Abstract
Background:
Extended depth-of-focus (EDOF) intraocular lenses (IOLs) provide a continuous range of uncorrected vision at different distances.
Objective:
To assess visual acuity, refractive outcomes, and patient satisfaction after cataract surgery using the LuxSmart EDOF IOL (Bausch+Lomb).
Design:
A two-center, prospective observational clinical study.
Methods:
This study includes patients who have undergone bilateral phacoemulsification with bilateral implantation of the LuxSmart EDOF IOL, with the aim of achieving emmetropia. Visual acuity (VA) and subjective refraction were obtained in all patients preoperatively and at 1 week, 1 month, 3 months, and 6 months postoperatively. Subjective measures were obtained using the Catquest-9SF questionnaire and the Patient-Reported Spectacle Independence Questionnaire (PRSIQ). Contrast sensitivity and a glare assessment were undertaken postoperatively. Safety outcomes were additionally assessed.
Results:
Sixty eyes from thirty patients were recruited for this study. The mean spherical equivalent was 1.04 ± 1.67 D preoperatively and −0.30 ± 0.46 D at 6 months. Monocular uncorrected distance VA was 0.57 ± 0.32 (logMAR) preoperatively and 0.09 ± 0.12 (logMAR) at 6 months. Mean 6-month monocular uncorrected VA was 0.11 ± 0.13, 0.12 ± 0.15, and 0.31 ± 0.15 (logMAR) at 80, 66, and 40 cm, respectively. More than 90% of patients were either fairly or very satisfied with their vision. There was one case of suspected vitreomacular traction postoperatively.
Conclusion:
The LuxSmart EDOF IOL offers good refractive, VA, and safety outcomes. The single elongated focal point designed to enhance the DOF shows good results for VA and patient satisfaction.
Introduction
Over decades, research has been ongoing to compensate for the natural gift of accommodation, which is lost in cataract surgery and presbyopia. Recently, extended depth-of-focus (EDOF) intraocular lenses (IOLs) have been developed to offer a large range of vision while preserving binocularity and minimizing photic phenomena observed in multifocal IOLs 1 by creating an elongated focal point, enhancing the quality over a range of distances rather than discrete points. 2
Several different EDOF IOLs are available on the market today. A classification of EDOF IOLs was proposed by Alió (then published in the scientific literature by Megiddo-Barnir and Alió 3 – with Alió as last author) and categorized EDOF IOLs into five types according to their optical design: type 1 uses spherical aberrations to elongate the DOF; type 2 utilizes the pinhole effect; type 3 is a multifocal IOL with a low addition for near vision; type 4 is a hybrid IOL that utilizes spherical aberrations, and it is combined with a near vision addition; and type 5 uses a special geometry that creates a power gradient from the center to the periphery.
The LuxSmart EDOF IOL (Bausch+Lomb, Rochester, NY, USA) uses fourth- and sixth-order spherical aberration of opposite signs, located centrally, to increase the subjective depth of field to approximately 1.5 D with a gradual reduction of vergence from the center to the periphery.4 –6 The lens is classified type 5 EDOF IOL according to Megiddo-Barnir and Alió. 3 This group of EDOF IOLs have either a center with higher refractive power that gradually declines in the periphery or use wavefront to extend the DOF. The authors hypothesized that using this lens would therefore offer patients an increased depth of vision with a minimal level of dysphotopsia.
This prospective case series sought to investigate visual and refractive outcomes as well as patient satisfaction after cataract surgery and LuxSmart EDOF IOL implantation.
Materials and methods
Study design
This is a two-center, prospective observational clinical study that included patients who underwent cataract extraction and implantation of a LuxSmart EDOF IOL (Bausch+Lomb, Rochester, NY, USA).
Patient selection
In total, 32 patients were recruited, treated, and followed up between the dates of 25th June 2020 and 21st March 2021. All patients over the age of 50 who had bilateral cataractous eyes with regular astigmatism up to 1.5 D, and no other significant ocular comorbidity, were eligible for inclusion in this study. Excluded ocular comorbidities were significant dry eye, keratoconus, advanced glaucoma, diabetic retinopathy, macular degeneration, or cystoid macular edema. Patients with regular astigmatism (>1.5 D) or irregular astigmatism on corneal topography were excluded. Patients who did not undergo bilateral LuxSmart implantation during the study duration were also excluded. Patients were required to have a well-dilated pupil after topical treatment to facilitate postoperative assessment of the IOL axis. Patients with previous intraocular or corneal surgery, evidence of instability on keratometry, or biometry measurements were also excluded.
