Abstract
Background
Guidewire retention (GWR) poses a patient safety risk during Central Venous Catheter (CVC) insertions and is listed as a ‘Never Event’ by the National Health Service England (NHSE). WireSafe™ is an intervention to prevent GWR, but its cost-effectiveness is uncertain. This study is an early economic evaluation comparing WireSafe™ to standard care from a UK healthcare perspective.
Methods
We conducted (i) a primary analysis of GWR related NHSE Never Events data in England between 2016 and 2020; and (ii) a cost-utility analysis, including healthcare costs and quality-adjusted life years (QALYS) for populations receiving CVC-insertions. We applied a cost-effectiveness threshold of £30,000 per QALY and considered three WireSafe™ costing scenarios (£18.50, £4.50, and £2.50).
Results
NHSE Never Events data showed 61 GWR cases, averaging 1 per month. Most incidents (92%) were identified during hospital stays, with one serious adverse outcome reported (peri-arrest). In a population of 200,000, we estimate WireSafe™ would prevent 59.92 wire retentions, 5.61 procedural adverse events, 0.3 cardiac adverse events, and 0.19 deaths, improving QALYs by 4.87. In the base case analysis WireSafe™ was not cost-effective and had an economically justifiable price of £2.44. There were high levels of uncertainty in the lowest cost-scenario (ICER 95% credible interval: Dominant; £793,398).
Conclusions
The health benefits of WireSafe™ are limited due to low GWR rates and high identification rates, making WireSafe™ viable only at low costs. Future research should prioritise obtaining more precise estimates of these parameter values which are key determinants of cost-effectiveness.
Introduction
Central venous catheters (CVC) enable easier monitoring and administration of regular medications and fluids to in-patients in intensive care units and emergency departments. They are commonly inserted using the Seldinger technique, where a guidewire guides the catheter into the vein before being removed. 1 Sometimes, guidewires are unintentionally retained. 2 Guidewire retention (GWR) is a patient safety risk as it can lead to complications such as catheter-related bloodstream infections (CRBSI), occlusion, thrombosis, arrhythmia, vascular punctures, 3 and potentially death.4–6
GWR typically requires a procedure to remove the guidewire, which may increase patients’ length of stay. It also places a personal burden on clinicians who make errors, 7 and imposes a financial burden on the NHS through substantial litigation costs. 8 GWR may pose a significant population-level risk given estimates of approximately 200,000 CVC-insertions annually in the UK. 9 The rate of GWR is unclear but has been approximated at two per month in England, 10 with similar findings in the USA. 11
Given the potential patient safety risk and associated costs, interventions to prevent GWR are important. Interventions have typically involved retraining and procedural modifications targeting human factors like distractions, interruptions, fatigue, and lack of experience that can lead to errors.11,12 Engineered solutions, such as WireSafe™, which are not dependent on human performance, may be more effective in preventing GWR. 13 WireSafe™ is as a boxed kit, containing all equipment for one guidewire insertion involving the Seldinger technique (typically CVC insertion or chest drain procedures). Importantly, while the insertion procedure can be completed up to the point of removing the guidewire, the clinician cannot progress unless the guidewire is inserted into the box, where it acts as the key to open it. This allows access to the rest of the kit and, thus, ‘forces’ the user to remove the guidewire. 14
Current epidemiological evidence on GWR is sparse, and the effectiveness of WireSafe™ has only been explored in small observation and simulation studies which found no instances of GWR when using the device.5,14,15 However, WireSafe™ is also likely to incur substantial intervention costs as devices would be required in all CVC-insertion procedures, not just those that result in GWR. Consequently there is a trade-off between the health gained due to preventing GWR and WireSafe™ device costs in relation to standard care. Due to the lack of robust effectiveness studies the current evidence base is not sufficient to support a cost-effectiveness analysis of WireSafe™ within a full economic evaluation, which is required to inform commissioning decisions for UK health technologies. 16
Where evidence is limited, early economic evaluations can provide insights into the affordability of interventions in development of healthcare systems by exploring plausible scenarios based on assumptions, rather than requiring specific point estimates from high quality sources. Early economic evaluations are particularly useful for setting research priorities by identifying key areas of uncertainty which have the greatest influence on cost-effectiveness results.17,18
This study is an early economic evaluation of WireSafe™ compared to standard care. Our objectives were to conduct: a data analysis of GWR incidents reported in centralised NHSE Never Events data; and a cost-utility analysis assessing the cost-effectiveness of WireSafe™ from a UK healthcare perspective.
Methods
Data analysis
We obtained NHSE Never Events data on ‘Guidewire’ incidents between 01 April 2016 and 31 March 2020. 19 This data is routinely collected as GWR is classed as a ‘Never Event’ by the National Health Service England (NHSE). Never Events are recognisable and wholly preventable safety incidents with potential to cause patient harm, 20 and all such incidents must be reported. The WireSafe™ device is designed to reduce central line and chest drain GWR. Therefore, only GWR Never Events following central line and chest drain procedures were included, whilst GWR following any other procedures were excluded. No additional exclusion criteria were applied.
The data analysis involved recoding free text entries on GWR incident dates, descriptions, and actions into discrete categories. Initially, one researcher (AH) summarised the relevant free text variables and distilled the initial codes, with a second researcher (MME) providing a review. Disagreements or uncertainties were resolved through discussions with a clinical expert (AC). A full description of text classification is provided in Appendix 1. To inform parameters in the cost-utility analysis, descriptive summaries (e.g. frequency counts and means) were produced for each of the derived variables (Appendix 2).
