Abstract
Objective
Hospital-acquired pressure injuries harm over 2.5 million patients at a U.S. cost of $26.8 billion. Sub-epidermal moisture scanning technology supports clinicians to anatomically identify locations at-risk of developing hospital-acquired pressure injuries. Our objective was to evaluate the cost-effectiveness of adopting sub-epidermal moisture scanners in comparison to existing hospital-acquired pressure injury prevention guidelines structured around subjective risk assessments.
Methods
A Markov cohort model was developed to analyze the cost-effectiveness of sub-epidermal moisture scanners in comparison to existing prevention guidelines, based on current clinical trial data from the U.S. health care sector perspective in the acute, acute rehabilitation and skilled nursing facility settings. A hypothetical cohort was simulated over a time horizon of one year. An incremental cost-effectiveness ratio was measured using U.S. dollars per quality-adjusted life year at a willingness-to-pay threshold of $100,000/quality-adjusted life year, and uncertainty was tested using probabilistic sensitivity analysis.
Results
Integration of sub-epidermal moisture scanners yielded cost-savings of $4054 and 0.35 quality-adjusted life years gained per acute care admission, suggesting that sub-epidermal moisture scanners are a dominant strategy compared to standard care and producing a net monetary benefit of $39,335. For every 1000 admissions in high-risk acute care, sub-epidermal moisture scanners could avert around seven hospital-acquired pressure injury-related deaths and decrease hospital-acquired pressure injury-related re-hospitalization by approximately 206 bed-days.
Conclusions
Acute care, acute rehabilitation and skilled nursing settings that adopt sub-epidermal moisture technology could achieve a return on investment in less than one year. Providers may want to consider these types of technology that aid clinical judgment with objective measures of risk in quality improvement bundles.
Introduction
Hospital-acquired pressure injuries (HAPIs) are costly to health systems and harmful to patients. In fact, these conditions harm over 2.5 million U.S. patients at a national cost of $26.8 billion. 1 Many patients face risk for these sometimes fatal conditions that are mostly preventable in many parts of the health system, ranging from hospitals, to rehabilitation, to long-term care. 2 In the last decade, U.S. Medicare has established reduced reimbursements and penalties to standardize the use of guidelines for pressure injury prevention.3,4 Perhaps, these injuries still occur in large numbers throughout health systems through inconsistent guideline initiation and iatrogenic error.
Pressure injuries are areas of localized damage to the skin and underlying soft tissue caused by pressure or in combination with shear, typically over a bony prominence, and occurring with prolonged exposure against an external surface or medical device. 5 The most common areas of tissue damage are over the patient’s sacrum and heels. Development of these injuries primarily depends upon the intensity and duration of the pressure, and the tolerance of the tissue to that pressure. The latter is determined by factors such as intrinsic integrity of the skin, and degree to which the skin is exposed to moisture, friction, and shear. 6 Damage from pressure injuries can develop in less than 24 h, but can take as long as five days to appear visually. 6 By the time the injury is detected, the damage to the underlying tissue is often more extensive than it appears. 6 Thus, the success of the risk mitigation strategies for pressure injuries greatly depends upon the timely detection of progressive tissue damage in patients’ anatomies.
The National Pressure Injury Advisory Panel (NPIAP) has published a series of guideline recommendations for pressure injury prevention between 2004 and 2019. 7 All guideline recommendations have encouraged bedside clinicians to use a standardized risk-assessment tool such as the Braden Scale in order to implement preventive strategies (e.g. repositioning, moisture/incontinence management, support surfaces, and nutritional supplements) to the highest risk patients. However, these risk assessment instruments do not have well-established inter-rater reliability, and there is a high degree of variability in the interpretation of how to score the risk of individual patients. 8 To compound this variability, risk-assessment tools ask clinicians to score patient risk for pressure injury on six subjective components that have not been well defined: sensory perception; moisture; activity; mobility; nutrition; and friction and shear. 9
The field of bedside care for patients in any facility, whether acute, rehabilitative, or long-term care, could benefit from technologies that objectify the measures of risk for pressure injury among patients in a timely manner. Such tools that remove subjective analysis of patient risk could improve risk-stratification so that clinicians spend the most time implementing preventive measures for pressure injuries and other iatrogenic injuries among patients who actually need additional care. Scanning diagnostic technology has been developed for bedside care that actually interprets moisture levels on the skin and underlying tissue of the patient, resulting in a more objective clinical assessment of “sub-epidermal moisture” (SEM) among pressure injury risk factors. 10 Such technology designed to scan the patient’s skin (“SEM scanner”), may not only provide a clinically effective analysis of patients’ localized inflammation-based risk for pressure injury development in alignment with current clinical trial data, but could be could also be cost-effective. This technology combined with clinical judgment and traditional risk assessment tools such as the Braden Scale would further enhance the accuracy of risk management for decubitus patients.
