Abstract
Background
Healthcare-related adverse events directly impact patient safety. Effective reporting of adverse events and workplace factors affecting the quality and quantity of reporting has been a recent focus. In Nova Scotia, pharmacists have been required to report quality-related events (QREs), errors, and near misses since 2010 through the Canadian Pharmacy Incident Reporting (CPhIR) database. This study aims to better understand how healthcare professionals who use the CPhIR system feel about their experience with QREs and the QRE reporting process.
Methods
A total of 1000 registered pharmacists and staff were contacted through the Nova Scotia College of Pharmacists. Five focus group meetings were conducted from May to October 2018, consisting of 17 community pharmacists, pharmacy technicians, and assistants.
Analysis
Thematic analysis was used to identify and define emerging themes in the transcripts by multiple readers. The Actor-Network Analytical Theory helped draw a web of connections in producing a safety culture that extends beyond the roles at the dispensing counter.
Results
It was found that participants were committed to minimizing and reporting errors, but using the CPhIR database system is both time-consuming and onerous. Additionally, there was a lack of continuity in communicating the QRE system protocols and compliance protocols for the system.
Conclusions
Community-based pharmacy culture needs to shift from a compliance-based culture, where error reporting is based on completion and volume, to a just culture that embraces quality and learning from mistakes, a critical element of safe dispensing.
Keywords
Introduction
An alarming 7.5% of patients who seek treatment in Canadian hospitals experience adverse effects. 1 Adverse effects are defined as complications that are caused by health care management as opposed to the patient's underlying illness. 1 Adverse events experienced by patients are preventable in 36.9% of cases (Baker, 2004). In Canada, of almost 2.5 million hospital admissions that occur yearly, 185,000 are due to an adverse effect. 1 The high frequency at which adverse effects occur in hospital settings is indicative that patient safety may not always be at the forefront in health care management.
In order to improve how adverse effects are handled and reported, investment is needed from federal, provincial, and territorial jurisdictions. Funding is needed to support the development of information technology infrastructures to provide a standardized system of identifying, reporting, and tracking patient safety data. In order for these information technology infrastructures to work, a culture that supports reporting, learning, and improving the transparency of patient safety data is needed. 2 Pharmacies are one of the first professions in healthcare to implement a culture of transparency. Pharmacy regulatory authorities are creating a culture of transparency by requiring their workforce to report adverse effects or “events and near-misses anonymously to an independent third party.” 2
In Nova Scotia, the regulatory organization for the profession of pharmacy (Nova Scotia College of Pharmacists—herein referred to as the College) employs the Institute for Safe Medicine Practices (ISMP) in Canada to manage a mandatory system of quality-related event (QRE) reporting. A QRE is any form of error that occurs along the value chain from prescriber to patient use. These errors may occur at any stage of the value chain. The resulting harm of a QRE ranges from none (e.g. misspelled name) to high (e.g. death). The majority of QREs do not result in harm because they are caught and corrected along the value chain. The College requires all community pharmacies to report all QREs.
Nova Scotia pharmacists have been required to report all QREs since 2010. 3 Between 2010 and 2017, pharmacists, technicians, and assistants reported 98,097 QREs, of which 82% (or 80,488) did not reach the patient. 4 These are considered to be near misses in the reporting system. 3 Incorrect dose or frequency was the most common QRE, accounting for 25.6% of reported QREs. 4 Most of the QREs occurred at order entry, followed by preparation and dispensing, and prescribing. Within the seven years of reporting, two deaths occurred, which account for 0.002% of all prescriptions completed in the 301 community pharmacies in the province. Although rare, high harm does occur, and the rate at which high harm occurs did not decline during the reporting period. This severity of the harm, despite the low frequency, poses a complex problem in organizational culture and management requiring innovative examination. 5
Objective
The goal of this research is to explore underlying issues in QRE reporting in Nova Scotia community pharmacies by describing the opinions and experiences of pharmacists, technicians, and assistants.
