Abstract
Background:
Selective laser trabeculoplasty (SLT) is an effective long-term option for the treatment of open-angle glaucoma.
Objectives:
To investigate the real-world efficacy and safety of SLT with the Zeiss VISULAS green laser in medically treated primary open-angle glaucoma (POAG).
Design:
Retrospective, single-center study.
Methods:
POAG patients ⩾18 years of age on at least one antiglaucoma medication prior to the procedure, who underwent SLT with the Zeiss VISULAS green laser (Carl Zeiss Meditec, Jena, Germany). Medications were terminated or reinstated at subsequent follow-ups depending on the intraocular pressure (IOP) control. The primary efficacy outcome measures were mean reduction in glaucoma medications and mean reduction in IOP at 3, 6, and 12 months compared to baseline. Safety outcomes were intra- or post-procedural complications.
Results:
One hundred fifty-six eyes of 156 patients, aged 66.5 ± 13.2 years, were recruited. The mean number of medications was reduced from 2.3 ± 1.0 at baseline to 0.8 ± 1.1 and 0.8 ± 1.2 at 3- and 6 months, respectively (p < 0.001). Despite the reduction of medications, the mean IOP (mmHg) was reduced from 16.6 ± 3.10 at baseline to 15.4 ± 3.82 at 3 months (n = 156) and 15.1 ± 3.30 at 6 months (n = 140) (p < 0.001). IOP reduction ⩾20% was observed in 28.8% of patients at 3 months and 27.9% of patients at 6 months. In the 12-month follow-up group (n = 22), mean medications and IOP (mmHg) at baseline were 2.3 ± 0.9 and 16.0 ± 2.80, respectively, which decreased to 0.9 ± 1.1 (p < 0.001) and 14.5 ± 1.92 (p = 0.103), respectively. At 3- and 6-month post-procedure, 59.6% of patients and 60% of patients, respectively, were medication-free compared to none at baseline. At 6 months, 69.3% of patients maintained IOP lower than or equal to pre-procedure IOP on fewer medications compared to baseline. No vision-threatening intra- or post-procedure complications were noted.
Conclusion:
SLT with the Zeiss VISULAS green laser resulted in a clinically and statistically significant reduction in number of medications needed for IOP control. The procedure had a good safety profile similar to that described in the literature for SLT, with no vision-threatening complications.
Plain language summary
Open-angle glaucoma is an eye disorder characterized by increased internal eye pressure, which can lead to optic nerve damage and consequent vision loss. Typically managed with medications that lower this pressure, alternative treatments like laser therapies are also available, also know as Selective Laser Trabeculoplasty (SLT). This study examined a new laser’s effectiveness as such an alternative. Researchers conducted a retrospective analysis of adult patients who had received the SLT green laser treatment. These patients had all been using at least one glaucoma medication before undergoing the procedure. The main goal was to assess changes in the number of medications required and eye pressure levels at several time points—three, six, and twelve months after the treatment. The findings were promising. Initially, patients used an average of 2.3 different medications. After the treatment, this average decreased significantly within just three to six months. By six months, about 60% of patients no longer needed any glaucoma medications. Importantly, this reduction in medication did not compromise the control of eye pressure. In fact, the majority of patients maintained lower or stable eye pressure compared to their levels before the treatment. Furthermore, the long-term outcomes (evaluated at twelve months for a smaller group) showed continued effectiveness of the laser treatment with sustained medication reduction and stable eye pressure. Regarding safety, the study underscored a low risk of complications. There were no severe or vision-threatening side effects reported following the laser procedure. Overall, this study supports that this green laser as a beneficial option for managing eye pressure in patients with open-angle glaucoma, offering a potential reduction in the dependence on medications. The treatment is not only effective in lowering eye pressure but also in maintaining these levels with minimal safety concerns.
