Abstract
Background
Adverse drug events (ADEs) are defined as any injuries due to medication use. We hypothesized that the incidences of ADEs and medication errors (MEs) could be associated with linguistic skills of pediatric patients.
Methods
We analyzed data from the Japan Adverse Drug Events study on pediatric inpatients. This study included inpatients aged one months and older and less than seven years old. We compared the primary outcome of ADEs and MEs between patients aged under three years and three years and older as children typically do not acquire sufficient linguistic skills until around three years of age.
Results
This study included 639 patients; 412 (64%) patients aged under three years and 227 (36%) patients aged three years and older. We identified 241 ADEs in 639 patients; 152 ADEs among patients aged under three years (37 ADEs per 100 patients) and 89 ADEs among those aged three years and older (39 ADEs per 100 patients). ADEs among patients aged under three years were less likely to be found (49 ADEs) during their hospital stay than those aged three years and older (20 ADEs) (P = 0.02). Among 172 MEs identified in 639 patients, 25 MEs (15%) resulted in ADEs; 23 (92%) occurred to those aged under three years and two (8%) occurred to those aged three years and older (P = 0.0008).
Conclusion
ADEs were less likely to be found and MEs resulted in ADEs more frequently in patients under three years old, and these differences could be explained by differences in their linguistic skill levels.
Introduction
Adverse drug events (ADEs) are defined as any injuries due to medication use, 1 and regardless of continuous international efforts over the past 60 years to prevent such events, 2 they still constitute a major public health issue. In 2017, the World Health Organization announced the Third Global Patient Safety Challenge as “Medication Without Harm”. 2 To achieve this global challenge, the effective strategy based on the detailed epidemiology of ADEs in various settings in various countries is essential.
Previous reports have shown that pediatric patients are especially vulnerable to ADEs and medication errors (MEs) because of their limited reserves of body fluid volume, differences in how they metabolize drugs as compared to adults, and the need to calculate drug dosage depending on body weight. 3 Another important factor that makes pediatric patients more vulnerable to ADEs and MEs may be that young children lack the linguistic skills to fully express themselves. We hypothesized that children's linguistic expression abilities could be related to the occurrence of ADEs and MEs, and sought to determine the age difference of ADEs and MEs. At three years of age, children generally begin to develop their linguistic skills, and by four years of age, they have acquired adult-like skills. 4 We compared the occurrence of ADEs and MEs in children aged under three years with those aged three years and older in our cohort of Japanese pediatric inpatients taken from the Japan Adverse Drug Events (JADE) study.5–8
Methods
Study design and patient population
The data for this sub-study were extracted from the JADE study of pediatric inpatients. 5 Briefly, the JADE study for pediatric inpatients included patients aged 15 years of age and below who were admitted to any wards including neonatal intensive care units (NICUs), pediatric ICUs, ICUs, and those aged over 15 years who were admitted to pediatric wards at two teaching hospitals in Japan during a three-month study period in 2009. This sub-study focused on children aged between one month old or more and less than seven years old. Children over seven years old were excluded, as they have established semantic processing, and their word processing mechanisms are qualitatively similar to adults.9,10 The study population was divided into two groups: the under three-year-old patients and the three-year-old and older patients.
The institutional review boards of the two participating hospitals approved the study. Because all data were obtained as part of routine daily practice, informed consent was waived by the institutional review boards.
Main outcome and definitions
The primary outcome of the study was the ADEs and MEs among patients, which we compared between patients aged under three years and three years and older.