Methods
Approximately 1000 registered pharmacists, registered pharmacy technicians, and pharmacy assistants were contacted through the college.
Five focus group meetings were conducted over six months, from May 2018 to October 2018 to facilitate the engagement of diverse perspectives from community pharmacies. A saturation point, where no new themes were surfacing, was reached after five groups. The focus groups ran between 60 and 90 min and used a semi-structured interviewing approach. The facilitator introduced the first topic and only introduced other topics if they did not come up in the discussion among the participants. In this manner, there is as little prompting of participants as possible.
The following questions were developed for discussion in the focus group based on the findings of previous work:
Is error reporting important? How does the QRE process work in their pharmacy (share of load among staff)? What is the relationship to corporate systems (for those in chain stores)? What is the relation to customer service? How long does it take? How are these medication error reporting processes communicated (e.g. bulletins, website, other)? How do you want these processes to be communicated? What is the system like (a website or printout from paper as a prop)? How are numbers reported? Is there differentiation between corrected error versus near miss? What is the resulting “harm”?
The transcripts were reviewed for themes present in the data. Multiple readers reviewed the transcripts, and the emerging themes were compared. There was general agreement on the themes present and how they related to the topics investigated. The transcripts were mined for exemplar quotes to represent the identified themes.
Data analysis
Thematic analysis was used to identify and define themes in the data and align with methods used in similar work.6,7
Humans, technologies, and social structures all exist within a recurring relationship. 8 In this relationship, the social and organizational actors and technological infrastructures are created and recreated as the human actors within the relationship use them. 8 This theory is the Actor-Network Theory (ANT) and can be applied to the use of technology within the healthcare system.
Individuals act based on their understanding of concepts espoused by organizations in which they are participating. 9 Pharmacists, therefore, enact safety culture, Continuous Quality Improvement (CQI), and Quality-Related Event (QRE) reporting in a manner consistent with their values and the interaction of those values with the regulations promulgated by the College in the context of the Pharmacy Association of Nova Scotia (PANS). We approached the data by surfacing the themes related to QRE reporting which our participants agreed upon.
The Actor-Network Analytical Theory applied in this research helps to draw a web of connections in the production of a safety culture that extends beyond the roles at the dispensing counter. The physician, the regulator, PANS, Corporate owners, drug manufacturers, the media, government, and the public are a part of the CQI and QRE web. Many stories of QREs cite physical devices as actors (computers, software, bottles, pill colors, etc.). The color of a pill could cause confusion between two different medications increasing the likelihood of a dispensing error. ANT provides a means by which to relate these non-human and non-corporeal things and constructs to humans and surface plausible explanations of error processes. Ultimately humans act, but how they act is influenced and constrained by things as well as the other humans with which they associate. 10
As shown in Figure 1, the bolded circles were the most prevalent themes from the focus groups. QREs, safe dispensing practices, communication practices and structures, and the emphasis on the relationship the pharmacy has with the community. The smaller circles introduce the influence of other professional roles, time, workload, and technology. The lines depict the interconnections in the network of actors. Society is only engaged through the communication channels; however, workload is connected to the four main themes in the network placing an emphasis on the role workload plays in influencing errors as a non-corporeal actant.