Keywords
Introduction
Glaucoma is a chronic optic neuropathy characterized by progressive retinal ganglion cell death, leading to characteristic optic nerve head changes and visual field constriction.1,2 Intraocular pressure (IOP) is the only modifiable risk factor for disease progression, and reduction of IOP remains the most effective treatment strategy in glaucoma. 3 Medical therapy with antiglaucoma eye drops is the standard first-line treatment in primary open-angle glaucoma (POAG). 4 However, antiglaucoma medications have several local and systemic adverse effects. 5 These side effects, combined with the long duration of treatment and the need for multiple medications, lead to reduced quality of life. 6 This can affect patient satisfaction and reduce compliance with medications. 7 There is a clear need to identify alternative treatment options that can reduce or replace antiglaucoma medications in POAG management.
Selective laser trabeculoplasty (SLT) involves treating the trabecular meshwork with a Q-switched, frequency-doubled 532 nm Nd:YAG laser. It induces the secretion of proinflammatory cytokines, namely IL-1α, IL-1β, TNF-α, and IL-8, which activate matrix metalloproteinases and recruit monocytes to the trabecular meshwork. This leads to trabecular meshwork remodeling and phagocytosis of cellular debris within the trabecular meshwork, resulting in increased aqueous outflow and reduced IOP. 8
SLT has an excellent safety profile 9 and has been proven noninferior to antiglaucoma medication in terms of IOP reduction and treatment success rates. 10 Several studies have demonstrated the effectiveness of SLT as an adjunctive treatment in reducing IOP and medication burden in open-angle glaucoma (OAG).10–12 There is also substantial evidence supporting its potential as first-line therapy in OAG and ocular hypertension (OHT).13–16 Six-year results from the LiGHT Trial demonstrated better long-term disease control and reduced need for incisional glaucoma surgery with SLT as compared to medication. 17 In a recent meta-analysis of randomized controlled trials comparing SLT and antiglaucoma medication in the treatment of OAG or OHT, both SLT and medical therapy achieved comparable IOP reduction at 12 months, while glaucoma surgery rates, number of antiglaucoma medications, and rates of ocular adverse effects were significantly reduced with SLT. 18
The Zeiss VISULAS green (Carl Zeiss Meditec, Jena, Germany) workstation for retinal photocoagulation was recently updated with a new SLT application mode for the VISULAS green laser. It utilizes a squared spot shape profile that ensures homogeneous laser energy distribution over the entire treated area. Unlike other SLT procedures that titrate laser energy based on the formation of cavitation bubbles, Zeiss SLT utilizes a simplified titration process based on trabecular meshwork pigmentation, which prevents undue energy application to the target tissue. 19 To the authors’ knowledge, only one study reports the prospective results of the device in a 34 eyes glaucoma cohort and with a 3-month follow-up. 20 This study investigated the efficacy and safety of the Zeiss SLT among South African patients with POAG on antiglaucoma medications in a real-world clinical setting.
Methods
Study design
This was a retrospective, observational, single-center study conducted at a private ophthalmology practice in Durban, South Africa. The electronic health records of patients who received SLT with Zeiss VISULAS green between May 2022 and December 2022 were reviewed.
The study included patients aged ⩾18 years, with mild to moderate POAG as per the Glaucoma Severity Staging system 21 with Humphrey visual field mean deviation not worse than −12 dB, on at least one antiglaucoma medication pre-SLT, and a minimum of 3 months follow-up post-SLT. SLT was performed in these patients with the intention to decrease the number of pharmacological agents needed for IOP control. Patients with a history of glaucoma surgery within 1 year prior to the Zeiss SLT or with a history of any ocular surgery within the period of review were excluded. Other exclusion criteria were history of previous SLT, abnormal angle anatomy or narrow angles (Schaffer grade 1 or 2) on gonioscopy, secondary glaucoma, congenital glaucoma, and pregnancy.