Cost-utility analysis
Population and clinical pathway
The cost-utility analysis used a decision tree to compare CVC-insertions with WireSafe™ to standard care, i.e. CVC-insertions without WireSafe™. We followed methods specified in the National Institute for Health and Care Excellence manual, including healthcare costs and health benefits measured as quality adjusted life years (QALYs), each discounted at a 3.5% per annum. 16 Results are reported as incremental cost-effectiveness ratios (ICERs) with WireSafe™ being deemed cost-effective for ICERs below £30,000 per QALY.
The decision tree (Figure 1) included a population from England, aged 57 (mean age of patients in the NHSE data). We assumed guidewire removal procedures carry comparable risks to CVC insertion procedures, thus entailing a minor risk of mortality and surgery-related adverse events (AEs), such as catheter occlusion and CRBSI. Where guidewires were retained beyond the original hospital stay, we include a risk of mortality and risk of cardiac related AEs informed by our NHSE data analysis.
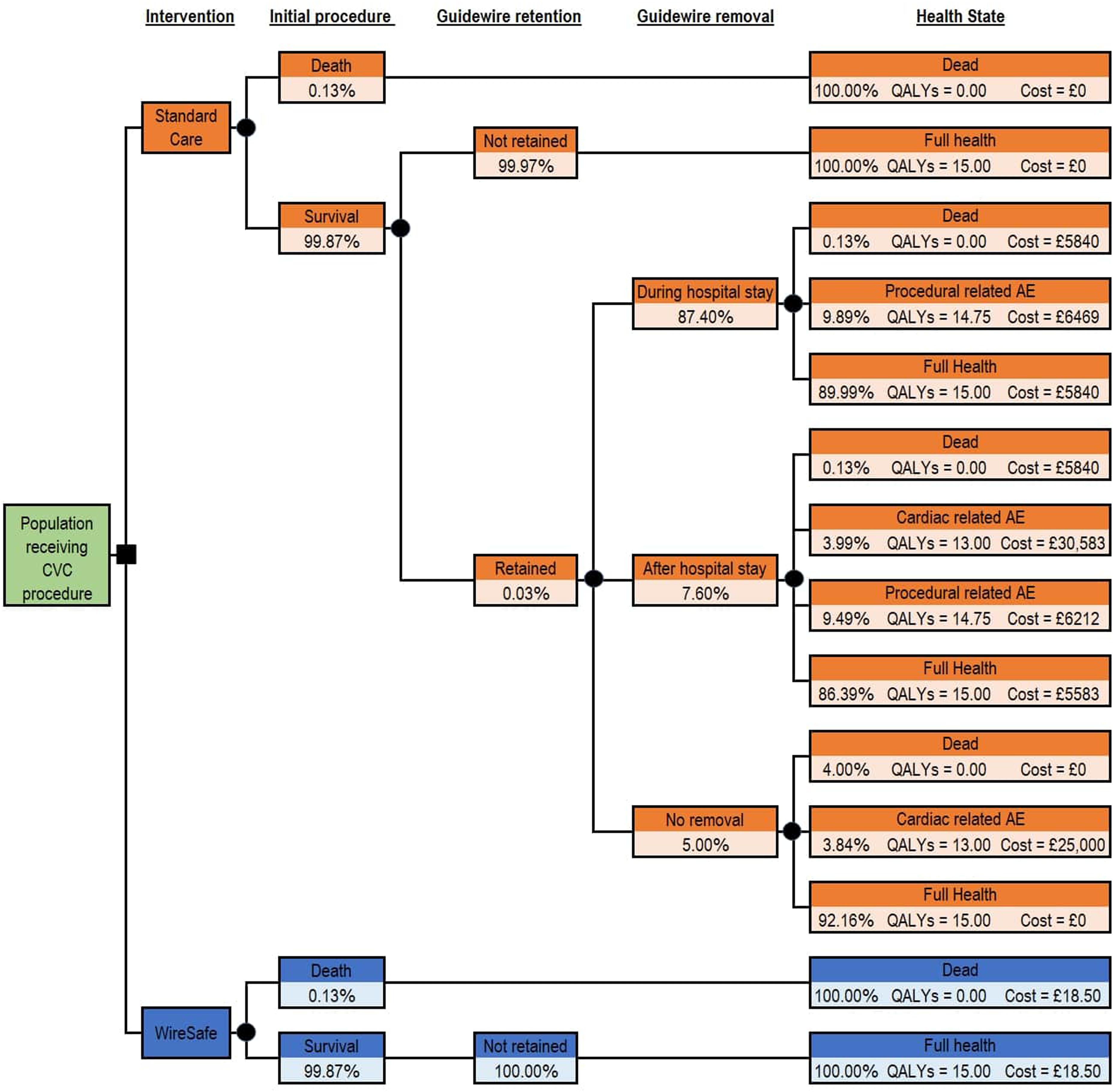
Decision tree.
Epidemiological parameters
All model parameters were obtained from the NHSE data analysis results, from published literature, or through informed discussion with clinicians and healthcare professionals with experience using WireSafe™. A full description of parameter calculations and estimates is provided in Appendix 3.
We identified three publications reporting GWR rates,6,21,22 and adopted the highest estimate of 3 per 10,000. 6 Due to being an engineered solution, our base case assumes WireSafe™ would prevent all instances of GWR, consistent with results from a forced-error simulation study where guidewires were removed 10 out of 10 times for WireSafe™ when compared to 2 out of 10 times for standard care. 5 The rate of GWR detection and removal (95%) and probability of removal during patients’ initial hospital stay (92%) were obtained from the NHSE data.
