness remains limited.
We report here the findings of the AHRQ Making Healthcare Safer IV rapid review on the effectiveness of programs for responding to harms experienced by patients during clinical care in terms of outcomes for patients, families, healthcare professionals, and healthcare systems. 35
Methods
For this rapid review, we streamlined traditional systematic review processes as proposed by AHRQ's Evidence-based Practice Center Program. 36 The protocol for this review is posted on the AHRQ website. 37 Adjustments included: limiting the number of databases searched; modifying search strategies to focus on finding the most valuable studies (i.e. being flexible on sensitivity to increase the specificity of the search); and restricting the search to studies published in English since 2010. We limited the search to studies published in 2010 or after for three reasons: increased relevance to contemporary healthcare; that was the first year a publication referred to an organization's program for responding after patients experience harm as being fully integrated with the organization's patient safety efforts 16 ; and for feasibility.
Study selection
We searched for original studies and systematic reviews on the review question according to eligibility criteria presented in Supplement Table 1—Study inclusion and exclusion criteria. The review included programs that are intended to provide a transparent explanation of what caused the harm, a focus on investigating and learning from harm events to determine corrective actions, and an apology when an error caused harm. To provide rigorous evidence of CRPs’ effectiveness, we focused on studies with a comparison group. We searched PubMed and the Cochrane Library, supplemented by a search for unpublished reports publicly available from AHRQ, CMS, the Patient Centered Outcomes Research Institute, the Patient Safety Movement, or professional organizations interested in the topic (i.e. Institute for Healthcare Improvement, and the Pathway to Accountability, Compassion, and Transparency [PACT] Collaborative, Betsy Lehman Center for Patient Safety). For details of the search strategy, see Appendix A of the AHRQ report. 35 We updated our search to capture studies up until November 2025 and included an article suggested during the peer review process.
Data extraction
For this report, we used the artificial intelligence (AI) feature of DistillerSR (AI Classifier Manager) as a second reviewer for title and abstract screening. Conflicts between team members and the AI Classifier Manager were resolved by team members. The full text of each remaining potentially eligible article was reviewed by two team members independently to confirm eligibility. One reviewer extracted information into standardized forms and a second reviewer checked the first reviewer's abstraction for completeness and accuracy. Data were compiled into evidence tables and narratively summarized. Some studies conducted more than one analysis for a given outcome. In such situations, we drew conclusions about the effect of the CRP from the most rigorous analysis the study reported.
Risk of bias (quality) and strength of evidence assessments
We used the ROBINS-I tool for assessing the Risk Of Bias In Non-randomized Studies (all studies were non-randomized). 38 The risk of bias assessments focused on the main outcome of interest in each study. Based on the robustness of their design and implementation, we categorized interventions into two groups: most robust [Seven Pillars,19,39 CARe,17,21 and the University of Michigan Health System claims management program or disclosure-with-offer program],16,18 and least robust.40–42 We graded the strength of evidence (SOE) on outcomes for the most robust interventions that had more than one study of effectiveness, using methods outlined in the AHRQ Effective Health Care Program Methods Guide for Effectiveness and Comparative Effectiveness Reviews. 43
Results
We found nine studies that met our eligibility criteria (Figure 1), and they reported on five unique CRPs implemented from 2001 to 2013, in a variety of settings in the United States. A summary of studies is provided in Supplement Table 2, including information about overlaps in data analyzed by studies focused on the same CRP. All settings involved acute care hospitals and/or health systems that included ambulatory clinics. No studies included other ambulatory settings, rehabilitation facilities, telemedicine, or long-term care.
