Abstract
Background:
Cytomegalovirus retinitis (CMVR) is a vision-threatening retinal infection that primarily affects severely immunocompromised individuals.
Objectives:
This study aimed to compare the clinical presentations, long-term visual outcomes, and predictors of visual prognosis in CMVR patients across different immunodeficiency subtypes.
Design:
Retrospective cohort study.
Methods:
This study analyzed 117 eyes from 91 patients with CMVR who were treated at a tertiary eye center between 2009 and 2024. Eyes were stratified by the underlying cause of immunosuppression into a human immunodeficiency virus (HIV)-positive group (71 eyes of 58 patients) and an HIV-negative group (46 eyes of 33 patients). The HIV-negative group included patients with hematologic malignancies, drug-induced immunosuppression (e.g., posttransplant or autoimmune therapy), solid organ transplantation, autoimmune disease, congenital immunodeficiency, and poorly controlled diabetes mellitus type 2 (DM2). Patient demographics and comprehensive ophthalmologic examination findings were recorded and compared across these subgroups.
Results:
The study included 91 patients (117 eyes) with a mean age of 40.1 ± 14.7 years; 62.6% were male. Of these, 58 patients (71 eyes) were HIV-positive (mean age 39.7 ± 10.9 years; 65.5% male) and 33 patients (46 eyes) were HIV-negative. Among HIV-negative patients, 19 eyes were in patients with hematologic malignancies (mean age 35.3 ± 21.1 years; 75% male), 20 eyes were in patients with drug-induced immunosuppression (mean age 38.6 ± 17.9 years; 40% male), and the remainder eyes were associated with solid organ transplant, autoimmune disease, congenital immunodeficiency, or poorly controlled DM (7 eyes). Eyes in HIV-negative patients exhibited more severe vitritis than those in HIV-positive patients (34.8% vs 19.7% with vitritis grade +2; p = 0.009). Conversely, HIV-positive patients presented with worse baseline visual acuity (mean 1.85 ± 1.14 logMAR vs 1.31 ± 1.17 logMAR; p = 0.045). Comparing the two main HIV-negative subgroups, the drug-induced immunosuppression group showed a significantly higher incidence of retinal vascular sheathing (30% vs 0% of eyes; p = 0.009), whereas the hematologic malignancy group demonstrated more cotton-wool spots (21.1% vs 0% of eyes; p = 0.030). Despite these phenotypic differences, long-term visual outcomes were similar across all groups. On multivariable regression, baseline best-corrected visual acuity (BCVA) (β = 0.527, p < 0.001) and retinal detachment (β = 0.254, p = 0.017) were independent predictors of poorer final visual acuity.
Conclusion:
CMV retinitis demonstrates distinct clinical phenotypes in different immunosuppressed populations. HIV-negative patients tend to show more pronounced vitritis and vasculopathic retinal features, whereas HIV-positive patients have worse baseline visual acuity. However, the long-term visual prognosis was not significantly influenced by the underlying cause of immunosuppression.
Introduction
Cytomegalovirus retinitis (CMVR) is a severe, vision-threatening opportunistic infection that predominantly affects immunocompromised individuals, including patients with advanced human immunodeficiency virus/acquired immunodeficiency syndrome (HIV/AIDS), solid organ transplant (SOT) recipients, and those receiving immunosuppressive therapy for autoimmune diseases or hematologic malignancies. 1 In HIV-associated CMVR, profound T-cell depletion, particularly impairment of CMV-specific CD4+ responses, permits uncontrolled viral reactivation from latency within CD34+ hematopoietic progenitor cells. 2
Non-HIV-associated CMVR arises in other immunocompromised populations, such as patients with hematologic malignancies undergoing chemotherapy, individuals with autoimmune diseases treated with immunosuppressive agents, and recipients of hematopoietic stem cell transplantation or SOT.3–5
In HIV-positive patients, antiretroviral therapy (ART) restores CD4+ T-cell function, usually controlling CMV replication within weeks and reducing relapse rates to below 10%. However, immune recovery uveitis (IRU) subsequently develops in approximately 10%–20% of cases, often leading to vision-threatening complications such as macular edema.6,7 By contrast, non-HIV patients, including transplant and cancer recipients, experience prolonged immunosuppression from chemotherapy or anti-rejection regimens. These patients demonstrate higher relapse rates (30%–54%), slower viral clearance, and greater risks of ganciclovir resistance (15%–30% compared with 5%–10% in HIV-positive patients). 8
Such differences highlight the need to investigate whether the underlying immune status shapes disease manifestations, such as lesion morphology, vitritis severity and retinal distribution, and influences long-term visual outcomes. Prior reports suggest that non-HIV patients often require more intensive intravitreal therapy and may exhibit atypical inflammatory features.9,10
The present study was therefore designed to evaluate how distinct etiologies of immunodeficiency, including HIV/AIDS, transplantation, oncologic therapy, and autoimmune suppression, affect the clinical presentation and outcomes of CMVR. Specifically, we aimed to determine whether fundamental differences in immune pathogenesis translate into clinically meaningful variations in retinal features and long-term visual prognosis.