Mortality following CVC-insertions (0.13%) were calculated using rates of CRBSI equal to 0.48 per 1000 device days, mean insertion periods of 15 days, and attributed mortality for CRBSI of 17.5%. 23 The same mortality risk was applied to guidewire removal surgeries. Probabilities of surgery-related AEs were 9.9% based on reported rates of catheter occlusion and CRBSI. 24 The mortality risk and probability of cardiac related AEs following prolonged GWR were both 4%, aligning with upper literature estimates. 6
QALYs
People in “full health” were assigned health utility scores of 0.85, 25 across 26.5 years of survival based on average life expectancy at age 57 in the UK, 26 resulting in 15 QALYs after discounting. Due to lack of available data, surgery-related adverse events (AEs) were assumed to incur a disutility of 0.25 QALYs. For cardiac AEs, a disutility of 0.1 QALYs per year (equivalent to 2 discounted QALYs across full lifetimes) was applied, based on utility values for myocardial infarction survivors. 25 Whilst the NHSE data analysis identified pain as an outcome from GWR no disutility was applied as effects were reported as minor and short lived.
Costs
Manufacturers indicate WireSafe™ can be used as a replacement to standard CVC packs if the device is packaged within current kits by suppliers. 13 However NHS trusts may incur costs for both the WireSafe™ and standard CVC packs if they cannot alter current contracts with suppliers, 27 or due to clinical wastage from clinicians using multiple packs. Due to this uncertainty three costing scenarios are presented: scenario A includes the full WireSafe™ pack cost (in addition to the CVC pack) of £18.50; scenario B and C assume WireSafe™ is packaged within standard CVC packs by suppliers. The reduced costs are equal to £4.50 for “standard” (scenario B), and £2.50 for “mini” (scenario C) WireSafe™ kits. Additionally we report the economically justifiable price (EJP), which is the maximum cost at which WireSafe™ is cost-effective. The EJP was calculated by varying the intervention cost parameter and re-calculating the ICER until it equalled the £30,000 per QALY threshold.
Procedure costs for guidewire removals were obtained from NHS reference costs (2021/22) for the healthcare resource group (HRG) “Removal of Central Venous Catheter, 19 years and over” [YR44A]. 28 Removals during original hospital stays were considered unplanned non-elective short stay procedures (£853), while removals post stay were calculated as weighted averages for elective, non-elective, and day case procedures (£596).
NHS costs for conducting root cause analyses following GWR incidents were equal to £3987 and were estimated by multiplying staff unit costs, 29 by time requirements reported in personal communication with clinicians, entailing 27 h for surgical consultants and 3.5 h for band 4 administrators.
The costs of procedure-related AEs were assumed equivalent to diagnosis and treatment costs for CRBSI 30 uprated to 2021/22 prices (£629) using the NHS Cost Inflation Index. 29 Costs for cardiac-related AEs were £25,000 encompassing: treatment costs for the initial event of £2386 calculated as a weighted average of NHS reference costs for cardiac arrest HRGs (EB05A to EB05C) 28 ; and discounted ongoing treatment costs of £1115 per annum obtained by uprating costs for survivors of non-fatal myocardial infarction. 31
Sensitivity analyses
Deterministic sensitivity analyses were conducted by changing uncertain parameter values and re-calculating the ICER: scenario 1 doubled GWR rates to 0.0006; scenario 2 reduced the effectiveness of WireSafe™ from 100% to (a) 95% and (b) 80%; scenario 3 added litigation costs of £32,063 based on average costs per NHS never event legal claim 8 for all GWR resulting in an AE; and scenario 4 limited costs and QALYs impacts following cardiac related AEs to the initial year only. To identify key areas of uncertainty we generated a tornado diagram, displaying the percentage change in the ICER following a 1% change in each model parameter.
We characterised the combined uncertainty in the model parameter values through probabilistic sensitivity analysis (PSA). 32 Random variables were assigned to parameter values following recommended distributions i.e. gamma for costs, beta [0, 1] for probabilities/utilities. 33 The PSA was run over 10,000 iterations. We assumed standard error values for several model parameters as estimates were not available from the literature, Appendix 3. Due to these limitations, we use the results of the PSA to generate 95% credible intervals around the ICER, rather than specifically reporting the probability of cost-effectiveness (i.e. the proportion of times WireSafe™ is cost-effective).
Results
NHSE analysis
A detailed summary of the NHSE analysis is provided in Appendix 2. Data were collected on 103 guidewire-related Never Events, with the analysis sample limited to 61 incidents (49 related to central lines and 12 to chest drains). The average patient age was 57 years. Of these incidents, and where timing of detection was reported (n = 48 cases), 92% occurred while patients were still in hospital. The length of time between insertion and detection was within 24 h for 59%, and longer than 1-month for only 10%. Four cases of GWR were potentially associated with adverse health outcomes comprising minor pain (two cases), impact on the effectiveness of a primary medical procedure, and a peri-arrest.
Cost-utility analysis
In the base case analysis, which included our best estimate for parameter values, WireSafe™ was not cost-effective relative to standard care in any costing scenario (Table 1), with an economically justifiable price of £2.44. This was due to there being only 60 cases of GWR in 200,000 CVC-insertions, consequently WireSafe™ avoided just 57 GWR removal procedures, 5.6 surgical AEs, 0.3 cardiac AEs, 0.2 deaths, and improved health by 4.9 QALYs (Appendix 4, A4.1).
Cost-effectiveness results per person: mean (95% credible intervals).