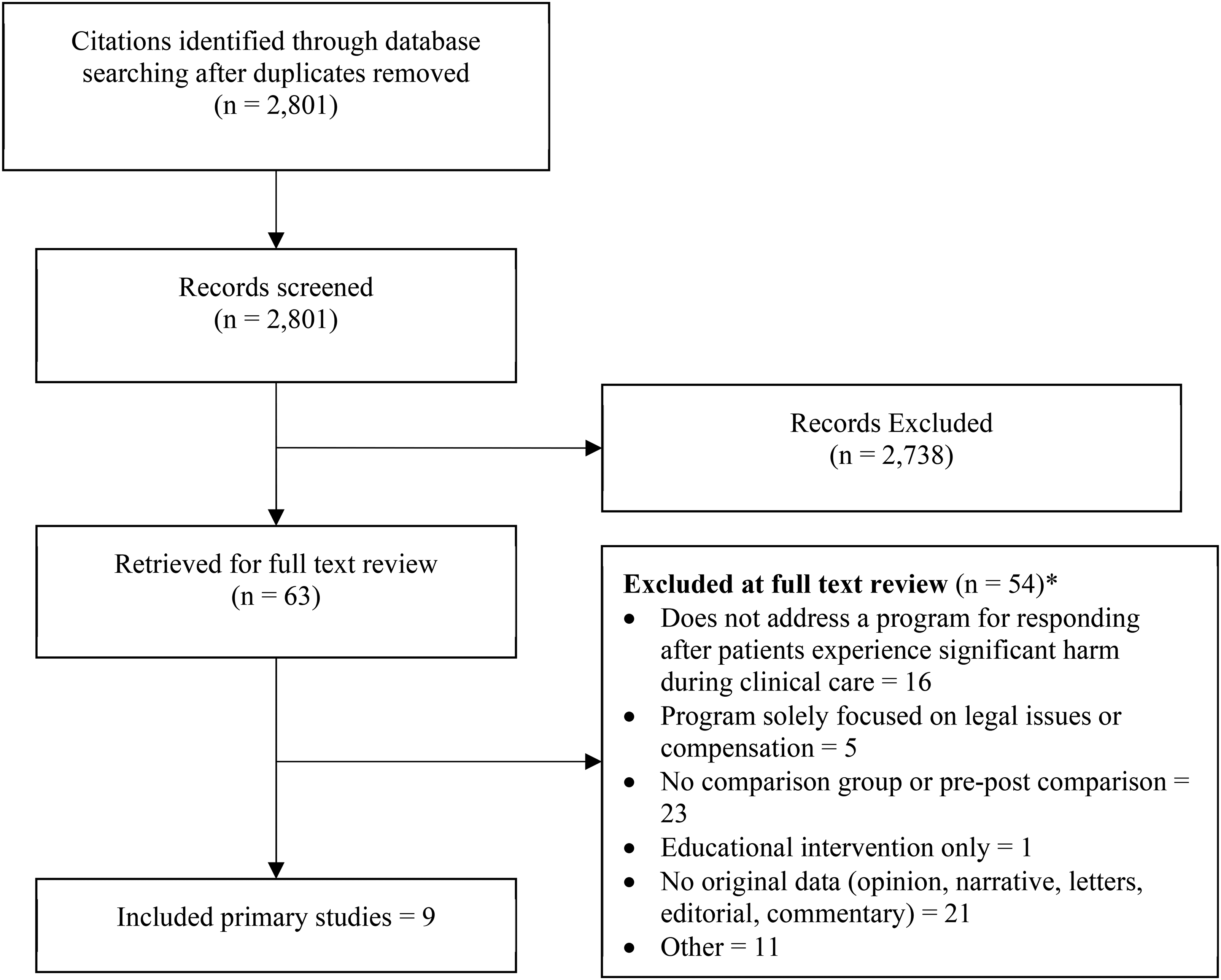
Results of the search and screening. *Total exceeds the number of records in the exclusion box because records could be excluded for more than one reason and both reviewers did not need to agree on one reason for exclusion.
Limited information was reported regarding the clinical characteristics of patient populations targeted by studies. We presumed all patients that had events eligible for a CRP were included unless otherwise specified. Three studies focused on sub-populations based on clinical conditions within their study setting (Supplement Table 2). Limited information existed regarding the demographics of populations studied. Four studies reported the proportion of patients that were female (range 52–59%) and their mean age (range 40.4–58.1 years),16,17,21,39 only two studies reported race (86–87% white),16,21 and one reported ethnicity (11.1% Hispanic). 21 Patients’ preferred language, socioeconomic status, and other demographic characteristics were not reported in any study. Although one study used a difference-in-differences analysis, 42 and another used an interrupted time series design, 17 most studies were retrospective pre-post quantitative analyses, with two involving qualitative analyses (Supplement Table 2). The number of events to which the CRPs were applied was variable (Supplement Table 2).
Most studies had concerns for bias due to confounding. One study had a critical risk of bias overall, 18 with five others having a serious risk of bias overall17,19,21,40,41 (Supplement Figure 1. Risk of bias assessments).