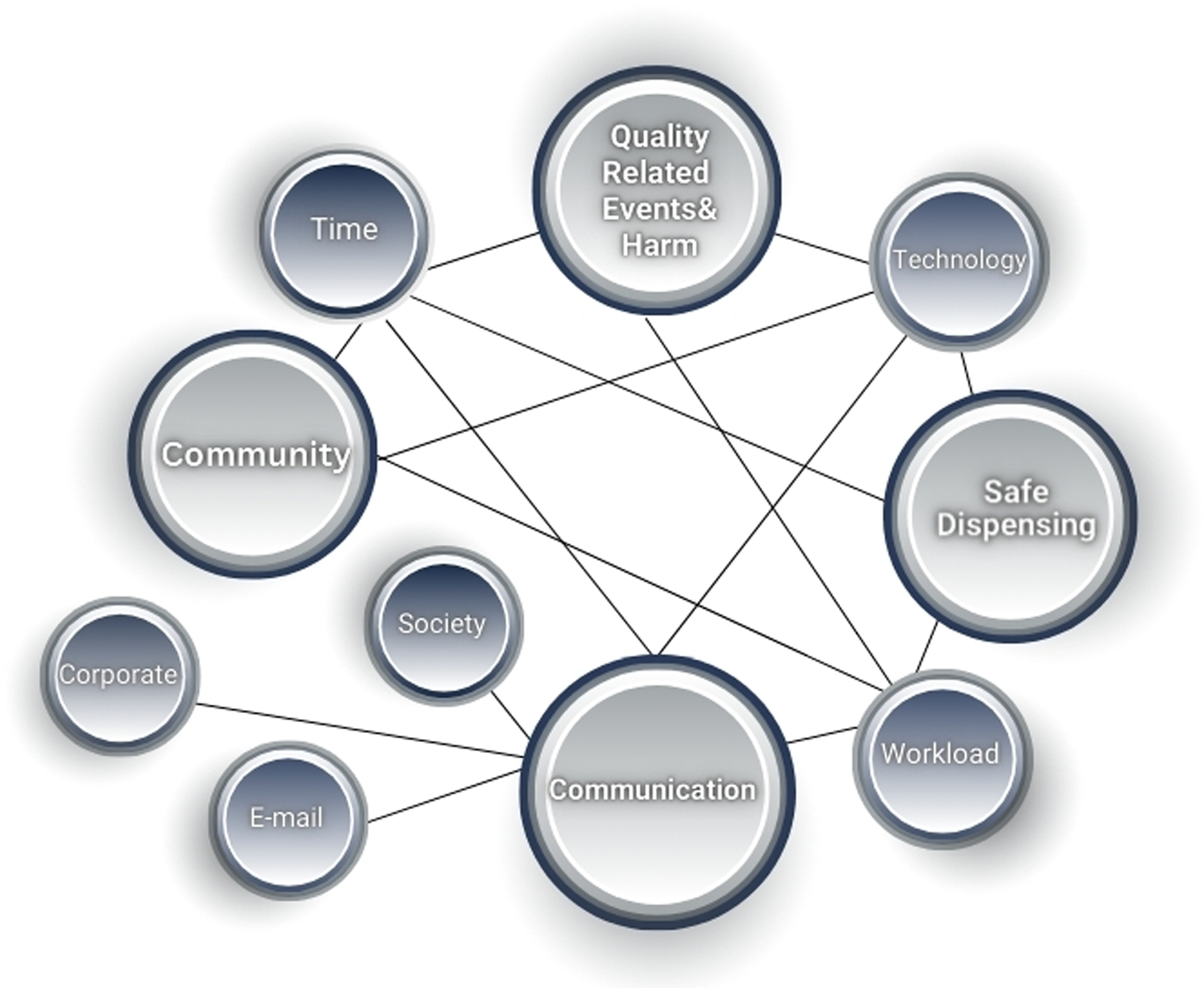
Focus group themes.
Results
The feedback from 17 community pharmacists, technicians, and assistants was collected over six months through focus group engagements. Participants were primarily female (all but one) and ranged in age between 30 and 56. No new themes emerged after the five engagements as the pharmacists, technicians, and assistants have consistent views related to QRE reporting. Key themes regarding participants’ experiences with QREs and the QRE reporting process are listed in Figure 1. These themes interrelate with one another, physical structures such as the College and the pharmacy chains as well as the individual pharmacists and their interrelations with one another in the Actor-Network. Each theme is supported by many comments made by participants which are represented by exemplar quotes.
Safe dispensing
“It's very important that medications go safely to the patient. And, anything that helps is important.” (Participant two—exemplar).