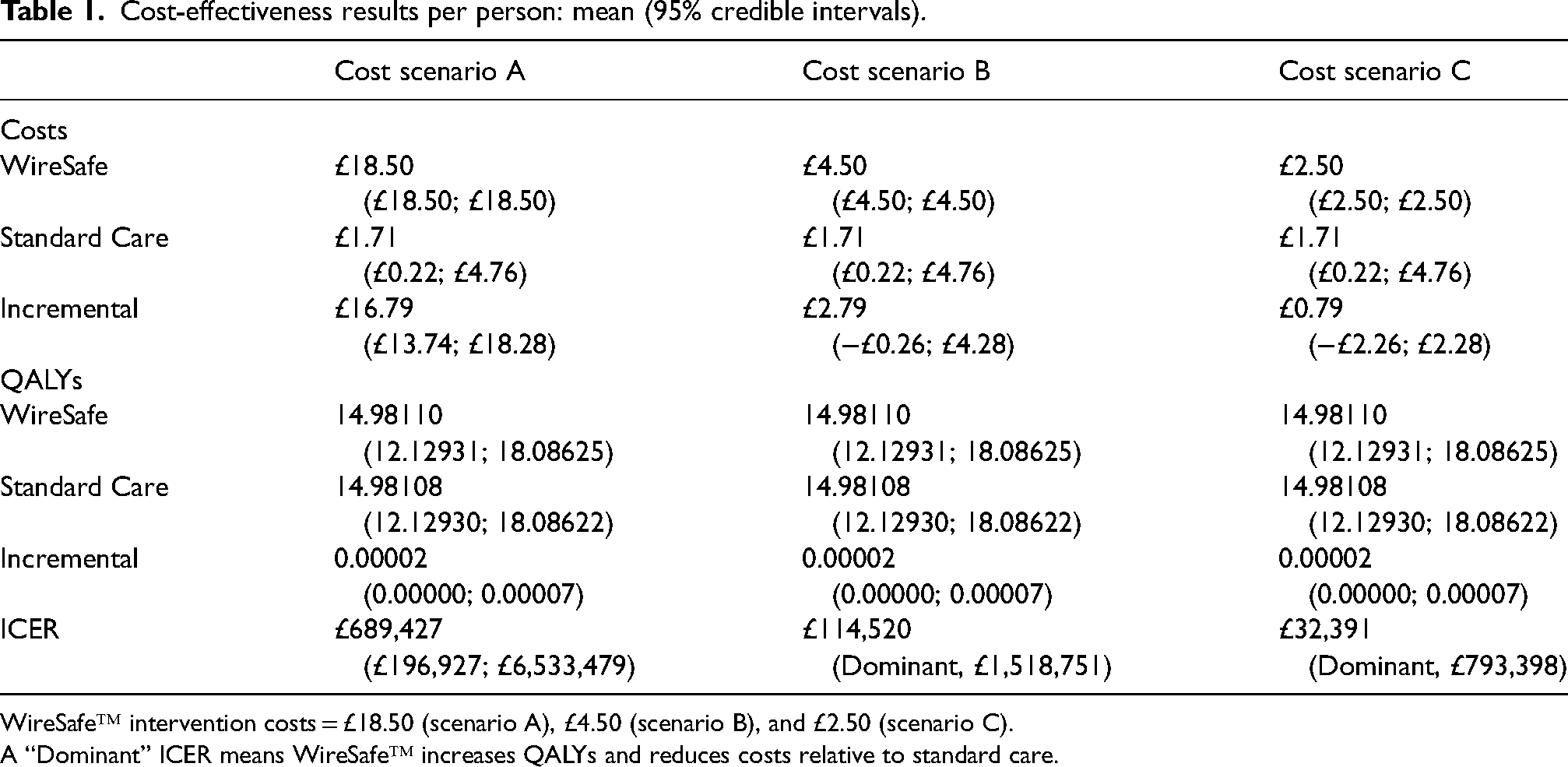
WireSafe™ intervention costs = £18.50 (scenario A), £4.50 (scenario B), and £2.50 (scenario C).
A “Dominant” ICER means WireSafe™ increases QALYs and reduces costs relative to standard care.
WireSafe™ was not cost-effective in any of the univariate sensitivity analyses (Appendix 4, A4.2) for cost scenario A (cost = £18.50). However, WireSafe™ was cost-effective when doubling GWR incidence rates for cost-scenario B (cost = £4.50) and C (cost = £2.50), and when including litigation costs for cost-scenario C only. Reducing intervention effectiveness to 95% and 80% reduced the EJP to £2.31 and £1.95 respectively. In cost scenario A, the lower 95% credible interval for the ICER exceeded £196,000 per QALY, indicating WireSafe™ was very unlikely to be cost-effective. The wide credible intervals for cost scenarios B and C reflect the considerable uncertainty in the parameter estimates (Table 1).
The parameter with the most substantial impact on the ICER was the probability of retained guidewires being detected, where a 1% change resulted in over a 7% change in the ICER. A percentage change in the GWR incidence rate, the effectiveness of WireSafe™, the proportion of cases detected during initial hospital stays, and QALYs following cardiac AEs all similarly changed the ICER by around 1% to 2%. Meanwhile, changes in healthcare costs only modestly affected the ICER (Figure 2).