The CRPs described in the studies used similar processes (Figure 2). All organizations learned of harm events through reporting by healthcare team members, and some programs included events reported by patients, families, or attorneys (Table 1). Some studies did not describe criteria used to determine whether events were eligible for their CRP, whereas the most recently implemented CRPs17,21,41 described increasingly detailed criteria (Table 1). In more recent studies, “CRP event” criteria included events reaching a severity of harm threshold, externally reportable events, provider or institutional preference, or receipt of a pre-litigation notice.
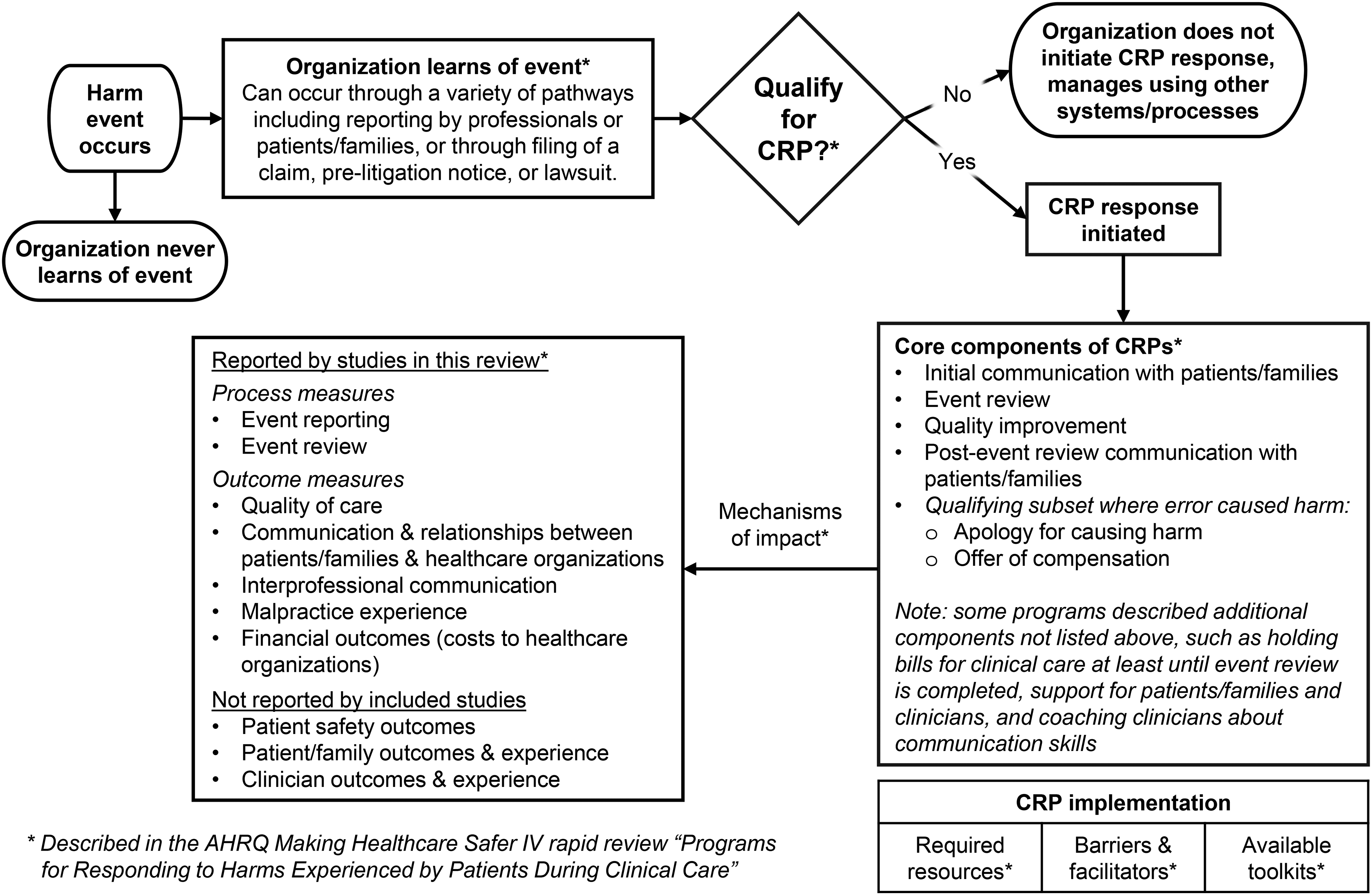
Structure and process of communication and resolution programs described in included studies.