Data collection
Patients attended one preoperative visit. They were also examined 1 or 2 days postoperatively. Follow-up visits were scheduled at 1 week, 1 month, 3 months, and 6 months postoperatively. Preoperative visits included the measurement of intraocular pressure (IOP) and visual acuity (VA) using a Snellen chart then converted to logMAR. A dilated fundus exam and an optical coherence tomography (OCT) scan of the macula were also performed, along with optical biometry using either an IOL Master 500 or 700, which measured anterior chamber depth (ACD), axial length (AL), white-to-white (WTW), and keratometry measurements. For monocular data, the first-operated eye was used regardless of laterality. The surgical procedure is presented in video format (see Supplemental Digital Video 1).
The main outcomes of the study are the visual and refractive results as well as patient satisfaction. Patients were checked for subjective refraction and slit lamp findings at each postoperative visit. Binocular and monocular uncorrected and corrected distance vision as well as intermediate and near vision at 80, 66, and 40 cm were also assessed. Safety outcomes, including all postoperative complications, were assessed in this study. Defocus curves were completed using a standard approach on an electronic Snellen chart where the subject’s eye is corrected to their best distance refraction followed by defocusing the eye by 0.5 D increments between −2.5 D and +1.5 D. Additionally, +0.25 D and −0.25 D lenses were used for fine-tuning. Letters are randomized between the different lens measurements.
Subjective outcome measures were obtained using the Catquest-9SF and the Patient-Reported Spectacle Independence Questionnaire (PRSIQ).7,8 Patients also had a subjective assessment of glare, halos, and sunburst using a halo and glare simulator 9 and were asked to explain the frequency and the severity of each phenomenon. Contrast sensitivity was evaluated in both mesopic and photopic conditions using the CSV-1000 device (VectorVision, TX, USA). Patient outcomes were compared with previously obtained healthy control data.
The LuxSmart IOL
The LuxSmart EDOF IOL (Bausch+Lomb, Rochester, NY, USA) is a biconvex, aspheric refractive optic with no diffractive rings. It is a type 5 EDOF IOL that uses fourth- and sixth-order spherical aberration of opposite signs, located in the central 2 mm, to increase the subjective depth of field to approximately 1.5 D with vergence gradually reduced from the center to the periphery.4 –6 The achieved DOF is due to: fourth-order SA (Z4,0) of about 0.88 D; and sixth-order SA (Z6,0) of the opposite sign of about 2.00 D, both combined lead to nearly 1.50 D DOF. 10 The periphery of the LuxSmart IOL is aberration-free due to its aspheric design. 10 The scientific literature reports that it may achieve significantly higher distance-corrected intermediate VA as well as higher performance for near vision, as compared with a conventional monofocal IOL, without increasing the risk of photic phenomena.3,11 It is a hydrophobic, acrylic copolymer containing an ultraviolet (UV) absorber. The lens has a four-point fixation and a 360° square-edge design. It is available in powers of 0 to +34 D. The 6 mm optic is formed from a 2 mm EDOF center surrounded by a transition zone and a monofocal periphery. Lenses with four-point fixation have been shown to provide better centration and reduced rotation compared to open-loop fixation or ‘haptic-less’ lenses, which is critical in the optimization of post-operative refractive outcomes with EDOF lenses.12 –15 Additionally, aspheric lenses have been associated with reduced aberrations and improved contrast sensitivity compared to spheric or multifocal lenses, whereas square edges have been shown to reduce the rates of cell proliferation in the lens epithulium.2,16,17
Procedure
All surgeries were performed at the Gemini Eye Clinics in Zlin and Vyskov, Czech Republic and were performed as standard phacoemulsification under intracameral anesthesia through a 2.2 mm limbal corneal incision on the steep corneal axis to reduce astigmatism. A 5.5 mm capsulorhexis was performed. The LuxSmart IOL was inserted in the bag in all cases. The patients had bilateral phacoemulsification and IOL implantation and the maximum time between the first and second eye implantation was 30 days. Patients were medicated with a tapered dose of steroid and non-steroid anti-inflammatory eye drops for 1 month and topical antibiotics for 2 weeks.