Previous studies analyzing the value of pressure injury prevention protocols have found them to be cost-effective across many health care setting types even using subjective measures of risk.11–14 We hypothesize that the SEM scanners are cost-effective technologies since it reduces variability in the subjective risk-assessment of high-cost pressure injuries that can harm patients. We analyzed the cost-effectiveness of SEM scanners in comparison to existing prevention guidelines, based on current clinical trial data at a willingness-to-pay threshold of $100,000 per quality-adjusted life year (QALY) from the U.S. health care sector perspective.
Methods
Design of the study
This study implemented a Markov model to analyze the cost-effectiveness of the SEM scanner as part of a complete prevention protocol compared to the current standard of care in preventing HAPIs on the sacrum and heels among a simulated patient cohort throughout several types of facilities (Figure 1). Model parameters on pressure injury outcomes related to SEM technology were extracted from published data about subepidermal moisture biocapacitance measurement device efficacy studied in a blinded clinical trial, a validated instrument to detect tissue damage on the sacrum and heels (“The SEM Scanner”, BBI LLC, Los Angeles, CA).6,15 Our model was adapted to analyze patients in acute care hospitals (ACH) and inpatient rehabilitation facilities (IRF), as well as residents of skilled nursing facilities (SNF). These models adjusted for variations in HAPI treatment duration and patterns, as well as general patient characteristics.
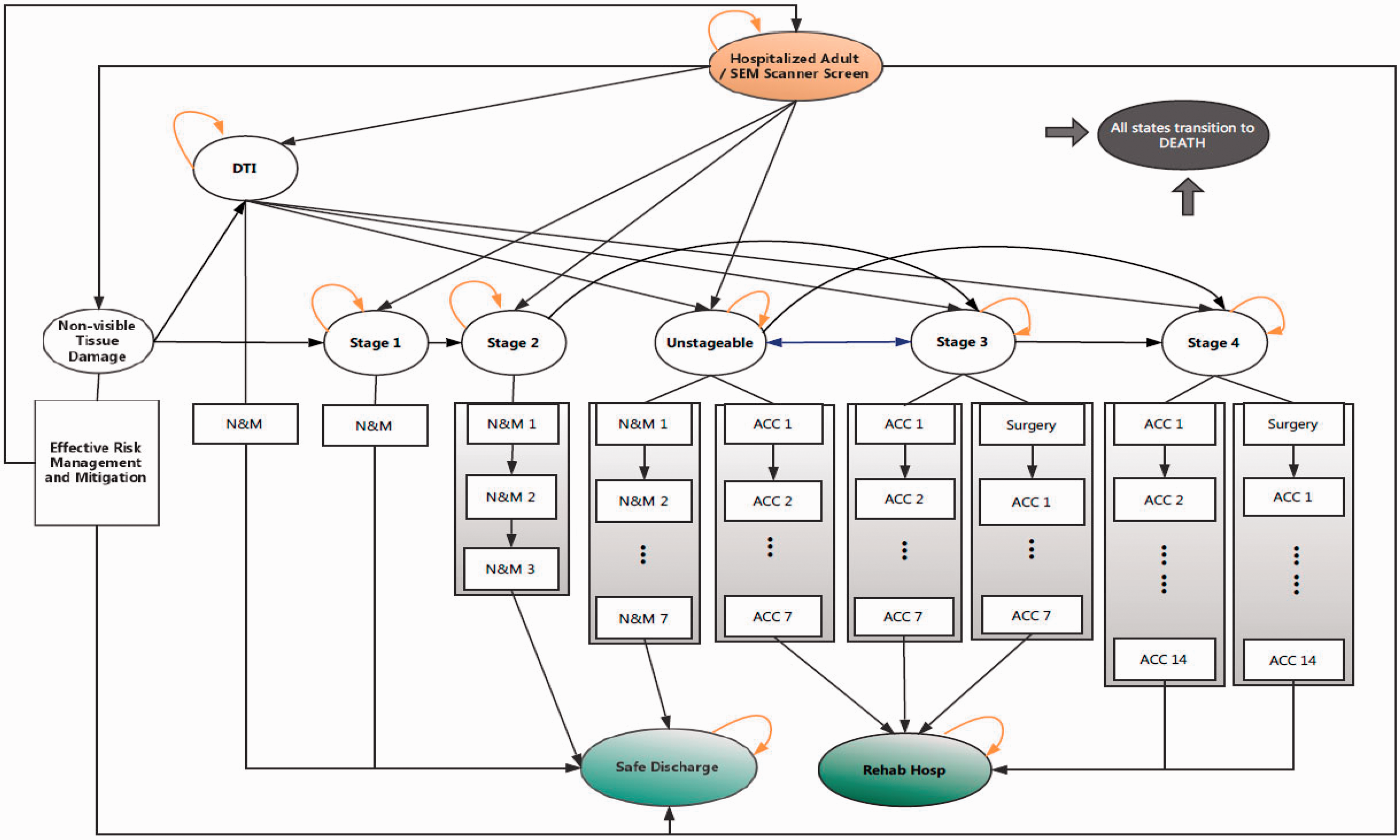