Identification of CRP events.
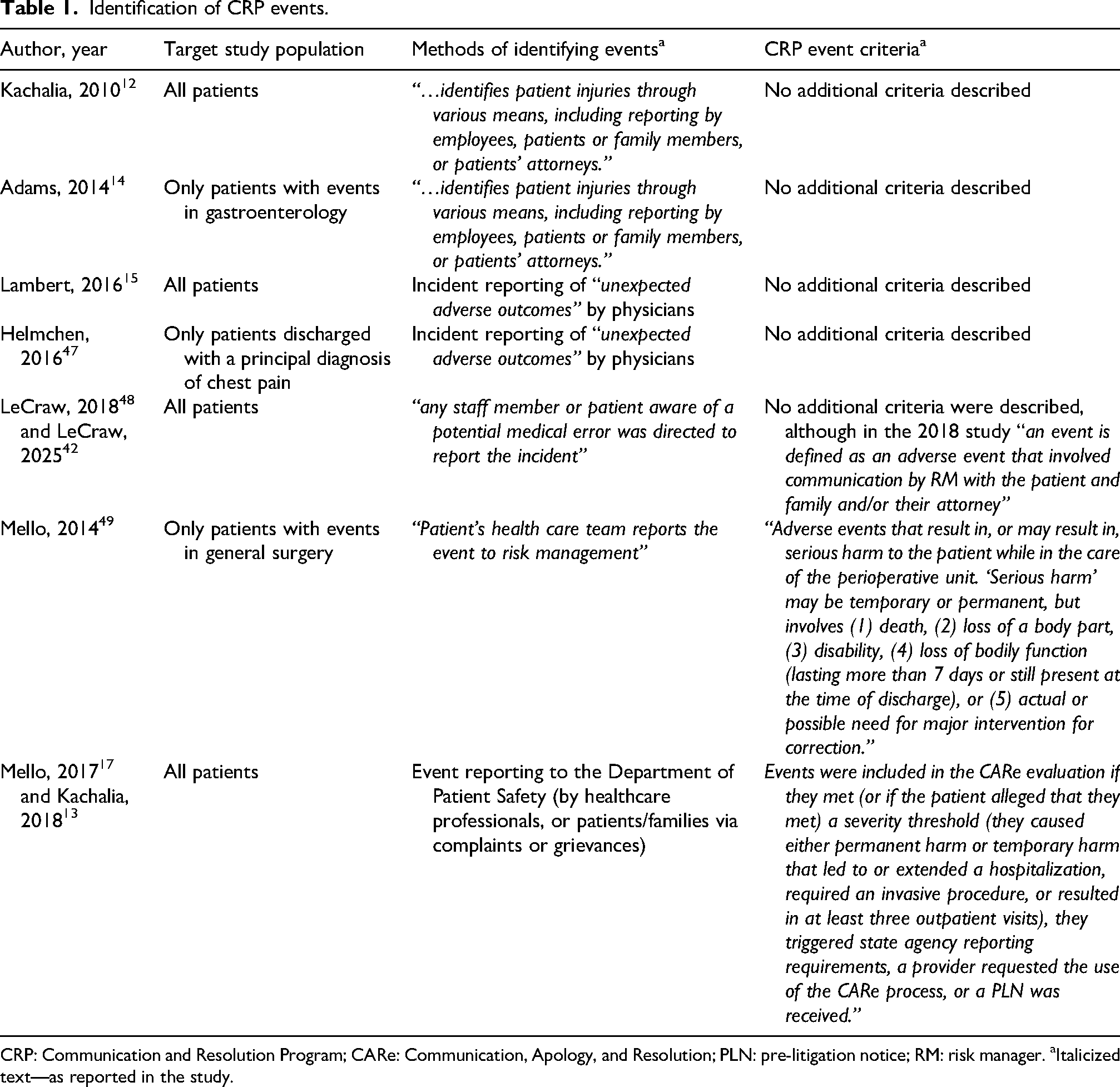
CRP: Communication and Resolution Program; CARe: Communication, Apology, and Resolution; PLN: pre-litigation notice; RM: risk manager. aItalicized text—as reported in the study.