In the JADE study of pediatric patients, an ADE was defined as any injury due to a medication use, and a ME was defined as any inappropriate use of a medication in the ordering, dispensing, administering or monitoring stages. Some ADEs were associated with MEs and were considered preventable ADEs. Some MEs did not result in ADEs but had the potential for harm, which were considered potential ADEs. If a potential ADE was intercepted before reaching the patient, it was considered an intercepted potential ADE. 1
Data collection and review process
The JADE study used a validated methodology reported in previous studies1,11 and established for pediatric patients. 5 Briefly, a board-certified pediatrician trained all research reviewers who comprised a board-certified pediatrician, pediatric nurses, and a dietitian. These trained reviewers screened all medical charts, laboratory results, incident reports, and prescription queries by pharmacists at the participating hospitals and collected potential events as well as the characteristics and administrative data of all patients in the cohort. Then, independent physician reviewers assessed the data and identified potential events as ADEs and MEs, as well as the details of events such as severity, stage at which the error occurred, and their preventability. If the reviewers reached different conclusions, they reached a consensus through a discussion. Interrater reliability between reviewers using κ statistics was calculated and was in the acceptable range (κ = 0.32–0.98). 1
Statistical analyses
Descriptive statistics are shown as medians with interquartile ranges for continuous variables and as numbers and percentages for categorical variables. We used a Wilcoxon rank sum test when the data were continuous and a chi-square test or Fisher's exact test when the data were categorical to compare patient characteristics and the details of ADEs and MEs between patients aged under three years and patients aged three years and older. We then compared patients with and without mental retardation as a stratified analysis for the confounding bias concerned with mental retardation. All reported P values were two-tailed, and P values less than 0.05 were considered statistically significant. We carried out all analyses using Stata 16.1 software (Stata Corporation, College Station, Tex., USA).
The institutional review boards of the two participating hospitals approved the study. Because all data were obtained as part of routine daily practice, informed consent was waived by the institutional review board.
Results
The present study used data from 639 of the 1189 patients in the JADE study (Figure 1). The median age was two years old (interquartile range (IQR) 0–4), 366 (57%) were male, and 512 (80%) were hospitalized in pediatric wards (Table 1). Among the 639 patients, there were 412 (64%) under three-year-olds and 227 (36%) three-year-olds and older. The patients aged under three years included fewer male patients (220; 53% vs. 146; 64%, P = 0.008), were more likely to have been admitted to PICUs (12; 2.9% vs. 1; 0.4%) and ICUs (18; 4.4% vs. 6; 2.6%) (P = 0.009), and had more major malformations (100; 24.3% vs. 37; 16.3%, P = 0.04). Patients aged three years and older were more often treated for cancer than those under three years (7; 1.7% vs. 10; 4.4%, P = 0.04) (Table 1).
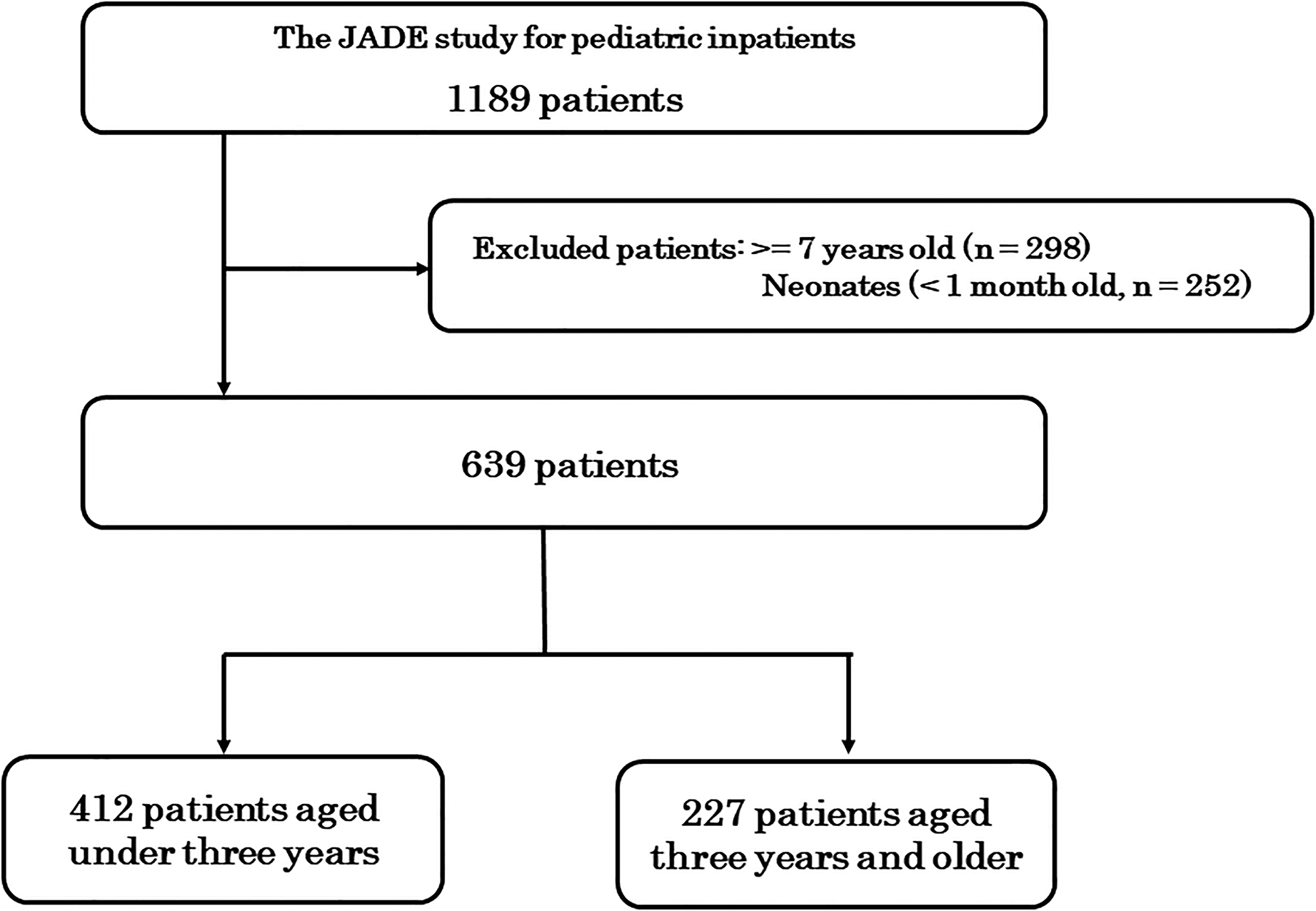
Study flowchart.
Patients' characteristics.
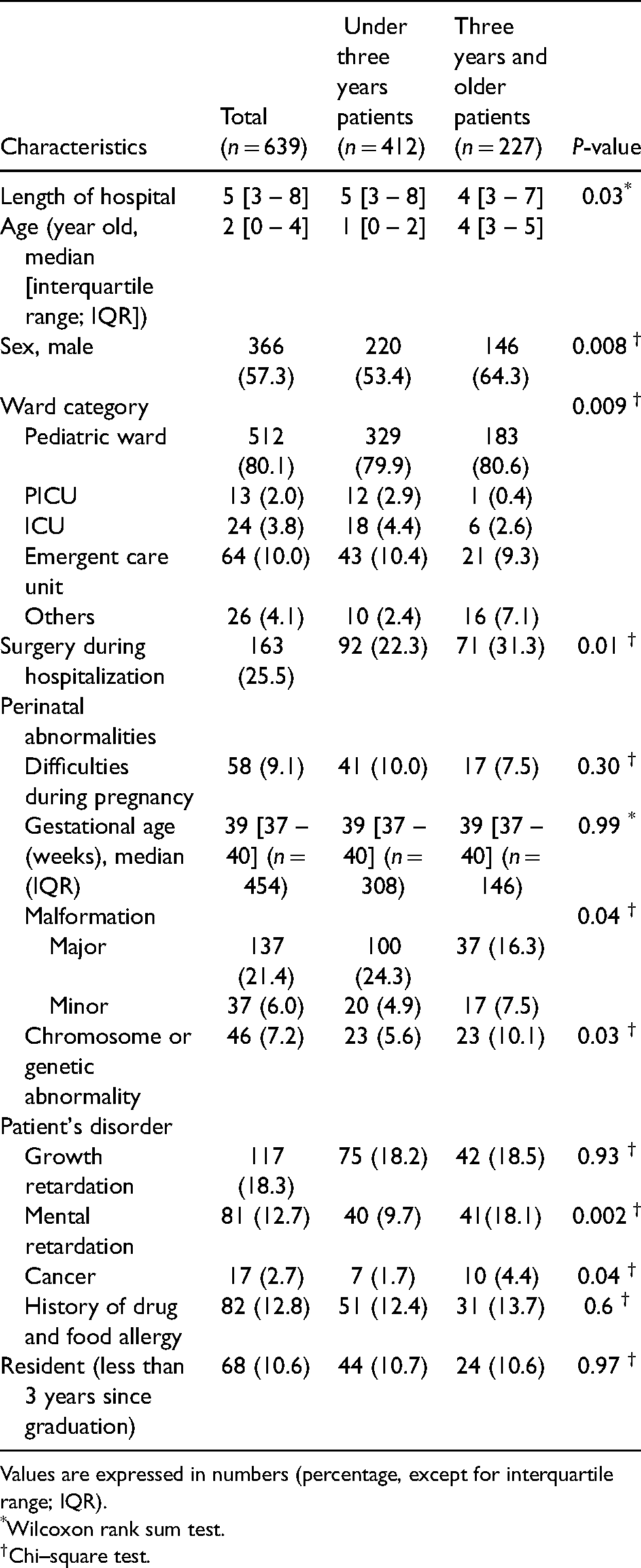
Values are expressed in numbers (percentage, except for interquartile range; IQR).