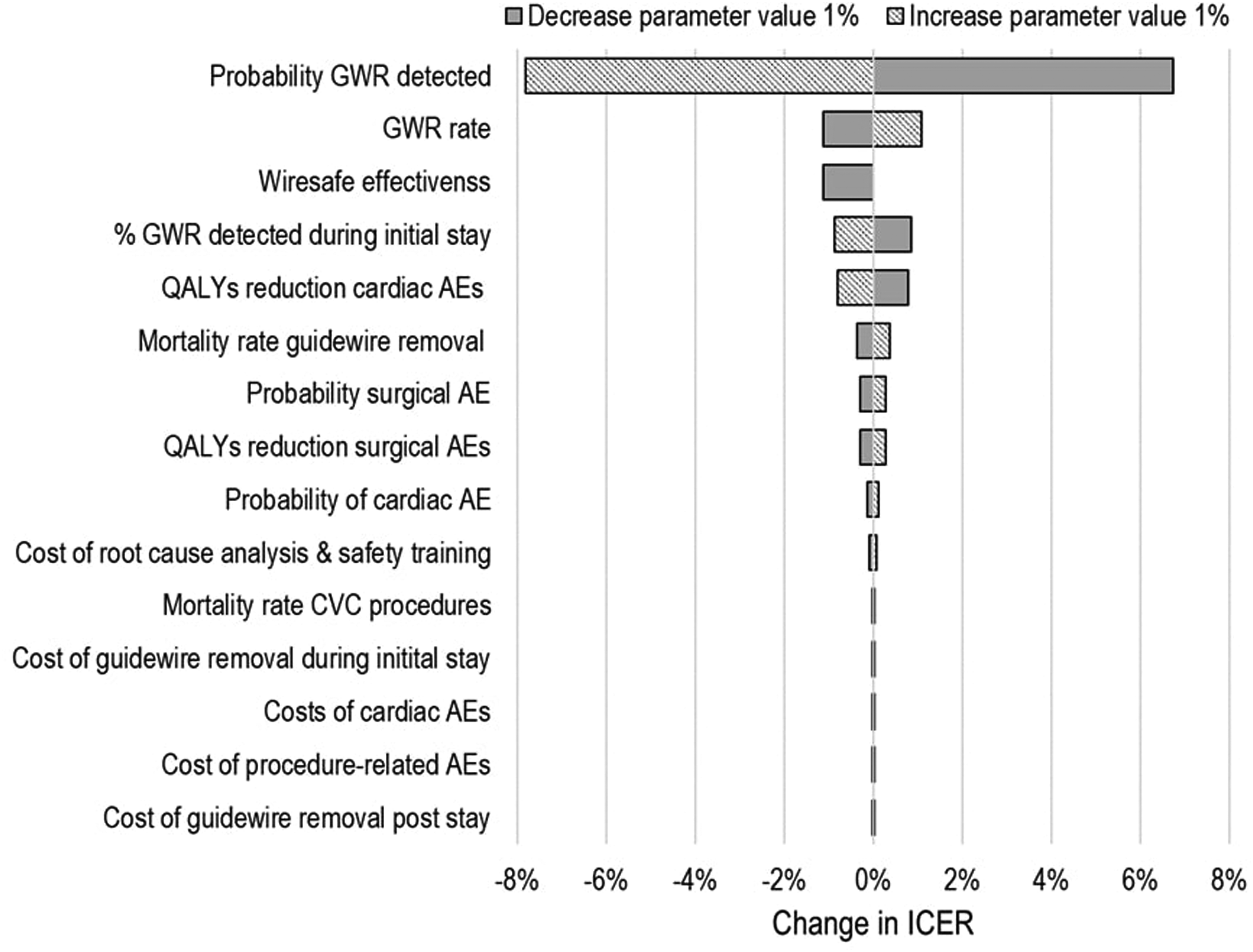
Tornado plot (% change in ICER from 1% change in parameter values).
Discussion
In the base case analysis, which applied our best approximation of parameter values, WireSafe™ was not cost-effective compared to standard care. Because of substantial uncertainties in the evidence (including intervention effectiveness), these results should not be interpreted as definitive recommendations or used as the sole basis for a stop/go decision for intervention development.17,18
Interventions like WireSafe™, which incur costs for all procedures, not just those that result in GWR, are unlikely to be cost-effective if GWR rates are low. This is because health benefits are limited to a very small number of people relative to the costs. We identified very low rates of GWR, with only 61 guidewire NHSE Never Events identified in England over four years. Estimates of GWR from large hospital studies in France and the USA are equally low, ranging from 1.5 to 3 cases per 10,000 CVC insertions,6,21,22 meanwhile smaller ward-level studies suggest higher rates in settings like intensive care and trauma.34,35
Our NHSE analysis also found almost all GWR cases were identified during patients’ initial hospital stay. There may be a reduced need for interventions to prevent GWR if retained guidewires are typically removed before causing significant harm. Both the occurrence and detection of GWR cases were key factors influencing cost-effectiveness and future research should prioritise obtaining reliable estimates for these parameters whilst also investigating heterogeneity across populations and clinical settings.
Assumptions regarding intervention costs were also key in determining cost-effectiveness. In the higher costing scenario, where NHS trusts incur costs for a full WireSafe™ pack, the ICER consistently exceeded £200,000 per QALY, well above the £30,000 per QALY threshold. Alternatively, WireSafe™ could be viable if the device is incorporated within standard CVC packages by suppliers. The lower cost scenario of £4.50 (WireSafe™ standard) and £2.50 (WireSafe™ mini) somewhat aligned with the economically justifiable prices which ranged from £1.95 to £4.88 across our sensitivity analyses.
Given WireSafe™ is an engineered solution, and in the absence of robust data, we applied a 100% effectiveness assumption. While initial evidence suggests high adherence to WireSafe™ protocol and improvements in procedural safety,10,14,15 complete elimination of GWR is uncertain, as guidewires may still fragment during insertion 4 and clinicians could override device mechanisms. Reducing effectiveness to 80% resulted in an economically justifiable price of £1.95. Research, including randomised controlled trials and larger observational studies of device-adherence/workarounds are needed to assess intervention effectiveness, and may require assessments on proxy outcomes due to low GWR incidence rate.
Our NHSE analysis identified fewer health impacts post-GWR than reported in broader literature.6,21 Only four cases reported adverse outcomes, consisting of pain, reduced procedure effectiveness, and potential peri-arrest. Other studies cited higher complication and mortality rates, though their estimates relied on historical data and personal communications. Varying assumptions in rates of health outcomes, however, had a minimal impact on the cost-effectiveness results. Similarly, the inclusion of litigation costs only modestly changed the ICER, although it did result in WireSafe™ becoming cost-effective for the lowest costing scenario. It is unclear what proportion of GWR cases result in litigation claims, whilst the inclusion of litigation costs in economic evaluation is debatable as NHS trusts pay insurance premiums rather than bearing the direct costs of claims. 36
Our findings have broad implications for patient safety in the NHS. As with all health technologies, safety interventions are only viable if they are effective and cost-effective. Since most NHSE Never Events are rare, widespread preventive measures like WireSafe™ are unlikely to be cost-effective unless they are low-cost. The NHS has committed to introduce safety measures with strong protective barriers protecting against Never Events, 20 but this may conflict with decisions by NHS trusts who are unwilling to invest in such interventions if they are not cost-effective.