A Markov model of hospital-acquired pressure injury outcomes in hospital care with and without the use of a sub-epidermal moisture scanner to detect early tissue deformation. Stages correspond to the standardized scale of pressure injury severity. Patients who undergo care for pressure injuries either receive nursing and monitoring (N&M) or acute and chronic care (ACC) in addition to surgery; numbering after N&M and ACC corresponds to the consecutive day(s) or additional care in a specific state prior to transition to other possible health states. All health states have Death as a potential outcome. The Markov model extends to 365 days in total.
In the ACH setting, the time horizon of the model was 365 days with one-day cycles. A rate of 3% was chosen for discounting when applicable. All costs were corrected for inflation to represent 2018 U.S. Dollars, and effectiveness was measured in units of QALYs in order to produce an incremental cost-effectiveness ratio (ICER) and net monetary benefit. All analyses were performed using Microsoft Excel® (Microsoft Corp., Seattle, WA, ©2016).
Model structure
The model consisted of 11 mutually exclusive health states (Table 1). The model comparator with the SEM scanner had an additional health state, “Non-visible Tissue Damage”, in which scanner-identified patients with potential HAPI development received in-time mitigation efforts to decrease the likelihood of further disease progression. Risk mitigation was based on moisture levels at an anatomical site of greater than or equal to an SEM delta equal to or greater than 0.6 (i.e. clinically meaningful differences between the highest and lowest absolute SEM values at a scanned anatomy, which indicates that the anatomy is at risk of developing a PI and that pressure injury risk mitigation should be provided to prevent further advancement to any stage of prevention injuries). 6
Model probabilities.