*Wilcoxon rank sum test.
†Chi–square test.
There were 81 (13%) patients with mental retardation. Among them, there were 40 (9.7%) aged under three years and 41(18%) (P = 0.002) aged three years and older (Table 1).
Adverse drug events
We found 241 ADEs in 639 patients in the data: 152 ADEs in the under three-year-old patients (37 ADEs per 100 patients) and 89 ADEs in the three-year-old and older patients (39 ADEs per 100 patients). Among these 241 ADEs, 172 ADEs (71%) were detected by physicians, nurses or pharmacists in clinical practice although 69 ADEs (29%) were not detected in clinical practice and were identified in this study (Table 2). Among the under three-year-old patients, pharmacists were more likely to identify ADEs, and physicians were more likely to identify ADEs among the three-year-old and older patients (P = 0.02) (Table 2).
Healthcare professionals who detected the adverse drug events on clinical practice.
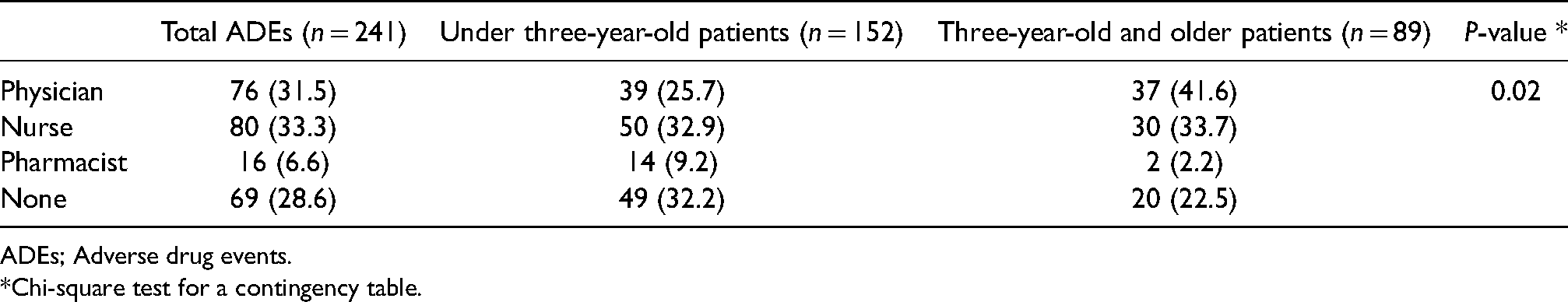
ADEs; Adverse drug events.
*Chi-square test for a contingency table.
In the stratified analysis, we found a total of 222 ADEs among 558 patients without mental retardation; 148 ADEs in 372 patients aged under three years (40 ADEs per 100 patients) and 74 ADEs in 186 patients aged three years and older (40 ADEs per 100 patients). Nineteen ADEs occurred among the patients with mental retardation; four ADEs in 40 patients under three years (10 ADEs per 100 patients) and 15 ADEs in 41 patients aged three years and older (37 ADEs per 100 patients).
Medication errors and preventable adverse drug events
We found 172 MEs in 639 patients in this study; 97 MEs in the patients aged under three years (24 MEs per 100 patients) and 75 MEs in the patients aged three years and older (33 MEs per 100 patients). Among 172 MEs, 25 (15%) resulted in ADEs, thus, these 25 MEs were considered as preventable ADEs. Among these 25 preventable ADEs, 23 (92%) occurred among the under three-year-old patients; 24% (23/97) of MEs resulted in preventable ADEs in the patients aged under three years, while only 2.7% (2/75) of MEs resulted in preventable ADEs among the patients aged three years and older. Among the remaining 147 MEs, 10 (5.8%) were not considered at risk for ADEs, although 137 MEs (79.7%) were considered as potential for ADEs and classified as potential ADEs (Table 3).