Methods
This retrospective cohort included 117 eyes from 91 patients diagnosed with CMVR who received treatment at the Farabi Eye Hospital, Tehran, Iran, from 2009 to 2024. This study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (Supplemental Material). 11
Sample size calculation
The required sample size for this study was determined using a Chi-square power analysis to ensure sufficient statistical power to detect meaningful effects. Based on an expected effect size (Cohen’s w) of 0.3 (representing a medium effect), an alpha level (α) of 0.05, and a desired statistical power (1–β) of 0.80, the minimum required sample size was calculated to be 88. The final sample size in the study consisted of 91 patients, ensuring the study maintained a power level of at least 80% to detect significant differences.
Diagnostic criteria
CMVR was diagnosed by uveitis specialists according to the Standardization of Uveitis Nomenclature (SUN) working group framework and required: (i) necrotizing retinitis with indistinct borders and satellite lesions; and (ii) either characteristic fundus features (e.g., wedge-shaped, hemorrhagic, or granular retinitis) in a compatible immunocompromised host or polymerase chain reaction (PCR) positivity for CMV DNA in intraocular fluid, with a threshold > 500 copies/mL considered positive. 1
In cases where inflammatory or masquerade conditions could mimic CMV retinitis (e.g., Behçet’s disease or lymphoma), diagnostic confirmation relied preferentially on intraocular fluid PCR testing and careful exclusion of alternative etiologies. Clinical diagnosis was supported by the absence of features typical of noninfectious uveitis, lack of response to immunosuppressive therapy, and prompt clinical improvement following initiation of anti-CMV antiviral treatment.
Eligibility
Inclusion criteria were adults (⩾18 years of age) with active CMVR, available baseline, follow-up data. Exclusion criteria were other infectious retinitis (e.g., toxoplasmosis, acute retinal necrosis), primary intraocular lymphoma, insufficient documentation of ophthalmic examinations or immune status, immunocompetent patients, and preexisting retinal disease likely to confound visual outcomes (e.g., advanced diabetic retinopathy, age-related macular degeneration, prior retinal detachment (RD), or retinal vascular occlusions).
Grouping by immune status
Patients were stratified into two groups: (1) HIV-associated CMVR, defined by laboratory-confirmed HIV infection, CD4+ T-cell counts were recorded when available; and (2) non-HIV CMVR, comprising immunocompromised patients with documented immunosuppressive conditions, including SOT recipients, hematologic malignancies, autoimmune diseases treated with immunosuppressive therapy, or poorly controlled diabetes mellitus type 2 (DM2) (HbA1c > 9%).
Data collection
Extracted variables included age, sex, eye laterality, systemic diagnosis, serologic, and antigen testing for HIV, details of immunosuppressive or chemotherapeutic regimens, and the type of antiviral therapy (systemic or intravitreal).
Ophthalmic examinations and definitions
Best-corrected visual acuity (BCVA) was measured using Snellen charts and converted to logMAR for analysis. Baseline, post-injection, post-vitrectomy, and final visual acuities were recorded. Vitritis was graded according to SUN criteria. 12 Ophthalmic examinations and grading of clinical findings were performed by multiple experienced graders. Using fundus photography and indirect ophthalmoscopy, retinal lesion location was categorized as macular, peripheral, or total retinal involvement; lesion size as 1, 2-, and 4-disc diameter (DD, defined as the largest contiguous area of active retinitis; lesions larger than 4 DD were classified as ⩾4 DD and included in the 4 DD category); and the clinical type of retinitis and presence of retinal vasculitis were documented. Macular involvement and CMV-related complications, including macular edema, retinal thinning or necrosis, and vitreoretinal interface abnormalities, as well as RD, optic atrophy, and retinal ischemia were assessed by clinical examination and optical coherence tomography when media clarity and clinical circumstances permitted. Pars plana vitrectomy (PPV) was performed when clinically indicated, including RD or severe vitreous opacification.
Data on CMV viral load, CMV serologic status, and detailed systemic immunologic markers were not consistently available in the ophthalmic records and were therefore not included in the analysis.
Treatment protocol
All patients diagnosed with active CMV retinitis received antiviral therapy based on clinical severity, lesion location, and immune status. Intravitreal ganciclovir (2 mg/0.1 mL) was administered as first-line local therapy, typically on a weekly induction schedule, followed by maintenance injections at longer intervals as clinically indicated. Systemic antiviral therapy (intravenous or oral ganciclovir/valganciclovir) was used in selected cases, particularly in patients with bilateral disease, extensive retinal involvement, or severe systemic immunosuppression. Additionally, patients who were HIV-positive were treated accordingly in the hospital, including treatment with ART therapy.