Procedure
The Zeiss VISULAS green is an integrated workstation for retina and glaucoma laser therapy that is equipped with a module for performing SLT. It uses a frequency-doubled 532 nm Nd:YVO4 laser to achieve selective photothermolysis of the trabecular meshwork. Each laser application consists of a fixed pattern made up of 52 squared spots, with a total application diameter of 400 µm. 19 Approximately 100 non-overlapping 400 μm laser applications were administered over 360° of the trabecular meshwork. Laser power was manually titrated between 15 and 40 µJ based on the degree of trabecular meshwork pigmentation as per the manufacturer’s recommendation. No post-procedural anti-inflammatory or additional IOP-lowering drops were used. Depending on patient’s pharmacological treatment at baseline and the level of IOP control post-SLT, one or more antiglaucoma agents were terminated at the discretion of their treating specialist 10 weeks after the procedure, allowing for a washout period of at least 2 weeks before their 3-month review. Antiglaucoma medications were terminated or reinstated at subsequent follow-up visits depending on the level of IOP control, at the discretion of the treating specialist. All treatments and follow-up consultations were conducted by two specialist ophthalmologists.
Data collection
Only one eye per patient was included. For patients who underwent bilateral SLT, a single eye was randomly selected using simple randomization based on the last digit of their clinic number. If the clinic number ended in an even number, the right eye was selected, while the left eye was selected if the clinic number ended in an odd number.
Patient charts were reviewed and demographic data (age, gender, race), relevant ocular history, and details regarding the SLT procedure (date of treatment, laser energy applied) were recorded. Clinical details of a comprehensive ocular examination (including slit lamp examination of the anterior segment, IOP measurement with Goldmann applanation tonometry, gonioscopic examination of the anterior chamber and classification of angle pigmentation according to SCHEIE grading, and fundus examination) and number of antiglaucoma medications used were noted for each patient at baseline and at 3, 6, and 12 months post-SLT. Any complications occurring during or after the procedure as well as any repeat SLT or subsequent filtration surgery were also noted.
The primary outcome measures were mean reduction in glaucoma medications at 3, 6, and 12 months compared to baseline and mean reduction in IOP at 3, 6, and 12 months compared to baseline. Secondary efficacy outcomes were defined for 3- and 6-month follow-up data as per the Guidelines on Design and Reporting of Glaucoma Surgical Trials 22 ; these included the proportion of eyes with IOP ⩽18 mmHg and ⩽15 mmHg, the proportion of eyes with IOP ⩽18 mmHg and ⩽15 mmHg on fewer medications compared to baseline, the proportion of eyes with reduction in both IOP and medications, the proportion of eyes with at least 20% IOP reduction versus baseline, the proportion of medication-free eyes, and the percentage reduction in IOP and medications versus baseline. Safety outcomes included any intra- or post-procedural complications. Pre-procedural factors such as history of cataract surgery, grade of anterior chamber angle pigmentation (according to SCHEIE grading of angle pigmentation), and number of pre-SLT medications were investigated for any effect on efficacy outcomes.
Statistical analysis
Statistical analysis was performed using the SPSS version 28 (IBM, Armonk, NY, USA). Descriptive statistics, in the form of mean ± standard deviation, was used to summarize the change in number of medications and IOP from the preoperative visit through 12 months postoperatively. Wilcoxon signed-rank test was used to compare the number of medications as well as the IOP at baseline versus each time point. A p < 0.05 was considered statistically significant. The graphs were created in accordance with the Guidelines on Design & Reporting Glaucoma Trials. 22
Results
Patient demographics
One hundred fifty-six eyes of 156 patients were recruited, with a mean age of 66.5 ± 13.2 years. The participants were 55.1% White, 34.6% Indian, and 10.3% Black; 52.6% of the patients were female (Table 1). Most patients were pseudophakic (61.5%, n = 96). All patients had normal angle morphology; majority of patients (89.1%, n = 139) had barely visible (Grade I) to no pigmentation (Grade 0) according to Scheie’s grading of angle pigmentation. Mean laser energy applied was 34.9 ± 6.07 µJ.
Patient demographics and preoperative measurements.