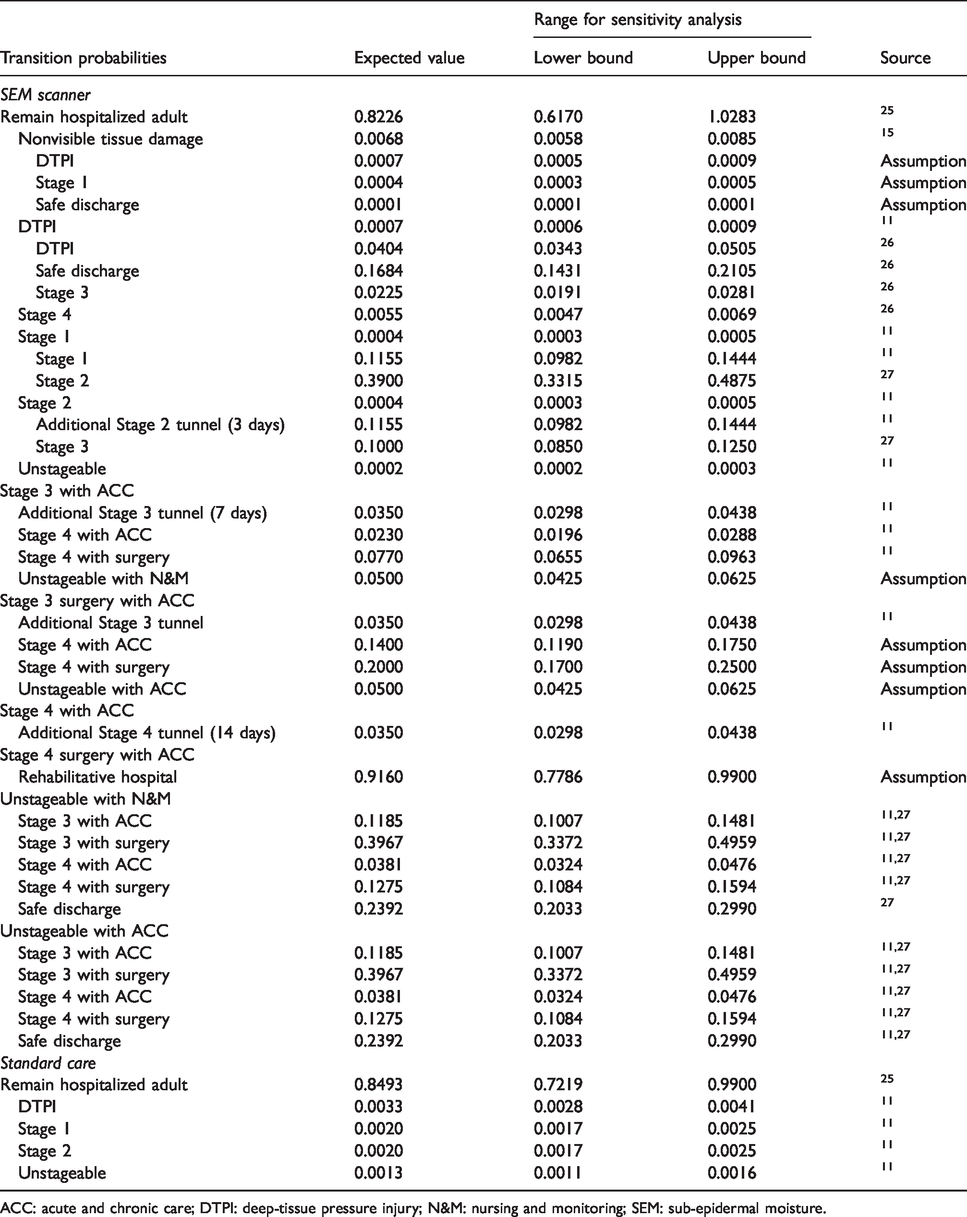
ACC: acute and chronic care; DTPI: deep-tissue pressure injury; N&M: nursing and monitoring; SEM: sub-epidermal moisture.
Pressure injury stages 2, 3, 4, and unstageable were each modeled as “tunneling states”. That is, patients remained in these stages continuously for a set number of cycles (days) based on common treatment durations before they transitioned to other states. “Pre-stage 1” and Stage 1 pressure injuries, as well as deep tissue pressure injuries (DTPIs) were also modeled as daily transitional occurrences. Pre-stage 1 refers to nonvisible tissue deformation either on the surface or in soft tissue underneath the skin, by which moisture levels can still increase.
Standard of care pathway
Admitted patients started from the hospital adult state with a standard prevention protocol provided. Hospitalized adults transitioned to any stage of pressure injury during the hospital stay. Those who did not develop HAPIs during the stay transitioned to safe discharge. At any time, these patients could have transitioned to all-cause death.
SEM scanner protocol pathway
In the intervention arm, admitted patients started at hospital adult with the SEM Scanner estimated as part of an effective prevention protocol. When an SEM scanner was used, patients could move to a nonvisible tissue damage state if an SEM scanner detected tissue deformation. If nonvisible tissue damage was not detected after screening, patients remained in the hospitalized adult state until they developed any state of pressure injuries. If no pressure injuries developed during a patient’s admission, they transitioned from the hospitalized adult state to safe discharge.
Model probabilities related to SEM scanners were obtained from clinical trial data (Registered Clinical Trial No. NCT02701101). 15 For parameters related to standard care or where data were not otherwise available, we abstracted data from peer-reviewed literature to inform model parameters.