Target refraction
Patients were targeted for emmetropia to permit good unaided distance and intermediate vision. To aim for these target refractions, IOLs were implanted from a range of 18.0–26.0 D. The IOL power was calculated using the SRK/T formula and the IOLMaster (Carl Zeiss Meditec AG, Jena, Germany). The A constant was changed from 119.2 to 118.6 from patient 24 (OS) onward in order to optimize postoperative outcomes.
Statistical methods
Data values are presented as a mean ± standard deviation unless otherwise specified. Graphical data was prepared using GraphPad (Prism, San Diego, CA, USA). A p value of <0.05 was considered statistically significant, and p values are indicated where applicable.
Results
Patient population
In total, 32 patients were recruited for this study. However, two patients did not have bilateral LuxSmart IOL implantation and were therefore excluded from the dataset: one preferred waiting before receiving the surgery in the fellow eye, which still had good vision—LuxSmart IOL was implanted in the second eye after the study; the other patient could not be implanted with LuxSmart in the fellow eye because the IOL power needed was not available. Of the 30 patients who underwent bilateral surgery (60 eyes in total), 13 were male and 17 were female. The mean age was 66.5 ± 7.8 (range 50–80) years. A summary of the patients’ preoperative biometry results is presented in Table 1.
Preoperative biometry data.

ACD, anterior chamber depth; AL, axial length; WTW, white-to-white; K, keratometry measurements.
Refractive outcomes
The mean spherical equivalent was 1.04 ± 1.67 D (range: −4.0 to +4.50 D, and median: 1.06 D) preoperatively and −0.30 ± 0.463 D (range: −1.50 to +0.75 D, median: −0.25 D) 6 months postoperatively. The absolute cylinder remained stable at 0.37 ± 0.352 D (range: 0–1.25 D, median: 0.50 D) preoperatively and 0.41 ± 0.32 D (range: 0–1 D, median: 0.50 D) 6 months postoperatively.
VA outcomes
Monocular uncorrected distance VA (UDVA) was 0.57 ± 0.32 (range: 0.13–1.3, median: 0.52; logMAR) at baseline and improved to 0.086 ± 0.12 (range: −0.06 to 0.48, median: 0.075; logMAR) 6 months postoperatively, whereas monocular corrected distance VA (CDVA) was 0.17 ± 0.18 (range: −0.08 to 0.6, median: 0.11) and −0.03 ± 0.065 (range: −0.16 to 0.11, median: −0.04) (logMAR) at baseline and 6 months, respectively. Monocular and binocular VA at different distances, 6 months postoperative, are presented in Table 2 as well as Figures 1 and 2.
Monocular and Binocular VA at different distances at 6 months postoperatively.