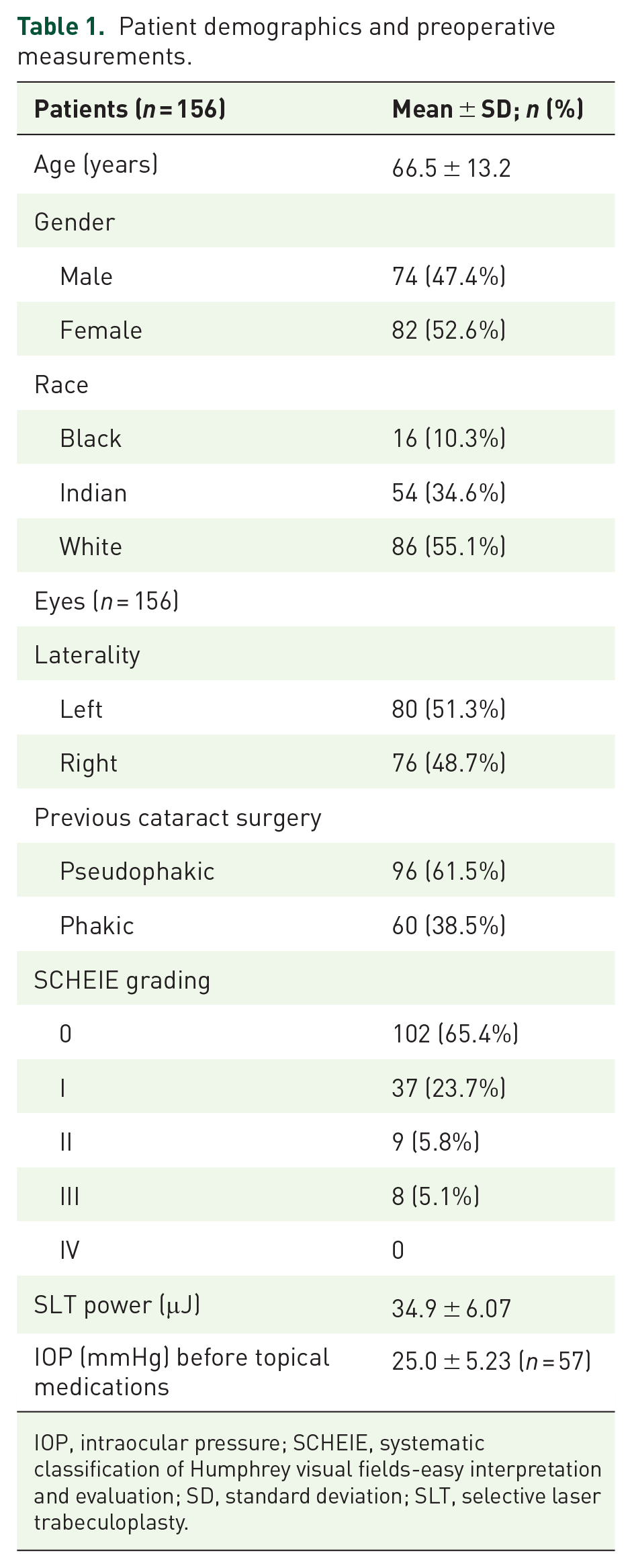
IOP, intraocular pressure; SCHEIE, systematic classification of Humphrey visual fields-easy interpretation and evaluation; SD, standard deviation; SLT, selective laser trabeculoplasty.
Efficacy outcomes
Data from all 156 patients were available for assessment at the 3-month follow-up. Outcomes of 140 patients were available for assessment at 6 months.
Average number of antiglaucoma medications used at baseline was 2.3 ± 1.0, of which the most frequently used agent was prostaglandin analogs, followed by beta antagonists, topical carbonic anhydrase inhibitors, alpha-2 receptor agonists, and oral carbonic anhydrase inhibitors. Mean number of medications reduced to 0.8 ± 1.1 at 3 months and 0.8 ± 1.2 at 6 months (p < 0.001) (Table 2(a)). Percentage reduction in mean number of medications was 75.5% at 3 months and 71.6% at 6 months (Table 3). More than half of the patients were medication-free at 3 months (59.6%, n = 93) and at 6 months (60%, n = 84). The proportion of patients on ⩾2 medications was 74.3% (n = 116) at baseline, decreasing to 30.1% (n = 47) at their last follow-up (Figure 1).
(a) IOP and medications outcomes of all eyes at baseline, 3 and 6 months. p values are versus baseline.