Model state costs were obtained from prior research (Table 2). Standard care prevention costs and previous studies mapped out stage 1 or 2 HAPI costs using a micro-costing approach. Other state costs were estimated from gross-costing methods.1,12 For the hospitalized adult state, only material and labor costs related to HAPI prevention were included since daily inpatient hospitalization costs are not attributable to HAPI. We assumed that the cost of treating DTPIs was the same as HAPI prevention. 12
Model costs and health utilities.
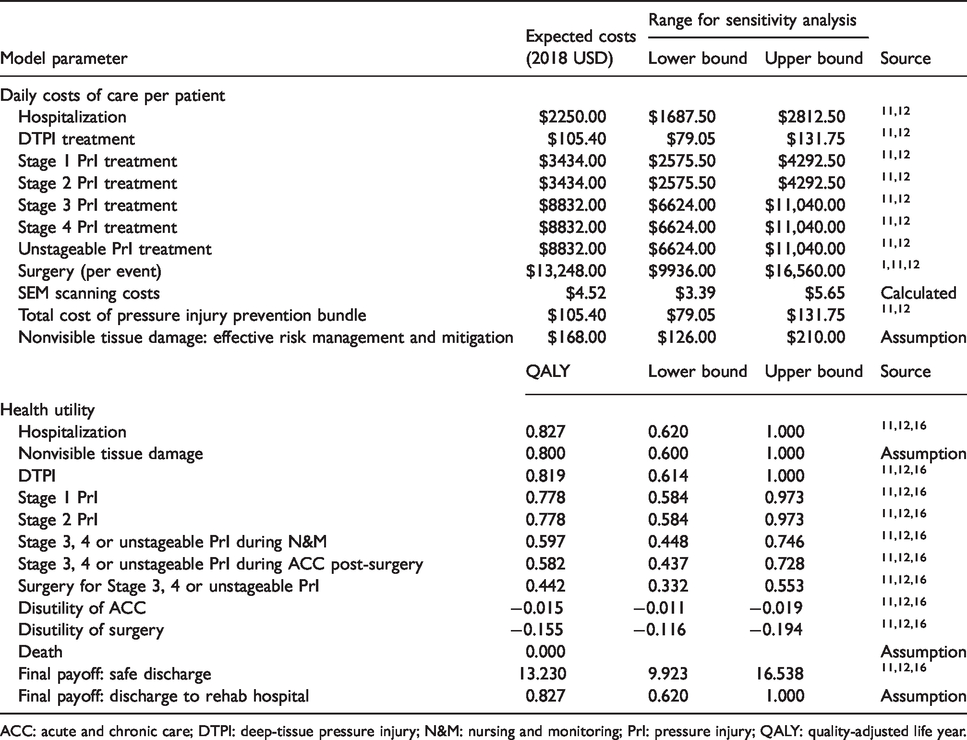
ACC: acute and chronic care; DTPI: deep-tissue pressure injury; N&M: nursing and monitoring; PrI: pressure injury; QALY: quality-adjusted life year.
The utilities used in the model were measured in units of QALYs based on EQ-5D index scores published in previous research with 0.0 QALYs representing death and 1.0 QALYs representing full health. 16 Patients diagnosed with a HAPI receiving acute and chronic care or surgery had adjusted disutilities to reflect less desirable health states. Final utility payoffs were awarded to patients either safely discharged or transferred to rehabilitative facilities, which assumed the addition of QALYs for remaining life expectancy after care at a facility.
Sensitivity analyses
The univariate sensitivity analyses were conducted to test model uncertainty by varying the expected parameter values by ±25% and assessing the impact on the model results. A Bayesian multivariate probabilistic sensitivity analysis (PSA) was done using 10,000 Monte Carlo simulations with standard care as the baseline approach. The PSA applied a distribution for each variable to characterize the impact of uncertainty on all parameters simultaneously. Beta distributions were used for variable with values ranging between 0.0 and 1.0 (i.e. probabilities and utilities), and gamma distributions were used for positive values >1.0 (i.e. costs).