Pharmacists indicated that their role is to use their knowledge of medications in order to aid and protect patients. The participants reported that incidents (e.g. incorrect medication and dosage) are regarded as critical QREs, but note that there is an acceptance that mistakes happen and need to be reported.
Compliance
“I work for [company] and we have an internal reporting system and that's critical. When staff pharmacists send it, it goes to the pharmacy manager and also my head office. So we report important things … So that's what we do and what we learn from. We share and we spend a lot of time on it. We enter it in CPhIR because we have to. And I ask my assistant not to spend more time than 30 seconds…the priority is patients waiting, or pill packs to package…CPhIR is the least priority. If it's an incident, we document it internally, with details in case legal things happen, [but for] CPhIR we just do it quickly.” (Participant four)
Participants indicated that the need to report even small errors (label incorrectly typed—name misspelled was a common “sig error”) by the regulatory body as fully as those which may have (if not caught) caused significant harm was identified as unnecessary workload and time pressures that are inconsistent across the industry.
Communication
“I usually look through the bulletins… but I get so many emails that I don’t often check them. But… every quarter… I’ll also look through the bulletins just to see if there's anything… helpful to my staff and I’ll disseminate that to them along with all of our error information. Lately, I haven’t found that there was much that was applicable. There's a lot of hospital incidents that just doesn’t apply to us… But there definitely have been incidents that seem applicable that I’ve shared with staff, but I honestly can’t think of any off the top of my head.” (Participant nine)
One participant mentioned a program binder they discovered at a pharmacy, while others would report that they had created their own binder of processes for the staff. The participants expressed frustration with the volume and utility of communication from the regulator and emphasized the problematic nature of the communication structure which is exacerbated by increasing workload and time pressures to manage patient demands. Many of the notices, bulletins, and other official communications come via email. Participants indicated that these communications add to workload and often go unnoticed.
QRE and harm
“We’ve certainly made some changes … based on incidents that occurred in our store and looking through what we’ve reported is a good way for us to review and see what areas we need to work on and identify times when we’ve made the same kind of error. …. But on the more local level, just looking at our own store and our own errors, we’ve identified lots of things we’ve been able to improve upon so for that I find it really useful.” (Participant seven)
Participants stated that pharmacists are trained to minimize harm and do what they can to prevent medications with the potential for harm from reaching patients. Participants emphasized that the punitive nature of the inspection system seems to interfere with the effective use of the QRE reporting system. Additionally, over-communication and the use of email have reduced the value of the bulletin process. Despite these barriers, pharmacists indicate they have become more alert to potential mistakes and have taken action.
Discussion
The findings of this study suggest that there is a need for a shift in the organizational culture in community pharmacies across Nova Scotia. Culture needs to shift from a compliance-based culture, 11 where error reporting is based on completion and volume, to a Just Culture 12 that embraces quality and learning from mistakes, a critical element of safe dispensing. 12 The pharmacists identified two key areas that present opportunities to help make this cultural shift through streamlined communication from the regulatory agency and standardized reporting practices.
Shifting to a just culture
Currently, the healthcare industry puts emphasis on production, efficiency, and cost control. Healthcare workers have expectations of perfectionism which fosters a compliance workplace culture where individuals may be scared to report errors. 11 A compliance culture not only results in poor reporting procedures but can also cause pharmacists undue stress about job security and can cause mistrust of the organization they are employed for.7,13 This becomes especially problematic as reporting an error is only one of many actions that should accompany the presence of an error. When errors are acknowledged, the events leading to and following the error should be analyzed to establish a just culture to apply the learnings to support patient safety. 13 Learning from errors and identifying potential issues will significantly improve the safety of the system. 14 But, if healthcare professionals do not trust the system, the entirety of the learning process cannot take place.