The CRPs in the studies had five common components, and two additional components were noted in three of the five (Table 2). After identification of CRP eligible events, all studies required initial communication with patients/families, which tended to include a timely discussion of what had happened, responding to patient/family emotions, questions, and support needs, and describing the CRP process. All organizations then conducted an event review with the goals of understanding what happened, why, and whether the event had been preventable, and this was followed by the third common component: efforts to improve quality and safety. Post-event review communication was the fourth common component across CRPs and involved explaining the event review findings to the patient/family. The fifth common component was an apology and referring the event to the insurer when the event involved a claim or litigation or it had been deemed to involve serious preventable harm. Two of the five CRPs only selectively conducted the fourth or fifth components, tending to base their actions on whether the patient/family requested compensation or not (Table 2).
Intervention characteristics.
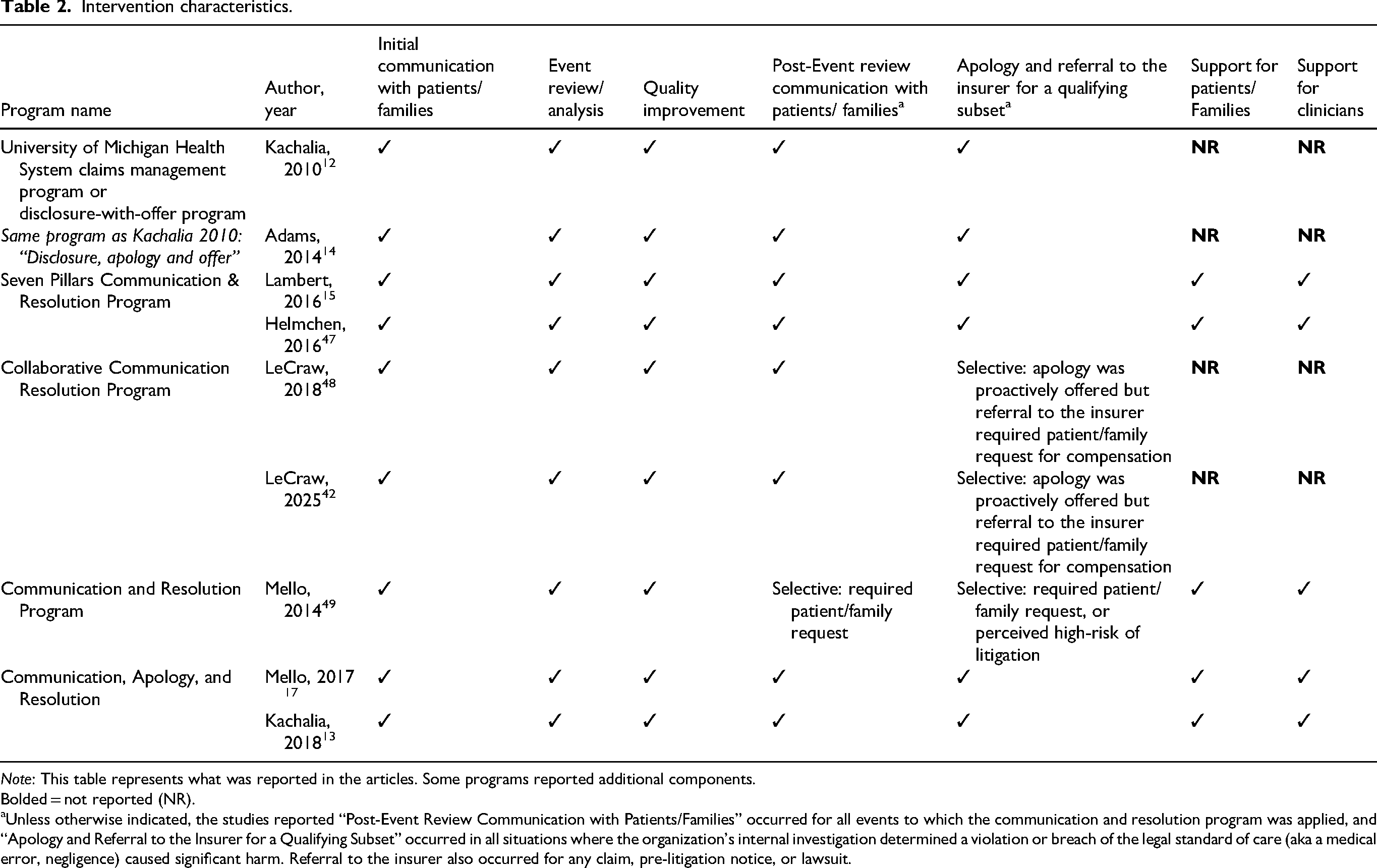
Note: This table represents what was reported in the articles. Some programs reported additional components.