(b) IOP and medication outcomes of eyes that reached 12 months follow-up (n = 22). P values are versus baseline.
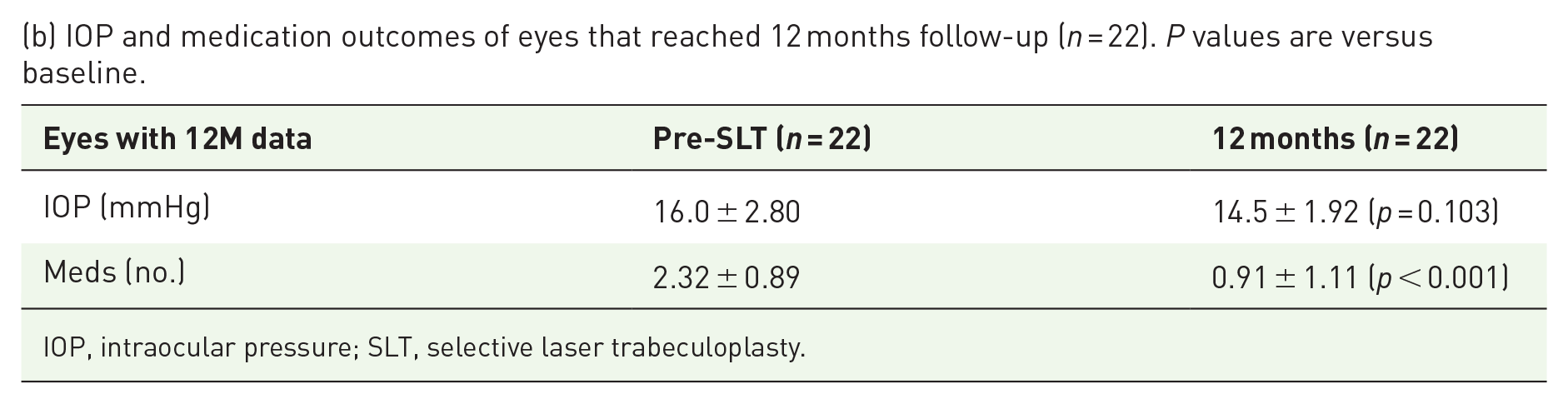
IOP, intraocular pressure; SLT, selective laser trabeculoplasty.
Success at each timepoint.

IOP, intraocular pressure; SLT, selective laser trabeculoplasty.

Bar diagram comparing medications at preoperative baseline and postoperative follow-up (latest timepoint).
Baseline IOP was 16.6 ± 3.10 mmHg, which decreased to 15.4 ± 3.82 mmHg at 3 months and 15.1 ± 3.30 mmHg at 6 months (Table 2(a), Figure 2(a)). Reduction in IOP from baseline was statistically significant at both timepoints (p < 0.001). Percentage reduction in mean IOP was 5.16% at 3 months and 7.83% at 6 months (Table 3). At least 20% reduction in IOP was observed in 28.8% (n = 45) patients at 3 months and 27.9% (n = 39) patients at 6 months (Figure 2(b)).

(a) Box plot of IOP outcomes from preoperative baseline to 6 months postop. (b) Scatterplot of IOP outcomes at preoperative baseline and postoperative follow-up (6 months). The diagonal line indicates no change in IOP; the horizontal line indicates 18 mmHg. The red line indicates IOP reduction of at least 20%.
At 3 months and 6 months post-procedure, 59% of patients (n = 92) and 60% (n = 84) patients, respectively, showed reduction in both IOP and number of medications (Table 3). At 6 months, 69.3% of patients (n = 97) maintained IOP lower than or equal to pre-procedure IOP on a fewer number of medications compared to baseline.
Consistent 12-month cohort (n = 22)
Twelve-month outcomes were available for 22 patients. Average number of antiglaucoma medications used pre-procedure by this group was 2.3 ± 0.9, which reduced to 0.9 ± 1.1 at 12 months (p < 0.001; Table 2(b)). Baseline IOP in these patients was 16.0 ± 2.80 mmHg, which decreased to 14.5 ± 1.92 mmHg at 12 months (p = 0.103).