Given the retrospective design and long study period, treatment regimens were not fully standardized and were individualized by the treating ophthalmologist. The total number of intravitreal injections per eye was therefore variable and is reported as a summary measure in the Results section. Pars plana vitrectomy was performed when indicated for complications such as retinal detachment or severe vitreous opacity.
Statistical analysis
Data were analyzed using IBM SPSS Statistics for Windows, version 22 (IBM Corp., Armonk, NY, USA). The Shapiro–Wilk test assessed normality of continuous variables. Categorical variables were compared using the Chi-square test. Continuous variables were compared using the independent-samples t-test or the Mann–Whitney U test, as appropriate. Predictors of final visual acuity (logMAR) were identified with multivariable linear regression. A backward selection procedure was applied: variables with univariate p < 0.20 entered the initial model, and variables were retained at p < 0.10 in the final model. All p-values were two-sided, with p < 0.05 considered statistically significant unless otherwise specified.
Results
Baseline characteristics
Ninety-one patients (117 eyes) with CMV retinitis were included. Mean age was 40.12 ± 14.67 years of age (median 38), and 62.6% were male. Disease was unilateral in 71.4% and bilateral in 28.6%. By immune status, 58 patients (63.7%; 71 eyes) were HIV-positive and 33 (36.3%; 46 eyes) were non-HIV. The non-HIV cohort comprised of hematologic malignancies (n = 12; 19 eyes); drug-induced immunosuppression (n = 15; 20 eyes) which includes solid organ transplant (n = 4), autoimmune disease (n = 9; systemic lupus erythematosus n = 3, Behçet’s n = 2, rheumatoid arthritis n = 1, psoriasis n = 1, inflammatory bowel disease n = 1, myasthenia gravis n = 1), congenital immunodeficiency (severe combined immunodeficiency (SCID) n = 1, hyper-IgE n = 1); and poorly controlled DM (n = 6). Lesions most commonly involved the macula (46.5%), followed by peripheral (39.6%) and total retinal involvement (13.9%). Fundoscopic features included white patches (41.0%) and hemorrhage (40.2%), with vascular sheathing (17.1%), branch angiitis (6.8%), necrosis (6.8%), and cotton-wool spots (5.1%) less frequent (Figure 1). Vitritis was +1 in 67.5%, +2 in 25.7%, and absent in 6.8%. RD occurred in 26.5% of eyes; PPV was performed in 14.5%. (Table 1). Among treated eyes, the number of intravitreal injections ranged from 1 to 5 per eye, with a mean of 1.76 ± 1.13 injections.

Fundus photography of CMVR in a young female with AIDS.
Baseline demographic and ocular characteristics of patients with CMV retinitis.

CMV, cytomegalovirus; DD, disc diameter; HIV, human immunodeficiency virus; N, number of patients; PPV, pars plana vitrectomy; RD, retinal detachment.
HIV-positive versus non-HIV comparisons
Age and sex distributions were similar between HIV-positive and non-HIV groups (HIV-negative). Non-HIV eyes exhibited more +2 vitritis (34.8% vs 19.7%, p = 0.009), whereas HIV-positive eyes more often had +1 vitritis (77.5% vs 52.2%). Lesion location and other fundoscopic features (branch angiitis, vascular sheathing, hemorrhage, cotton-wool spots, necrosis, white patches, ischemia, scar, and atrophy) did not differ significantly. Lesion size distributions were also similar when categorized as small (1 DD), medium (2 DD), and large (4 DD) (p = 0.413). RD (21.7% vs 29.6%; p = 0.397) and PPV rates (13.0% vs 15.5%; p = 0.793) were comparable (Table 2).
Comparison of demographic and clinical data across patients with HIV and non-HIV patients.

Fisher’s exact test for categorical variables and independent t test for continuous variables.
DD, disc diameter; HIV, human immunodeficiency virus; N, sample size; PPV, pars plana vitrectomy; RD, retinal detachment.
Visual acuity over time (HIV vs non-HIV)
HIV-positive eyes presented with worse baseline BCVA (logMAR 1.85 ± 1.14 vs 1.31 ± 1.17; mean difference 0.54 ± 0.22; p = 0.045). At post-injection, post-vitrectomy, and final visits, no significant between-group differences were observed (all p > 0.20; Table 3, Figure 2).
Comparison of BCVA between HIV+ and HIV– patients across time points.