Bolded = not reported (NR).
Unless otherwise indicated, the studies reported “Post-Event Review Communication with Patients/Families” occurred for all events to which the communication and resolution program was applied, and “Apology and Referral to the Insurer for a Qualifying Subset” occurred in all situations where the organization's internal investigation determined a violation or breach of the legal standard of care (aka a medical error, negligence) caused significant harm. Referral to the insurer also occurred for any claim, pre-litigation notice, or lawsuit.
Some CRPs described support for patients, families, and/or clinicians, and holding hospital and professional fee bills. More details about each component and CRP-specific variations are available in the AHRQ report. 35
Effectiveness of programs for responding after harm
A wide range of measures were reported by the studies and their findings are summarized in Table 3.
Summary of reported process and outcome measures.
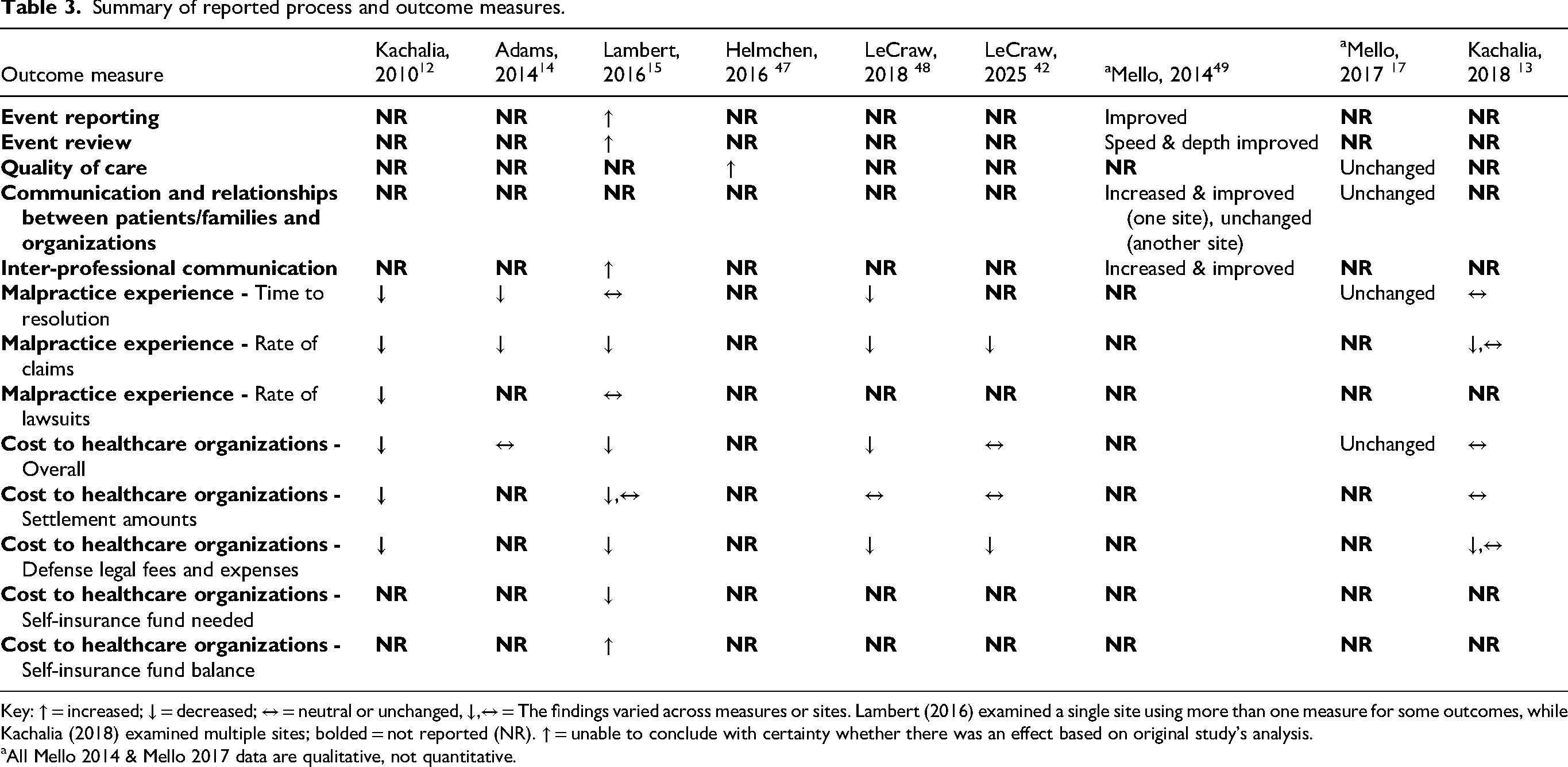
Key: ↑ = increased; ↓ = decreased; ↔ = neutral or unchanged, ↓,↔ = The findings varied across measures or sites. Lambert (2016) examined a single site using more than one measure for some outcomes, while Kachalia (2018) examined multiple sites; bolded = not reported (NR). ↑ = unable to conclude with certainty whether there was an effect based on original study's analysis.