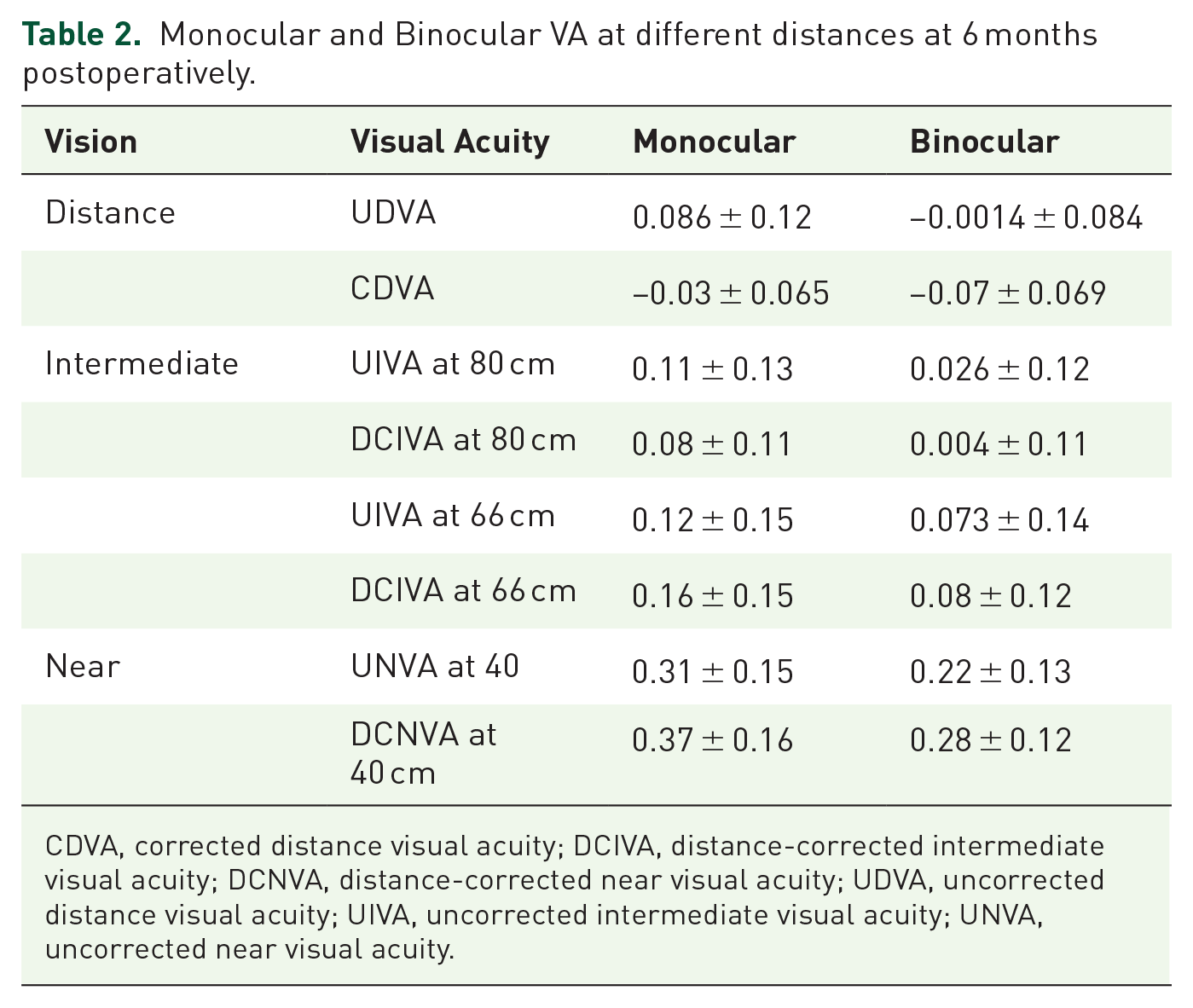
CDVA, corrected distance visual acuity; DCIVA, distance-corrected intermediate visual acuity; DCNVA, distance-corrected near visual acuity; UDVA, uncorrected distance visual acuity; UIVA, uncorrected intermediate visual acuity; UNVA, uncorrected near visual acuity.