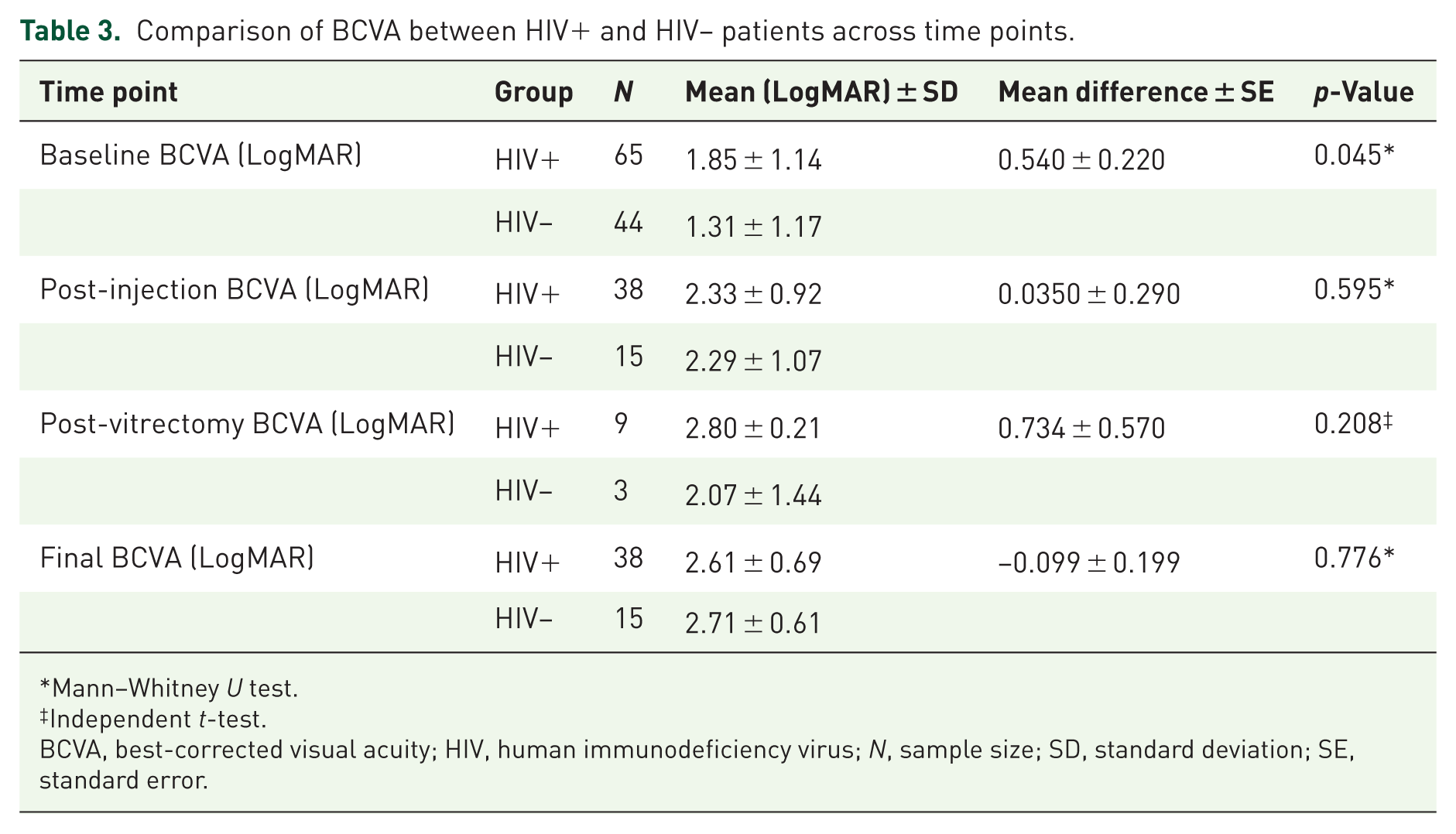
Mann–Whitney U test.
Independent t-test.
BCVA, best-corrected visual acuity; HIV, human immunodeficiency virus; N, sample size; SD, standard deviation; SE, standard error.

Trend of BCVA at different time points including baseline, post-injection, post-vitrectomy, and final BCVA in HIV+ and HIV– patients.
Non-HIV subgroup analysis (drug-induced vs hematologic)
Demographics and laterality were similar. Vascular sheathing was more frequent in drug-induced immunosuppression than in hematologic malignancy (30.0% vs 0.0%; p = 0.009), whereas cotton-wool spots were more frequent in hematologic malignancy than in drug-induced immunosuppression (21.1% vs 0%; p = 0.030). Location, size (small/medium/large), and most other features did not differ significantly. Hemorrhage (68.4% vs 40.0%; p = 0.075) and necrosis (15.0% vs 0%; p = 0.079) showed nonsignificant trends (Table 4).
Comparison of demographic and clinical data across patients with hematologic malignancies and immunosuppressant receiving patients.