This study had limitations due to available evidence, necessitating an early economic evaluation focusing only on key events, costs, and outcomes related to WireSafe™ and GWR. Not all relevant costs or savings associated with WireSafe™ were included, such as training requirements, impact on procedural time, and multi-function as a sharps container which may reduce needle stick injuries. 27 Due to a lack of evidence, we also did not include costs related to reablement care after hospital discharge, which may have been required in some patients who received surgeries to remove retained guidewires. The exclusion of these costs is not likely to impact findings for the higher cost scenarios but could result in WireSafe™ being cost-effective for the lowest cost scenario where the ICER was close to the £30,000 per QALY threshold. Additionally, our NHSE analysis only included reported GWR cases, there may be unreported cases that result in more substantial patient harm. Incomplete or early reporting of patient outcomes after GWR cases without long-term follow-up may equally explain low AE rates. Finally, lack of evidence on parameter variances and distributions limited findings from the PSA resulting in very wide credible intervals.
Footnotes
Acknowledgements
We would like to thank the device inventor- Dr Peter Young- and David Thompson from the device manufacturing company –Venner Wire- for their time and provision of information regarding the differing device costs and issues. We would also like to thank Dr Will Lea for his advice regarding root cause analyses and associated staffing and costs.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This report is independent research funded by the National Institute for Health and Care Research Yorkshire and Humber ARC (NIHR 200166). The views expressed in this publication are those of the author(s) and not necessarily those of the National Institute for Health Research or the Department of Health and Social Care.
Ethical approval
This study did not require ethical approval. The data analysis used routinely collected and anonymised data made publically available by NHSE. The cost-utility analysis used secondary data from published sources.
Nominated author
Richard Mattock is the nominated author and guarantor for this work. The guarantor accepts full responsibility for the work and/or the conduct of the study, had access to the data, and controlled the decision to publish.
Appendix 1: Data Analysis Methods
Methods for variable derivation and text classification.

| Variable | Data type | New variables |
|---|---|---|
| Region | Nominal | - |
| Date of birth | Date | Used in combination with date of incident to deduce age at time of incident |
| Date of incident, and date detected | Date | Used to deduce length of time between insertion and detection, with some checks also made by reference to the free text data which sometimes mentioned timings as well. |
| Description of what happened | Free text | Distilled to deduce length of time between detection and removal (where mentioned), when removal took place (whilst still in hospital versus after original discharge), and patient health outcomes. |
| Immediate action taken | Free text | |
| Key findings | Free text |
Appendix 2: Data Analysis Frequency Tables
Patient health outcomes.

| Patient health outcome | Frequency | Details |
|---|---|---|
| No mention was made of patient health outcomes | 38 | |
| Patient reported as having no adverse outcomes directly linked to the GWR | 19 | Examples include: |
| Pain (unknown duration) | 1 | Patient reported right-sided chest pain following drain, which clinician reviewer advised could be due to GWR. |
| Possible pain | 1 | Patient attended A&E and had their arm X-rayed. Clinician reviewer advised GWR may have caused the admission as the decision was taken to X-ray the arm. |
| Reduced effectiveness of primary medical procedure | 1 | The clinician reviewer reported that the drain was effective enough to relieve the tension of the lung but not to resolve the pneumothorax. |
| Possible peri-arrest | 1 | The clinician reviewer advised that a guidewire can cause arrhythmia which could result in arrest. But, unclear as to whether caused by the GWR. |