Key assumptions
We made multiple assumptions in order to complete this analysis. First, we assumed that the cost of DTPI was the same as maintaining a full HAPI prevention protocol, since there is no other evidence on cost of treatment for DTPI management other than population average estimates in the literature cited in this study. Second, this analysis is not done alongside the clinical trial, but rather a simulation of a “what-if” scenario for SEM scanner value if the scanner accurately triages patient risk with expected performance.
Results
SEM scanners as part of a prevention protocol are a dominant strategy compared to standard care since it lowers costs and increases QALYs. Over a period of one year, prevention protocols with an SEM scanner costs $912 and add 12.55 QALYs per admission, whereas standard care costs $4966 and yields 12.19 QALYs per admission (Table 3). For every 1000 admissions in high-risk acute care, an SEM scanner could potentially avert around seven HAPI-related deaths and decrease HAPI relate re-hospitalization by approximately 206 bed-days.
Expected results of the cost-effectiveness analysis comparing SEM scanning to standard care for pressure injury prevention in hospitals.
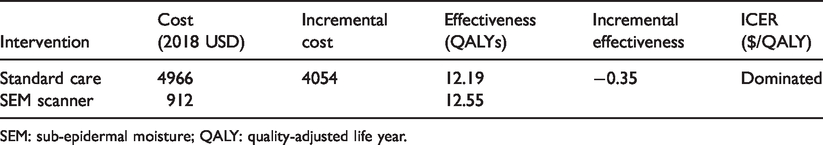
SEM: sub-epidermal moisture; QALY: quality-adjusted life year.
At a willingness-to-pay threshold of $100,000/QALY, the SEM scanner protocol generated a net monetary benefit of $39,335. Using an SEM scanner also resulted in comparably rapid return on investment (ROI) to achieve breakeven in only about one week of use. For every dollar invested in a prevention protocol with an SEM scanner, the annual return was $141.
Sensitivity analyses
We performed a univariate sensitivity analysis for each variable. The ±25% variation around the value of all cost, probability, and utility estimates did not change the results of the model from the main conclusions. However, the analyses identified three parameters with the most impact on the model results: (1) the probability of safe discharge; (2) the probability of safe discharge with non-visible tissue damage; and (3) the probability of “pre-stage 1” from hospitalized adult.
Probabilistic sensitivity analysis
The PSA suggested that the prevention protocol using an SEM scanner was cost-effective approach in all simulations with a mean cost of $913.94 (95% confidence interval [CI]: 900.60–927.28) and mean utility of 12.50 QALYs (CI: 12.30–12.69). Whereas, the standard care alternative resulted a mean cost of $4965.99 (CI: 4890.21–5041.76) and mean utility of 12.15 QALYs (CI: 11.96–12.34). The ICER remained negative on average, since there was a cost-savings of $11,577.29 per QALY gained (CI: $10,881.29 to $12,273.29 saved per QALY gained).
Discussion
According to the economic evaluation of SEM scanner technology relative to standard care, SEM scanners are cost-effective as a value-added component of a pressure injury prevention protocol. This is due to the fact that SEM scanning reduces variability in risk-assessment among high-risk patients, which leads to more targeted implementation of prevention guidelines that reduce the risk of harm. Health systems considering the adoption of new technologies as part of a quality improvement bundle to reduce pressure injury incidence should consider investing in technology that improves standardized risk assessments such as SEM scanners. While such technology may add up-front costs to pressure injury prevention protocols, these costs are offset by the long-term costs of treating a pressure injury, or for that matter, paying for related penalties or the impending malpractice lawsuits. As of 2015, CMS institute a policy that penalized facilities 1% of total reimbursements which fell into the bottom-quartile with respect to composite rates of HAPIs and 11 other hospital-acquired conditions. 3
The cost-effectiveness of SEM scanning technology is not specific to any one facility type, but perhaps holds value in most types of U.S. health system facilities. We analyzed the cost-effectiveness across acute, rehabilitative and SNF care, and results were similar. SEM scanners have implications for all health system facility types in terms of value-added. This is particularly important to integrated health systems such as accountable care organizations, which may contain different facility types within a single continuum. Thus, SEM scanners add value not just to individual facilities, but continuity in quality improvement efforts to improve HAPI prevention with a potential for compounded ROI in larger health systems. Systems that track patient risk using SEM scanning technologies can track risk of complicated patient cases between facilities less concern about the inter-rater reliability.