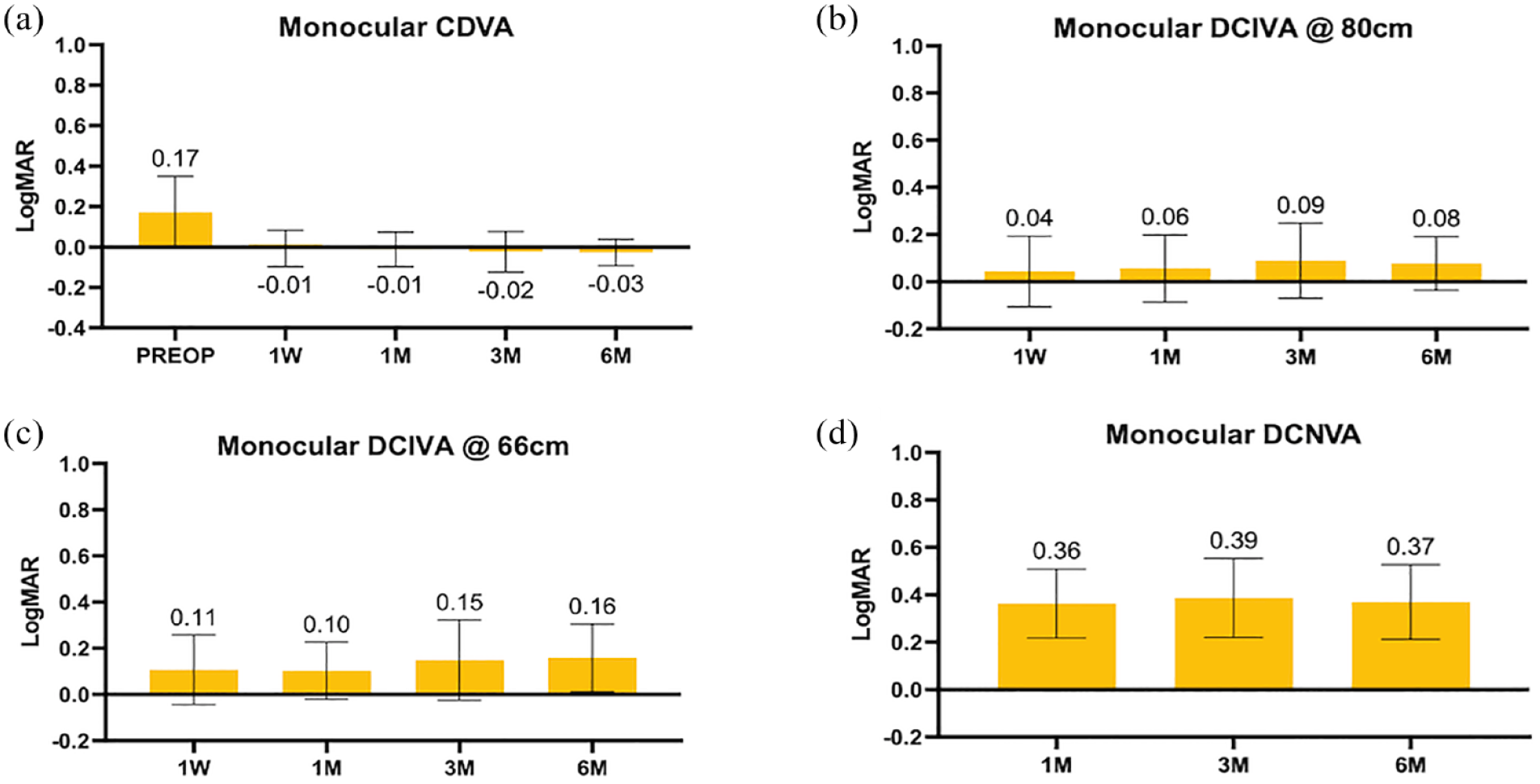
Monocular corrected visual acuity (VA) outcomes: (a) Monocular corrected distance visual acuity. (b) Monocular corrected intermediate visual acuity at 80 cm. (c) Monocular corrected intermediate VA at 66 cm. (d) Monocular corrected near visual acuity. Graph values represent means, and error bars represent standard deviations.

Binocular corrected visual acuity (VA) outcomes: (a) Binocular corrected distance visual acuity. (b) Binocular corrected intermediate VA at 80 cm. (c) Binocular corrected intermediate VA at 66 cm. (d) Binocular corrected near visual acuity. Graph values represent the mean and error bars indicate standard deviation.
Patients were tested for their range of acceptable uncorrected vision using a monocular defocus curve (Figure 3). Mean values across the number of patients tested (n = 29) and acceptable vision was taken as <0.2 logMAR, which was shown between −1.5 D and +0.5 D.