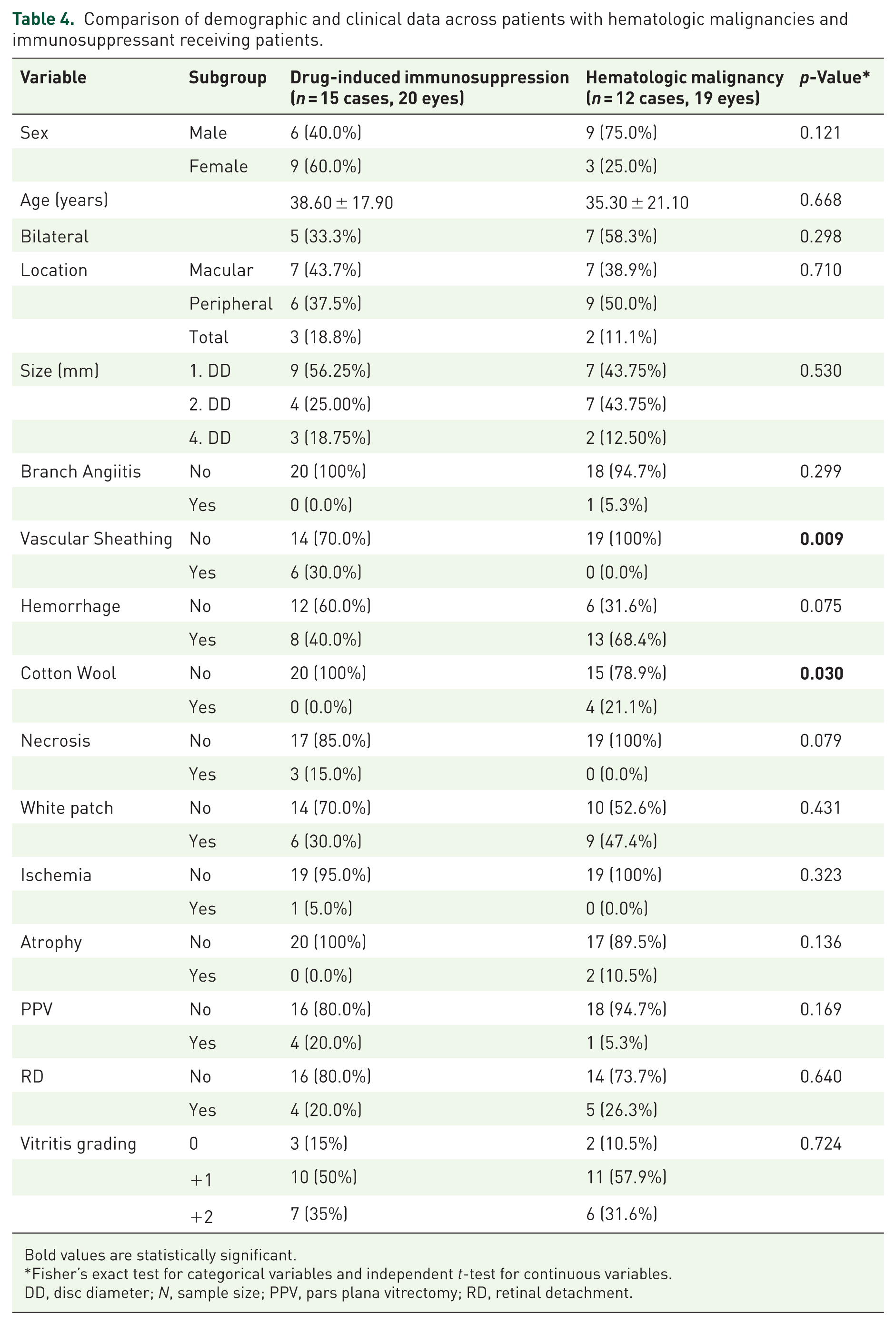
Bold values are statistically significant.
Fisher’s exact test for categorical variables and independent t-test for continuous variables.
DD, disc diameter; N, sample size; PPV, pars plana vitrectomy; RD, retinal detachment.
Visual acuity over time (non-HIV subgroups)
Hematologic malignancy cases had better baseline BCVA than drug-induced cases (0.97 ± 1.12 logMAR vs 1.67 ± 1.14 logMAR; p = 0.063). Post-injection and final visual outcomes were not significantly different (all p > 0.05; Table 5 and Figure 3).
Comparison of BCVA between drug-induced immunosuppression versus hematologic malignancies.