SEM scanner technology contributes to a larger concern expressed about the predictive validity of pressure injury risk assessment in a report by the American College of Physicians. 17 If evidence to support subjective risk-assessment with clinical judgment is poor, then technologies that standardize the practice of risk-assessment and score all patients equally are the best solution. This is especially the case among the elderly, morbidly obese, and patients of darker skin tones for which traditional methods of risk-stratification can be challenging. 18 Technologies such as SEM scanners should be developed in greater variety in order to improve all dimensions of risk that the field of hospital safety deems important (e.g. moisture, mobility, activity, friction and shear, etc.). 19
This study has several limitations. First, not all parameters are based on clinical trials of SEM scanner technology, but rather from a targeted literature review to obtain other model parameters. Second, QALYs in the economic evaluation are based solely on a U.S. representative survey of patients, including those with pressure injures, but do not reflect the direct effectiveness of SEM scanners on patient outcomes. Third, there is model uncertainty related to limited sampling of available trials used to illustrate SEM scanning efficacy; additional trial data from U.S. applications will reduce uncertainty. We performed sensitivity analyses to test model uncertainty.
The field could benefit from wide-scale research on the clinical effectiveness of SEM scanning in other facility types, either through clinical trial designs or observational research. Nonetheless, handheld, wireless medical device that assess patient-specific measures of risk such as moisture reduce variability in subjective risk-assessment. The field of patient safety has much to gain from widespread development of instruments such as an SEM scanner that reduce this variability in general. 20 Further steps can be taken to integrate such technologies with electronic health record (EHR) systems in facilities so that information on patient risk is documented efficiently and analyzed for patient risk using internal algorithms. The need for clinicians to transcribe risk scores by hand into an EHR adds additional, unnecessary potential for user-error that patients are better off without.
Why is the information about this study important to providers? Providers often monitor the changing health status of patients throughout an admission, which may include transfer between units. Given the limited availability of an SEM scanner throughout a facility, providers are best suited to note continual use of such risk-assessment tools between procedures or progression to different units. Providers are often involved directly in the innovation of quality improvement bundles for bedside care. Thus, providers are well-suited to consider the integration of an SEM scanner with clinical workflow, such as triggering targeted intervention of an SEM scanner for a specific type of patient.
Most providers are no longer limited to monitoring patient progress in a single setting such as acute care. Many providers today monitor patients who transition regularly between acute and long-term care for ongoing complications, such as in the Comprehensive Care Program. 21 Their ability to quarterback designated use of standardized risk assessment tools between transfers to different types of facilities can mitigate the overall risk to ensure that a patient readmission does not occur at an even greater cost than these models calculate. Again, the value-added of SEM scanning across this continuum may increase the financial bandwidth for medical divisions, which can in turn support investments in other quality improvement programs or allow provider to manage additional, complicated patient cases without the worry of a harmful pressure injury.
The final point that providers have to consider in employing an effective pressure injury prevention protocol is risk management. Patients who are incorrectly assessed to have a low degree of pressure injury risk using traditional methods of clinical judgment may miss high-risk patients who will ultimately develop pressure injuries. Technologies such as SEM scanners give providers objective measures of risk so that they can effectively manage highest-risk patients towards improved outcomes. Technology-directed care leads to opportunities for reduced waste by assisting providers direct health care delivery for patients of highest need, as opposed to all patients or at random.
In conclusion, evidence identified in a clinical systematic review supported the use of SEM measurement devices as effective tools for early pressure injury detection. 22 In this health economic analysis, SEM scanners are a cost-effective means of documenting pressure injury risk based on moisture levels on patients’ skin and underlying soft tissue. This technology circumvents the high cost of most pressure injuries in facilities and may achieve an ROI in less than one year. This ROI is important since most facility medical directors operate on 12-month fiscal budgets. 23 The opportunity cost of a pressure injury averted provides medical directors the financial bandwidth to invest in other areas of patient safety in the future so that overall, health care becomes more cost-efficient and patients are safer from harm. 24
Footnotes
Authors’ note
All the authors meet the requirements for authorship according to ICMJE standards.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BBI LLC (Bruin Biometrics) gave an unrestricted grant to the University of Southern California in support of Dr William Padula. BBI retained Monument Analytics as a paid consulting firm between 2018 and 2019.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