Streamlined communication practices
In Halsall's (2012) focus group study on healthcare quality within a community pharmacy, personnel would often claim to be overwhelmed with their workload, limiting the amount of patient care they were able to provide. 15 Pharmacists have limited time to provide patients with counsel or be required to have another member of staff relay information on their behalf. 15 Medication errors can cause significant stress for pharmacists and also increase workload. 16 Through the focus groups, pharmacists revealed that the reporting of small errors (such as a misspelled name) takes extensive time to complete. To proactively prevent errors, there is a need for clear and standardized communication on system protocols, prevention practices, and support for pharmacists. The communication practices currently in place cause barriers to supporting a learning culture. Email seems to be a low success method. 17 There seems to be little time to read routine emails. Through effective communication with pharmacists and a change in beliefs about error reporting practices, community pharmacies can strive to create an environment where mistakes are viewed as opportunities for learning, resulting in improved patient safety. Creating a standardized reporting system, where pharmacists only report errors to one system, could alleviate unnecessary workload and provide pharmacists more time to meet the needs of their patients.
Standardized reporting system
The lack of a standardized error reporting system is not only inefficient but also directly impacts the quality of information being recorded. 18 Many pharmacists seem to desire a one-stop reporting structure integrating the company and the regulatory systems. One central database should be used for all medication incidents and near misses to allow the sharing of information between pharmacies. 19 The willingness to accept that mistakes happen and that the potential exists for a dispensing error to result in death reinforces a willingness to participate in a QRE prevention program. The participants shared that the current reporting structure takes significant time to complete resulting in filling in required fields and skipping the detailed text descriptions of the incident. Pharmacists understand that mistakes occur, but pharmacists must be given tools to learn from one another to prevent the recurrence of mistakes.
The problem of reporting simply because you have to is exacerbated by the large presence of corporate pharmacies in the market. Corporate pharmacies require extensive reporting when a QRE may have resulted in harm but none for sig errors. The result is a duplication of reporting when something important happens as well as signposting of the time-waste that sig error reporting is perceived to be. Many pharmacists seem to desire a one-stop reporting system integrating the company system and the regulatory system. This would also help to alleviate the workload issue identified by participants as a contributor to error occurrences.
The facilitation of a Just Culture steeped in learning and sharing across open communication channels and standardized reporting systems creates an opportunity for a patient-first approach to pharmacy.
Limitations
This study has some limitations. First, the study may include a biased set of participants as pharmacists, pharmacy technicians, and pharmacy assistants with well-established interests and knowledge may have been more eager to participate. Therefore, a selection bias cannot be excluded. Nevertheless, participants mentioned several limitations that they encountered in QRE reporting, which was the aim of the study. Second, the sample size of this study included 17 participants through five focus groups; however, saturation was reached quickly.
The concerns regarding community pharmacies’ stressful working conditions and workload increase resulting in personnel burnout, demonstrate a need to better understand how current working conditions affect patient safety. 20 Data generated from CQI programs can assist in that process.
Conclusion
The community pharmacy is embedded in the healthcare industry that places emphasis on production, efficiency, and cost control. Registered pharmacists and their staff are not immune to the realities of business and the pressures of demanding workloads and public scrutiny The Nova Scotia QRE system was founded on an ideal of no-fault reporting to enable analysis and error reduction/quality improvement. Like many such systems, blame culture and under-reporting have crept in and derailed the values as espoused. A focus on a just culture provides a foundation for employees to learn from mistakes and focus on the transparency of patient safety culture in community pharmacy, putting the patient first
Footnotes
Acknowledgements
In Memoriam of Dr James R. Barker. Scholar, mentor, and leader Dr James (Jim) R. Barker passed away at his home in Halifax, Nova Scotia, on 2nd December 2023. He was a graduate student at Purdue University and the University of Colorado Boulder and used his innate curiosity to advance knowledge in his professoriate positions at Marquette University, the University of New Mexico, the United States Air Force Academy, the University of Waikato, and Dalhousie University.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This work was approved by the Health Sciences Research Ethics Board on 16 March 2018 (REB#2018-443).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nova Scotia Health Research Foundation (grant # PSO-EST-2014-9593).