Outcomes by baseline medications, lens status, and SCHEIE grading
Patients were stratified according to number of medications used pre-procedure (Table 4). Patients on one medication at baseline were almost free of medications at 6 months. Patients on ⩾2 medications at baseline maintained IOP reduction at 6 months even with a reduction in number of medications used (Table 4).
IOP and medication outcomes based on number of medications at preop.

SLT, selective laser trabeculoplasty.
Patients were also stratified according to lens status and SCHEIE grading (Table 1). There were no statistically significant differences between pseudophakic and phakic eyes in IOP and number of medications at baseline (p = 0.164 and p = 0.184, respectively) and at 6 months postoperatively (p = 0.719 and p = 0.583, respectively). Similarly, no significant differences were observed between eyes grouped by degree of angle pigmentation in terms of IOP and number of medications at baseline (p = 0.457 and p = 0.693, respectively) and at 6 months (p = 0.122 and p = 0.393, respectively).
Safety
Three patients (1.9%) experienced a transient rise in IOP greater than 5 mmHg during the first 5 days after SLT. This self-limiting IOP spike was not found to be associated with either success or failure at 3 months post-procedure. Two patients (1.3%) developed a corneal epithelial defect after vigorously rubbing their anesthetized eyes to remove the coupling gel post-procedure; the defect was minor in both cases and resolved without further complications. No other side effects or complications were noted. Three eyes were excluded from the study population and the data analysis as they received additional glaucoma surgery within 6 months of the SLT procedure.
Discussion
This study examined the efficacy and safety of SLT with the Zeiss VISULAS green laser in POAG patients on medication. As mean IOP was already under control at baseline, the main aim of the procedure was to reduce the number of medications needed to keep IOP under control. A clinically and statistically significant reduction was observed in the number of glaucoma medications, dropping from 2.3 ± 1.0 at baseline to 0.8 ± 1.1 at 3 months and 0.8 ± 1.2 at 6 months (p < 0.001). Proportion of medication-free patients increased from zero at baseline to 59.6% at 3 months and 60% at 6 months. In the 12-month cohort, number of medications reduced significantly from 2.3 ± 0.9 at baseline to 0.9 ± 1.1 post-procedure (p < 0.001). Additionally, even if IOP was already under control, mean IOP decrease was statistically significant (p < 0.001), along with a reduction in number of therapeutic agents used.
In a randomized controlled trial by Lee et al. comparing adjuvant therapy with SLT to therapy with medication alone in POAG patients, baseline number of medications was 2.3 ± 1.1 in the SLT group, which reduced to 1.5 ± 1.2 at 6 months (p < 0.0001). 11 The percentage reduction in mean number of medications was 34.8%. Better medication-lowering efficacy was seen in the current study, where mean number of medications at baseline decreased from 2.3 ± 1.0 to 0.8 ± 1.2 at 6 months (p < 0.001)—a percentage reduction of 71.6%.
Francis et al. 23 conducted a prospective, non-randomized trial evaluating efficacy of SLT in medically controlled patients of OAG. The mean number of medications was 2.8 ± 1.1 at baseline, which reduced to 0.7 ± 0.9 at 6 months and 1.5 ± 0.9 at 12 months (p < 0.0001). Among the patients on one medication pre-SLT, 63% were medication-free at 12 months. Comparable medication-lowering efficacy was seen in the current study.
In a prospective study by Yener et al. comparing SLT as adjunct or initial therapy in POAG patients, number of medications in the adjunctive treatment group reduced from 2.11 ± 0.88 at baseline to 0.9 ± 0.15 at 12 months (p < 0.001). 24 Proportion of eyes on ⩾2 medications at baseline was 76.1%, which reduced to 38% at 12 months. This was comparable to medication reduction seen in the current study: in the 12-month follow-up group, number of medications reduced from 2.3 ± 0.9 at baseline to 0.9 ± 1.1 (p < 0.001). Proportion of patients on ⩾2 medications was 74.3% at baseline, which decreased to 30.1% at the latest timepoint.