All Mello 2014 & Mello 2017 data are qualitative, not quantitative.
Two studies evaluated
Malpractice experience and financial outcomes for healthcare organizations were the most commonly measured outcomes. Six studies evaluated
Seven studies evaluated
Discussion
This review identified nine studies that evaluated effects of CRPs. While the CRPs shared core components, their design and implementation varied. Studies primarily assessed healthcare organizations’ malpractice experience and financial outcomes, such as time to resolution, rates of claims and lawsuits, and cost to organizations. No studies provided data on CRPs’ effects on patient or family experiences, clinician outcomes, or patient safety.
The most robust CRPs showed decreases in rates of claims, defense legal fees, and expenses, although the SOE was low. Other results included lower or unchanged rate of lawsuits, overall cost to healthcare organizations, settlement amounts, and improved self-insurance funding. Variability in the findings may reflect differences in CRP design, CRP utilization, or evaluation methods. Three studies reported that they selectively applied their CRP in ways that would be expected to maximize malpractice experience and cost benefits for healthcare organizations.40–42 Beyond the included studies, inconsistent implementation and utilization of CRPs is a recognized challenge for the field. 44
The medicolegal and financial effects of CRPs may carry different implications for different parties. Healthcare leaders may see reduced costs as beneficial, while patients and families might question whether such savings come at the expense of adequate compensation. Importantly, no studies examined how much compensation reached injured patients and families. The notable patient-centered benefit identified in the studies was a shorter time to resolution. Although two CRPs showed no significant change in time to resolution, three CRPs showed 30–54% decreases.
Many of the studies were designed to focus on malpractice and financial outcomes so they could address a common barrier to implementation: the concern that CRPs might increase costs or legal risk. However, in the studies that reported it, most CRP events appeared to not involve compensable harm,21,40 and long-term data suggest many claims involve reasonable care. 45 Therefore, CRPs may be best understood as important parts of organizations’ clinical and patient safety functions with legal and financial implications, rather than primarily as a risk management or financial strategy.
Beyond malpractice and finances, evidence of CRPs’ effects was limited. Some studies noted improvements in event reporting and inter-professional communication, but the role of incentives in those studies complicates interpretation. Although the mechanisms by which CRPs can prevent or mitigate harms to patients/families are well described, 35 patient and family experiences were not directly studied in the included research. Beyond this review, some literature suggests CRPs can improve emotional, psychological, and communication outcomes compared to historical approaches.10,11,46,47 Evidence about clinicians’ experiences pre- and post-CRP implementation was also lacking.
This is the first rigorous review of CRPs’ effectiveness, and we found that CRPs had positive or neutral effects on the measured outcomes as compared with historical responses, with no evidence of harm. Given this and their ethically grounded person-centered approach, a recent Technical Expert Panel (TEP) convened by AHRQ voted to recommend implementation of robust CRPs, 48 noting that stronger evidence is needed on patient, family, clinician, and safety outcomes. Our review and the TEP's recommendation are timely given the recently enacted CMS PSSM and its CRP-related components. To inform implementation, we explored the mechanisms of CRPs’ effects and identified publicly available CRP implementation toolkits (details available in the full AHRQ report), and noted barriers and facilitators of successful CRP implementation (Supplement Table 3. Summary of common barriers and facilitators to implementation, and extensive details available in the full AHRQ report). 35
This review has several limitations, including heterogeneous study populations, interventions, methods, and outcomes, as well as a narrow focus on acute care hospitals and health systems that limits generalizability across other care settings. Patient populations were poorly described, but the available data suggests they lacked diversity, raising concerns about equity since marginalized groups are at higher risk for safety events and may experience worse outcomes.11,49,50 Descriptions of primary CRP components were often vague, varied widely, and lacked standardization, making comparisons difficult. Six of the nine studies included in our review carried at least a serious risk of bias. The rigor of the statistical methods varied widely from simple pre-post analyses to interrupted time series with sensitivity analyses and difference-in-differences analysis. These limitations, and the studies’ inconsistent findings, meant data were insufficient to grade the SOE for all but two outcomes—rate of claims and defense legal fees and expenses—which were both graded as having low SOE. Many important outcomes were not studied, and some findings were only qualitative. Older malpractice and financial outcomes may be outdated. Lastly, it is possible that negative consequences from CRP implementation have occurred but are underrepresented in the literature because of publication bias, which may further limit generalizability.