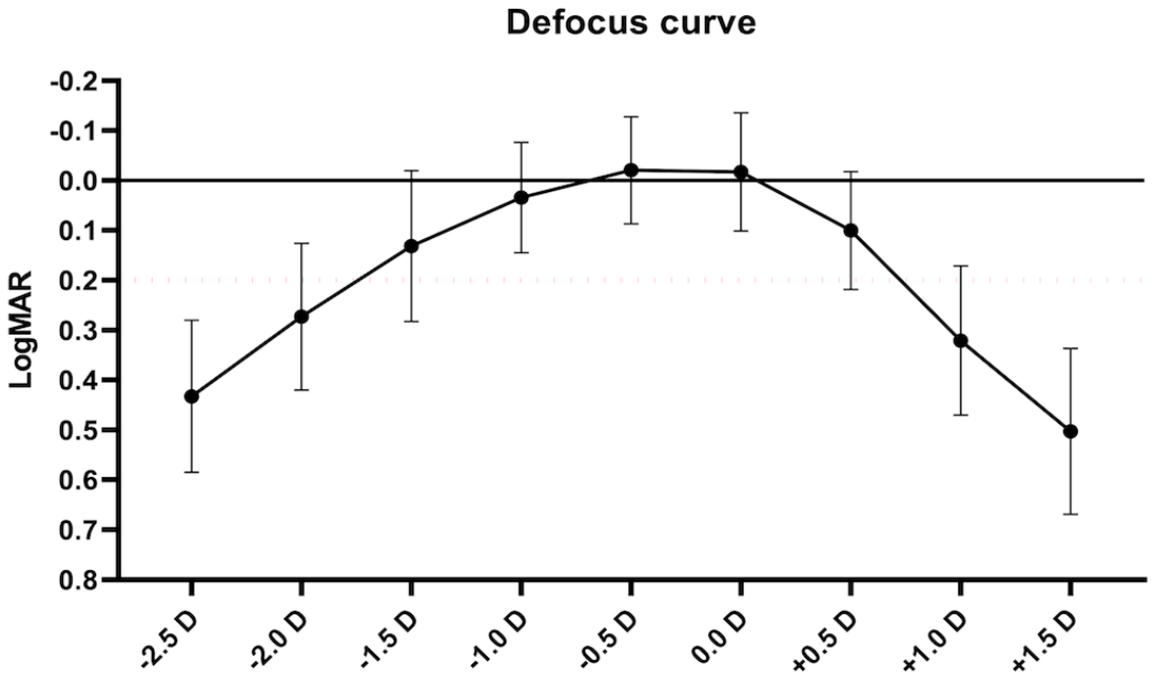
Monocular manual defocus curve. Figure represents mean values across all treated patients. 0.2 logMAR was determined as acceptable visual acuity.
Patients (n = 29) were also assessed for contrast sensitivity under photopic, mesopic, and mesopic with glare situations (Figure 4). Mean contrast sensitivity for the photopic conditions was within the normal range except for the 12 cycles per degree (cpd) test, which was slightly below the normal range. Mesopic conditions were slightly below the normal for all tests except for the smallest spatial frequency, where both were at the lower end of the normal range. However, the normal range available is only applicable to the younger population.

Contrast sensitivity curves. (a) Photopic contrast sensitivity at varying 3–18 cpd spatial frequencies. (b) Mesopic contrast sensitivity at varying 3–18 cpd spatial frequencies. Patients are tested in both mesopic and mesopic + Glare scenarios. Normal ranges are presented in green as per previous studies (Pomerance GN, Evans DW, 1994).
There were statistically significant differences between monocular and binocular distance-corrected visual assessments of CDVA, DCIVA at 80 cm, DCIVA at 66 cm, and DCNVA at 40 cm (all p values <0.001, Mann–Whitney test). Similarly, there were significant differences between monocular uncorrected and distance-corrected visual acuities for distance VAs (p < 0.001, Wilcoxon test), intermediate VAs at 80 cm (p = 0.006), intermediate VAs at 66 cm (p = 0.006), and near VAs at 40 cm (p < 0.001). Binocular differences between uncorrected and distance-corrected VAs were only noticed between distance VAs (p < 0.001, Wilcoxon test), intermediate VAs at 80 cm (p = 0.04), and near VAs at 40 cm (p < 0.001). Differences between binocular UIVA and DCIVA at 66 cm were not significant (p = 0.10).
Patient-reported outcome measures
Two patient-reported outcome measures were used to assess spectacle independence (PRSIQ) as well as visual disability (Catquest-9SF). Patients demonstrated a high level (>80%) of glasses independence for distance and intermediate vision. Additionally, >90% of patients were either fairly or very satisfied with their postoperative vision.