In the only other study on SLT performed with the same device, the Zeiss VISULAS green laser, Pillunat et al. prospectively examined a cohort of medically treated as well as treatment naïve patients of POAG. 20 Interestingly, their study’s aim was opposite to the current study, as their primary objective was IOP reduction and they did not change the number of medications to isolate the reduction in IOP. Baseline IOP reported in the Pillunat et al. study was higher than that observed in our study (21.0 ± 2.69 mmHg vs 16.6 ± 3.10 mmHg, respectively); accordingly, IOP-lowering efficacy of Zeiss SLT obtained in their study was greater than that in our study, as higher baseline IOP is associated with greater IOP reduction post-SLT.25,26 Conversely, in their study, number of medications did not reduce as per protocol, while in our study, a clinically and statistically significant reduction in the number of medications was obtained while keeping IOP under control. A further explanation could be the predominance of eyes with light or no trabecular meshwork pigmentation in the current study, as greater angle pigmentation has been found to be associated with success post-SLT. 27
Similarly, other studies evaluating IOP-lowering efficacy of SLT have reported greater IOP lowering than what was observed in our study. Katz et al. 14 observed a 26.4% IOP reduction post-SLT while Nagar et al. 15 reported a > 20% IOP reduction from baseline in 8T of patients. However, as with the study by Pillunat et al., the mean baseline IOP was higher in these studies compared to the current study (24.5 ± 2.1 mmHg and 29.3 mm Hg, respectively, vs 16.6 ± 3.10 mmHg in the current study).
Our study did not find any association between efficacy outcomes and pre-procedure factors such as patient demographics, degree of trabecular meshwork pigmentation, or history of cataract surgery. This corresponds with previous studies that have found higher pre-SLT IOP to be the only factor consistently affecting post-SLT efficacy.25,26
The safety outcomes for the Zeiss SLT observed in this study correspond with the reported safety of SLT in prior studies. 9 Adverse events following Zeiss SLT observed by Pillunat et al. were few, mild, and transient with up to 3 months of follow-up; 20 our study, with a percentage of patients in the 12-month analysis, validates their findings. A transient IOP spike greater than 5 mmHg was observed only in 1.9% of the total study population. Corneal epithelial defects were observed in two patients but these were not related to the procedure itself and were caused due to patients rubbing their anesthetized eyes to remove the coupling fluid used during the procedure. No vision-threatening complications were noted.
The limitations of our study include its retrospective design, lack of a control group, and short period of follow-up for most patients. However, a smaller cohort was followed consistently for 12 months, and this group demonstrated significant medication reduction comparable to that achieved by SLT in previous studies, suggesting that medication-lowering efficacy was sustained throughout 1-year post-Zeiss SLT. Another limitation was the exclusion of patients with further glaucoma surgery, which could have introduced some bias toward treatment success; however, their number was limited to 3 eyes as opposed to 156 eyes included in the study population. Prospective, comparative studies with larger sample sizes and longer follow-up periods are needed to evaluate the long-term efficacy and safety of the Zeiss SLT. Future studies should assess rates of treatment failure due to inadequate IOP reduction, increase in medication burden, or need for additional glaucoma surgery. Quality of life outcomes after medication reduction can also be investigated.
This study was the first to investigate the efficacy and safety of SLT with the Zeiss VISULAS green laser in routine clinical practice. The Zeiss SLT was found to achieve a clinically and statistically significant reduction in the number of antiglaucoma medications needed for IOP control, with most patients becoming medication-free at 6 months post-procedure. Medication-lowering efficacy and safety profile of the Zeiss SLT were found comparable to that reported in previous SLT studies. In conclusion, SLT with the Zeiss VISULAS green laser is an effective and safe treatment modality in medically treated primary open-angle glaucoma.
Footnotes
Acknowledgements
Editorial assistance was provided by GP Communications and was funded by Carl Zeiss Meditec Inc. The concept of the study was created by the principal investigator and not influenced by the funder. The funder was not involved with the data collection, analysis, or interpretation, and did not influence the results of the study.