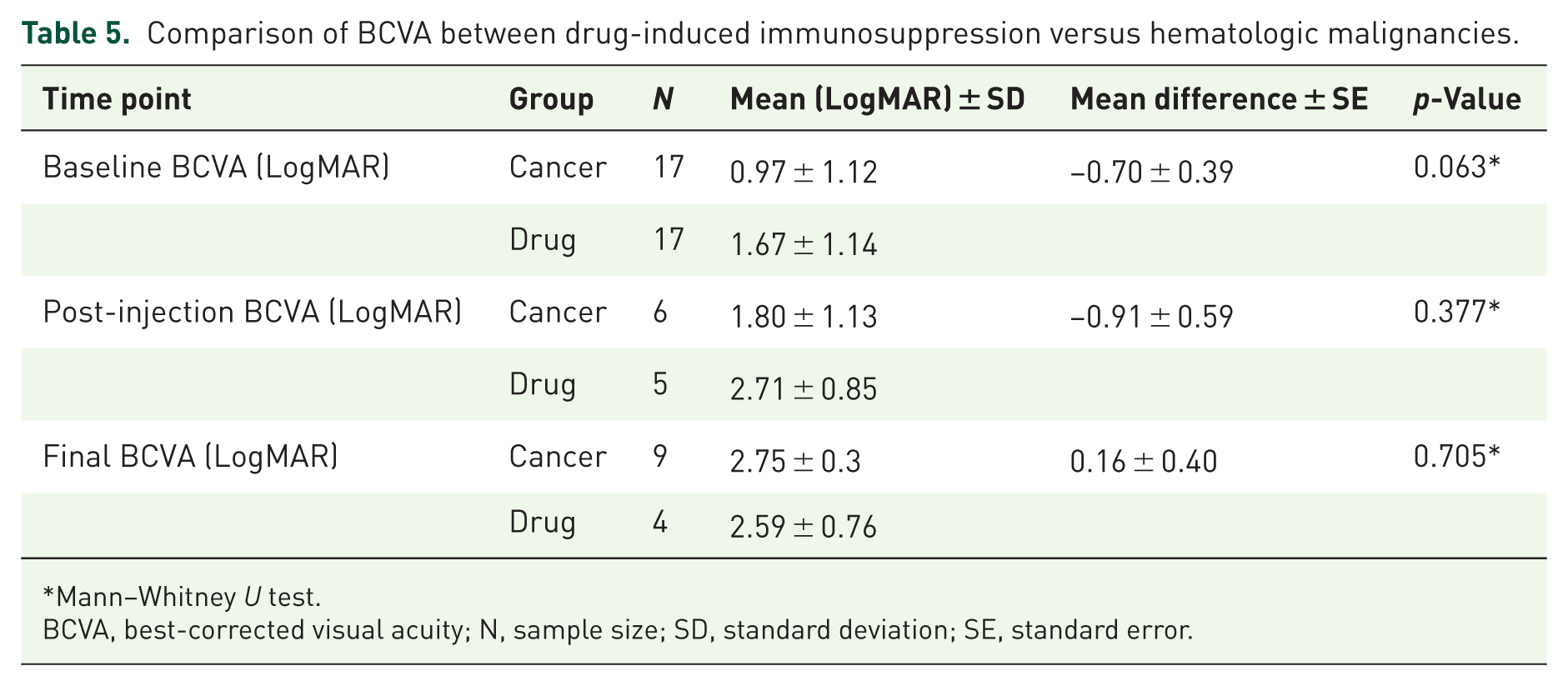
Mann–Whitney U test.
BCVA, best-corrected visual acuity; N, sample size; SD, standard deviation; SE, standard error.
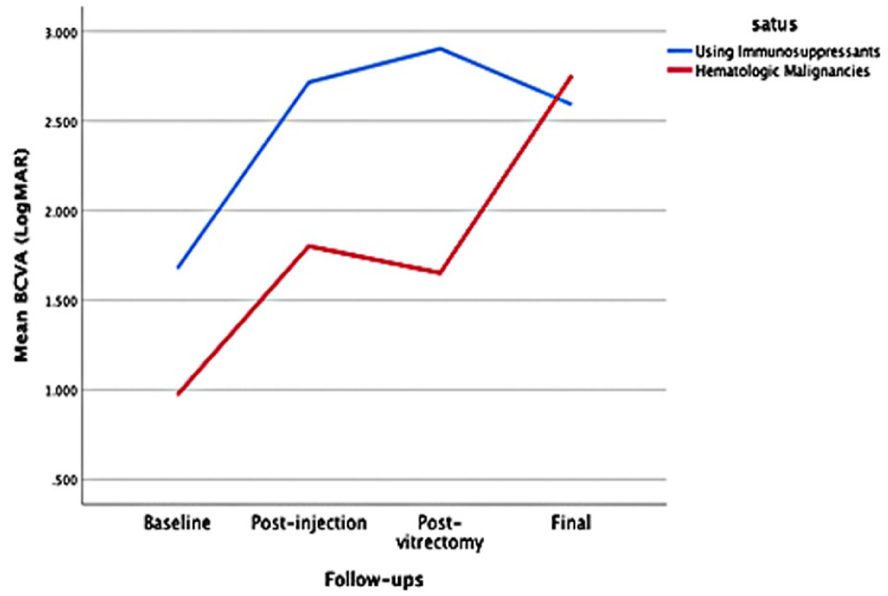
BCVA trend at baseline, post-injection, post-vitrectomy, and final BCVA between CMVR with hematologic malignancies and those receiving immunosuppressive drugs.