Patient halo, starburst, or glare were assessed using a questionnaire. The median values for subjective halo were modeled using a Halo & Glare simulator (ViSU-L GmbH, Hannover, Germany). 9 When assessed by questionnaire, halo, starburst, and glare were present in less than 50% of patients. Only one patient (1/28) frequently experienced these phenomena. Of those who experienced these visual symptoms, most cases had mild symptoms. Only 2/28 experienced severe halos, 1/28 had severe starburst, and 1/28 reported severe glare (Figure 5).

Patient-reported severity of halo, starburst, and glare.
Complications
Postoperatively, one patient was observed to have suspected vitreomacular traction, which was considered unrelated to the IOL. No patient had persistent anterior chamber inflammation, cystoid macular edema, or ocular hypo/hypertension.
Discussion
EDOF IOLs offer a larger focal range with respect to monofocal lenses while limiting the glare and contrast sensitivity issues associated with multi-focal/trifocal lenses. 18
The LuxSmart IOL is based on the principle established by Benard et al., which states that the fourth- and sixth higher-order aberrations with opposite signs can increase the DOF.4,6 In the LuxSmart IOL, these higher-order aberrations are located in the central 2 mm with a power gradient from the center toward the periphery, which is optimized for distance. 19
This study recruited a total of 60 eyes to undergo routine phacoemulsification cataract surgery with the LuxSmart EDOF IOL implantation. The study included patients with astigmatism ⩽1.5 D, unlike most studies that limited the inclusion criteria to ⩽0.75 D. This was decided considering that the aim of the study was to evaluate LuxSmart IOL in a ‘real-world’ patient population. Moreover, the corneal astigmatism was managed by an incision on the steep corneal axis.
Patients demonstrated significant distance, intermediate, and near VA improvements from baseline to 6 months (Table 2, Figures 1 and 2). Median monocular DCIVA was 0.2 (0.06–0.24, IQR) (logMAR), which would fulfill the American National Standards Institute ANSI Z80.35-2018 co-primary effectiveness endpoint. 20 Median logMAR binocular UNVA at 6 months was 0.21 (0.15–0.3, IQR), whereas mean logMAR binocular DCNVA was 0.27 (0.21–0.37, IQR). This decrease in DCNVA may be due to the patients becoming slightly myopic postoperatively (−0.30 ± 0.46 D, median: −0.25, IQR: −0.5 to −0.09), thus distance correction moved the focal point further from the eye.
These VA results are comparable with the results of Tahmaz et al., 19 and Campos et al. 11 who studied LuxSmart IOL in comparison with monofocal IOLs. Our results are also comparable with Vivity (Alcon Laboratories, Inc, Irvine, CA, USA), a commercially available EDOF IOL of similar design (type 5 in the Megiddo-Barnir classification): in a recent study by Al-Amri et al., UDVA, CDVA, and UNVA were comparable to the results of the current study. 21 Another IOL of similar design is the Elon IOL (Medicontur); however, to the authors’ knowledge, the scientific literature does not yet report results for this IOL. Table 3 shows the visual outcomes of LuxSmart IOL, Vivity IOL, as well as a number of EDOF IOLs of different designs.11,19,21 –28
Comparison between LuxSmart EDOF, Vivity, Mini WELL Ready, TECNIS Synergy, and Isopure.

CDVA, corrected distance visual acuity; DCIVA, distance-corrected intermediate visual acuity; DCNVA, distance-corrected near visual acuity; IOL, intraocular lenses; UDVA, uncorrected distance visual acuity; UIVA, uncorrected intermediate visual acuity; UNVA, uncorrected near visual acuity.
Monocular defocus curve analysis demonstrates a defocus of +1.5 D while maintaining a VA of 0.2 logMAR or better (Figure 3). This is more than expected for a typical monofocal lens but less (+2.0 D) than that reported in a meta-analysis for other EDOF and multifocal lenses (+2.0 D and +3.0 D), respectively. Given the different methodology for measuring contrast sensitivity, it is difficult to compare our results with that of other studies. Mean contrast sensitivity for the photopic conditions is within the normal range except for the 12 cpd test.