Future research should prioritize understanding CRPs’ effects on patients, families, clinicians, safety outcomes, and organizational culture. The evidence base would be strengthened by developing standardized outcome measures, conducting longitudinal studies, and exploring issues of equity. While randomized controlled trials are not feasible, more insights could be gained from multimodal research approaches, including mixed methods and cluster randomized trials. Specific aspects of CRPs that could be strengthened include more explicit linkage to quality improvement by promoting consistent engagement of patients and families in learning processes, stronger corrective actions, and more robust accountability. Lastly, future work should explore factors that inhibit and promote CRP implementation and sustainability, and optimize the systems, processes, people, and other resources necessary to improve outcomes.
Conclusions
The findings of this review support implementation of CRPs, which offer a proactive, ethical, and person-centered alternative to traditional responses after patients are harmed by their healthcare. Evidence to date shows only positive or neutral effects, though most studies focused on malpractice and financial impacts rather than patient, family, clinician, or safety outcomes. Future research on CRPs should assess their effects on patients, families, clinicians, safety outcomes, and organizational culture.
Supplemental Material
sj-docx-1-cri-10.1177_25160435261422384 - Supplemental material for Programs for responding after patients are harmed by their healthcare: A making healthcare safer IV rapid review
Supplemental material, sj-docx-1-cri-10.1177_25160435261422384 for Programs for responding after patients are harmed by their healthcare: A making healthcare safer IV rapid review by Lauge Sokol-Hessner, C. Matthew Stewart, Ritu Sharma, Allen Zhang, Thomas H. Gallagher, Allen Kachalia, Eric B. Bass and Marisa Clifton in Journal of Patient Safety and Risk Management
Footnotes
Authors’ note
The authors of this manuscript are responsible for its content. Statements in the manuscript do not necessarily reflect the official views of or imply endorsement by AHRQ or the HHS.
Acknowledgments
The authors gratefully acknowledge the following individuals for their contributions to this project: Karen S. Cosby M.D., FACEP, CPPS, Heather Sherman M.S., M.P.H., PhD, and Monika Haugstetter, M.H.A., M.S.N., R.N.,CPHQ, of the Agency for Healthcare Research and Quality (AHRQ) Center for Quality Improvement and Patient Safety; and the peer reviewers for the AHRQ Rapid Review publication, including Sigall K. Bell, M.D., Carole Hemmelgarn, M.S., Stephen Pearlman, M.D., M.S.H.Q.S., and Eric J. Thomas, M.D., M.P.H., FACP.
ORCID iDs
Ethical considerations
Ethical approval was not required.
Consent to participate
Not applicable
Consent for publications
Not applicable
Authorship contributions
All authors made substantial contributions to this study in accordance with the ICMJE requirements. All authors conceived of the research design. LSH and AZ performed analyses and take responsibility for the integrity of the data and the accuracy of the analysis. All authors participated in the interpretation of the data. All authors helped to draft and revise the paper, and approved the final version of the article prior to submission.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Agency for Healthcare Research and Quality, (Contract No. 75Q80120D00003 Task Order 75Q80122F32009).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Sokol-Hessner reports personal fees and non-financial support from the Institute for Healthcare Improvement, outside the submitted work. Dr. Gallagher reports receiving Federal and foundation grants to the University of Washington for research and implementation activity related to CRPs. None of the other authors declared potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
As a review, no primary research data was analyzed in this work. The full set of data coded from primary studies and reviews are available as appendices to the full and publicly available ARHQ report.
Supplemental material
Supplemental material for this article is available online.