Predictors of final visual acuity
On multivariable linear regression, worse baseline BCVA (logMAR; β = 0.527, p < 0.001) and RD (β = 0.254, p = 0.017) independently predicted poorer final vision. HIV status and other covariates were not independent predictors. (Table 6).
Predictors of final visual acuity in CMV retinitis patients.

Bold values are statistically significant.
BCVA, best-corrected visual acuity; CMV, cytomegalovirus; DD, disc diameter; HIV, human immunodeficiency virus; PPV, pars plana vitrectomy; RD, retinal detachment.
Final visual acuity outcomes were additionally analyzed using clinically meaningful functional categories. Among the 54 eyes with available final BCVA data, no eyes achieved vision better than 20/40 (logMAR < 0.30). Five eyes (9.3%) retained ambulatory vision (final BCVA ⩽ 20/200, logMAR ⩽ 1.0), while the majority of eyes (49/54, 90.7%) met criteria for legal blindness (final BCVA > 20/200, logMAR > 1.0).
Discussion
Our investigation of clinical and visual outcomes across the immune-status spectrum of CMVR revealed both shared and divergent manifestations shaped by the underlying etiology of immunosuppression. Non-HIV immunocompromised patients demonstrated a propensity for more severe intraocular inflammation, presenting more often with higher-grade vitritis, whereas HIV-positive patients exhibited worse baseline visual acuity. Despite these baseline differences, long-term visual outcomes were comparable across immune-status groups. Furthermore, baseline BCVA and RD emerged as the only independent prognostic indicators, underscoring the need to identify and treat patients at higher risk promptly with tailored regimens.
We found no significant differences in several retinal features, zonal involvement (macular/peripheral), lesion size, RD rates, and other fundoscopic findings, between HIV and non-HIV patients, consistent with prior reports.8,13 While immune status clearly influences disease susceptibility and inflammatory phenotype, our findings suggest that within this clinically defined cohort, long-term visual outcomes were more strongly associated with structural disease severity at presentation, specifically baseline BCVA and retinal detachment, than with immune-status category (HIV vs non-HIV). Importantly, this conclusion is based on clinical prognostic modeling rather than direct immunologic or virologic measurements, which were unavailable in a substantial proportion of patients. In line with previous work by Ho et al. 14 , vitreous inflammation was more prominent in non-HIV CMVR, supporting the concept that these patients may retain relatively greater inflammatory capacity than those with advanced HIV. This pattern plausibly reflects differences in intraocular immune responses: HIV patients with severe CD4+ T-cell depletion may show minimal vitritis despite active retinitis, while non-HIV patients with iatrogenic or disease-related immunosuppression can still mount noticeable inflammatory reactions.15,16 Importantly, in non-HIV settings, particularly when lymphoma is in the differential, vitreous sampling with CMV PCR remains crucial to distinguish CMVR from masquerade entities such as primary intraocular lymphoma.
Longitudinally, although HIV-infected patients presented with significantly worse baseline BCVA, subsequent intravitreal therapy, vitrectomy when indicated, and follow-up care yielded similar visual outcomes to non-HIV patients. Nonsignificant trends toward greater macular involvement and slightly higher RD frequency in HIV-positive cases may help explain their poorer presentation. 17 Prior studies also link higher plasma/intraocular CMV DNA with worse outcomes. 7 In advanced HIV (often CD4+ < 50 cells/μL), delayed diagnosis and higher viral burden likely contribute to the initial visual deficit. 18 While we did not quantify time-to-diagnosis or viral load, these factors remain plausible explanatory mechanisms.
Within the non-HIV group, we observed distinct phenotypes between drug-induced immunosuppression and hematologic malignancies: vascular sheathing was more frequent with drug-induced immunosuppression (e.g., calcineurin inhibitors/corticosteroids), whereas cotton-wool spots were more frequent in hematologic malignancies. These differences may reflect medication-related microvascular injury and endothelial dysfunction on the one hand, and microvascular ischemia related to hematologic disease on the other. Prior work has emphasized the clinical challenge of recurrent disease and treatment escalation in these populations.16,19 Recognition of these subgroup-specific features can expedite targeted testing (e.g., aqueous/vitreous PCR) and guide differential diagnosis when overlapping microangiopathic processes are possible.