The contrast sensitivity testing performed in this study demonstrated comparatively good visual outcomes in both mesopic and photopic conditions. Normal ranges are presented in Figure 4, as per previous studies. 29 Contrast sensitivity results in mesopic conditions are slightly below normal for all tests, except for the smallest spatial frequency, which is within the normal range. However, the normal range is only available for the younger population, which may not allow for an adequate comparison of our data obtained from the older population. Moreover, the contrast sensitivity is expected to be affected in EDOF IOLs as a trade-off to the elongation of focus due to distribution of light energy rather than focusing it in one focal point.
As for halos and glare, a recent study has shown that the incidence of halos and glare with LuxSmart IOL is comparable to monofocal IOL where in both IOLs 66.7% never had photic phenomena and only 8.3% had them often. 11 In the current study, more than 50% of patients did not have photic phenomena, and only one patient (1/28, 3.5%) had them frequently and only 2/28 had severe symptoms (7%).
A recent study reveals that the lens Modulation Transfer Function (MTF) measurements, which represent the magnitude of response of an optical system in the imaging process, have shown to be a good metric to predict the visual performance at different levels of focus in pseudophakic patients and are consistent with a wide and smooth transition between far as well as intermediate distances vision. 5 The MTF curve for the LuxSmart at the intermediate focus reaches relatively higher values than the MTF curve at the far focus, thus confirming the high optical quality of the intermediate focus and is consistent with the visual performance expected from an EDOF lens.
The present study demonstrated a good and tolerable safety profile of the LuxSmart EDOF lens. There was one event of suspected vitreomacular traction reported at the 6-month assessment, which is a rarely reported complication of cataract surgery and is unlikely to have been directly caused by the IOL. 30
Patient satisfaction was assessed using the Catquest-9SF as well as the PRSIQ.7,8 All patients except one (3.6%) reported that they were either fairly satisfied (53.6%) or very satisfied (42.8%) after the treatment. All activities that had caused some difficulty because of eyesight have improved after the surgery. Of the patients, 90% are spectacle-free for distance and intermediate vision, whereas 76% of patients still require glasses for reading. Visual phenomena complications such as glare, halos, and starbursts were low in this cohort. Realistic expectations contribute to patient satisfaction, since EDOF IOLs are known to provide excellent distant and intermediate vision, as well as ‘good enough’ near vision. 21
The primary aim of EDOF lenses is to improve the depth of field of vision while avoiding the potential complications of multifocal lenses, 2 and there are now several EDOF lens options available. The authors have previously reported the visual outcomes of the ISOPURE, PhysIOL, in a similar study design. 31 The use of this lens in a study of 18 patients with bilateral cataract showed similar improvements in uncorrected and corrected vision, although similar quality-of-life assessment was not made. There is currently a larger trial comparing the use of this lens against a monofocal lens, 32 and further research is required to investigate longer-term outcomes of EDOF lenses such as the LuxSmart, the ISOPURE lens, the Tecnis Eyhance, and the Acrysof IQ Vivity. 23
This study has a number of limitations. First, sample size analysis was not performed when it was decided to implant 30 patients (60 eyes). Second, the A constant was changed during the course of the study to optimize postoperative refractive outcomes for patients. Therefore, it is difficult to directly compare the results of those patients with different A constants. Third, patients with an astigmatism of <1.5 D were included in this study. This level of astigmatism may lower postoperative uncorrected visual acuity. Finally, this study did not have a control arm to compare the EDOF lens implantations with. Nevertheless, there is a significant literature base describing the visual, refractive, and quality-of-life outcomes with standard IOLs, which can serve as a comparator to this data.33,34
In summary, the data presented in this study forms the first case series of patients who have received the LuxSmart EDOF (Bausch+Lomb) intraocular lens after phacoemulsification surgery. The data demonstrates good VA outcomes. The LuxSmart IOL single elongated focal point enhances the DOF as per its design. Additionally, patient satisfaction as assessed by validated patient-reported outcome measures was high. These findings suggest that the LuxSmart EDOF IOL bridges the gap between monofocal and multifocal IOL for patients who desire an increased depth of vision.
Footnotes
Acknowledgements
Medical writing assistance provided by GP Communications and funded by B+L. The authors have authorized the submission of their manuscript by GP Communications and approved the statements and declarations in this submission.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