Error classifications.
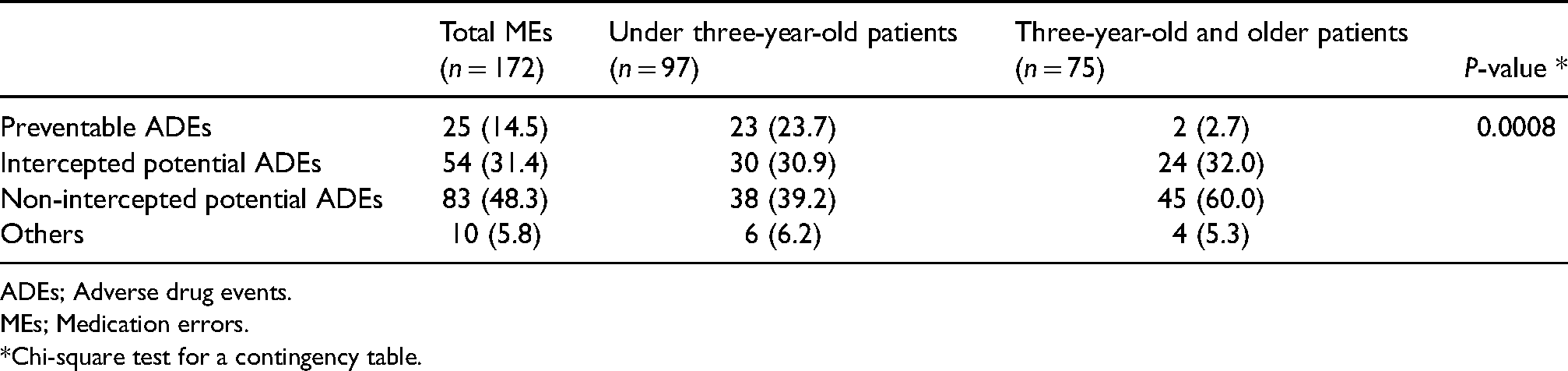
ADEs; Adverse drug events.
MEs; Medication errors.
*Chi-square test for a contingency table.
The error stage was significantly different between the patients aged under three years and those aged three years and older (P = 0.002) (Figure 2). More MEs occurred at the monitoring stage in the patients aged under three years (23 MEs; 24% in the under three-year-old patients vs. three MEs; 4% in the three-year-old and older patients), while more MEs occurred at the ordering stage in the three-year-old and older patients (69 MEs; 71% in the under three-year-old patients vs. 67 MEs; 89% in the three-year-old and older patients).
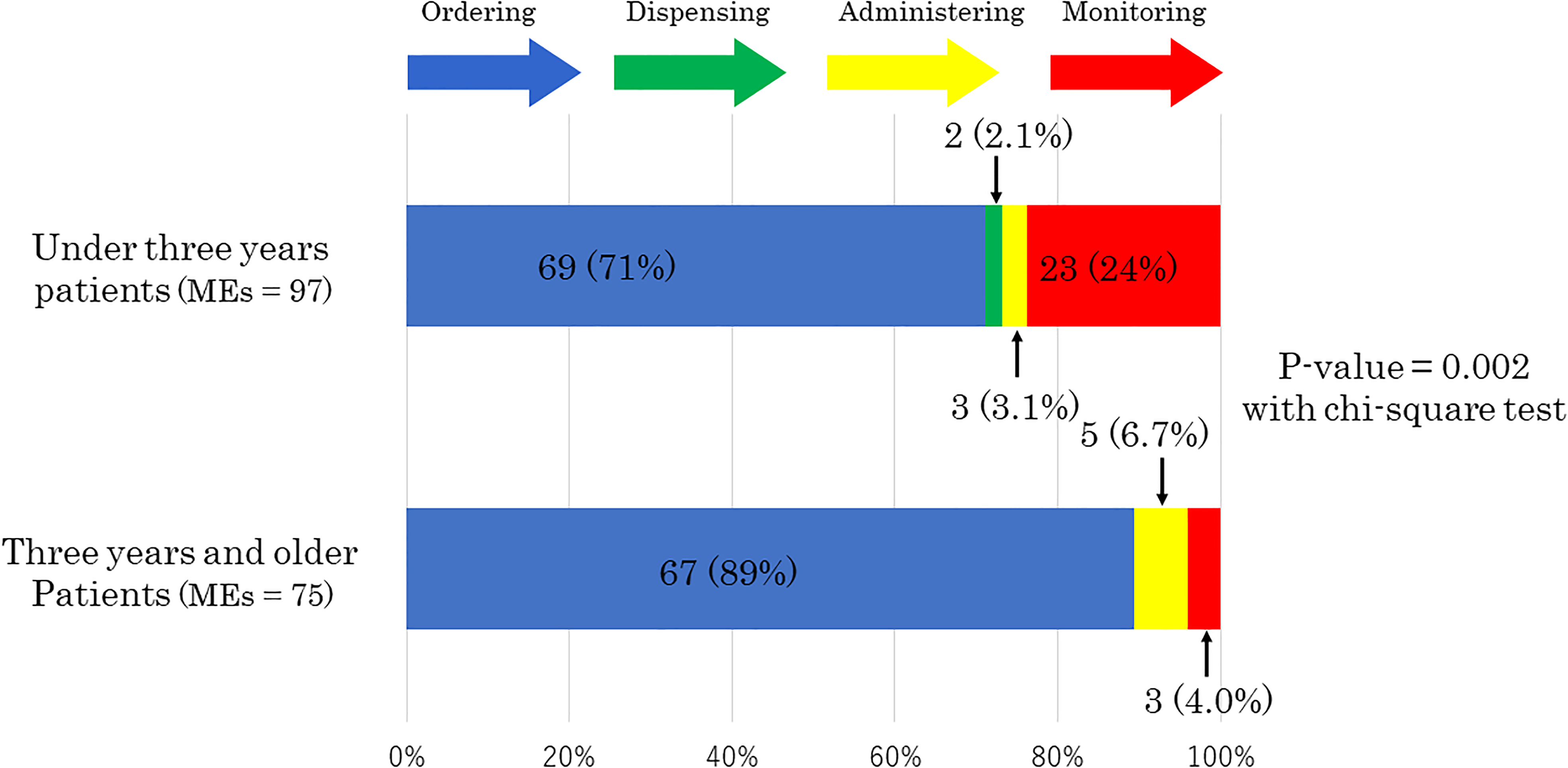
Stages of medication errors.
There were 21 MEs (22%) among the total 97 MEs that occurred when the doctors in charge were resident physicians for the under three-year-old patients, while only six MEs (8.0%) occurred when resident physicians were in charge of the three-year-old and older patients (P = 0.02).
In the stratified analysis, we found a total of 143 MEs and 25 preventable ADEs among 558 patients without mental retardation; 79 MEs in 372 patients aged under three years (21 MEs per 100 patients) and 64 MEs in 186 patients aged three years and older (34 MEs per 100 patients), while 23 preventable ADEs in 372 patients aged under three years (6.2 preventable ADEs per 100 patients) and two preventable ADEs in 186 patients aged three years and older (1.1 preventable ADEs per 100 patients). Twenty-nine MEs occurred among the patients with mental retardation and none of the patients with mental retardation experienced preventable ADEs; 18 MEs in 40 patients aged under three years (45 MEs per 100 patients) and 11 MEs in 41 patients aged three years and older (27 MEs per 100 patients)
Discussion
We found that ADEs among the under three-year-old patients were less likely to be found during their hospital stays than those among the three-year-old and older patients. In particular, physicians less often recognized ADEs in the under three-year-old patients. We also found difference at the stage of medication use where MEs occurred; more MEs occurred at the monitoring stage in the under three-year-old patients, while most MEs occurred at the ordering stage in the three-year-old and older patients. While about 30% of MEs were intercepted, the rest of MEs were not intercepted overall and these non-intercepted MEs resulted in preventable ADEs more often in the under three-year-old patients than the three-year-old and older patients. The results of the stratified analysis showed that there were no patients with mental retardation who experienced preventable ADEs.