The overall RD prevalence was 26.5%, within the 20%–30% range reported in the literature, with similar RD and PPV rates across immune-status groups, echoing prior findings.14,20 Despite phenotypic differences at presentation, long-term visual outcomes did not differ by immune etiology.13,14,21 Multivariable analysis reinforced that baseline BCVA and RD, not immune-status category, drive final visual prognosis, suggesting that disease stage and vasculitic activity at presentation are the principal determinants of outcome. Although RD was not statistically significant in univariate testing, we prespecified it as a clinically important covariate and retained it in the multivariable model to control for confounding by baseline BCVA, vitritis grade, and lesion location. In the adjusted model, including RD changed the estimates of other predictors and provided a more clinically coherent specification, indicating that its lack of univariate significance likely reflected confounding-suppression rather than absence of effect.
Clinical trajectories nonetheless differ between HIV and cancer/transplant patients because of immune reconstitution dynamics. In HIV, effective ART often restores CMV-specific T-cell responses within months, driving durable viral control and low relapse rates once CD4+ counts recover; however, this can unmask IRU with sight-threatening sequelae.6,7,22–25 In contrast, cancer/transplant recipients experience sustained or cyclical immunosuppression, predisposing to higher relapse, slower viral clearance, and greater ganciclovir resistance (15%–30% vs ~5%–10% in HIV), sometimes necessitating foscarnet, cidofovir, or maribavir.22,26–29 Clinical series, including those from tertiary centers, underscore the need for more intensive intravitreal regimens and prolonged systemic therapy in such patients, especially amid chemotherapy-related myelosuppression or graft versus host disease activity.19,30
The uniformly poor final acuity across groups in our cohort helps explain why lesion size, macular involvement, and RD did not associate with final BCVA. A plausible interpretation is a ceiling effect: once CMVR has produced extensive retinal necrosis, additional structural differences may exert limited incremental impact on vision. Earlier detection and treatment, before extensive photoreceptor and RPE damage, remain central to improving prognosis.
Our series also included complex cases, one patient with concurrent multiple myeloma and HIV, categorized within the HIV group for analysis, and three pediatric patients (SCID and congenital HIV). These highlight the breadth of CMVR presentations across the lifespan and immune defects.
Although our findings are broadly consistent with prior comparative studies of HIV- and non-HIV-associated CMV retinitis, our cohort provides complementary data from a Middle Eastern tertiary referral center, a population that remains underrepresented in the literature. The Iranian cohort reflects a heterogeneous spectrum of immunosuppression, including autoimmune disease–related therapy, hematologic malignancy, transplantation, and poorly controlled diabetes, which may differ in relative frequency and referral patterns from East Asian or Western series. In addition, the advanced visual impairment at presentation observed in our cohort likely reflects delayed diagnosis and limited routine ophthalmic screening in systemically ill patients, highlighting an important real-world challenge in similar healthcare settings. Finally, our subgroup analysis within non-HIV patients identified distinct vasculopathic and microangiopathic retinal features that may aid clinical recognition across diverse immunosuppressed populations.
Strengths of this work include standardized diagnostic criteria (SUN), a uniform treatment paradigm (intravitreal ganciclovir plus systemic antivirals), and explicit subgroup analyses within non-HIV etiologies, areas underexplored previously. Limitations include the retrospective single-center design, incomplete data on CD4+ nadir, immunosuppressant levels, CMV viral loads, and the lack of systematic IRU assessment and genotypic resistance testing, particularly relevant in cancer/transplant cohorts. Visual outcome analyses may be influenced by incomplete post-treatment visual acuity data, despite the fact that intravitreal antiviral therapy was administered to most patients in the cohort.
In summary, CMVR shows distinct phenotypes across immunodeficiency subtypes, yet final visual outcomes are driven chiefly by baseline BCVA and RD rather than by immune etiology. These findings support early detection, rapid antiviral initiation, and phenotype-informed monitoring, prioritizing early intervention in HIV and vigilance for inflammatory/vasculitic complications in non-HIV, while emphasizing shared prognostic determinants that transcend the specific cause of immunosuppression.
Supplemental Material
sj-docx-1-oed-10.1177_25158414261440308 – Supplemental material for Cytomegalovirus retinitis in diverse immunocompromised populations: clinical presentation, treatment outcomes, and prognostic factors
Supplemental material, sj-docx-1-oed-10.1177_25158414261440308 for Cytomegalovirus retinitis in diverse immunocompromised populations: clinical presentation, treatment outcomes, and prognostic factors by Seyed Ali Tabatabaei, Arash Mirzaei, Kimia Daneshvar, Ghazal Ghochani, Nader Mohammadi, Mohammadreza Mehrabi Bahar, Zahra Mahdizad, Kosar Esmaeili, Mehdi Aminizade, Morteza Karimi, Pedram Afshar, Amirhossein Valizadeh, Kosar Namakin, Mohamad Sanei, Sarah Miller, Kareena Chawla, Nazanin Ebrahimi Adib, Jaron Sanchez and Mohammad Soleimani in Therapeutic Advances in Ophthalmology