Appendix 3
Full list of model parameter values.
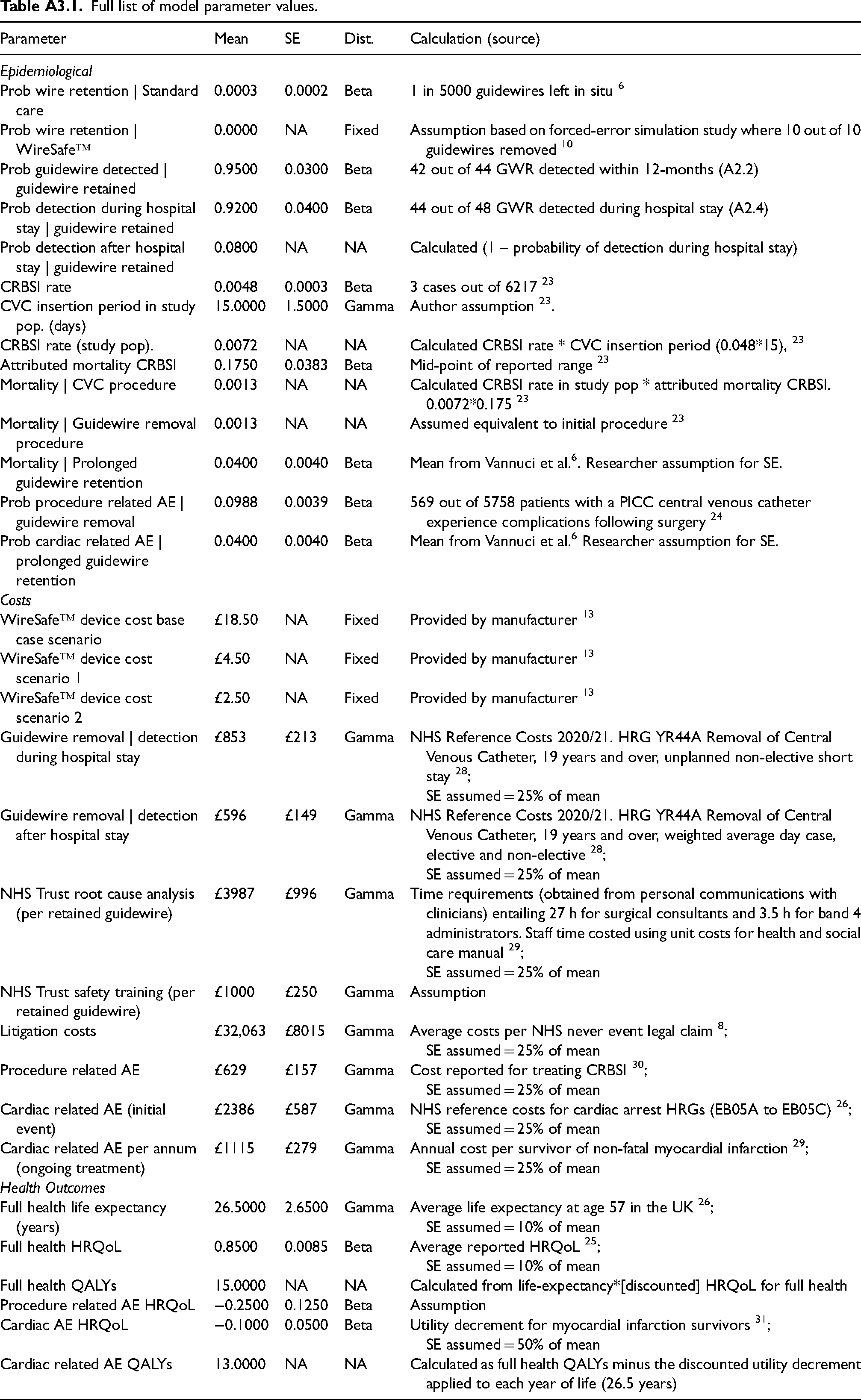
| Parameter | Mean | SE | Dist. | Calculation (source) |
|---|---|---|---|---|
| Epidemiological | ||||
| Prob wire retention | Standard care | 0.0003 | 0.0002 | Beta | 1 in 5000 guidewires left in situ 6 |
| Prob wire retention | WireSafe™ | 0.0000 | NA | Fixed | Assumption based on forced-error simulation study where 10 out of 10 guidewires removed 10 |
| Prob guidewire detected | guidewire retained | 0.9500 | 0.0300 | Beta | 42 out of 44 GWR detected within 12-months (A2.2) |
| Prob detection during hospital stay | guidewire retained | 0.9200 | 0.0400 | Beta | 44 out of 48 GWR detected during hospital stay (A2.4) |
| Prob detection after hospital stay | guidewire retained | 0.0800 | NA | NA | Calculated (1 – probability of detection during hospital stay) |
| CRBSI rate | 0.0048 | 0.0003 | Beta | 3 cases out of 6217 23 |
| CVC insertion period in study pop. (days) | 15.0000 | 1.5000 | Gamma | Author assumption 23 . |
| CRBSI rate (study pop). | 0.0072 | NA | NA | Calculated CRBSI rate * CVC insertion period (0.048*15), 23 |
| Attributed mortality CRBSI | 0.1750 | 0.0383 | Beta | Mid-point of reported range 23 |
| Mortality | CVC procedure | 0.0013 | NA | NA | Calculated CRBSI rate in study pop * attributed mortality CRBSI. 0.0072*0.175 23 |
| Mortality | Guidewire removal procedure | 0.0013 | NA | NA | Assumed equivalent to initial procedure 23 |
| Mortality | Prolonged guidewire retention | 0.0400 | 0.0040 | Beta | Mean from Vannuci et al. 6 . Researcher assumption for SE. |
| Prob procedure related AE | guidewire removal | 0.0988 | 0.0039 | Beta | 569 out of 5758 patients with a PICC central venous catheter experience complications following surgery 24 |
| Prob cardiac related AE | prolonged guidewire retention | 0.0400 | 0.0040 | Beta | Mean from Vannuci et al. 6 Researcher assumption for SE. |
| Costs | ||||
| WireSafe™ device cost base case scenario | £18.50 | NA | Fixed | Provided by manufacturer 13 |
| WireSafe™ device cost scenario 1 | £4.50 | NA | Fixed | Provided by manufacturer 13 |
| WireSafe™ device cost scenario 2 | £2.50 | NA | Fixed | Provided by manufacturer 13 |
| Guidewire removal | detection during hospital stay | £853 | £213 | Gamma | NHS Reference Costs 2020/21. HRG YR44A Removal of Central Venous Catheter, 19 years and over, unplanned non-elective short stay
28
; |
| Guidewire removal | detection after hospital stay | £596 | £149 | Gamma | NHS Reference Costs 2020/21. HRG YR44A Removal of Central Venous Catheter, 19 years and over, weighted average day case, elective and non-elective
28
; |
| NHS Trust root cause analysis (per retained guidewire) | £3987 | £996 | Gamma | Time requirements (obtained from personal communications with clinicians) entailing 27 h for surgical consultants and 3.5 h for band 4 administrators. Staff time costed using unit costs for health and social care manual
29
; |
| NHS Trust safety training (per retained guidewire) | £1000 | £250 | Gamma | Assumption |
| Litigation costs | £32,063 | £8015 | Gamma | Average costs per NHS never event legal claim
8
; |
| Procedure related AE | £629 | £157 | Gamma | Cost reported for treating CRBSI
30
; |
| Cardiac related AE (initial event) | £2386 | £587 | Gamma | NHS reference costs for cardiac arrest HRGs (EB05A to EB05C)
26
; |
| Cardiac related AE per annum (ongoing treatment) | £1115 | £279 | Gamma | Annual cost per survivor of non-fatal myocardial infarction
29
; |
| Health Outcomes | ||||
| Full health life expectancy (years) | 26.5000 | 2.6500 | Gamma | Average life expectancy at age 57 in the UK
26
; |
| Full health HRQoL | 0.8500 | 0.0085 | Beta | Average reported HRQoL
25
; |
| Full health QALYs | 15.0000 | NA | NA | Calculated from life-expectancy*[discounted] HRQoL for full health |
| Procedure related AE HRQoL | −0.2500 | 0.1250 | Beta | Assumption |
| Cardiac AE HRQoL | −0.1000 | 0.0500 | Beta | Utility decrement for myocardial infarction survivors
31
; |
| Cardiac related AE QALYs | 13.0000 | NA | NA | Calculated as full health QALYs minus the discounted utility decrement applied to each year of life (26.5 years) |
Appendix 4
ICERs for WireSafe™ versus standard care, mean (95% credible interval).