Our findings could be explained by the less developed level of linguistic expression in the under three-year-old patients. The MEs that occurred at the ordering stage were generally due to erroneous dose calculations based on body weight or inappropriate prescriptions. These MEs were likely to be intercepted by pharmacists and were not associated with the linguistic expression levels of patients. On the other hand, the MEs at monitoring stage were mostly due to delayed or missed identification of ADEs, and these MEs could be ameliorated or even prevented if patients were able to express their complaints clearly. Thus, the MEs were more likely to result in preventable ADEs in the under three-year-old patients. Previous reports showed that linguistic barriers among pediatric inpatients whose mother language was not English and stayed at the hospital of English speaking environment significantly increased risk for ADEs and longer lengths of hospital stay.12,13 Other research show that three and four-year-old children have adult-like grammatical expressions.4,14 These findings support our findings though this study is the first that analyzed the characteristic differences for ADEs and MEs by age and association with linguistic expression levels.
To improve patient safety in daily clinical practice, healthcare professionals need to be aware of these findings; however, the observation of pediatric inpatients necessitates effort and resources. Novel monitoring technologies could prove to be useful. For instance, a wrist band with accelerometry for under three-year-old patients could be used to detect small changes in vital signs and alert healthcare workers to potential ADEs. 15 Another effective strategy could be intensive monitoring by pharmacists and implementation of clinical decision support systems (CDSS). If a pharmacist recognizes that a child under three years old is at a higher risk for preventable ADEs, they can provide counselling for patients and their families to prevent or ameliorate preventable ADEs. 16 CDSSs support physicians by suggesting appropriate prescriptions and monitoring based on known risks for various types of patients. 17 Healthcare professionals could focus on the under three-year-old patients with monitoring stage that prevent or ameliorate preventable ADEs in this high-risk group as a high risk strategy. 18
This study did not find any occurrences of preventable ADEs in patients with mental retardation. One of the possible interpretations could be that healthcare professionals should monitor such patients intensively because, based on evidence and their own experiences, they know that patients with mental retardation often lack the ability to recognize health problems and some environmental and individual barriers prevent them from receiving necessary care. 19 Thus, this finding also supports our belief that healthcare professionals need to be aware of who is at a high risk for preventable ADEs to improve patient safety.
A limitation of the study is that some ADEs and MEs may have not been detected because they were not noted in the medical charts; however, our approach used the standard method to detect ADEs and MEs because more robust measurements have not yet been developed. We did not detect effect modifiers such as nursing status or guardians’ attendance that might influence ADEs and MEs. Finally, this study used data from JADE study which was conducted in 2009; however, the clinical situation and practice such as monitoring procedure or medical device at that time would not be very different from the current standard.
Conclusions
The present study clarified that the occurrence of ADEs was less likely to be found and MEs resulting in preventable ADEs were more likely in under three-year-old patients, and that this may be related to their less developed levels of linguistic expression.
Footnotes
Acknowledgements
The JADE study for pediatric inpatients was conducted by the following investigators: Dr David W. Bates, Dr Tsukasa Nakamura, Dr Kiyoshi Kikuchi, Ms. Eriko Waki, Ms. Sanae Ohashi, Ms. Masako Shibuya, Ms. Kayoko Hiroi, Mr Takashi Kawakubo, Ms. Kaori Yamamoto, Ms. Makiko Ohtorii, Ms. Ai Mizutani, Ms. Mika Sakai, Ms. Yuko Ueo, Ms. Yuka Iwamoto, Ms. Hitomi Ito, Ms. Chikako Senou, Ms. Megumi Hara, Mr Tomohiro Sonoyama, and Mr Taro Saito.
Authors’ contributions
All authors declare that they have contributed to this article. JT acquired the funding of this study, performed the analysis, interpreted the data, and wrote the draft manuscript. SM designed the study, acquired the funding of this study, performed whole project administration, implemented the survey, interpreted the data, and performed critical revisions. YO implemented the survey and performed critical revisions. HI performed critical revisions and supervised the whole study process. TM designed the study, acquired the funding of this study, interpreted the data, performed critical revisions, and supervised the whole study process. All authors read and approved this version of the manuscript version for submission.
Data availability statement
The data underlying this article will be shared on reasonable request to the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by JSPS KAKENHI [Grant Numbers JP18H03032 to T.M., JP22790494 to M.S., JP24689027 to M.S., JP15K08862 to M.S., JP20K10364 to M.S.] and The Japan Foundation for Pediatric Research [Grant Number No19-010 to J.T.].