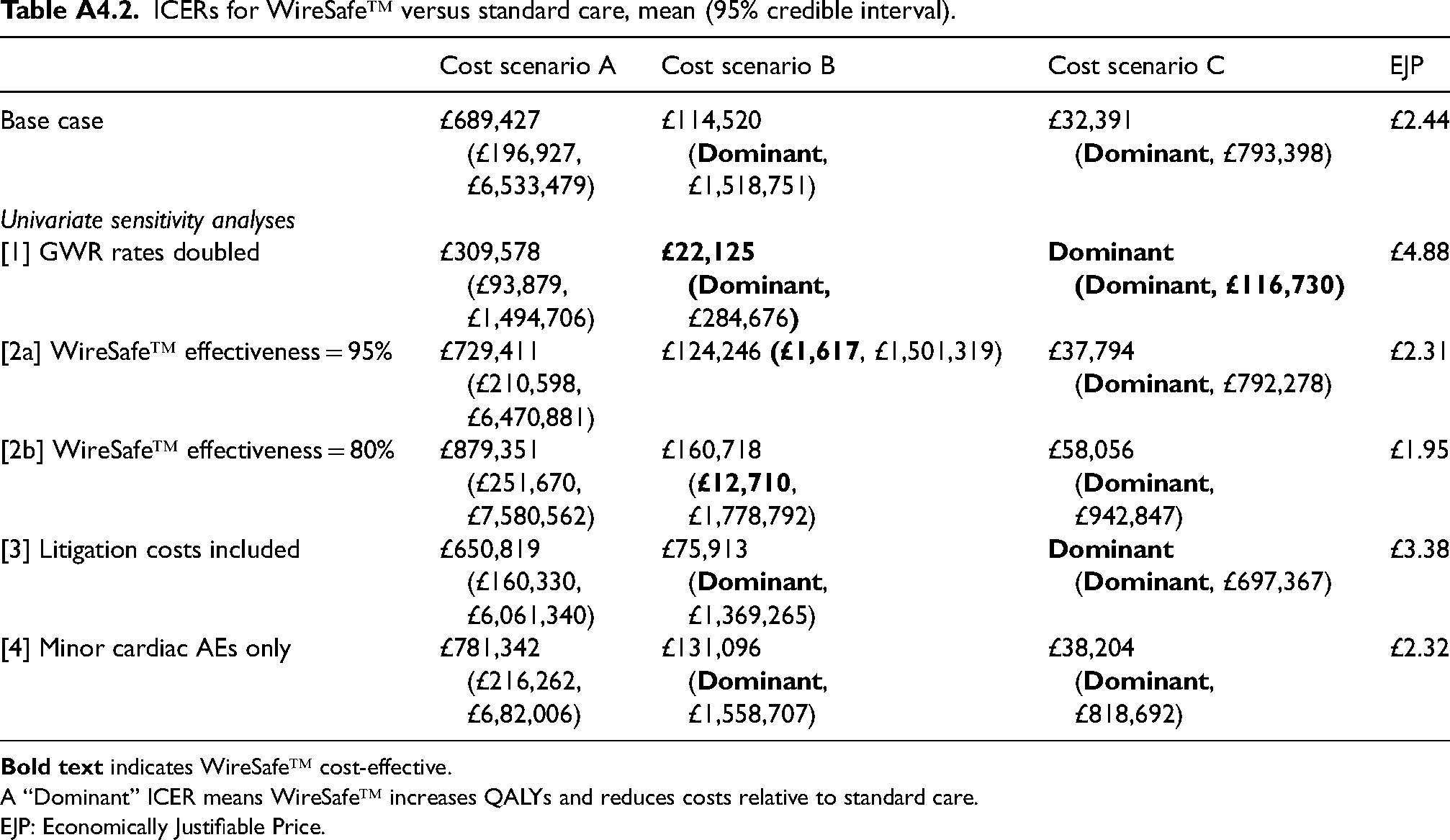
| Cost scenario A | Cost scenario B | Cost scenario C | EJP | |
|---|---|---|---|---|
| Base case | £689,427 (£196,927, £6,533,479) |
£114,520 ( £1,518,751) |
£32,391 ( |
£2.44 |
| Univariate sensitivity analyses | ||||
| [1] GWR rates doubled | £309,578 |
|
£4.88 | |
| [2a] WireSafe™ effectiveness = 95% | £729,411 |
£124,246 |
£37,794 |
£2.31 |
| [2b] WireSafe™ effectiveness = 80% | £879,351 |
£160,718 |
£58,056 |
£1.95 |
| [3] Litigation costs included | £650,819 |
£75,913 |
£3.38 | |
| [4] Minor cardiac AEs only | £781,342 |
£131,096 |
£38,204 |
£2.32 |
A “Dominant” ICER means WireSafe™ increases QALYs and reduces costs relative to standard care.
EJP: Economically Justifiable Price.
